Abstract
Median arcuate ligament syndrome, or celiac artery compression syndrome (eponym: Dunbar syndrome), has historically been attributed to pathophysiologic vascular compression causing downstream ischemic symptoms of the organs supplied by the celiac trunk. However, the more we learn about the histology, clinical presentation, and treatment outcomes, health care providers are increasingly correlating the symptoms of MALS with the long-term, repetitive compression of the celiac ganglion rather than the celiac trunk. This article provides a comprehensive review of current MALS literature, emphasizing the multidisciplinary approach these patients require in all phases of their care. With most patients with MALS waiting an average of 10.5 months to 2.6 years, 9, 10 our need for better diagnostic protocols and clearer understanding of the pathophysiology of the disease is paramount. Further investigation into patient outcomes, associated conditions, and linked pathophysiology would help better characterize this disease with hopes of moving it from a diagnosis of exclusion to one of standard work-up with decreased time to treatment and symptom relief for patients.
Definition
Median arcuate ligament syndrome (MALS) is also known as celiac artery compression syndrome (CACS) or Dunbar syndrome. First described anatomically by Lipshutz in 1917 1 during cadaveric dissections, MALS has historically been thought to arise from external compression of the celiac artery and surrounding structures by the median arcuate ligament. This ligament is a fibrous band that forms the anterior wall of the aortic hiatus of the diaphragm at the T12/L1-2 spinal level, allowing the passage of the aorta, azygous, and hemiazygos veins, as well as the thoracic duct from the thoracic cavity to the abdominal cavity. Just inferior to this hiatus, the aorta gives rise to the celiac trunk and its downstream vessels, supplying blood to the esophagus, stomach, proximal duodenum, liver, gallbladder, pancreas, and spleen. Therefore, MALS is defined as the clinical signs and symptoms that manifest from this often prolonged and overlooked compression.
Harjola reported the first resolved case of postprandial epigastric pain and epigastric bruit following decompression of the celiac artery from a fibrosed celiac ganglion in 1963. 2 2 years later, in 1965, Dunbar et al 3 reported a case series of surgical treatment of MALS. Since this time, MALS has been a controversial diagnosis mostly due to the unclear consistent pathophysiological link between patient symptoms and objective findings.
Epidemiology
Despite its long-documented history, MALS continues to largely be a diagnosis of exclusion due to its non-specific symptoms being more attributable to other more common upper gastrointestinal and abdominal pathologies. Additionally, being cited as a rare disease indicates that it occurs in less than 1 every 200,000 people in the US following the Orphan Drug Act of 1983. Incidences of celiac artery narrowing from a handful of studies in the mid to late 1900s have ranged from 5 to 27 percent, though many of these studies did not distinguish between intrinsic and extrinsic compression.4-7
The European Society for Vascular Surgery (ESVS) Clinical Practice Guidelines for the Management of the Diseases of Mesenteric Arteries and Veins 8 cite MALS as the “most common cause of single-vessel abdominal arterial stenosis.” Still, in a 1972 study published by Levin and Baltaxe, 24% of asymptomatic adults were found to have severe narrowing of the proximal celiac axis with the compression occurring above the MAL of the diaphragm and celiac plexus. 4 With such a high incidence of celiac artery compression in an asymptomatic population, this sparked necessary hesitation among health care providers in diagnosing MALS/CACS, for which patients with MALS can go undiagnosed for extended periods of time.
Diab et al noted a mean duration of symptoms of 10.5 months (range: 3 months-7 years) prior to diagnosis in their single-site 10-year retrospective study. 9 Sultan and Hynes reported an even longer mean duration of symptoms prior to surgery at 2.6 years. 10
In terms of patient demographics, MALS appears to affect females more than males7,9,11,12 and can occur in any stage of life, though most articles will cite an average occurrence in the 4th to 6th decade of life.9,11,12 In a Nationwide Inpatient Sample of 33,951 patients diagnosed with MALS from 1999 to 2011, Rezigh et al noticed an increasing incidence of MALS diagnosis since 2000 but showed that only approximately 2.44% of patients undergo celiac artery decompression. They also highlighted an increased correlation of those diagnosed with MALS having concurrent depression. 12 Further, Kozhimala et al found that those diagnosed with MALS are less likely to have diabetes (12.5% vs 26.9%), renal disease (4.6% vs 8.2%), hypertension (41.5% vs 70.3%), mesenteric atherosclerotic disease (14% vs 61.9%), and peripheral arterial disease (15% vs 39.7%) when compared to patients diagnosed with celiac artery compression most commonly due to atherosclerosis. 11 It is worth noting that they distinguish MALS as a syndrome with associated symptoms, and celiac artery compression is an anatomical diagnosis made through imaging including mesenteric ultrasound, CT angiography, MR angiogram, and conventional angiography.
In a 10-year cross-sectional study using the American College of Surgeons-National Surgical Quality Improvement Project database, Romero-Velez et al identified 763 patients who had undergone surgical release of the MAL for CACS. Among this cohort of patients, the majority were white (87%) and female (74%) with a mean age of 44 years old and an average BMI of 24 kg/m2. Sixty-one percent were classified as American Association of Anesthesia (ASA) Categories 1 and 2, with a low prevalence of comorbidities. 7
Anatomy and Pathophysiology
Despite its original description primarily focusing on the vessel compression causing ischemic symptoms, we now understand that the amalgamation of both vascular and neurogenic compromise is more likely closer to the whole story for patients with MALS. Vascularly, there is chronic repetitive compression of the celiac trunk that is partially relieved during inspiration as the celiac artery moves caudally and the diaphragm moves cephalad, and then recompression during expiration constricting the celiac axis in the cephalad direction and leading to downstream signs and symptoms of upper gastrointestinal ischemia. The continual inculcation of the diaphragm upon the celiac trunk results in intimal hyperplasia, as the walls of the celiac artery attempt to strengthen and stenose against the repetitive squeeze.
Due to extensive collateral circulation supplied from the mesentery, it was thought that at least 2 of the 3 major arteries to the gut must be partially or fully occluded, and/or diseased for symptoms of ischemia to occur. 8 The main collateral pathway between the celiac axis and superior mesenteric artery is through the gastroduodenal artery. When prograde flow through the celiac axis is compromised, the superior mesenteric artery can compensate with retrograde flow through the anteroinferior and posteroinferior pancreaticoduodenal arteries. These vessels communicate with their counterparts, the anterosuperior and posterosuperior pancreaticoduodenal arteries, to form a collateral network called the pancreaticoduodenal arcade. 13 Similarly, Rosenblum et al describe the arc of Barkow, a potential communication between omental branches from both the SMA and celiac axis, as well as an “uncommon but well-described communication,” the arc of Buhler, defined as a direct communication between the celiac artery and SMA, which is a persistence of the embryonic ventral segment arteries. 14 This collateral network is able to supply flow to the otherwise compromised areas of the celiac axis and is the basis for the hesitation in diagnosis and disconnect between clinical symptoms and objective findings in patient with MALS, when there are asymptomatic patients with the same level of external MAL tightness.
Despite the vast collateral network supply, Hollier et al 9 found that 23% of patients diagnosed with chronic mesenteric ischemia had single artery disease, most commonly isolated to either the superior mesenteric or the celiac artery. In 2016, Weber et al suggested that MALS was not a vascular disease, positing the celiac ganglion compression was more likely the cause of the altered sensation in pain from the somatic nerves in the plexus, much like other nerve compression diseases. 15
Neurogenically, the celiac ganglion and plexus sit atop the celiac trunk (Figure 1) and are exposed to the same chronic repetitive external compression as the celiac artery with respiratory variation. The celiac, or solar plexus, is the largest of the sympathetic nervous system and is responsible for relaying sensory information of the gastrointestinal tract from stomach to splenic flexure. Balaban et al were among the first to theorize a neurogenic basis for the symptoms associated with MALS, dating back to 1997.
16
In a case report in the American Journal of Gastroenterology, they explain that the regularization of the gastric electrical rhythm in their patient after surgical decompression of the celiac axis supports a neurogenic basis for MALS symptoms. Anatomical relationship of celiac ganglion and celiac trunk with aortic hiatus (https://www.nysora.com/pain-management/ultrasound-guided-celiac-plexus-block-and-neurolysis/).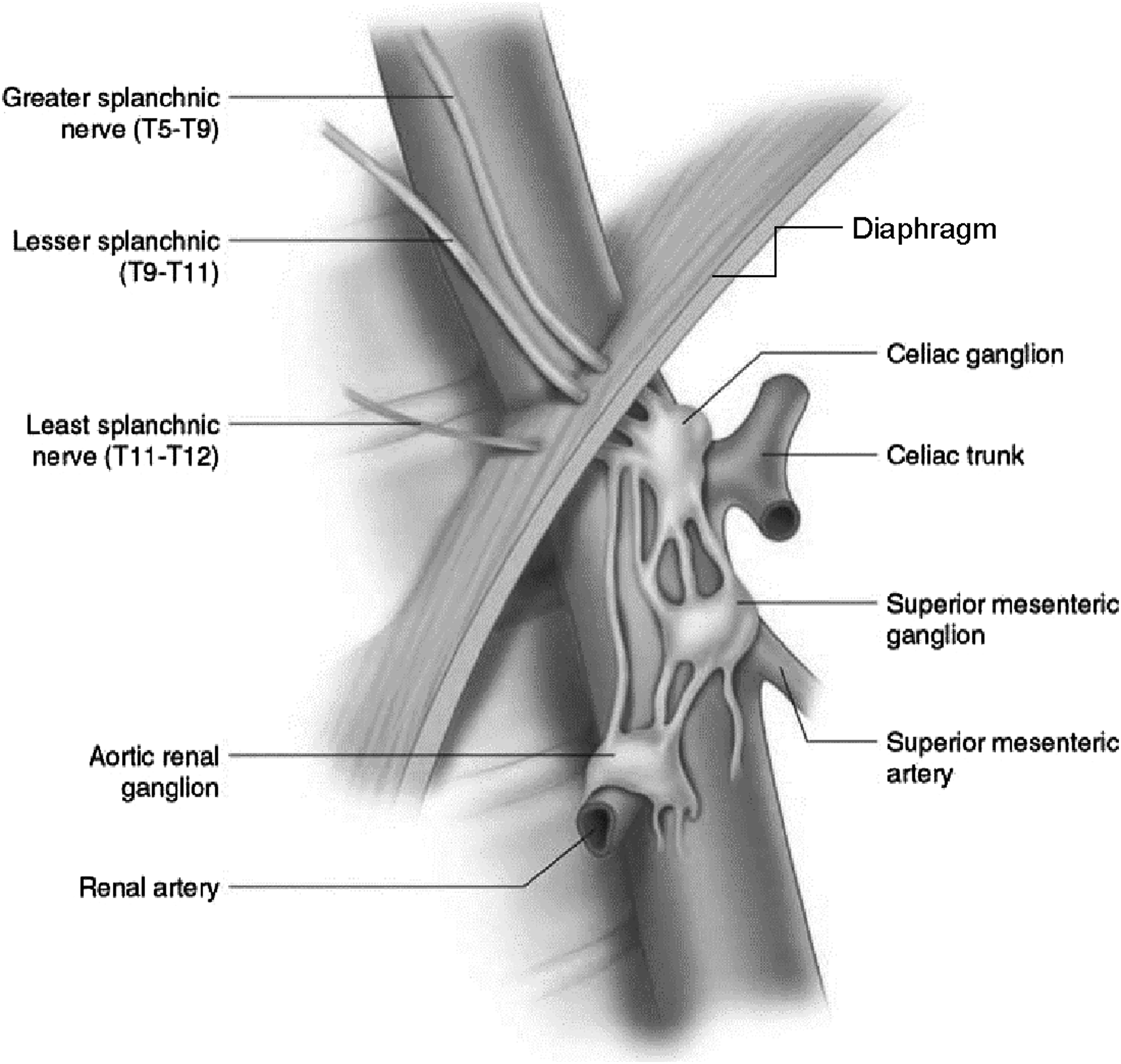
In 2021, Barbon et al published a single-institution, retrospective cohort study in patients clinically diagnosed with MALS where most patients experienced postprandial pain relief and decreased GI symptoms after a celiac plexus block. 17 Notably, this pain relief was not correlated with the presence of celiac artery abnormalities and was suggestive of the primary etiology of MALS being due to neuropathy.
Clinical Presentation
Patients with median arcuate ligament syndrome typically present with symptoms such as nausea, vomiting, postprandial epigastric pain, and sitophobia, or the extreme aversion to food and/or eating (often mistaken as anorexia18,19), that if left undiagnosed for long enough can lead to weight loss. However, Harr et al also published a case series of 3 athletes who presented with exercise-related transient abdominal pain, diagnosed with MALS, and had complete resolution of their symptoms following treatment. 20
Several authors cite the symptoms of MALS as flow-related and due to organ ischemia in the upper abdomen.21,22 This symptomatology has been well documented for years and is well understood. In Qamar’s study published in 1985, they were able to demonstrate that once food reached the gastric mucosa, there was immediate rise in blood flow through the celiac axis, a response that is instantaneous and short lived with peak systolic velocity returning to baseline after 10-15 min. 23 In a patient with MALS, extrinsic compression (and intrinsic stenosis from prolonged compression) of the celiac artery prohibits this increase in blood flow through the celiac axis and the same peak systolic velocity is unable to be obtained, and can lead to symptoms of organ ischemia.
As these symptoms overlap with more common conditions such as biliary colic, gastroparesis, Helicobacter pylori disease, gastroesophageal reflux disease (GERD), peptic ulcer disease (PUD), and Inflammatory Bowel Disease (IBD), other diagnoses must first be excluded and lead to a physical/temporal workup delay in diagnosis for these patients. This can contribute to a substantial delay between presentation and diagnosis for a patient with MALS that averages 10.5 months to 2.6 years.9,10
Having further characterization of the syndrome and its clinical profile might help diagnosticians solidify their medical decisions earlier. Huynh et al released a retrospective case series of eleven patients from a single-center institution highlighting possible associated conditions for patients diagnosed with MALS. Of those identified as patients with MALS who underwent surgery within 5 years (2013-2018), 71.4% were found to have evidence of delayed gastric emptying, 36.4% has anatomic abnormalities of their visceral vasculature, 27.3% had postural orthostatic tachycardia syndrome, and 18.2% had Ehlers-Danlos syndrome. 24
Dyches et al later published a cadaveric study examining anatomic differences in celiac axis morphology between cis males and cis females, finding the distance between the celiac trunk and median arcuate ligament to be considerably shorter with twice the likelihood of the median arcuate ligament overlapping the celiac trunk in female cadavers than in male cadavers. 25 Further investigation into associated conditions and linked pathophysiology has not been completed.
Trinidad-Hernandez et al proposed one of the first algorithms for evaluating patients with suspected CACS/MALS in 2006. 26 In this algorithm, patients were expected to undergo 8 procedures or imaging studies prior to meeting criteria for diagnosis of MALS. In 2019, Goodall et al published an updated algorithm that added 3 more tests to the routine abdominal investigations (colonoscopy, blood tests [including liver function tests and amylase], and H. pylori testing) prior to up to 2 more specialized tests assuming all the prior examinations were normal and only a possible incidental finding of celiac artery stenosis was found. 27
Diagnosis and Diagnostic Protocol
Simply put, a patient with MALS is diagnosed based on their clinical presentation and exclusion of other more common causes of abdominal pain (see above). So, for patients presenting with postprandial and/or post-exertional epigastric abdominal pain, episodic regurgitation, possible sitophobia, and weight loss, a health care provider will complete a history and physical exam (potentially noticing an epigastric bruit), and initiate a comprehensive gastrointestinal workup pathway including bloodwork, esophagogastroduodenoscopy (EGD), upper gastrointestinal series (UGI), right upper quadrant ultrasound (RUQ US), abdominal CT or MRI, and nuclear medicine gastric emptying study. Esophagogastroduodenoscopy helps rule out mucosal pathology, gastric emptying study helps rule out gastroparesis, and cross-section imaging (US/CT/MRI) helps rule out upper GI anatomic or functional pathologies. 15
After these studies are completed, a diagnosis of MALS may be considered plausible prompting more specialized evaluation. A specialist may obtain inspiratory and expiratory duplex ultrasonography and angiography of the celiac axis and SMA to better characterize the anatomy. 27 For a patient with MALS, duplex US characteristically shows a normal SMA and an abnormal celiac axis with velocity measurements varying with respiration.
Moneta et al demonstrated that the mesenteric duplex ultrasound for flow through the celiac artery is a validated screening test to detect for 70% or greater SMA and CA stenosis in their 1993 blinded prospective study. In patients with MALS, there is an increased velocity in the celiac artery, typically greater than 200 cm/second, that correlates with 70% stenosis on angiography. 28 Rodriguez cites a peak systolic velocity of greater than 249 cm/sec in the expiratory phase with normalization during inspiration as pathognomonic for celiac artery compression. 29 Computed tomography angiogram of the celiac axis in a patient with MALS will demonstrate characteristic hooking, or a focal narrowing in the proximal celiac axis, helping to distinguish this condition from intrinsic causes of celiac artery narrowing such as atherosclerotic disease. 30 Kim et al showed post-stenotic dilatation may be observed as a consequence of external compression of the celiac artery in patients with MALS. 22 Brody et al proposed a predictive model for patients with MALS using age and preoperative celiac artery expiratory velocities to predict clinical outcomes. 30 It is worth noting that there was a 60% correlation in their dual-variable logistic regression model between predicted probability of success and percent improvement. 31
New evidence from Barbon et al shows no correlation in celiac artery abnormalities and those patients who were clinically diagnosed with MALS and received pain relief with a celiac plexus block (CPB), further suggesting the absence of stenosis should not be used as an exclusion criterion. 17 As the neurogenic aspect of MALS is better understood, many have moved towards using the celiac plexus block as a confirmational diagnostic tool.15,17,32 These studies include both pediatric and adult patients diagnosed with MALS and have shown CPBs to be a predictor of symptomatic improvement after surgery. The block is used to target the visceral afferent pain fibers from several abdominal organs. Several approaches have been described, but most commonly, the posterior or anterior para-aortic approach is used.
Though there are not yet dedicated studies for the safety of CPB in the diagnosis of MALS, Davies et al published safety data on 2730 neurolytic blocks with either phenol or alcohol over a 5-year period (1986-1990) with a rate of permanent paraplegia less than 0.25% (4 patients, 0.15%), and notably 3 of these same patients with loss of anal and bladder sphincter function; therefore, the incidence of major complications following neurolytic celiac plexus block was cited at one case per 683 blocks. 33 Eisenberg et al published a meta-analysis of the safety and efficacy of neurolytic celiac plexus blocks in the treatment of cancer pain (63% pancreatic and 37% nonpancreatic). 34 Within this meta-analysis, they cite severe adverse effects occurring about 2% of the time (13/628 patients), including neurologic complications (1%) such as lower extremity weakness and paresthesias, and lumbar puncture; and additional 1% of non-neurological complications such as pneumothorax, pleuritic pain, hiccoughing, and hematuria. Taking these 2 studies in combination, health care providers can have an informed conversation with their patients when discussing a celiac plexus block for preoperative diagnostic verification or therapeutic symptom relief of MALS.
Chaum et al expanded the evidence for a neurogenic etiology for MALS in their recent clinico-pathologic case series. 19 They found 3 of 4 patients improved after robotic MAL release and celiac gangliectomy when surgical specimens showed periganglionic and perineural fibrosis with proliferation of small nerve fibers, consistent with that of an early traumatic neuroma. Given the relief of pain after removal of this damaged structure, this study further supports neurogenic compression as the primary etiology for MALS. The authors also suggest all excised tissues be submitted to pathology, especially all associated celiac ganglionectomies, for further characterization and study of this disease.
Treatment
The mainstay of treatment for patients with MALS/CACS is to address the associated symptoms thought to be induced from compression of the median arcuate ligament upon the celiac trunk and plexus. Options for alleviation of symptoms have expanded since the first open MAL release described in 1963. 2 Today, treatment options include celiac plexus block for therapeutic management, celiac angioplasty, celiac bypass, and open or minimally invasive (robotic/laparoscopic) median arcuate ligament release (or some combination of these), with 97% of procedures for MALS performed being the median arcuate ligament release according to literature reports. 22
When deciding who is a good candidate for surgical treatment for MALS, Weber et al found that those with atherosclerotic risk factors may have poorer outcomes than their non-atherosclerosis counterparts; however, symptom relief was seen in most patients undergoing an MAL release regardless of open or laparoscopic approach. 15 Additionally, Ho et al demonstrated a correlation with increased response to surgical decompression if patients presented with post-exertional pain (P = .022) but noted a lower likelihood of improvement post-decompression for those patients presenting with unprovoked pain (P = .006) or vomiting (P = .046). 35 Patel et al was also unable to identify conventional imaging (duplex ultrasound or computed tomography angiography) findings that could consistently predict the clinical response to laparoscopic release of the MAL. 36 This correlates with our increasing knowledge that MALS may be primarily more of a neurogenic disease over a vascular etiology.
Surgical Decompression
The most traditional method of treatment for MALS is decompressing the median arcuate ligament through an open approach. Open decompression begins with an upper midline laparotomy to access and decompress the MAL and the diaphragmatic cura away from the celiac artery. The diaphragmatic fibers are incised approximately 5 cm cephalad, exposing up to 4 cm of the aorta. Visual inspection or intraoperative ultrasonography demonstrating a return to normal peak systolic velocities can confirm efficient MAL release. Based on our understanding of MALS pathophysiology today, further neurolysis and wide excision of the involved celiac plexus are also recommended at this point to address the neurogenic component of this syndrome. 22 In a retrospective review of prospectively collected patient data over 10 years at the University of North Carolina at Chapel Hill, Kohn et al found both open and laparoscopic approaches to be safe with durable mid-range follow-up results. 37 Moreover, in a multi-institutional, international 20-year cohort study published in 2023, the Vascular Low Frequency Disease Consortium compiled data from 516 MAL release procedures (227 open, 44%; 235 laparoscopic, 45.5%; 54 robotic, 10.5%) showing no statistical difference in long-term failure after open vs laparoscopic MAL decompression but did note an associated higher perioperative morbidity with the open release procedure. 38 Moreover, it is important to note this study published a 3-year freedom from treatment failure rate at 51.9%, (95% CI, 46.1%-57.3%) with factors associated with increased treatment failure including robotic MAL release, a history of gastroparesis, a history of abdominal cancer, dysphagia or odynophagia, no relief from a preoperative CPB, and increasing number of pain locations. 38 A treatment failure rate as high as 48% is something to discuss with patients openly when going through the risks and benefits for this procedure.
Due to multiple studies showing no long-term differences in failure rates (see above), laparoscopic decompression of the celiac artery is quickly becoming the mainstay approach for MALS patients because of the benefits associated with any laparoscopic procedure leading to decreased postoperative morbidity (such as decreased rates of ileus, pain, blood loss, adhesions, and shorter recovery times). To our knowledge, Roayaie et al 39 was the first to describe the laparoscopic approach for treatment of MALS, which typically involves 4-5 ports to divide the MAL and skeletonize the celiac artery (with or without vascular intervention) with postoperative angiography for celiac artery flow assessment. Some considerations specific to the laparoscopic treatment of MALS include whether to use intraoperative ultrasonography and the potential for persistent stenosis in the celiac axis immediately following MAL release. Proponents of intraoperative ultrasound advocate for its use to verify the anatomy and adequate decompression by watching the celiac artery flow rate decrease upon ligament release; however, persistent internal compression from stenosis could leave a falsely elevated flow rate following decompression. Furthermore, Roseborough et al 40 reported improvement of symptoms in 93% of patients (14/15) following laparoscopic treatment with only intraoperative direct visualization of the celiac artery following MAL release.
In a direct comparison of open and laparoscopic treatment of MALS, Jiminez et al published a review of 400 patients who underwent surgical (279 open and 121 laparoscopic) treatment for MALS and were able to demonstrate 85% immediate postoperative symptom relief with only 26 patients later having recurrence of symptoms (19 patients in the open group [6.8%] and 7 patients in the laparoscopic group [5.7%]). 41 Notably, 9.1% of laparoscopically treated patients (11/121) required conversion to laparotomy secondary to bleeding (5 celiac artery bleeding, 2 aortic punctures, 1 phrenic artery laceration, 1 gastric artery bleeding, and 1 suprarenal artery bleeding). There were 3 cases of pneumothorax in the laparoscopic group but no procedure-related deaths. Most common complications cited in the open group were thrombosed bypass grafts (n = 6; 2%), stroke (n = 4; 1.4%), GERD (n = 3, 1%), pancreatitis (n = 3, 1%), hemothorax (n = 1; .3%), and splenic infarctions (n = 1; .3%), with no procedure-related deaths reported. 41
With the novelty of robot-assisted surgeries, we are still learning the best techniques for robotic MAL release. Do et al 42 demonstrated minimal mortality and morbidity with both the laparoscopic and robotic approaches in treatment of 16 patients with MALS in their single-institution retrospective review but noted a significantly longer operative time for the robotic approach than for the laparoscopic approach (145.8 min vs 101.7 min; P = .02). Until the robotic approach is able to reduce surgical times or improve patient outcomes, the laparoscopic approach to median arcuate ligament decompression will continue to be the standard of care.
Revascularization
Even with the shift in our understanding of the pathophysiology of MALS, physician-surgeons are still pursuing revascularization or vascular reconstruction of the celiac trunk on a patient-by-patient basis. In a 1997 article published in the Journal of American College of Surgeons, Takach et al described a tailored intraoperative approach to CACS based on individual operative findings over a 15-year period. 43 In the 5 (of 7 total) patients who underwent revascularization, there were 3 patients who received an aorto-celiac bypass (2 with polytetrafluoroethylene grafts and 1 with a vein graft), 1 patient received an angioplasty with a kitted polyester fiber patch, and 1 patient has aortic reimplantation of the celiac artery. This tailored approach to MALS treatment provided complete immediate and long-term relief from preoperative symptoms in all 7 patients.
The decision for vascular reconstruction is also based in making all attempts to alleviate compression with as little surgical intervention as possible and is often performed in patients with persistent thrill, compression, malformation, or pressure gradient in the celiac artery despite initial external MAL decompression. 43 With long-term compression, the intimal and adventitial layers of the celiac artery undergo hyperplasia with proliferation of abnormal smooth muscle and elastic fibers and can lead to substantial narrowing of the lumen. 6
An alternative to open revascularization is laparoscopic release of the MAL with percutaneous transluminal angioplasty (PTA). Seen as an adjunct to MAL release, the PTA has proven to achieve good patient outcomes for patients with persistent stenosis after initial external MAL decompression in a limited number of studies.40,44,45
Celiac Ganglion and Plexus Involvement
The profound results from CPB in addressing pain associated with MALS build a strong argument for celiac ganglion involvement of the disease.15,17,32 Sultan et al found that MAL decompression with celiac ganglion sympathectomy (CGS) is associated with improvement in mesenteric blood flow and marked symptomatic improvement when using the Quality-Adjusted Time Spent Without Symptoms of Disease and Toxicity of Treatment survey to evaluate patient outcomes following surgical treatment for MALS patients. In this study, with a mean follow-up interval of 2.8 years, 71% of patients were able to achieve freedom of symptoms despite repeat postoperative duplex ultrasound of the celiac artery showing a high velocity in 50% of patients. 10 Rationales used for ganglionectomy include resection compared with simple division may better inhibit reformation of a compressive band and that ablation of the ganglion will address some of the pain associated with MALS. 22
Summary
Median arcuate ligament syndrome, or celiac artery compression syndrome (eponym: Dunbar syndrome), has historically been attributed to pathophysiologic vascular compression causing downstream ischemic symptoms of the organs supplied by the celiac trunk. However, the more we learn about the histology, clinical presentation, and treatment outcomes, health care providers are increasingly correlating the symptoms of MALS with the long-term, repetitive compression of the celiac ganglion rather than the celiac trunk. With most patients with MALS waiting an average of 10.5 months to 2.6 years,9,10 our need for better diagnostic protocols and clearer understanding of the pathophysiology of the disease is paramount.
As demonstrated with Barbon et al’s study, there is increasing evidence showing no correlation between the celiac artery abnormalities and patients diagnosed with MALS who receive relief from a CPB, further suggesting the absence of stenosis not be used as an exclusion criterion for diagnosis. 17 Furthermore, the regularization of the gastric electrical rhythm in patients after surgical decompression of the celiac axis supports a neurogenic basis for MALS symptoms. With the repetitive damage caused by the diaphragm rubbing against the celiac ganglion, Chaum et al found histologic changes consistent with early traumatic neuromas. 19
After concluding a patient has MALS through a long series of diagnostic imaging and laboratory tests ruling out other diseases, a provider is still left with selection of an appropriate treatment plan without the benefit of firm guidelines. Today, options include celiac plexus block for therapeutic management, celiac angioplasty, celiac bypass, and open or minimally invasive (robotic or laparoscopic) median arcuate ligament release (or some combination of these) with 97% of patients receiving a ligament release at minimum. 22 Moving forward, the addition of a celiac ganglion sympathectomy (CGS) might demonstrate continued improvement and freedom from symptoms, as seen in the Sultan et al study. 10 By resecting the celiac ganglion, compared with simple division, there is thought that this will better inhibit reformation of a compressive band and pain associated with MALS. 22 Further investigation into patient outcomes, associated conditions, and linked pathophysiology would help better characterize this disease with hopes of moving it from a diagnosis of exclusion to one of standard work-up with decreased time to treatment and symptom relief for patients.
Supplemental Material
Supplemental Material - Median Arcuate Ligament Syndrome: Where Are We Today?
Supplemental Material for Median Arcuate Ligament Syndrome: Where Are We Today? by Kayden Maddox, Timothy M. Farrell, and Luigi Pascarella in Journal of the American Surgeon™.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
