Abstract
Background
Appendicitis in children can be treated with operative management (OM) or non-operative management (NOM) depending on patient risk factors. Our goal was to evaluate for differences in being offered NOM before and after the implementation of a standardized clinical pathway among patients of different demographic backgrounds.
Methods
This was a single center retrospective study of patients under the age of 18 years with appendicitis. Univariate regression was used to assess for associations between demographic factors and patients who were offered NOM.
Results
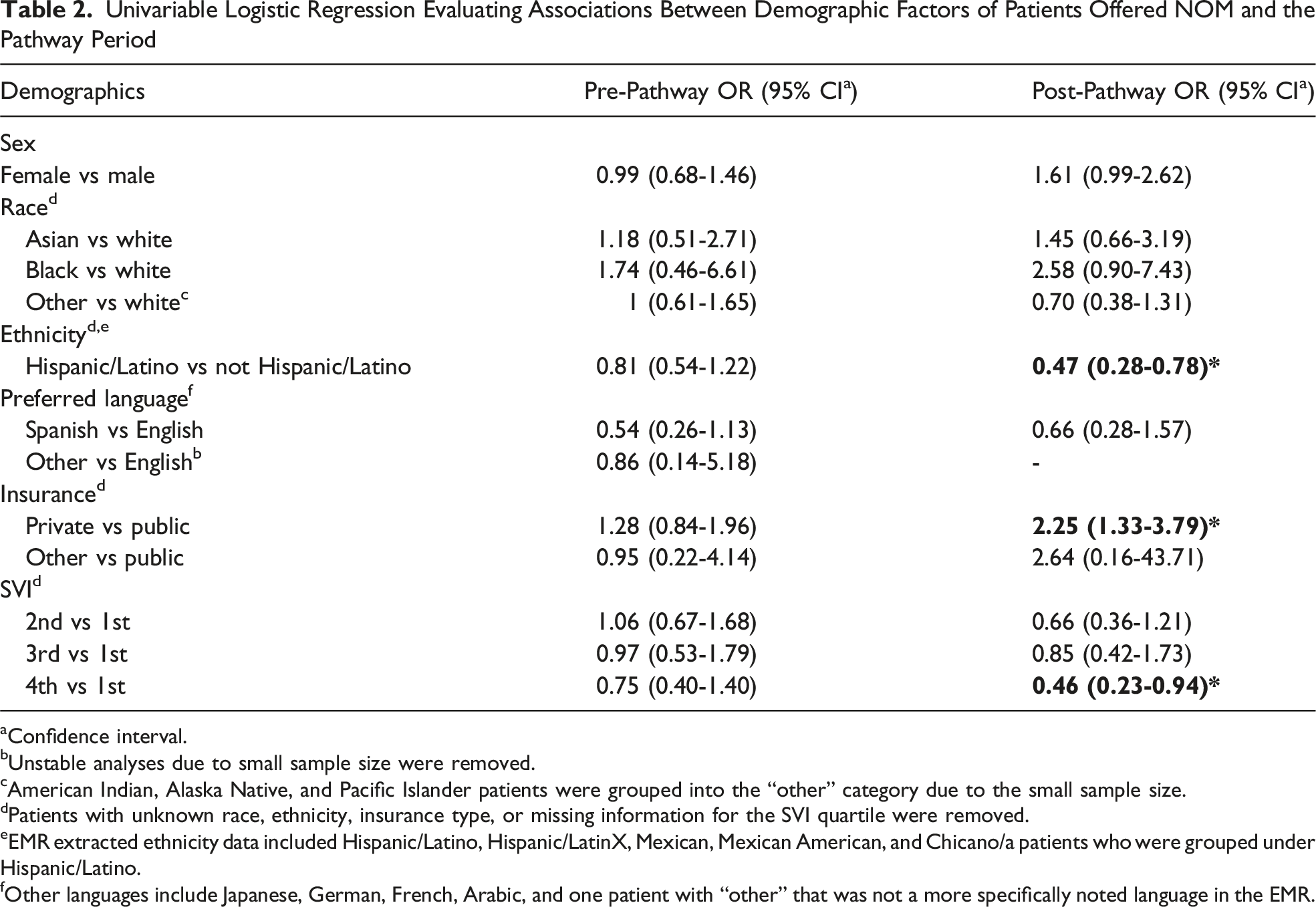
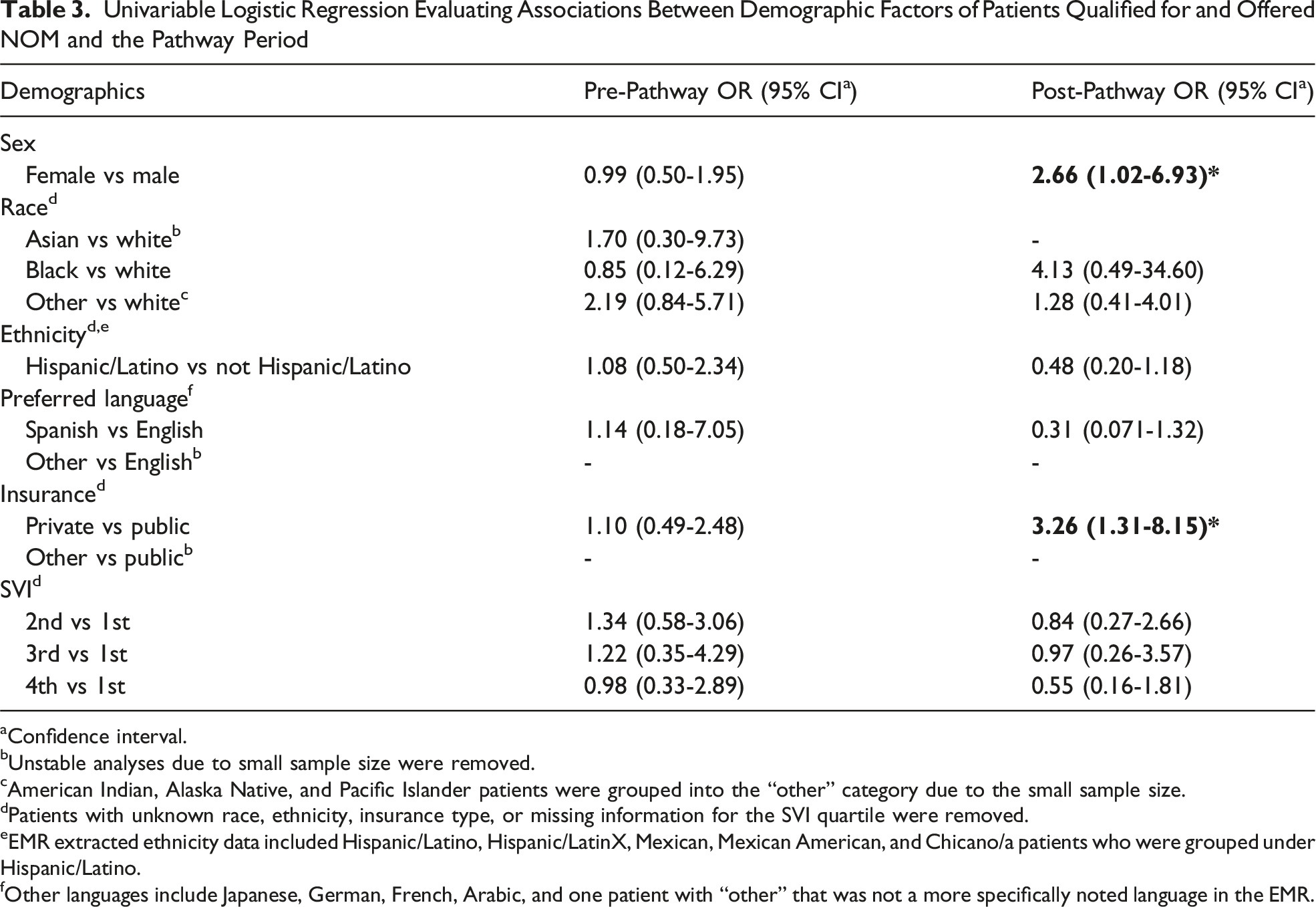
There were 730 unique patient encounters for appendicitis during the study period. Qualified patients had significantly increased odds of being offered NOM in the post-pathway period than in the pre-pathway period (OR = 2.21, 95th CI 1.28-3.82). In the post-pathway period, Hispanic/Latino patients (OR = 0.47, 95th CI 0.28-0.78) and patients in the 4th social vulnerability index quartile (OR = 0.46, 95th CI 0.23-0.94) had decreased odds of being offered NOM. Patients with private insurance (OR = 2.25, 95th CI 1.33-3.79) had increased odds of being offered NOM. When restricted to patients who qualified for NOM, female patients (OR = 2.66, 95th CI 1.02-6.93) and patients with private insurance (OR = 3.26 95th CI 1.31-8.15) were more likely to be offered NOM.
Conclusion
A clinical pathway for appendicitis increased the odds that qualified patients were offered NOM. However, differences in who was offered NOM based on demographic features were seen. More research on the effect of clinical pathways and factors impacting differential care is needed.
Key Takeaways
• A clinical pathway for the non-operative management of appendicitis may improve selection of the appropriate patients for a more conservative strategy. • Differences in care among patients can occur, even with clinical pathways, and re-evaluation is needed to understand how pathways can be revised to provide more equitable care.
Introduction
Pediatric appendicitis is a common reason for presentation to the emergency department, with 250 000 cases reported yearly. Most of these cases occur between 10 and 19 years of age. 1 Until recently, appendectomy has been the curative gold standard for the management of acute appendicitis. Although less invasive techniques have been developed, abdominal surgery with general anesthesia carries a risk for complications as well as disability during the post-operative recovery period. More recent literature supports the use of non-operative management (NOM) with antibiotics for a select subset of patients with uncomplicated appendicitis. For these patients, shared decision-making with families to pursue NOM has been shown to reduce morbidity and cost associated with acute appendicitis. 2
Clinical pathways are evidence-based road maps for care developed by institutions to help standardize treatment plans. Pathways have been shown to increase adherence to evidence-based guidelines, reduce variations in care, improve clinical outcomes, and decrease costs.3-7 However, evaluating for differences in the demographics of patients who receive care remains important, as clinical pathways do not always eliminate disparities.
Disparities in appendicitis care based on demographic factors such as race, ethnicity, and preferred language have been found, including in time to diagnosis, length of stay, and complications.8-13 In a study by Benedict et al, a clinical pathway for appendicitis was able to reduce demographic related disparities in the post-operative management of acute appendicitis but did not address the pre-operative decision on whether to pursue operative management. 14
A clinical pathway for appendicitis was published at our institution in 2018 that outlined criteria for which patients could safely be offered NOM as well as recommendations for appropriate antibiotics and analgesics. Our primary objective was to evaluate the effects of the pathway on the treatment type and to assess for differences in the demographics of patients who were offered NOM before and after implementation.
Methods
This was a single center retrospective study of patients under the age of 18 years who were evaluated and treated for appendicitis. Although the study was completed at a single institution, data were collected for patients across two campuses where the surgical and pediatric teams are the same. Our institution’s Office of Human Research Protection Program determined that the study met criteria for exemption from IRB review (IRB #22-001385).
Data Extraction
Electronic medical record (EMR) data were retrospectively collected for patients with appendicitis between November 2012 and July 2022. The ICD-9 and ICD-10 diagnosis codes for appendicitis and CPT procedural codes for appendectomy were used to identify possible encounters. The data included demographics (sex, age, race, ethnicity, preferred language, insurance type, and social vulnerability index (SVI)) and clinical information (labs and imaging). SVI was developed by the Centers for Disease Control and Prevention and the Agency for Toxic Substances and Disease Registry to identify and quantify communities experiencing social vulnerability. 15 The index is based on 16 US census variables grouped into 4 themes including socioeconomic status, household characteristics, racial and ethnic minority status, and housing type and transportation.
Chart Review
Patient charts were reviewed to determine whether they met inclusion or exclusion criteria. Inclusion criteria were age under 18 years, evaluation by the pediatric general surgery team, and diagnosis of appendicitis during an emergency department visit or admission at 1 of our 2 campuses. Exclusion criteria were past history of appendicitis, complicated appendicitis requiring interventional radiology procedures, intensive care unit admission, and if the patient had missing data due to being transferred from an outside hospital.
Patients were designated as “qualified” for NOM if they met the objective criteria from the institutional appendicitis clinical pathway. Objective criteria included age 5 years or older, white blood cell count less than 18 000 per microliter, appendix size 1.1 cm or less, absence of fecalith, and no fluid collection or abscess. In the pathway, there were other more subjective factors that affected the decision for NOM. These factors were history or exam findings that could be interpreted differently by providers (chronic abdominal pain, significant preexisting medical condition, peritonitis) or potentially misremembered (length of time of abdominal pain). They were not included in the analyses as they were less reliably documented.
The same objective criteria were used in the pre- and post-pathway periods to designate patients as qualified. Initial surgical team documentation was used to determine whether patients were offered NOM. Language that specifically referred to NOM or treatment with intravenous antibiotics only were classified as an offer for NOM; less specific language, such as “alternatives” was not.
Statistical Analysis
Chi-squared tests were used to determine whether there were significant differences in sociodemographic characteristics between patients treated during the pre- and post-pathway periods. Univariable logistic regression models were used to assess whether there were significant associations between individual sociodemographic factors and being offered NOM, with separate models for the pre- and post-pathway periods. Next, additional univariable logistic regressions models were run to assess for pre- and post-pathway associations between the individual sociodemographic factors and being offered NOM, with these models restricted to only patients who qualified for NOM based on the clinical criteria described above. Within the subsample of patients who qualified for NOM, a separate logistic regression model was run to examine if qualified patients were more likely to be offered NOM in the post- compared to the pre-pathway period. Lastly, to assess whether the pathway period modified the association between sociodemographic factors and being offered NOM, we also fit separate logistic regression models with an interaction term between each individual factor and an indicator for pre- vs post-pathway. All statistical analyses were completed in SAS 9.4 (SAS Institute, Cary NC).
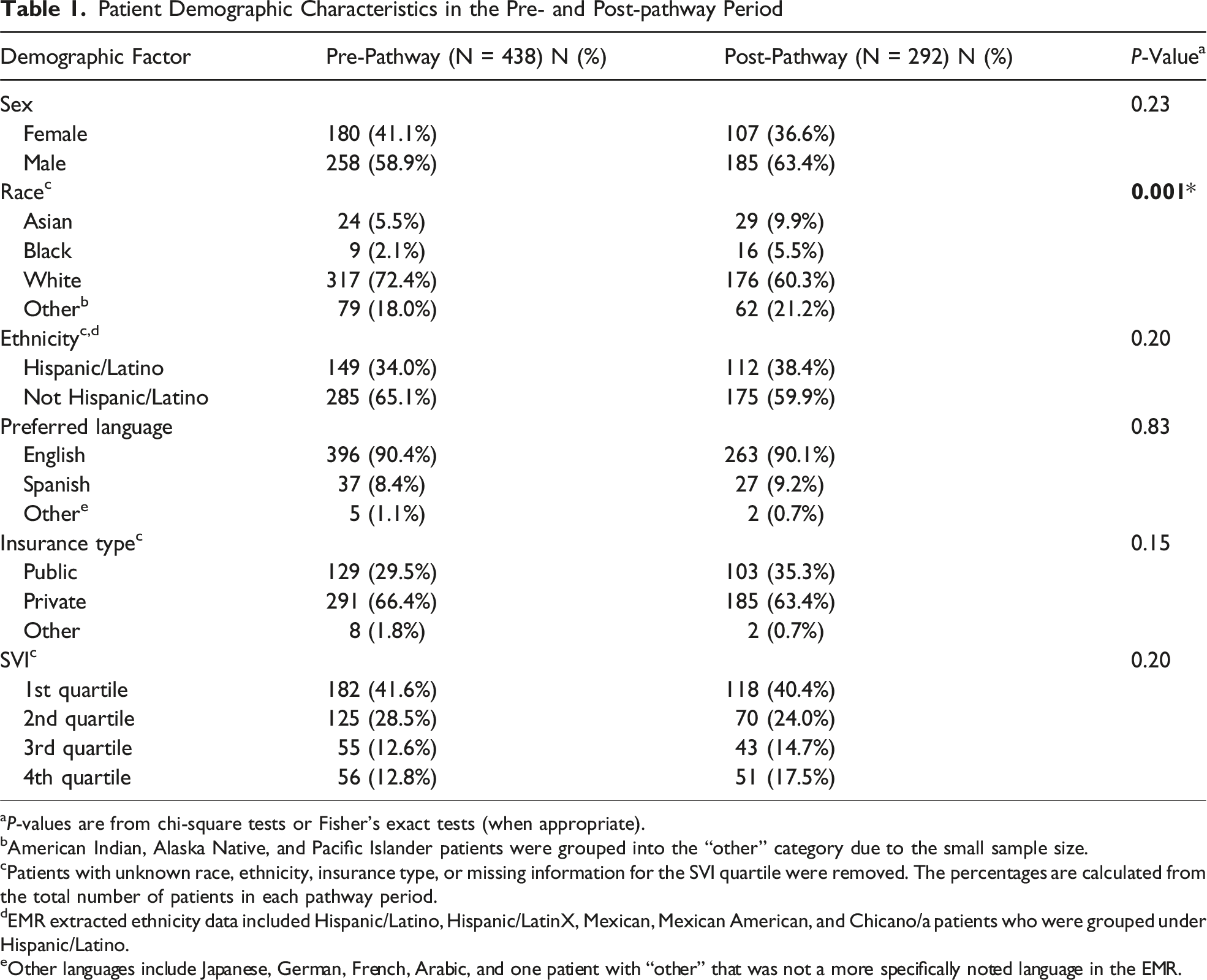
Results
The EMR data query resulted in 935 charts; after applying exclusion criteria and eliminating duplicate charts, 730 charts remained (Figure 1A). Of those, 60% (n = 438) of encounters occurred during the pre-pathway period and 40% (n = 292) during the post-pathway period. Demographic factors (sex, ethnicity, preferred language, insurance type, and SVI) were similar across the pre- and post-pathway groups (P > .05) except for patient race in the post-pathway period (P = 0.001) (Table 1). In the pre-pathway period, 42% (n = 186) of patients were offered NOM and in the post-pathway period, 40% (n = 116) were offered NOM. When pre-pathway patients were evaluated based on the pathway’s objective criteria, 32% (n = 138) qualified for NOM and 57% (n = 79) of those patients were offered NOM. Post-pathway, 38% (n = 111) of patients met the objective criteria for NOM and 75% (n = 83) of those patients were offered NOM (Figure 1B). Overall, qualified patients were more likely to be offered NOM in the post-pathway than in the pre-pathway period (OR = 2.21, 95th CI 1.28-3.82). Study design: A. Included and excluded appendicitis patient encounters. B. Comparison of pre- and post-pathway patients who were offered NOM, qualified for NOM, and the overlap of patients who were offered and qualified for NOM. Patient Demographic Characteristics in the Pre- and Post-pathway Period aP-values are from chi-square tests or Fisher’s exact tests (when appropriate). bAmerican Indian, Alaska Native, and Pacific Islander patients were grouped into the “other” category due to the small sample size. cPatients with unknown race, ethnicity, insurance type, or missing information for the SVI quartile were removed. The percentages are calculated from the total number of patients in each pathway period. dEMR extracted ethnicity data included Hispanic/Latino, Hispanic/LatinX, Mexican, Mexican American, and Chicano/a patients who were grouped under Hispanic/Latino. eOther languages include Japanese, German, French, Arabic, and one patient with “other” that was not a more specifically noted language in the EMR.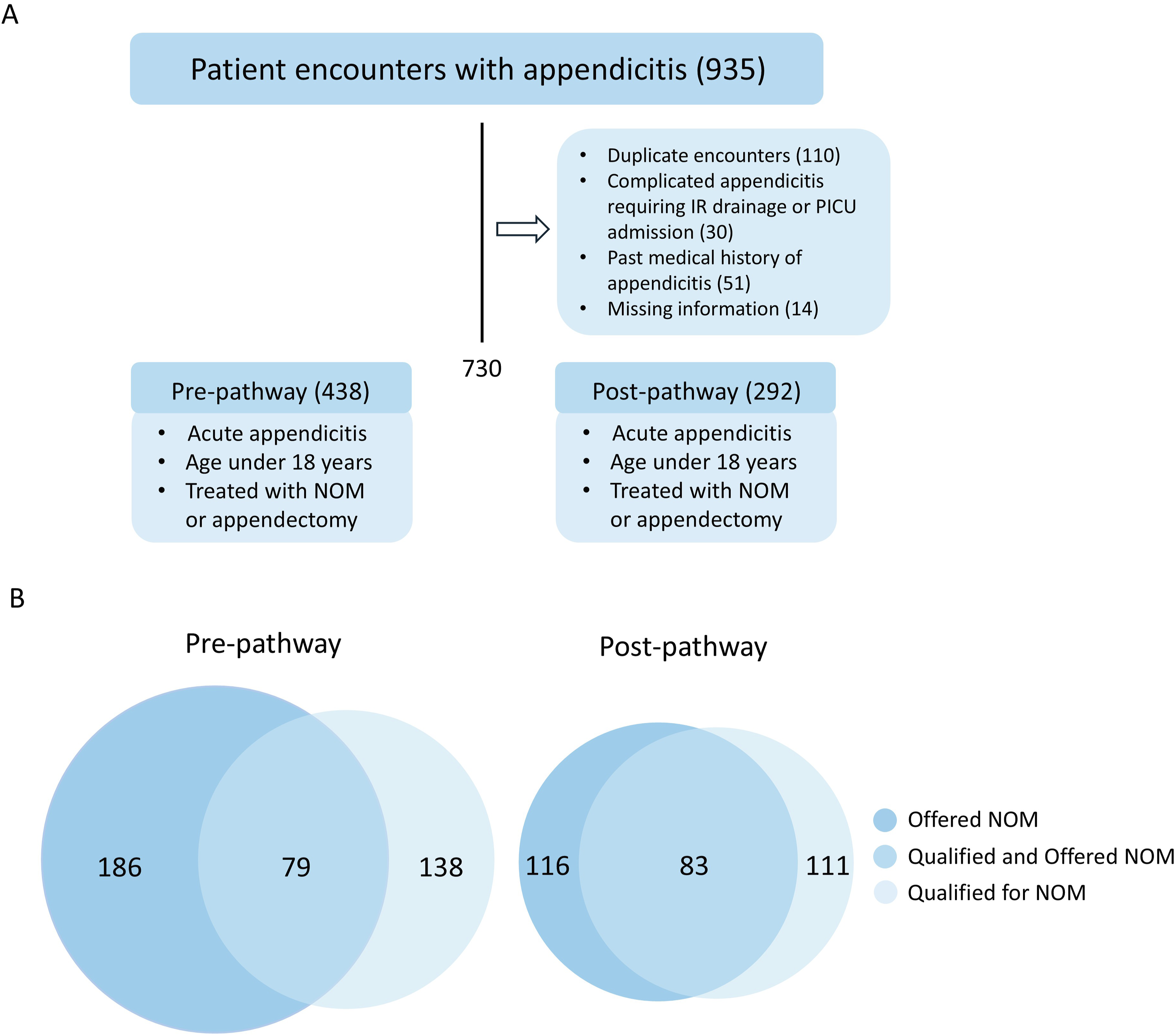
Sociodemographic Factors and Being Offered NOM
Univariable Logistic Regression Evaluating Associations Between Demographic Factors of Patients Offered NOM and the Pathway Period
aConfidence interval.
bUnstable analyses due to small sample size were removed.
cAmerican Indian, Alaska Native, and Pacific Islander patients were grouped into the “other” category due to the small sample size.
dPatients with unknown race, ethnicity, insurance type, or missing information for the SVI quartile were removed.
eEMR extracted ethnicity data included Hispanic/Latino, Hispanic/LatinX, Mexican, Mexican American, and Chicano/a patients who were grouped under Hispanic/Latino.
fOther languages include Japanese, German, French, Arabic, and one patient with “other” that was not a more specifically noted language in the EMR.
Sociodemographic Factors and Being Offered NOM Among Patients Qualified for NOM
Univariable Logistic Regression Evaluating Associations Between Demographic Factors of Patients Qualified for and Offered NOM and the Pathway Period
aConfidence interval.
bUnstable analyses due to small sample size were removed.
cAmerican Indian, Alaska Native, and Pacific Islander patients were grouped into the “other” category due to the small sample size.
dPatients with unknown race, ethnicity, insurance type, or missing information for the SVI quartile were removed.
eEMR extracted ethnicity data included Hispanic/Latino, Hispanic/LatinX, Mexican, Mexican American, and Chicano/a patients who were grouped under Hispanic/Latino.
fOther languages include Japanese, German, French, Arabic, and one patient with “other” that was not a more specifically noted language in the EMR.
Effect Modification of the Pathway Period
We did not find statistical significance of any interaction terms between sociodemographic factors and the indicator for pathway period (all P > 0.05), suggesting that the pathway did not significantly modify the effect of any demographic characteristic on being offered NOM.
Discussion
Previous studies have assessed whether NOM is an effective treatment for pediatric appendicitis. Various meta-analyses on NOM indicate that short- and long-term success rates range between 68%-90.5%.16-19 Factors that have been associated with NOM failure include presence of an appendicolith, increased and longer duration of pain, rebound tenderness, muscle guarding, appendiceal diameter >9 mm, and intraluminal appendiceal fluid.16,20-22 NOM has been associated with fewer disability days, improved quality of life measures, faster return to activity, and fewer missed days of work for parents,23,24 but also possibly increased length of stay, increased rates of ED visits or hospitalizations, and higher rates of complicated appendicitis if there is recurrence, though studies have had conflicting results.19,25 For some patients and families, concern for recurrence or future complication may be a deciding factor, while for others, avoiding general anesthesia, surgery, and possible surgical complications aligns with their goals. Ultimately, engaging in shared decision-making enables the clinical team to develop a plan that best reflects the patient’s and families’ values and preferences.
As the field of NOM continues to evolve, clinical pathways with a set of criteria can be developed to help guide the selection of patients. Prior studies evaluated pediatric appendicitis pathways designed to standardize diagnosis, surgical management, and antibiotic treatments for patients.26-28 Our study adds to this work by demonstrating that an appendicitis pathway may help facilitate choosing the appropriate low risk patients with uncomplicated appendicitis to treat with NOM. Patients who qualified were more likely to be offered NOM in the post-pathway period. However, when evaluated by demographic background, differences in being offered NOM became apparent after the pathway.
In the post-pathway period, there were differences in the odds of being offered NOM, regardless of whether patients qualified for NOM. When evaluating for associations between demographics and all patients who were offered NOM, Hispanic/Latino patients and patients in the lowest SVI quartile were less likely to be offered the treatment and patients with private insurance were more likely to be offered it. When the population was narrowed only to patients who qualified for NOM, there was no longer a difference in likelihood based on ethnicity or SVI quartile, but there remained an increase in odds of being offered NOM for patients with private insurance and a finding of increased odds for female patients. This suggests that if only patients who were qualified for NOM were offered it, that differences in being offered NOM may occur.
We also sought to determine whether the pathway period, pre- vs post-pathway, modified the effect of any sociodemographic factors on being offered NOM. There were no significant interactions, indicating that implementation of the pathway did not significantly impact whether sociodemographic factors were associated with being offered NOM.
Although standardization through clinical pathways can mitigate differences in care, 7 they are not always able to eliminate them. Other studies in the literature have shown persistent or new differences in care after pathway implementation, including in identifying patients with sepsis, 29 evaluations for non-accidental trauma, 30 and interventions and hospitalizations for acute gastroenteritis. 31 In our study, new differences in the odds of being offered NOM arose after the pathway. These differences are likely impacted by multiple factors including systemic, conscious, or unconscious biases in the healthcare system, though further research is required to better understand the contributors. A similar effect was seen in a national quality improvement project for the evaluation of febrile infants. 32 During the intervention period, a lower proportion of non-Hispanic Black infants and Hispanic/Latino infants had documented shared decision-making for obtaining cerebral spinal fluid, and a lower proportion of Hispanic/Latino infants had appropriate inflammatory markers sent and appropriate post-ED visit follow-up. Our study adds to these by demonstrating that clinical pathways do not ensure equity. These tools require reassessments to detect and understand disparities and revisions to reduce differential treatment.
There were several limitations due to the retrospective nature of the study. We relied on the EMR and documentation to determine whether patients qualified for NOM based on objective criteria and had limited ability to gauge whether patients qualified based on more subjective criteria, as they were not always clearly delineated in the notes. Additionally, we relied on the notes to determine if there was an offer for NOM. If providers did not document an offer, then it was unknown if NOM was not discussed and therefore not documented, or if NOM was discussed, but for conciseness and efficiency, only the discussion regarding operative management was documented since it was the family’s final choice. Though November 2018 was used to define the pre- and post-pathway period, there was likely a gradual change in use of the criteria for NOM, which could impact how patients are grouped into the pre- or post-pathway periods. This gradual change is also why the same objective criteria for NOM were applied to the pre- and post-pathway periods. Though the pre-pathway period did not have explicit criteria for NOM, the pediatric surgical team was offering NOM based informally on some of the same objective criteria. Additionally, the study was completed at two campuses and there may have been some variable practices due to local differences. However, the same surgical and pediatric providers worked at both locations and are from a single larger institution, which likely mitigated those differences. Lastly, the study was limited by relatively small sample sizes in some of the groups, causing instability in the effect estimates for the association between sociodemographic factors and all patients who were offered NOM, and the estimates for the subpopulation of qualified patients.
Future directions for research include a broader evaluation of appendicitis pathways for differences in patient and family preferences for the management of appendicitis. Both quantitative research to evaluate rates of family choice of NOM and qualitative research on family perspectives can help us understand the factors families consider when making such an important choice as surgery for their child. These studies would also help us understand the specific barriers to care that families experience, and once these have been identified, tools such as clinical pathways can be amended to provide more equitable care to patients.
Conclusion
A clinical pathway can increase the odds of a patient who qualifies for NOM being offered the treatment. However, when evaluated by sociodemographic backgrounds, there can be differences in patients who are offered NOM. After implementation of the pathway, when all patients were evaluated, Hispanic/Latino patients and patients in 4th SVI quartile were less likely and patients with private insurance were more likely to be offered NOM. When evaluated by whether the patient qualified for NOM, female patients and patients with private insurance were more likely to be offered NOM. Further research is needed to understand the effects of clinical pathways on equitable care.
Footnotes
Acknowledgments
Research reported in this publication was supported by the National Center for Advancing Translational Science (NCATS) of the National Institutes of Health under the UCLA Clinical and Translational Science Institute grant number UL1TR001881.
ORCID iDs
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data is available to share upon request.
