Abstract
Pediatric thoracic trauma is a major cause of pediatric morbidity and mortality. Pediatric patients have unique anatomy and physiology that account for differences in injury patterns when compared to adult patients. These differences warrant special consideration in injury recognition and intraoperative management. The initial management of pediatric thoracic trauma should follow the principles of the primary survey in that life-threatening injuries should be ruled out. Hemodynamically unstable patients with serious thoracic injury warrant thoracotomy for expeditious management of life-threatening conditions. However, the management of hemodynamically stable patients with thoracic injury has evolved with the advent of minimally invasive surgery and the well-documented benefits of video-assisted thoracoscopic surgery (VATS) compared to traditional thoracotomy. Multiple studies have shown that VATS can be performed safely and effectively for traumatic injuries in children in both the acute and delayed setting. In this article, we provide an overview of pediatric thoracic trauma and the use of VATS in the management of these conditions.
Introduction
Pediatric thoracic trauma is relatively uncommon but is considered a significant contributor to morbidity and mortality in children. 1 It accounts for an estimated 4-8% of all pediatric trauma cases and is the second leading cause of death in this population, surpassed only by brain injuries. 1 According to a national pediatric database study, thoracic injuries are associated with the highest fatality rate among all body regions, with mortality approaching 10%. 2 Similarly, Cooper et al demonstrated that thoracic trauma increases the risk of death by at least 20%, compared to abdominal injuries. 1
Blunt trauma is responsible for approximately 85% of pediatric thoracic injuries, with motor vehicle collisions being the most common mechanism. 1 However, penetrating thoracic trauma, although less frequent, carries a higher mortality risk. 3 Additionally, 50-80% of children presenting with thoracic trauma have injuries to other organ systems, further complicating their management. 3
The management of pediatric thoracic trauma requires a multidisciplinary approach involving prompt resuscitation, imaging, and surgical intervention when necessary. While non-operative management is sufficient in many cases, complex injuries may require operative solutions. Historically, thoracotomy was the standard approach for operative management. However, with advances in minimally invasive techniques, video-assisted thoracic surgery (VATS) is now a reasonable alternative in hemodynamically stable pediatric patients with thoracic injuries. 4 VATS offers reduced surgical morbidity, shorter hospital stays and improved postoperative recovery compared to open thoracotomy.4-6 This review aims to examine the role of VATS in pediatric thoracic trauma, highlighting its indications, techniques, and outcomes.
Demographics
The presentation of pediatric thoracic injuries varies significantly by age and mechanism. Infants and toddlers (ages 0-4) are most commonly injured due to non-accidental trauma or motor vehicle collisions.7,8 Children aged 5-9 are more frequently involved in pedestrian-struck incidents. 9 Among older children or adolescents (ages 10-17), thoracic trauma is often associated with high-impact recreational activities such as skateboarding, bicycling, and contact sports, as well as intentional injuries related to self-harm or interpersonal violence, including stab wounds and gunshot wounds. 7 Notably, penetrating thoracic trauma disproportionately affects males, and an age of less than 12 years has been identified as an independent risk factor for mortality in this population. 7 Furthermore, firearm violence and/or associated deaths continues to increase across numerous populations, especially in the post COVID-era.10-13
Anatomic and Physiological Considerations
The unique anatomic and physiological characteristics of pediatric patients influence both the patterns and severity of thoracic injuries. The pediatric chest wall is highly compliant due to greater bone elasticity, reduced muscle mass, and incomplete ossification of the ribs. As a result, rib fractures are less common in children than in adults. 14 However, the increased chest wall compliance and diminished musculature allow for greater transmission of force to the underlying thoracic structures, often resulting in significant intrathoracic injury without visible external trauma. 4
Pediatric patients also have increased mediastinal mobility due to their relatively loose connective tissue. 15 This predisposes them to complications such as tension pneumothorax and contralateral vena cava compression following diaphragmatic rupture. Additionally, the smaller caliber and increased collapsibility of the pediatric airway make children more susceptible to airway compromise following trauma.15,16 These anatomic distinctions necessitate a high index of suspicion for severe thoracic injury, even in cases where external signs of trauma appear minimal.
Injury Patterns in Children
Pulmonary Injuries
Pulmonary contusion is the most common intrathoracic injury in pediatric trauma, occurring in approximately 50% of children with thoracic trauma. 17 Unlike in adults, these injuries often present without associated rib fractures, reflecting the unique compliance of the pediatric chest wall. Pneumothorax and hemothorax are also common, with an incidence of nearly 30%. 4 Management typically involves tube thoracostomy for decompression, lung re-expansion, and quantification of blood loss to assess the need for surgical intervention. 15
Tension pneumothorax occurs more often in children than in adults due to increased lung compliance and mediastinal mobility. 14 Similarly, the highly mobile mediastinum places children at high risk for rapid hemodynamic deterioration due to shifting of abdominal contents into the thoracic cavity which in turn result in vena cava compression and impaired cardiac output. 14
Tracheobronchial Injuries
The unique tracheobronchial anatomy of children further amplifies the risks associated with thoracic trauma. The shorter and more compressible trachea increases the likelihood of airway compromise following external compression. 18 Major thoracic trauma can also lead to tracheobronchial injuries, including tracheobronchial rupture. A proposed mechanism of injury is anteroposterior compression of the chest wall and sternum against the spine, which displaces the lungs laterally and generates shear forces at the carina. 19
Most tracheobronchial injuries occur within 2.5 cm of the carina, with the right mainstem bronchus being more commonly affected than the left. 20 This predisposition is attributed to the shorter length of the right mainstem, a relatively unprotected position compared to the left mainstem, and a heavier right lung which it is positioned near. 21
Cardiac Injuries
Myocardial contusions are the most frequent cardiac injury in pediatric trauma patients, accounting for 90% of cardiac injuries in children and often occurring in conjunction with pulmonary contusions due to the compliant chest wall. 22 As such, there should be a higher index of suspicion for cardiac contusion in pediatric patients with pulmonary contusion. 22 Cardiac contusions are managed conservatively with pain control, telemetry monitoring and serial troponin I laboratory testing. 23
Penetrating cardiac injuries, although less common, carry a significantly higher mortality rate with firearms being the most frequent cause. These injuries are often associated with hemothorax and pneumothorax. 22 The right ventricle is most commonly involved due to its anterior position in the chest. 24 In a stable patient, a chest x-ray, Focused Assessment with Sonography for Trauma (FAST) exam or echocardiogram and contrast enhanced chest computed tomography (CT) scan can be performed to identify trajectory and diagnose cardiac injury. Once an injury is confirmed, the patient should proceed to the operating room. Unstable patients with high suspicion for cardiac injury should proceed directly to the operating room for sternotomy. 24
Diaphragm Injuries
Traumatic diaphragmatic rupture is a rare diagnosis with an estimated incidence of 0.07% of all pediatric trauma cases. 25 This injury is highly associated with blunt abdominal trauma and demonstrates a male predilection, with a reported 2:1 male to female ratio in children, compared to a 4:1 ratio in adults.25,26
Diaphragmatic ruptures most commonly occur at the junction of the muscular and tendonous portions of the diaphragm, with the left side affected more frequently due to the protective effect of the liver on the right. 27 In pediatric patients, these injuries are often diagnosed late, as concerns over radiation exposure and the increasing trend toward non-operative management of blunt abdominal trauma can delay definitive diagnosis. 25
Role of VATS in Trauma
VATS offers a means to address traumatic thoracic injuries that require more than a tube thoracostomy and provides the benefit of decreased morbidity compared to thoracotomy. In a hemodynamically stable patient, VATS can be performed safely for most traumatic thoracic injuries, including hemothorax and diaphragm injuries, while reducing the complications associated with open thoracotomy.4-6
Indications for VATS in the Acute Setting
Retained Hemothorax
For patients with retained hemothorax, VATS allows for direct visualization of the pleural cavity, evacuation of clotted blood, and identification of active bleeding when present. This approach is most beneficial in patients with approximately 500cc of retained blood or when 1/3 of the pleural cavity is occupied with blood. 28 The Eastern Association for the Surgery of Trauma (EAST) guidelines recommend early VATS over a second chest tube for persistent retained hemothorax, 28 but this recommendation is specifically made for adult patients. However, several single-center studies demonstrate that VATS can be performed safely for retained hemathoraces in pediatric patients.5,6
The optimal timing for VATS for retained hemothorax in pediatric patients is still to be determined. However, most adult studies suggests that intervention within 72 hours of trauma reduces conversion to thoracotomy, minimizes the need for additional procedures, decreases the risk of empyema and shortens hospital length of stay (LOS).29-31 Lin et al found that the best outcomes are achieved when VATS is performed within 3 days of injury, with a cutoff of no more than 6 days if early intervention is not feasible. 32 Morales et al corroborate these findings, showing increased conversion rates to thoracotomy and higher reintervention rates when VATS is delayed beyond 6 days. 33
However, recent studies suggest that timing alone may not be the primary determinant of VATS success. Instead, factors such as inability to tolerate single-lung ventilation, concomitant diaphragm injury, lack of periprocedural antibiotics, and hemothorax volume >900 mL have been identified as stronger predictors of conversion to thoracotomy.34,35 While early VATS is generally preferred, delayed intervention may still be appropriate in cases of severe multisystem trauma where immediate surgical management is not feasible. 34
Persistent Air Leak
VATS is also indicated for persistent air leaks following chest tube placement. A persistent air leak is defined as continued air leak for 5-7 days after initial chest tube placement. 36 Prolonged air leaks can lead to prolonged hospitalizations and increased complications. Multiple studies in adults demonstrate that early VATS (performed ≤5 days after injury) for this injury pattern is associated with lower rates of conversion to thoracotomy, reduced number of chest tube days and reduced LOS.37,38
During VATS, underwater submersion is used to identify the source of the air leak, allowing localization and targeted repair via stapling of the affected lung segment. 39 This approach provides a definitive solution while avoiding the morbidity of prolonged chest tube drainage.
Diaphragm Injuries
Diaphragm injuries are typically addressed transabdominally in the acute setting if the patient is hemodynamically unstable or has concomitant intra-abdominal injuries. 40 However, transthoracic approaches can be taken when intra-abdominal injuries have been excluded. The use of VATS in this setting was first reported by Ochsner et al 41 Compared to open approaches, VATS provides enhanced visualization of the diaphragm, particularly on the right side, and allowing for simultaneous management of associated thoracic injuries. 40 VATS has been shown to have excellent sensitivity (98-100%) in diagnosing a diaphragm injury and is particularly helpful in evaluating the posterior recess.27,42 Examination of the entire hemidiaphragm is best achieved with the camera in the fourth or fifth interspace. 43 The presence of 2 or more of the following injuries is a strong predictor of diaphragmatic injury: abnormal chest x-ray, associated intra-abdominal injury, high velocity mechanism, entrance wound inferior to the nipple line, and right sided penetrating trauma. 40
Indications for VATS in the Delayed Setting
Post-Traumatic Empyema
Post-traumatic empyema typically arises from an infected retained hemothorax. Initial management consists of antibiotic therapy and pulmonary toilet. 44 However, if the infection persists or results in fibro-purulent organization, decortication is required. 45 VATS can be used to perform decortication, allowing for the removal of the fibrinous adhesions and the visceral peel to promote lung re-expansion.
Despite the advantages of VATS, conversion to thoracotomy may be required if adhesions are extensive or if minimally invasive techniques fail to achieve adequate lung re-expansion. 46 Early intervention is important as prolonged infection, and fibrinous organization can make the procedure more technically challenging and increase the likelihood of requiring open surgery. 44
Delayed Recognition or Deferred Repair of Diaphragm Injury
In cases where a diaphragmatic injury is missed during the acute phase or when surgical repair is deferred, VATS can be an advantageous alternative to a transabdominal approach. Chronically herniated abdominal contents can form adhesions to the lung parenchyma and mediastinal structures, making reduction more complex. 47 The thoracic approach offers the advantage of direct visualization and release of intrathoracic adhesions, facilitating the safe reduction of herniated viscera and allowing for primary diaphragmatic repair. 48 By addressing these adhesions from within the thoracic cavity, VATS can potentially minimize the risk of abdominal organ injury during hernia reduction. Alternatively, a hybrid repair using VATS can be performed alongside an abdominal operation. 49
When to Choose VATS vs Thoracotomy
The decision between VATS and thoracotomy in pediatric trauma depends on multiple factors including the mechanism of injury, patient stability, injury pattern and surgeon experience. While VATS offers significant advantages in select cases, thoracotomy remains an important tool in the armamentarium of surgeons treating pediatric patients with life-threatening thoracic injuries.
A scoring system developed by Kazempoor et al aimed to predict the need for early VATS in pediatric trauma patients, identifying cardiac injury as the strongest independent predictor. 50 However, given the need for direct cardiac exposure, these injuries often necessitate sternotomy rather than VATS. Other significant predictors favoring early VATS included hypotension, penetrating trauma, and injuries involving the lung or diaphragm, reinforcing the role of VATS in select cases where open surgery is not immediately required. 50
VATS has gained widespread acceptance due to its numerous advantages over thoracotomy, including reduced postoperative pain, lower complication rates, shorter hospital stays, and faster recovery.51,52 VATS also facilitates enhanced visualization of thoracic structures, enabling precise diagnostic and therapeutic interventions. These benefits make VATS particularly attractive in stable patients requiring evaluation or management of hemothorax, pneumothorax, retained foreign bodies, or diaphragmatic injuries. However, patient selection remains key, as unstable or critically injured children may not tolerate the procedural constraints of VATS such as single-lung ventilation, necessitating immediate conversion or upfront thoracotomy.
Indications for Upfront Thoracotomy or Conversion
Certain scenarios necessitate upfront thoracotomy or conversion from VATS due to clinical instability, technical limitations, or injury complexity. Thoracotomy is indicated in cases of hemodynamic instability refractory to blood product resuscitation, uncontrolled hemorrhage, inability to tolerate single-lung ventilation, and inability to progress in the operation via VATS. Re-operative surgery and delayed presentations complicated by empyema or dense pleural adhesions are a relative contraindication to VATS.37,53,54
Drainage thresholds for thoracotomy include an initial chest tube output exceeding 20 cubic centimeters per kilogram (cc/kg) (or 1000-1500 cc in older adolescents) or ongoing bleeding at a rate of 2-3 cc/kg/h over 4 hours, suggesting active hemorrhage requiring operative intervention. 15 However, the most important determinants of operative intervention are hemodynamics and response to blood product resuscitation. A hypotensive patient refractory to blood product resuscitation with thoracic trauma should undergo immediate thoracotomy, regardless of chest tube output. 15
Specific thoracic injuries also favor thoracotomy due to their complexity or anatomical considerations. Tracheobronchial injuries often require posterolateral thoracotomy for direct visualization and repair, as these injuries typically occur within 2.5 cm of the carina and are easier to access via an open approach. 20 Tracheobronchial injuries should be suspected when a pneumothorax persists despite proper chest tube placement or the ongoing air leak is large. 20 While some recent literature suggests the potential for VATS in managing tracheobronchial injuries, its use remains limited to highly specialized centers. 55
Esophageal injuries identified early are typically managed with thoracotomy depending on their location, with left thoracotomy preferred for distal injuries and right thoracotomy for mid-esophageal injuries. 56 Injuries identified later in the course that present with extensive mediastinal contamination may necessitate irrigation and pulmonary decortication, which may be more effectively performed through an open approach. 56
Hilar and pulmonary injuries present another set of challenges. Persistent bleeding from the lung parenchyma or hilar structures requires precise control, often achieved through parenchymal-sparing techniques or direct hilar repair via thoracotomy. 57 Although blunt aortic injuries were historically managed with thoracotomy, endovascular approaches have become increasingly common, offering shorter hospital stays and comparable mortality rates. 58 Nevertheless, the long-term durability of endografts in growing children remains a concern and requires careful patient selection and follow-up. 58
Thoracotomy is also indicated in the setting of pediatric thoracic trauma with refractory shock or circulatory arrest following trauma. According to the EAST guidelines, resuscitative thoracotomy is conditionally recommended for pediatric patients presenting pulseless with signs of life following penetrating thoracic or abdominopelvic injuries. 59 There is a conditional recommendation against resuscitative thoracotomy for pediatric patients presenting pulseless following penetrating thoracic or abdominopelvic injury without signs of life and a strong recommendation against resuscitative thoracotomy for pediatric patients presenting pulseless with blunt thoracic injury without signs of life.59,60
VATS Technique in Trauma
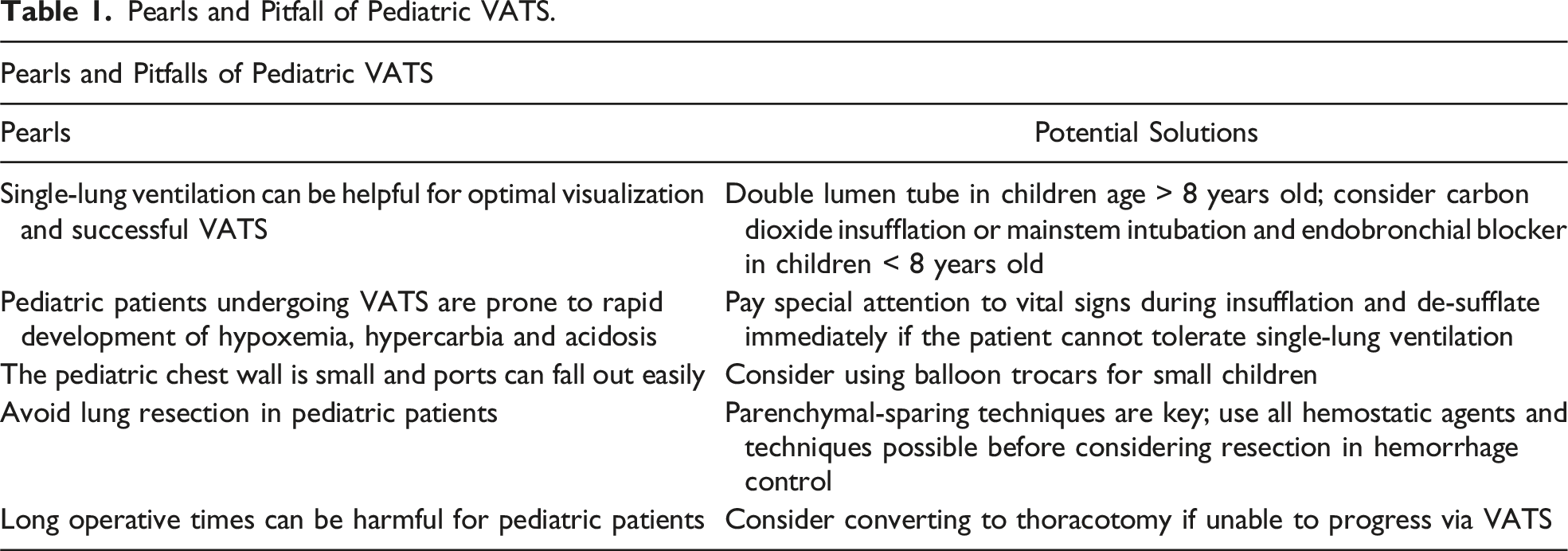
Pearls and Pitfall of Pediatric VATS.
Patient Preparation and Airway Management
Obtaining optimal visualization and working space via single-lung ventilation is required for successful thoracoscopic intervention. This is often achieved using a double lumen endotracheal tube (DLT) in adults and adolescents. However, the smallest available DLT (26fr) is unsuitable for children younger than 8 years old. 61 For infants and younger children, carbon dioxide (CO2) insufflation has been described as a means to improve visualization. 62 However, this technique must be used with caution, as it can lead to hemodynamic instability due to the increased mobility and compressibility of the pediatric mediastinum. 63 Hypercarbia and acidosis are more frequently observed in children under 10 years old, necessitating careful regulation of insufflation parameters. 64 A low flow rate (1 L/min) and minimal pressure (≤4 mmHg) are recommended. 65 If desaturation or hemodynamic instability occurs, immediate desufflation should be performed. Alternatively, single-lung ventilation can be achieved in small children using an endobronchial blocker with a balloon tip or a Fogarty catheter. 66 A bronchoscope is used to guide these devices into the desired main stem bronchus, where the balloon is inflated to occlude the lumen and facilitate lung collapse. 66
Patient Positioning and Port Placement
The patient is then positioned in the lateral decubitus position. The arms are extended for venous access, the bed is flexed at the hip for larger children and the body is secured using beanbags. The surgeon stands on the anterior side of the patient. 67 Optimal port position will vary by pathology. In most cases, the initial 5 mm trocar will be inserted in the posterior axillary line at the 5th or 6th intercostal space with additional ports placed under direct visualization.68,69 For most indications, the camera port should ideally be positioned as inferiorly as possible to provide a broad, panoramic view of the thoracic cavity. 66 Balloon trocars can be used to secure the trocars in children with small chest cavities. Curved tip ring forceps and suction devices help minimize instrument crowding and optimize maneuverability within the limited working space. 70
Thoracic Exploration
After single-lung ventilation is achieved, a 30-degree thoracoscope is introduced to provide a panoramic view of the thoracic cavity. 70 This angled scope allows for thorough examination around the lung lobes and hilar structures. Lung retraction may be performed using sponge sticks, and if further exposure is needed, the inferior pulmonary ligament can be released to improve mobilization.
Control of Active Hemorrhage
For ongoing hemorrhage, the first step is the application of direct pressure, this may occur with hemostatic gauze or sponge sticks. Suction can be used to improve visualization. If massive bleeding continues despite compression, then a conversion to thoracotomy is required. Pulmonary lacerations can be treated with electrocautery and topical sealants. 71 Bleeding from small vessels such as bronchial arteries can be controlled using vascular clips. 72 Bleeding that persists despite the previously mentioned techniques requires direct suture repair. Lung resection in the setting of trauma is uncommon and typically reserved for central injuries, extensive pulmonary lacerations, significant hemorrhage from a peripheral laceration or persistent hemorrhage despite attempts at parenchymal-sparing repair. 73 In a national, retrospective analysis of pediatric patients, wedge rection was associated with increased mortality with increasing mortality for patients requiring lobectomy and pneumonectomy. 74 Injuries to the pulmonary artery and vein are preferably controlled using polypropylene suture or endoscopic vascular staplers. 75 Alternatively, these vessels can be ligated using clips and an energy sealing device which can be easier to manipulate in small chest cavities. 76 While a ligated pulmonary arterial branch can sometimes be tolerated due to the dual blood supply to the lung (pulmonary and bronchial), a ligated pulmonary vein will always lead to vascular congestion and eventual pulmonary necrosis. A formal anatomic resection is therefore required in the setting of a ligated or sealed pulmonary vein. Bleeding due to penetrating pulmonary parenchymal injuries can be better visualized and controlled using a stapled tractotomy. 77
Management of Common Injuries Using VATS
Evacuation of Retained Hemothorax
The patient is positioned in the lateral decubitus position with single-lung ventilation. The thoracoscope is placed into the chest at the site of the chest tube with additional ports placed under direct visualization. Adhesions and blood clots are removed with ring forceps and a suction device. The chest is irrigated with warm saline and a chest tube is left in place in the chest cavity. 78
Repair of Diaphragm Injury
Diaphragm injuries occur more frequently on the left side, as the liver provides partial protection to the right hemidiaphragm. The typical approach in the acute setting is a diagnostic laparoscopy or laparotomy as this allows for inspection of herniated viscera and the ability to address concomitant abdominal injuries. 25 For stable patients where intra-abdominal injuries have been ruled out, VATS can be used in the acute setting to diagnose and address diaphragm injuries. Diaphragm injuries in the delayed setting are best managed via a thoracic approach due to the formation of intrathoracic adhesions. 47 Parelkar et al describe a technique for repair in children placing the initial camera port in the 3rd intercostal space at the mid-axillary line and 2 additional ports placed under direct visualization. High insufflation pressure and flow are used to assist with reduction of herniated contents and the diaphragm is repaired with nonabsorbable 2-0 suture. 43 Patch or mesh repair can be considered for defects > 3 cm that do not come together primarily. 79
Repair of Bronchial Injury
Bronchial injuries are rare injuries in children and are usually addressed via thoracotomy.80,81 This is performed at the 4th intercostal space with the patient in lateral decubitus position. Fiberoptic bronchoscopy of the injured bronchus is used to identify the area of injury. Lacerations can be closed with interrupted Prolene suture while complete transections can be brought together using an end-to-end anastomosis. A pedicled pleural or intercostal flap should then be used to buttress the repair. Guo et al describe a novel technique for addressing main bronchial injuries in children using VATS. 55 Incisions are placed between the fourth and sixth intercostal spaces in the left axillary line with the patient in the lateral decubitus position. The inferior pulmonary ligament is released, and the pulmonary artery and aorta are retracted laterally. The proximal end of the ruptured bronchus is identified using fiberoptic bronchoscopy and the 2 ends are brought together using continuous Prolene or PDS suture.
Outcomes of VATS vs Thoracotomy
The advantages of VATS over thoracotomy are well documented in the literature. However, data comparing these approaches in pediatric patients remain limited, and much of the evidence supporting VATS in children is extrapolated from adult studies.
Historically, thoracotomy required splitting the serratus anterior muscle, which led to a high incidence of winged scapula and chest wall deformities in children. 82 Even with the advent of muscle sparing techniques, thoracotomy has remained associated with poor cosmesis, prolonged wound site discomfort, and an increased need for perioperative blood transfusions when compared to VATS.83-85 Postoperatively, VATS is associated with decreased hospital LOS, reduced chest tube duration, and decreased reliance on epidural anesthesia. 86 These findings align with adult studies, which show that thoracotomy patients require more narcotic analgesia, experience longer recovery times, and face higher rates of pulmonary and infectious complications. Additionally, VATS patients tend to report greater satisfaction with postoperative cosmesis.52,87
Despite its advantages, there are some limitations to VATS. Prerequisites to VATS include optimal visualization with the thoracoscope via single-lung ventilation or the ability to tolerate carbon dioxide insufflation particularly in small infants in whom double lumen endotracheal tube intubation is not an option. Any evidence of respiratory or hemodynamic compromise would necessitate desufflation and conversion to thoracotomy. Furthermore, the technical demands of VATS in trauma require specialized expertise, and there is a steep learning curve for surgeons to develop proficiency in managing complex injuries thoracoscopically. 67
Conclusion
Pediatric thoracic trauma is a rare entity that carries a high risk of morbidity and mortality due to the unique anatomic and physiologic characteristics of this population, as well as the frequent presence of associated polytrauma. The role of VATS in managing these injuries continues to expand, with well-documented advantages such as reduced postoperative pain, fewer complications, faster recovery, and improved long-term cosmesis compared to traditional thoracotomy. While the principles of VATS remain the same, the unique anatomy and physiology of the pediatric population warrant special consideration at the time of operation. VATS can be used in both the acute and delayed setting provided that the patient remains hemodynamically stable. If visualization is inadequate or the patient becomes hemodynamically unstable, the provider should consider conversion to thoracotomy. Nevertheless, trauma surgeons should become adept in VATS as it has become more commonplace in the management of traumatic thoracic injuries in the pediatric population.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
