Abstract
Background
Enhanced Recovery After Surgery (ERAS) programs have spread after initial success in colorectal surgery decreasing length of stay (LOS) and decreasing opioid consumption. Adoption of ERAS specifically for ventral hernia patients remains in evolution. This study presents the development and implementation of an ERAS pathway for ventral hernia.
Methods
A multidisciplinary team met weekly over 6 months to develop an ERAS pathway specific to ventral hernia patients. 75 process components and outcome measures were included, spanning multiple phases of care: Preoperative-Clinic, Preoperative Day of Surgery (DOS), Intraoperative, and Postoperative. Preoperative components included education and physiologic optimization. Pain control across phases of care focuses on nonopioid, multimodal analgesia. Postoperatively, the pathway emphasizes early diet advancement, early mobilization, and minimization of IV fluids. We compared compliance and outcome measures between a Pre Go-Live (PGL) period (9/1/2020-8/30/2021) and After Go-live (AGL) period (5/12/2022-5/19/2023).
Results
There were 125 patients in the PGL group and 169 patients in the AGL group. Overall, ERAS compliance increased from 73.9% to 82.9% after implementation. Length of stay decreased from an average of 2.27 days PGL to 1.92 days AGL. Finally, the average daily postoperative opioid usage decreased from 25.4 to 13.5 MME after the implementation.
Discussion
Enhanced Recovery After Surgery can be successfully applied to the care of hernia patients with improvements in LOS and decreased opioid consumption. Institutional support and multidisciplinary cooperation were key for the development of such a program.
Key Takeaways
• Development of an ERAS program for hernia can be successful with multidisciplinary involvement from anesthesia, surgery, and other providers involved in perioperative care. • Enhanced Recovery After Surgery can be applied to the care of hernia patients with successful reduction in opioid use and LOS.
Introduction
Incidence of ventral hernia repair (VHR) continues to increase and accounts for substantial health care cost. 611,000 VHRs were performed annually in the United States in 2019, nearly twice the volume relative to 2006, with an estimated total annual cost of $9.7 billion. 1 For this large patient population, focused efforts to optimize perioperative care represent an important part of achieving the Institute for Healthcare Improvement’s (IHI) Triple Aim for high-value care: improving the care experience, improving population health, and reducing costs. 2
The advent of Enhanced Recovery After Surgery (ERAS) in 2001 marked a paradigm shift in perioperative care-focused principles of multimodal care, multidisciplinary collaboration, evidence-based practice, and continual reevaluation that are embedded in four stages of perioperative care: preadmission, preoperative, intraoperative, and postoperative. 3 The ERAS society has subsequently published guidelines in over 20 surgical specialties. 4 Benefits of these protocols including decreased LOS and decreased opioid use have been demonstrated across multiple disciplines including colorectal surgery, bariatrics, urology, and gynecology.3,4
There are several published evaluations of ERAS protocols for VHR with mixed outcomes. Colven et al demonstrated no significant decrease in LOS for patients undergoing abdominal wall reconstruction after implementation of an ERAS protocol. 5 However, Jensen et al demonstrated a decrease in LOS from 5.5 to 3 days after initiating ERAS. 6
Beyond the composite evaluation of Hernia ERAS protocol implementation, individual ERAS components have demonstrated effectiveness in the care of hernia patients. Specifically, the use of multimodal analgesia to minimize opioid use after VHR is well established.7,8
While evidence of the benefit of ERAS protocols for particular surgical disciplines is well established, and the evidence supporting ERAS pillars is well described, the practical steps of implementing a sustainable and effective program for ERAS in VHR are still evolving. This study describes the development of our ERAS protocol for VHR. We examine how several process measures changed after ERAS implementation and how opioid use and length of stay for hernia patients changed over the course of the study period.
Methods
This study was a quality improvement initiative and was deemed exempt from Institutional Review Board approval per Vanderbilt University Medical Center.
Hernia ERAS Pathway Development
A multidisciplinary ERAS team was developed with surgeon and anesthesia champions as leaders. Other members of the team included nurses, advanced practice providers, hospital administrators, and data analysts. Team members were selected from across the phases of perioperative care. The team met weekly over a 6-month period to create the ERAS hernia pathway. Established ERAS pathways for other disciplines at our institution were used for a framework. Additionally, the team performed a literature review of hernia-specific perioperative care. Ultimately, relevant best care standards were agreed upon. Track-able process measures of compliance were selected.
The team collaborated with health information technology specialists to build electronic order sets for preoperative, intraoperative, and postoperative phases of care and facilitate implementation. Data tracking began several months prior to launching the new ERAS protocol to establish a baseline from which to compare outcomes after implementation and quarterly ERAS compliance, and outcome measures are made available to surgeons, anesthesiologists, and other stakeholders for continual process evaluation. Surgeons, anesthesiologists, and nursing staff in all phases of care were educated on the components of this new program two months before launch.
Patient Population
Adult patients undergoing elective ventral hernia repair at Vanderbilt University Medical Center were included in the ERAS pathway. Patients undergoing outpatient surgery and those admitted postoperatively either for extended recovery or inpatient admission were included. Outpatient VHR patients in whom a primary hernia repair was performed and mesh was not placed, or in whom mesh smaller than 8 cm diameter was placed, were excluded. We excluded patients who underwent any concomitant procedure (urologic, colorectal, oncologic, etc) at the time of VHR from the hernia data set, but these patients still received ERAS pathway treatment. Open, laparoscopic, and robotic approaches were included, as were complex hernia with component separation, as part of the surgical treatments.
Description of Hernia ERAS Pathway
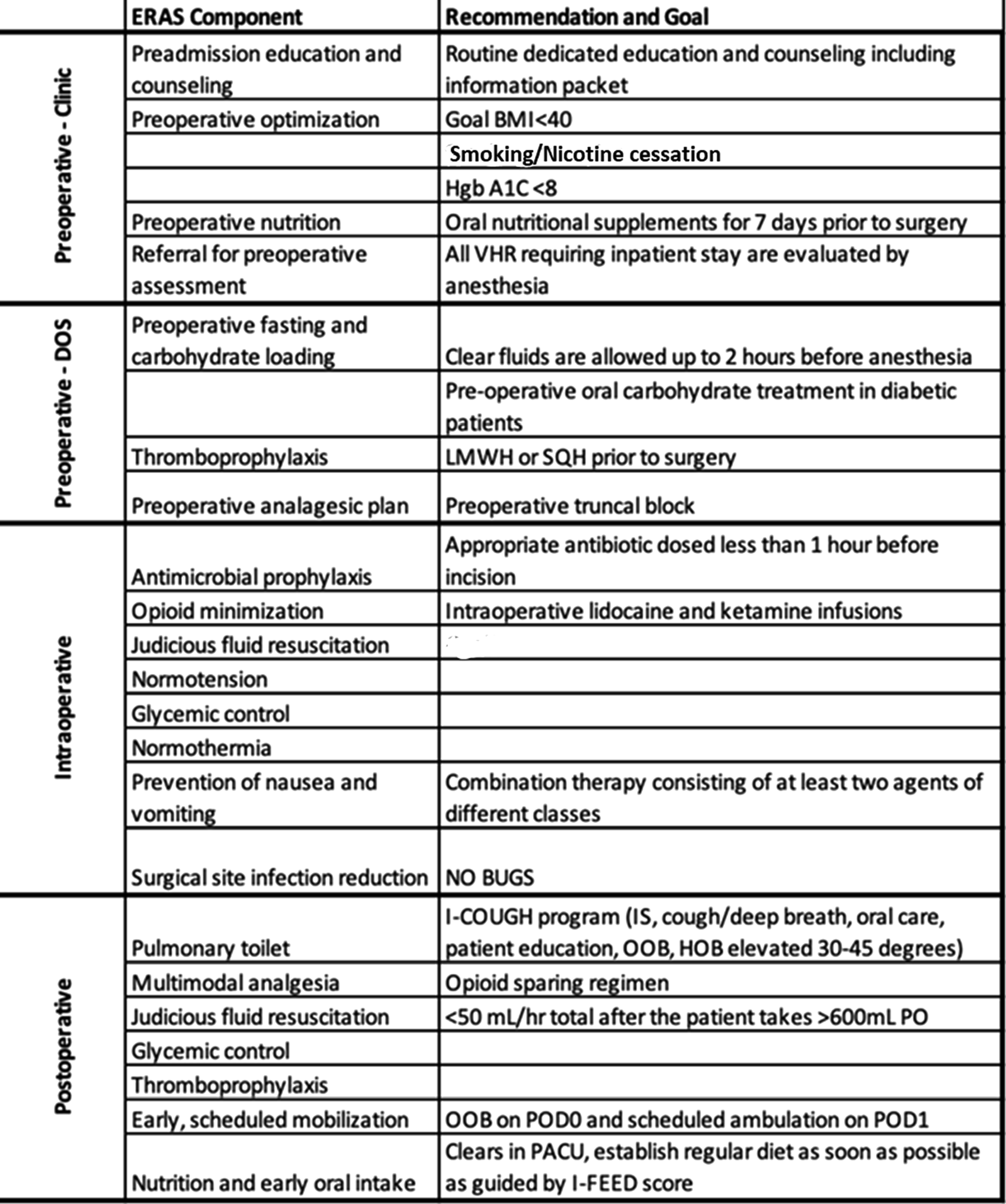
VHR ERAS Framework Outlining the 4 Phases of Care (Preoperative-Clinic, Preoperative-DOS, Intraoperative, and Postoperative).
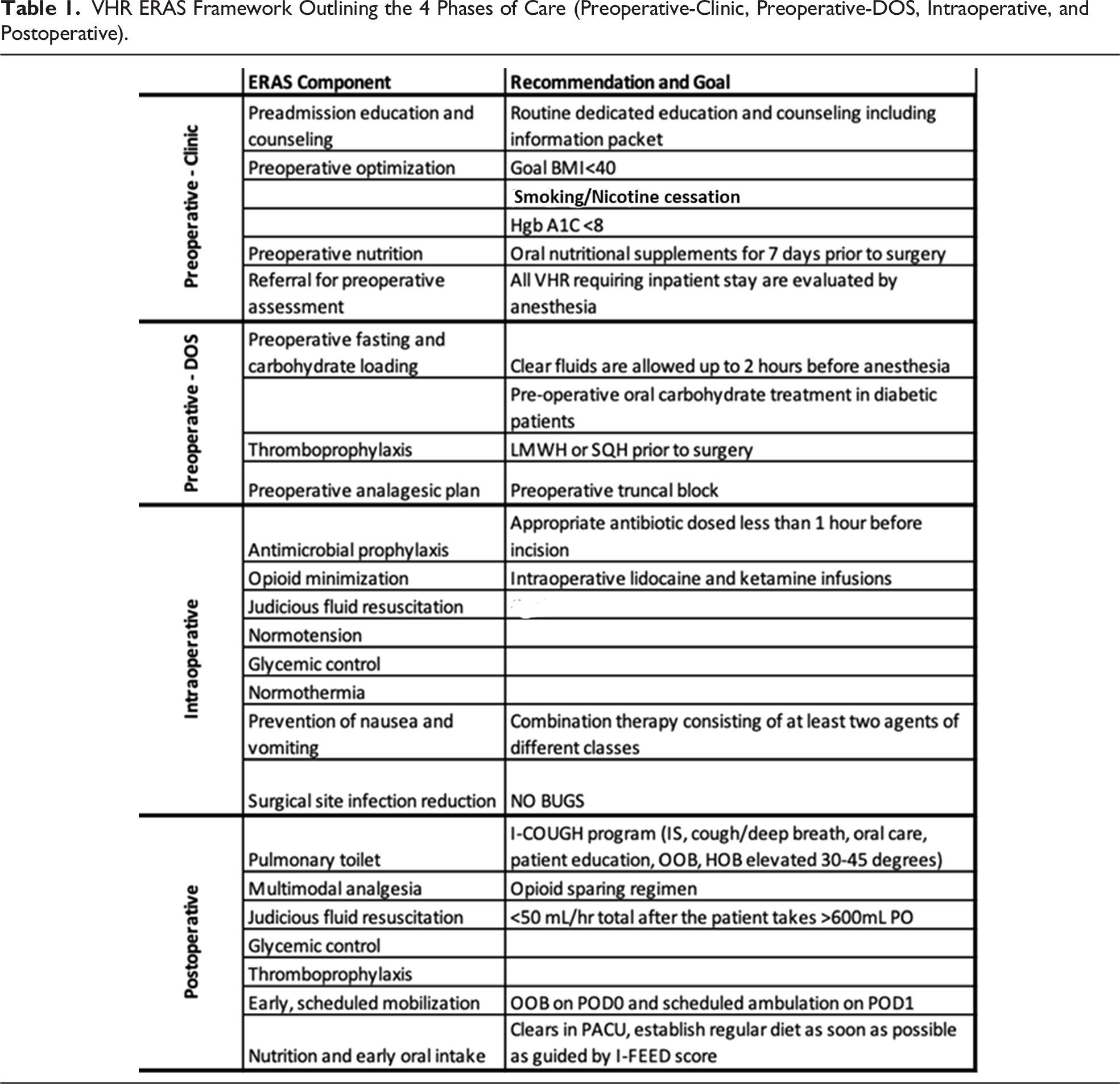
Preoperative-Clinic
Preoperative management of patients scheduled for VHR in clinic is focused on setting expectations and optimizing physiology. Physicians and clinic nurses counsel patients about ERAS components relevant to their operation. An educational packet outlines patients’ expected course and includes detailed instructions for before and after surgery. All patients are screened for goal BMI <40, smoking/nicotine cessation, and goal Hgb A1C <8. For patients with large hernia or hernia with loss of abdominal domain, botulinum toxin is considered to assist with intraoperative fascial closure. All patients are referred to a preoperative anesthesia clinic for comorbid risk stratification and optimization. One final component of this physiologic preparation is nutritional assessment and supplementation. All patients are screened for nutrition risk (>10% weight loss in the prior 6 months, BMI <18.5, and albumin<3 g/dL). Those at a risk of malnutrition are evaluated by a nutritionist and their procedure is delayed. All others receive oral immunonutritional supplements for 7 days prior to surgery.
Preoperative Day of Surgery
Day of Surgery management aims to prepare patients for the postoperative period. To prevent dehydration and subsequent excessive intravenous fluid administration, patients are allowed and encouraged to drink clear liquids (to include a protein clear liquid supplement) until 2 hours before the procedure. The operative site is clipped and cleaned with chlorhexidine wipes in the holding area to mitigate the risk of infection. Chemical thromboprophylaxis is administered. Multimodal opioid-sparing pain management is initiated with the administration of gabapentin, acetaminophen, and celecoxib in preoperative holding unless contraindicated.
Across phases of care, our multimodal approach to pain management relies on enteral medications, intravenous infusions, and regional blocks to avoid opioids. Enteral medications include scheduled acetaminophen, NSAIDs, and gabapentin. When used together, acetaminophen and NSAIDs have been shown to decrease pain approximately 35%. 9 While evidence of gabapentin’s use for analgesia is conflicting, a recent meta-analysis showed a trend for reduced pain. 10 We included gabapentin in our protocol as the sole agent specific to neuropathic pain. Many published ERAS bundles include scheduled muscle relaxants. No literature supports the use of muscle relaxants on a scheduled basis for surgical pain, so we prescribe them only as needed, due to the risk of sedation and orthostasis. Methocarbamol is our standard as-needed muscle relaxant.
Lidocaine and ketamine infusions are another helpful adjunct for managing postoperative pain. Weibel et al describe that lidocaine infusion tends to decrease ileus, nausea, and opioid consumption. 11 Additionally, a meta-analysis of ketamine infusions demonstrates reduction in postoperative opioid consumption and pain intensity with minimal adverse effects. 12
Epidurals are effective adjuncts to pain control in ERAS protocols, but they may prolong length of stay due to decreased ambulation and urinary retention.13,14 Alternatively, transversus abdominis plane (TAP) blocks are highly effective in VHR, 14 and regional blocks serve as our primary method of regional pain control. TAP, rectus sheath, and quadratus lumborum blocks are utilized depending on the location and anatomy of the hernia.
Intraoperative
All patients are managed according to Multicenter Perioperative Outcomes Group (MPOG) Anesthesiology Performance Improvement and Reporting Exchange (ASPIRE) Quality Improvement Project goals aimed to prevent acute kidney injury, avoid hypotension, maintain euglycemia, limit intravenous fluid administration, and achieve normothermia. 15 Our standard is for opioid minimization of less than 15 morphine milliequivalents (MMEs) to be administered intraoperatively for opioid naïve patients. Ketamine, lidocaine infusions, and either inhaled or intravenous agents are used to achieve this goal. Sugammadex is used as our standard neuromuscular blockade reversal agent. Postoperative nausea and vomiting prophylaxis is standard with 4 mg IV ondansetron prior to emergence. Dexamethasone is utilized in the regional blocks for a second antiemetic. SSI reduction is continued with bundled components of appropriate pre-incision antimicrobial prophylaxis and use of two applications of chlorhexidine gluconate prep. The outer gloves are exchanged prior to mesh implant handling.
Postoperative
Postoperative management is focused on early normalization. Multimodal opioid-avoidant pain management strategies of scheduled oral acetaminophen, gabapentin, and NSAIDs continue. Continuous IV lidocaine and ketamine use is employed selectively depending on the degree of pain control and targeted discharge time. Abdominal binders are also used as an adjunct for pain control. Methocarbamol is utilized as needed for muscle spasms, especially in component separation patients.
Prevention of complications includes medical comorbidity management with resumption of key home medications, avoidance of hyperglycemia, and judicious fluid management with a standardized initial maintenance rate of 50 mL/hr with goal of discontinuation on POD#1 if tolerating oral intake. Diet starts with clear liquids in PACU and transitions to regular diet with supplements after clear liquids are tolerated, preferably on POD#1. Infection prevention includes Foley catheter avoidance and early catheter removal (by POD#1) and prevention of postoperative pneumonia with the I-COUGH bundle. 16 Steps for early normalization consist of scheduled mobilization (QID) and out of bed in a chair for meals.
Details of Interim Analysis
The ERAS protocol implementation “Go-Live” occurred on 9/21/2021. We compared process and outcome measures before and after implementation. The 1-year Pre Go-Live (PGL) period examined was 9/1/2020 through 8/30/2021. The 1-year After Go-live (AGL) period examined was 5/12/2022 through 5/19/2023, several months after implementation to allow for a more complete adoption of the ERAS protocol. The two cohorts were compared using descriptive statistics using Excel, Version 2311, Microsoft, and Tableau.
Results
Population Descriptive Data.
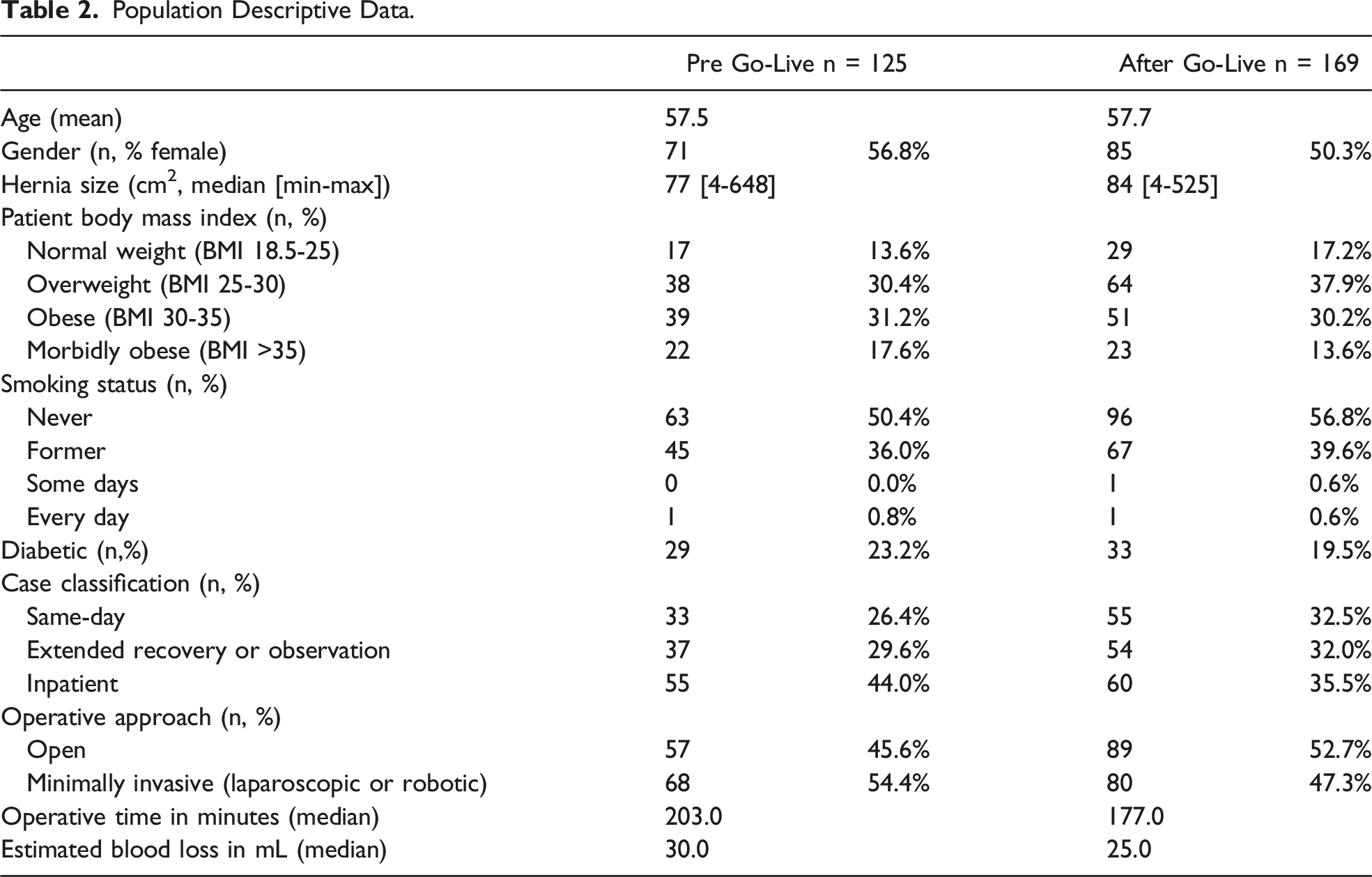
We examined how various process measures varied between the PGL and AGL groups across phases of care. Beginning with the Preoperative-Clinic phase of care, in the AGL period 89.9% of patients completed in-clinic ERAS education, and 76.3% of patients completed an in-person anesthesia clinic evaluation prior to surgery. After Go-live there was a higher rate of A1c screening for diabetic patients, and more patients achieved an A1c score below the set limit of 8.0 (97.7% AGL vs 88.2% PGL of those patients who were screened). In the Preoperative-DOS phase of care, there was 75.7% compliance with use of the Preoperative Order set in the AGL period. From the PGL to AGL periods, there was a notable increase in the use of enteral multimodal analgesia in the holding area. Preoperative acetaminophen administration increased from 54.6% PGL to 77.9% AGL and gabapentin administration increased from 50.5% to 77.5%. Celecoxib was not prescribed preoperatively in the PGL period, and 82.7% of patients received celecoxib in the AGL period. For many intraoperative process measures, interestingly, there was not much difference between the compliance with various MPOG process metrics between the PGL and AGL periods like glucose measurement, intraoperative temperature management, and VTE prophylaxis. Compliance rates for PONV prophylaxis administration, PACU rescue opioid avoidance, and intraop opioid minimization were similar between PGL and AGL periods. Use of lidocaine and ketamine infusions was 74.6% and 78.3%, respectively, in the AGL period, similar to the PGL period. Of note, there was a notable difference in the intraoperative intravenous fluid administration, with a median of 1.5 L in the PGL period and 1.2 L in the AGL period.
In the postoperative ward phase of care, 75.7% of cases employed the postoperative order-set. After Hernia ERAS implementation, there was an increase in the administration of nonopioid analgesics; specifically, NSAID prescription increased from 52.5% to 82.2% of patients and muscle relaxant prescription increased from 54.0 to 85.1%. Rates of acetaminophen and gabapentin prescription did not increase substantially relative to the preoperative period, but a high rate of compliance was maintained with acetaminophen at 90.3% and gabapentin at 91.4% AGL. Intravenous fluid administration over POD 0-2 decreased by 34.8% from a median of 2.2 L PGL to 1.4 L AGL. In the AGL period, patients were advanced to a regular diet on average 1.1 days after surgery (IQR 1,1), which was an improvement from a mean of 1.4 days after surgery (IQR, 1, 2) in the PGL period. In the PGL period, patients ambulated 6.8 hours (IQR 0.34-8.4) after surgery, on average, and performed a mean 1.6 (IQR 1.0-2.3) ambulations daily. After Go-live patients ambulated on average 5.1 hours (IQR 0.5-8.1) after surgery and performed 1.9 (IQR 1-2.6) ambulations daily. Wound infection rates were 4.1% in the PGL. These were all superficial infections and one deep infection. In the AGL group, the infection rate was 2.4% and all the infections were all superficial. Both groups had 1 seroma requiring procedural intervention. There were no mesh infections in either group. There were no perioperative deaths in the PGL group and 1 death in the AGL group on POD#63 due to a severe COVID-19 infection.
ERAS Compliance and Outcome Measures.
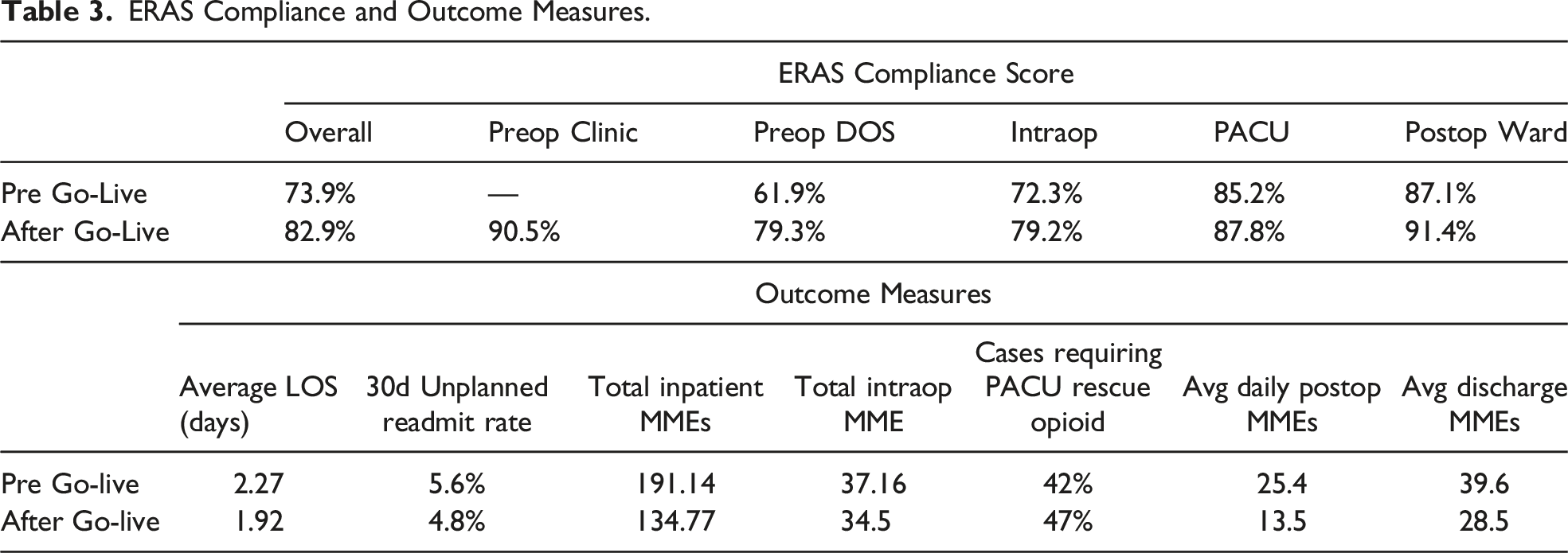
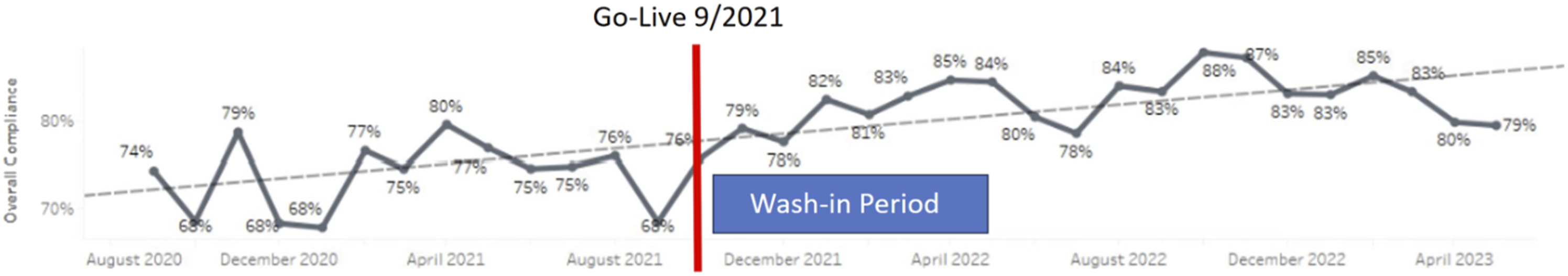
Overall ERAS compliance over time. This figure plots the change in compliance with all ERAS components combined from across the 4 phases of perioperative care.
Finally, there was a meaningful improvement in outcome measures targeted, as demonstrated in Table 3. There was an improvement in LOS from 2.27 to 1.92 days on average with no increase in the 30d unplanned readmission rates, which were 5.6% PGL and 4.8% AGL. Additionally, the average inpatient opioid MME decreased 30% from 191.1mme PGL to 134.7mme AGL, and the average daily postop MMEs decreased almost by half from 25.4 to 13.5.
Discussion
Our study demonstrates a comprehensive approach to implementing ERAS for ventral hernia patients. This can serve as a framework to establish local programs for hernia patients. Institutional support and identification champions from surgery, anesthesia, nursing, and leaders in all phases of care are key for development of a meaningful program. We describe key components of implementation across phases of perioperative care. Institutional support was already in place for ERAS programs, and several of the measures have become part of standard surgical care for all patients. Upon starting a formal program for ventral hernia with tracked measures and processes, we were able to make improvements in LOS and a decrease in opioid utilization. Our institution already has well-established ERAS programs in other specialties, and we were using these in a non-organized manner prior to this program. This has allowed us to make improvements from a baseline of good outcomes with continued room for enhancement.
As with most enhanced recovery programs, individual metrics alone are not what makes the difference in outcomes, but rather the sum of the parts. Compliance with ERAS components was evident in many of our tracked process measures, and there was a demonstrated trend of decreased LOS and decreased opioid use after implementation. Key interventions were preoperative education and physiologic optimization, nonopioid, multimodal analgesia beginning preoperatively and continuing through the postoperative phase, and early normalization. Institutional support and identification of champions from surgery, anesthesia, and leaders in all phases of care is key for development of such programs. Creation of a formal program with tracked measures allows the program to undergo continuous evaluation and evolution of best practices. Ongoing reinforcement and education, along with data analysis, are needed to ensure compliance and demonstrate successful clinical impact.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.