Abstract
Introduction
Bile spillage (BS) is common during laparoscopic cholecystectomy (LC) and has been shown to be associated with an increased risk of surgical site infection (SSI). We hypothesized that positive bile cultures (PBCs) increase the risk of postoperative complication including SSI.
Methods
A retrospective chart review was conducted including all patients older than 18 years undergoing urgent LC from January to September 2019. Charts were reviewed for the index admission and postoperative visits. We compared those who had PBCs with those who did not. Our primary endpoint was the rate of SSI. Univariate analysis and multivariate logistic regression were used to identify predictors of SSI.
Results
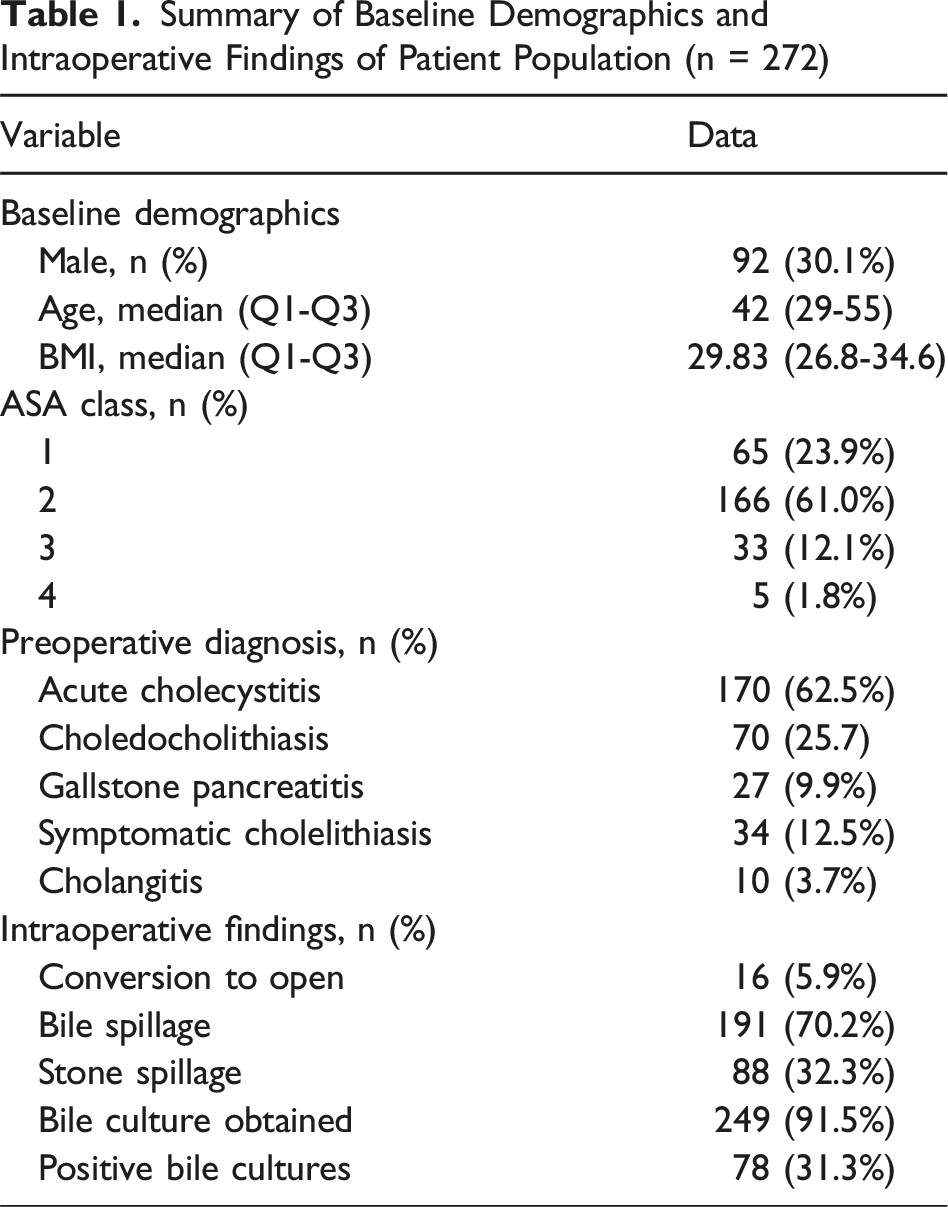
272 patients underwent LC. Indications for operation included acute cholecystitis (62.5%), symptomatic cholelithiasis (12.5%), and other indications (25%). Bile was spilled in 191 patients (70.2%). Positive bile cultures were obtained in 78 of 249 (31.3%) patients and were associated with preoperative endoscopic retrograde cholangiopancreatography (ERCP; 44% vs 26%, P = 0.014) and drain placement (50% vs 28.9%, P = 0.031). Eleven postoperative complications were noted, including 6 SSI (2.2%). Positive bile culture (3.8% vs 1.8%, P = 0.38), BS (3.1% vs 0%, P = 0.18), ERCP (1.4% vs 2.5%, P = 1.0), and drain placement (6.7% vs 1.7%, P = 0.13) were not associated with SSI. Multivariate analysis demonstrated that positive cultures were not predictive of complication (P = 0.13) or SSI (P = 0.91).
Conclusion
Positive bile cultures are not inherently associated with an increased risk of SSI and therefore should not lead to ongoing postoperative antibiotic therapy.
Background
Laparoscopic cholecystectomy (LC) remains the gold standard for the management of acute cholecystitis. Large-cohort studies have demonstrated that emergency laparoscopic cholecystectomy (EMLC) is associated with increased postoperative morbidity, including a higher incidence of subtotal cholecystectomy, prolonged hospital stay, SSI (surgical site infection), and the need for additional interventions.1-3 Although EMLC presents technical challenges due to active inflammation, immediate intervention has been shown to have decreased overall morbidity compared to elective interval cholecystectomy. 4 However, the active inflammation of acute biliary pathology causes frequent intraoperative bile and gallstone spillage with reported rates of bile spillage (BS) ranging from 10% to 40%. 5
The impact of BS on postoperative (SSI) remains inconclusive and controversial. Despite significant advancements in minimally invasive techniques, postoperative infectious complications are a significant concern following EMLC, albeit relatively rare. Independent risk factors for SSI, including patient age, gender, severity of symptoms, longer operative duration, and open surgical approach, have all been well documented across multiple studies.1-3 Previous studies have also shown an increased association between BS and postoperative SSI.6-8 While BS categorically changes the wound class of an EMLC from class two to class three, the presence of positive bile cultures (PBCs) may indicate an increased infectious burden akin to non-contained stool spillage, thereby contributing to adverse postoperative outcomes. However, it remains unclear whether the presence of PBC, independent of BS, further elevates the risk of SSI and other postoperative complications.
This study aims to investigate whether PBCs, in conjunction with BS, are associated with heightened SSI rates. Furthermore, we assess whether targeted antibiotic therapy, guided by intraoperative microbiological findings, reduces the incidence of SSI and other complications.
Methods
We conducted a prospective analysis of 272 consecutive patients who underwent cholecystectomy for symptomatic acute biliary tract disease over 1 year at Harbor-UCLA Medical Center. This study was reviewed and approved by the Institutional Review Board at Harbor-UCLA Medical Center. Patients older than 18 admitted for acute biliary tract disease (biliary colic, symptomatic cholelithiasis, cholangitis, acute cholecystitis, gallstone pancreatitis, and choledocholithiasis) and who underwent laparoscopic cholecystectomy and laparoscopic converted to open cholecystectomies were included. Open cholecystectomy, prisoners, cognitively impaired participants, or those not having any inclusion criteria were excluded from further analyses.
All patients were admitted through the emergency department and underwent same-admission cholecystectomy. At our institution, patients are admitted to the acute care surgery team. Acute care surgery is routinely performed by senior residents under the supervision of the primary attending who is present throughout the operation. Of note, all patients with acute cholecystitis or cholangitis were started on preoperative antibiotics upon admission to the hospital. The standard regimen at our hospital was ceftriaxone and Flagyl, but both preoperative and postoperative antibiotics were not protocolized within this study and were up to attending discretion based on severity of presentation or intraoperative assessment. All other patients were given antibiotics (cefoxitin) within 1 hour of skin incision. Microbiology bile specimens were collected by either aspirate via a laparoscopic cyst-aspirator or a swab from the gallbladder specimen which was opened on the back table immediately after removal from the abdomen. These were then sent for definitive pathology review following the conclusion of the operation. If bile was spilled during the operation, normal saline was used for irrigation, but the total amount of fluid was not standardized. If a case had significant inflammation, purulent bile, or necrosis, per surgeon discretion, a Jackson-Pratt drain was left in the gallbladder fossa at the end of the case. Severity of acute cholecystitis was based off of Tokyo 2018 criteria. 9 Tokyo grades II and III were grouped together in the analysis for SSI. Clinical evaluations were made throughout their length of stay in the hospital and during scheduled 2-week postoperative clinic visits, when appropriate. It takes 1 day for the Gram stain to come back from the bile cultures and 2 days for speciation of the biliary cultures. Antibiotic choice postoperatively was initially empirically chosen and then was dependent on the culture data.
Our primary outcome measure was surgical site infection (SSI), classified as either superficial or deep. Superficial SSI was limited to only the skin or subcutaneous tissue of the surgical site. Deep SSI was limited to the muscle or organ space infections. Organ/space infections were confirmed with imaging studies. A positive confirmation of SSI required antibiotic prescription during the patient’s admission or during the postoperative clinic visit.
The following data were recorded: baseline demographics (age and gender), comorbidities (BMI, ASA, hypertension, history of renal failure, or liver disease), intraoperative details (bile spillage, presence of empyema or hydrops, conversion to open, common bile duct exploration (CBDE), and intraoperative cholangiogram (IOC)), postoperative complications (SSI, bile leakage, and biloma), and length of stay. To examine whether bile spillage was associated with SSI, we recorded whether growth was present in the microbiology submissions and, if pertinent, described the specific bacteria.
Statistical analysis was performed using Stata Statistical Software (Release 17). We compared patients who had positive bile cultures and spillage with those who did not.
We defined a value of P < 0.05 as statistically significant. Our primary endpoint was the rate of SSI. A univariate analysis was used to identify predictors of SSI. We used the Mann-Whitney U test to compare continuous variables and the chi-square or Fisher’s exact test for the categorical variables, as appropriate multivariate logistic regression was used to identify independent predictors of SSI including the following variables: age, sex, history of DM, severity of cholecystitis, required ERCP, intraoperative drain placement, conversion to open surgery, postoperative complications, bile spillage, positive bile cultures, postoperative bile leak, and postoperative biloma.
Results
Summary of Baseline Demographics and Intraoperative Findings of Patient Population (n = 272)
Summary of Postoperative Outcomes (n = 239)

Comparison of Patients Who had Positive Bile Cultures to Those Who Did Not
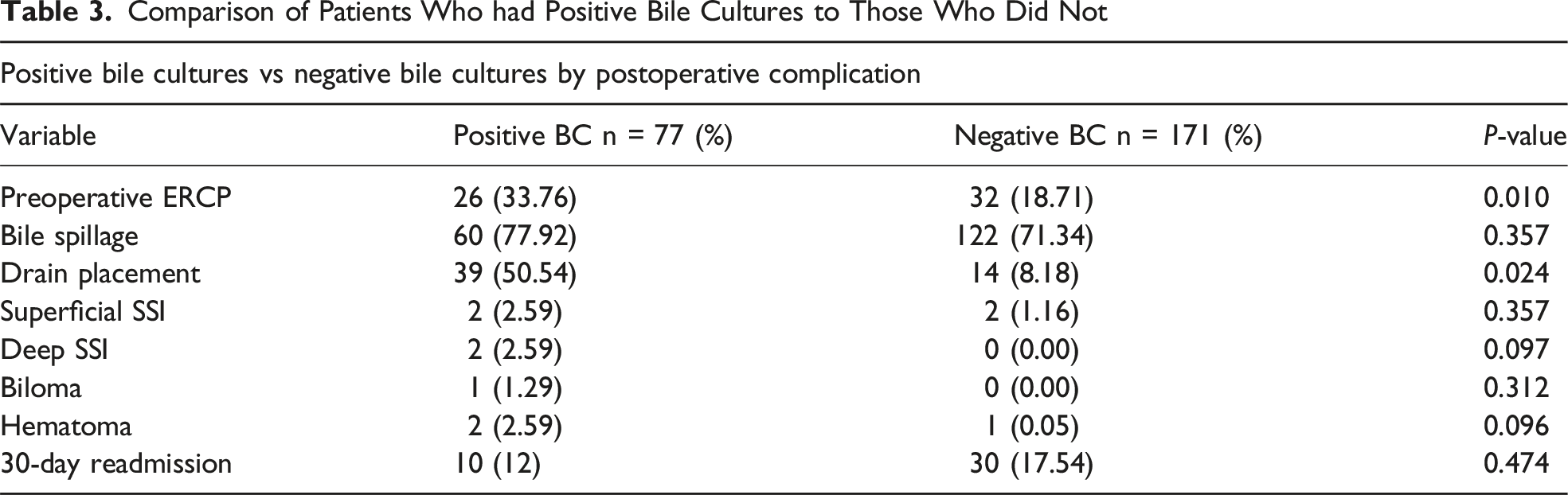
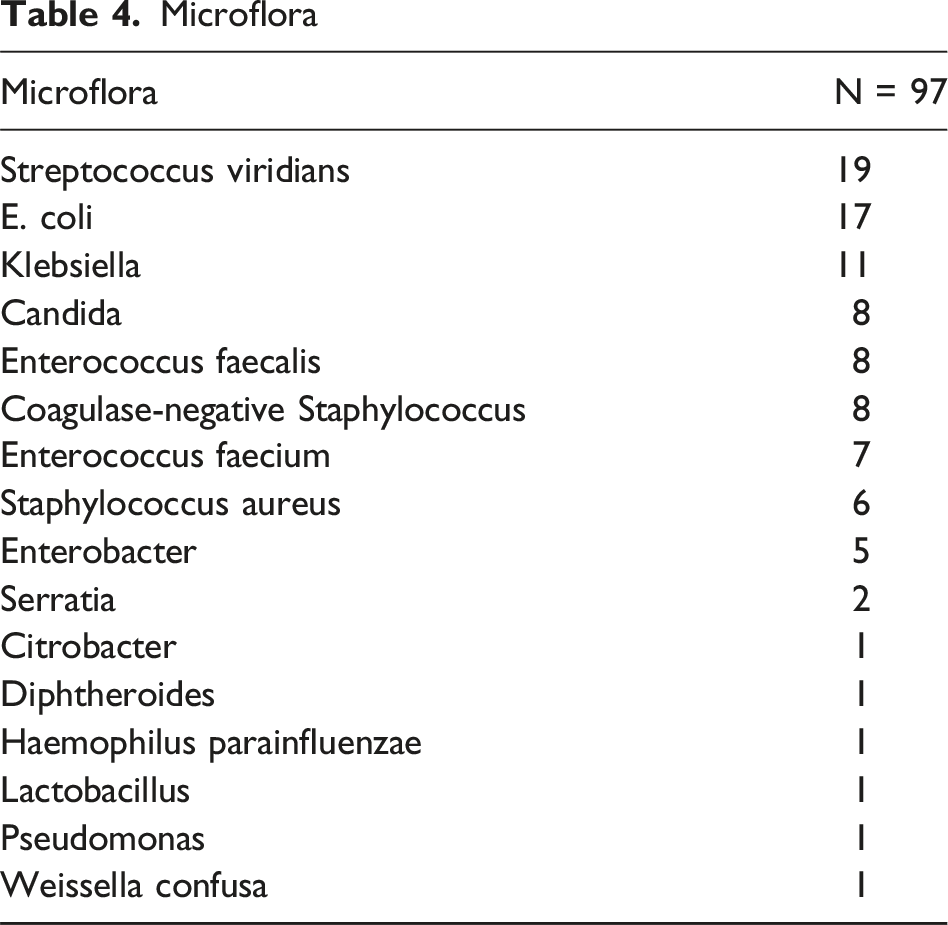
Microflora
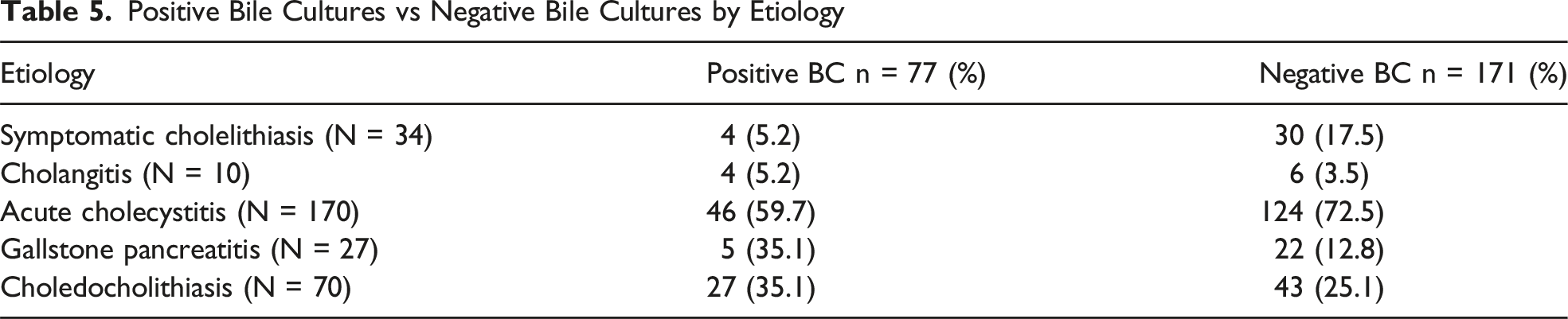
Positive Bile Cultures vs Negative Bile Cultures by Etiology
Discussion
In this study of acute symptomatic biliary pathology, we found no significant association between PBC and SSI or other postoperative complications. Additionally, we observed a correlation with history of DM, male sex, and severity of cholecystitis, but no correlation between bile spillage, drain placement, and ERCP with SSI. Notably, 31% of all patients with varying acute biliary pathologies presented with PBC. Among these patients, 50% underwent intraoperative drain placement, compared to only 8% of negative bile cultures, exhibiting a strong correlation between gallbladder inflammation, necrosis, and bacterial growth. Similarly, a substantial proportion of patients with choledocholithiasis demonstrated PBC (29.8%) in this study, emphasizing the risk for ascending cholangitis and sepsis in this population. Despite this high prevalence of PBC, our findings suggest that postoperative antibiotics are not warranted in PBC patients given the low risk of SSI and postoperative infectious complications.
Postoperative infectious complications remain a concern following EMLC. There are multiple well-validated independent risk factors for developing both superficial and deep postoperative skin and soft tissue infections after EMLC including advanced age, male sex, severity of acute cholecystitis, prolonged operative duration, prior history of cholecystitis, preoperative ERCP, DM, and conversion to open surgery.1-3,10,11 In our study, we also found that male sex, Tokyo grades 2-3 acute cholecystitis, and a history of DM correlated with SSI. We did not find a correlation between PBC, BS, drain placement, or ERCP with an increased risk of SSI. Conversion to open cholecystectomy also had no increased risk of SSI. Intraoperative JP drain placement had an OR of 1.97 but did not reach significance, which could correlate the surgeon’s judgement in particularly infectious gallbladders due to inflammation, purulence, or necrosis. These findings align with existing literature and underscore the importance of patient selection and perioperative management in mitigating infectious complications.
The impact of BS on SSI has been widely debated, with conflicting data on whether it independently increases the risk of postoperative complications or merely serves as a marker of disease severity. Peponis et al identified BS itself as an independent risk factor for SSI, reporting an OR of 2.29. 6 However, confounding variables including an older patient population, higher ASA scores, greater rates of acute cholecystitis and EMLC, and higher conversion to open cholecystectomy within their BS cohort may have influenced their findings. In contrast, Sarli et al and Jain et al demonstrated no significant association between BS and SSI provided adequate prophylactic antibiotics and peritoneal irrigation.7,8 Our study, which reported a higher BS rate (70%) than prior studies, aligns with these findings.9-11 In our study, all SSIs occurred in cases with BS (6 out of 191 cases with spillage and 0 out of 81 without spillage). However, this was not statistically significant. Because SSI only occurred in BS cases, it was not able to be utilized in our multivariate analysis since it was 100% correlated. The elevated BS rate in our cohort might be explained by the level of autonomy afforded to trainees at our institution and the increased complexity of cases due to delayed patient presentations and a high proportion of severe cholecystitis. Despite the frequency of BS in our study, our SSI rate remained within the reported range of 0.5% to 6.2% following LC, reinforcing the notion that BS alone is not a significant predictor of infectious morbidity.11-16
While PBCs have been frequently associated with severe biliary disease, their clinical relevance in guiding postoperative management, particularly regarding postoperative infections, remains uncertain. Ramírez-Giraldo et al reported a PBC rate of 52% in their prospective cohort of 700 patients, with common isolates including Escherichia coli, Klebsiella pneumoniae, Enterococcus, and B.Fragilis, as well as a 53% risk for multi-drug resistance. 17 This paralleled the microbiological findings within our study as well. However, we also had a large percentage of Streptococcus viridans, Candida, and coagulase-negative Staphylococcus. PBCs were associated with cholecystitis severity, older age, diabetes, and choledocholithiasis. 17 This was similar to the reported rate of PBC (20%-70% of all cases) as a byproduct of cholestasis and cholecystitis by Dyrhovden et al. 18 Our study reported a lower rate of 31%, yet no correlation between PBC and increased risk of SSI in both univariate and multivariate analyses. When looking at cases with BS, there was no significant correlation between PBC with SSI with an equal number of patients with SSI having PBC and negative bile cultures. It is our practice pattern to give preoperative antibiotics, retrieve all stones, and use copious irrigation in cases with BS. These methods could mitigate the risk of BS, especially in BS with PBC, and help explain our cohort’s low rate of SSI. Positive bile cultures with BS represent an increased risk of infectious contamination as a class-three wound if not class four in select cases. 19 Galili et al identified PBC as a significant factor in infectious postoperative complications, including SSI, in acute cholecystitis. 20 However, confounding variables for SSI including a higher percentage of older, diabetic, male patients with more advanced cholecystitis in the PBC grouping could have influenced their results. In contrast, Yoon et al found no significant association between PBC and postoperative infections in a cohort with moderate to severe cholecystitis. 21 Our findings contribute to this growing body of evidence, challenging the assumption that BS and PBC necessitate prolonged antibiotic therapy, highlighting the need for a more nuanced approach to infection risk stratification in EMLC patients.
Excessive antibiotic use contributes to bacterial resistance and increased health care costs, underscoring the importance of selective postoperative antibiotic administration. While the necessity of postoperative antibiotics remains controversial, existing literature suggests their role in SSI prevention is limited in low-risk patients undergoing EMLC.22-25 A randomized control trial by Regimbeau et al demonstrated that restricting postoperative antibiotics in mild and moderate acute cholecystitis was non-inferior in postoperative infectious outcomes when compared to their treatment group of 5 days of postoperative Augmentin. 24 For severe cholecystitis, the Surgical Infection Society’s Therapeutics and Guidelines Committee recommends a maximum of 4 days of postoperative antibiotics. 22 In our cohort, postoperative antibiotics were administered in 62 patients out of the 272 EMLC performed for severe cholecystitis with only two of those patients having positive blood cultures. Of the 6 patients diagnosed with postoperative SSI, 5 had received postoperative antibiotics highlighting the association between severe cholecystitis and increased SSI risk rather than the presence of PBC alone. Given the low incidence of SSI despite frequent BS, these findings support the judicious use of postoperative antibiotics, aligning with current principles of antimicrobial stewardship.
Our study has several limitations. As a single-center study at a county hospital with low patient medical literacy, poor health care access, and low socio-economic population, its generalizability may be limited. Additionally, the small sample size and low SSI rate within this cohort reduce statistical power in detecting the true impact of PBC on overall SSI. This leads to a higher chance of type II error. We also did not analyze bacterial resistance patterns or stratify cholecystitis severity, which could represent confounding variables. Furthermore, a 12% loss to follow-up may have affected postoperative complication analysis. However, this study represents one of the most comprehensive prospective evaluations of PBC on postoperative infectious complications across a spectrum of acute biliary pathologies. Further prospective studies are warranted to better define the role of intraoperative microbiological findings in guiding targeted interventions and optimizing postoperative outcomes in biliary surgery.
Conclusion
Positive bile cultures were not independently associated with an increased risk of surgical site infection.
Footnotes
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
