Abstract
The use of endoscopic retrograde cholangiography (ERCP) for diagnostic and therapeutic interventions on the pancreaticobiliary system has steadily increased, but the standard approach through the oropharynx is prohibited after Roux-en-Y (RYGB) gastric bypass surgery. Laparoscopic access to the gastric remnant allows for the completion of ERCP using the standard side-viewing duodenoscope to facilitate the completion of standard and advanced endoscopic maneuvers. Here, we describe our experience with the technical aspects of safe and effective performance of laparoscopic-assisted ERCP.
Keywords
Endoscopic retrograde cholangiography (ERCP) is a commonly performed procedure which allows diagnostic and therapeutic interventions on the pancreaticobiliary system via the gastrointestinal tract. Its use has steadily increased over recent years for indications including treatment of bile duct or pancreatic duct obstructions, as well as biopsies of masses or strictures, among others. 1 In standard performance, the endoscope is inserted into the oropharynx, passed through the esophagus and stomach and into the duodenum to access the ampulla of Vater.
After Roux-en-Y gastric bypass (RYGB), which is performed nearly 50 000 times annually in the United States, this anatomic conduit is eliminated.
2
The gastric pouch is connected to a limb of jejunum to form the Roux limb of usually 100-150 cm, while the remnant stomach and duodenum drain into the biliopancreatic (BP) limb of around 50 cm (Figure 1). As such, the distance needed to travel with the endoscope is generally prohibitive in performing this procedure with a duodenoscope. Efforts at balloon- or spiral-assisted deep small bowel enteroscopy can be technically hampered by the angulation of the jejuno-jejunal anastomosis irrespective of limb length and, even when the papilla is accessible, by the lack of a side-viewing apparatus or elevator mechanism. Anatomy of laparoscopic-assisted ERCP.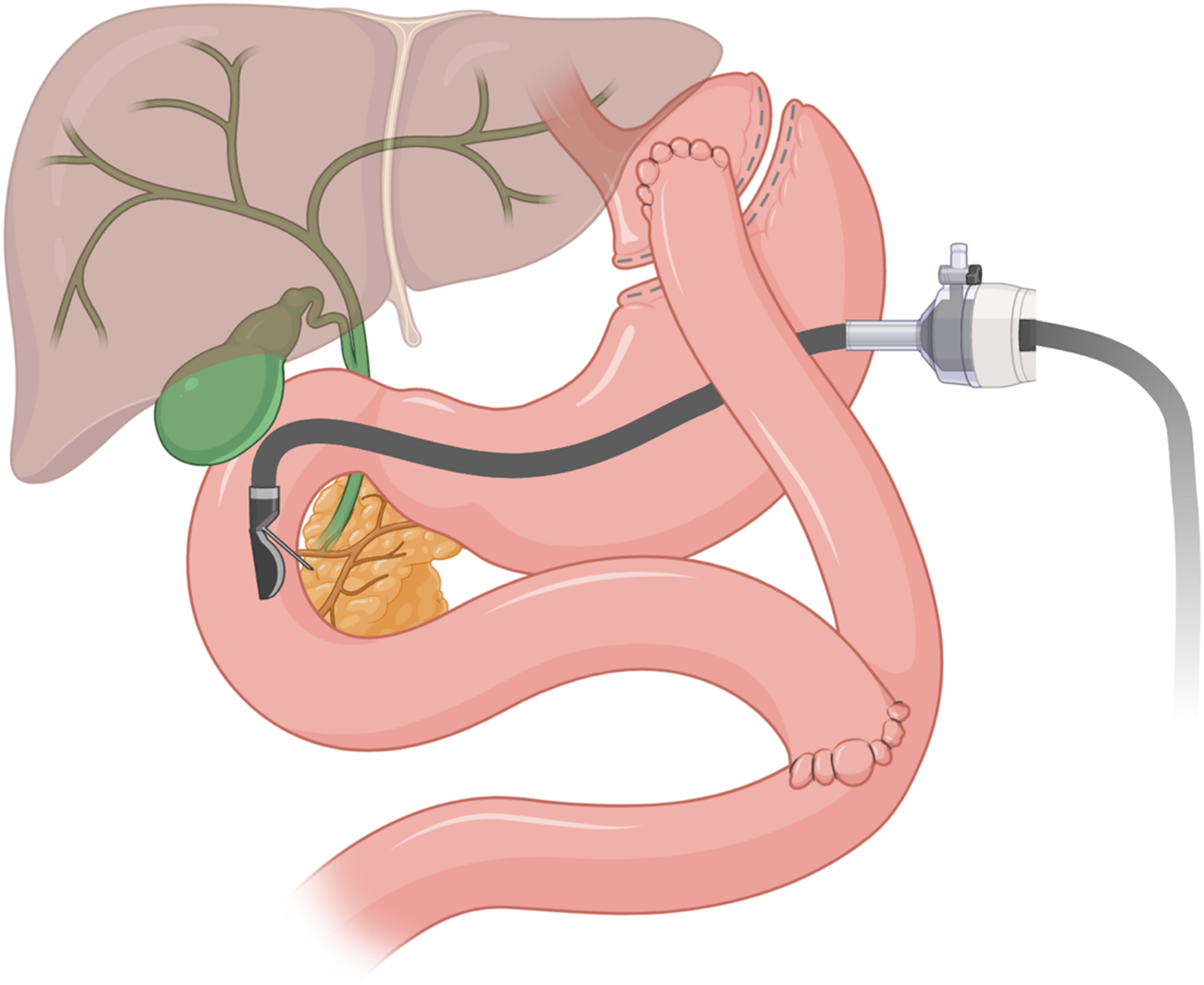
While laparoscopic-assisted ERCP requires additional resources in the use of the operating room instead of the endoscopy suite, and coordination between surgeon and endoscopist, laparoscopic-assisted ERCP has been demonstrated to have higher success rates and shorter procedure times than other advanced endoscopic techniques accessing the duodenum through the Roux limb and retrograde up the BP limb. 3 Here, we describe the technical considerations for performing laparoscopic-assisted ERCP in a patient who had previously undergone RYGB.
The patient is a 57-year-old woman who was referred to gastroenterology for evaluation of recurrent pancreatitis. She had previously been hospitalized multiple times with pancreatitis, elevated liver tests, and MRI findings of biliary dilatation concerning for choledocholithiasis. Her history was notable for a prior RYGB and prior laparoscopic cholecystectomy. She underwent an MRCP that demonstrated mild intrahepatic and extrahepatic biliary ductal dilatation with a 10 mm common bile duct, T2 hyperintense filling defect in the CBD consistent with an obstructing stone, and mild pancreatic ductal dilatation. The options for biliary drainage were discussed with her, and she elected to proceed with laparoscopic-assisted ERCP.
Materials.

The outer tip diameter of most commercially available duodenoscopes is between 13 and 14 mm, which can be easily advanced through a 15 mm trocar. Despite the short distance to travel from the trocar entry site in the stomach to the pylorus, traversing the pylorus and intubating the duodenum is not always straightforward. These efforts can be hampered by limited gastric distensibility with endoscopic insufflation in the setting of pneumoperitoneum, angulation of the trocar tip away from the pylorus, or entry of the trocar too close to the antrum/pylorus.
Once the duodenoscope is advanced to the papilla, a stable scope position is typically readily achievable. A portable C arm can be positioned for fluoroscopic imaging and ERCP can then proceed in standard fashion. Typical therapeutic maneuvers of ERCP including sphincterotomy, stone removal, and stent placement can be performed as indicated. Advanced techniques such as cholangioscopy and intraductal lithotripsy can be performed as indicated and if equipment and expertise are available.
An important consideration at completion of ERCP is need for endoscopic re-access. If elective future ERCP is anticipated, for instance, for stent removal/exchange or repeat stone therapy, an externally removable gastrostomy tube can be left at the trocar entry site. Once a mature gastrocutaneous fistula has formed, typically a minimum of 4-6 weeks following gastrostomy, the gastrostomy tube can be removed, and the tract can be dilated with standard endoscopic dilating balloons to permit percutaneous duodenoscope re-access to the gastric remnant.
This maneuver does not, however, permit endoscopic re-access within the early post-ERCP period. Delayed post-sphincterotomy hemorrhage requiring endoscopic intervention, as an example, may develop within several days following sphincterotomy. Treatment in this scenario would require either surgical re-access to the excluded gastric remnant or non-endoscopic (ie, angiographic) intervention. Limitations on early endoscopic re-access also carries important implications for prevention of post-ERCP pancreatitis (PEP), for which placement of a prophylactic pancreatic stent is a recommended strategy for patients at high risk for PEP. 4 These stents are intended for short-term indwell when used for PEP prophylaxis and may sometimes dislodge and pass spontaneously but in many instances require endoscopic removal—ideally in a time frame to prevent iatrogenic stent-induced pancreatic duct injury, typically sooner than time to maturation of the gastrostomy tract. This may diminish enthusiasm for prophylactic pancreatic stent placement in this patient population in favor or alternative PEP-pancreatitis strategies including stringent patient selection and rectal indomethacin.
After completion of the ERCP, the 15 mm trocar is removed. If a stent is utilized, or the endoscopist feels repeat ERCP is necessary, a gastrostomy tube can be placed in a Stamm fashion to facilitate percutaneous access. If there is no need for repeat procedure, the gastrotomy site is stapled using a laparoscopic stapler. The fascia of the 15 mm and 12 mm ports are closed with 0 braided absorbable suture, and all port sites are closed with 4-0 monofilament absorbable suture and skin glue.
During her ERCP, the patient was found to not have a common duct stone but rather a periampullary mass from which biopsies were taken and a stent was placed. After completion of the procedure, she was taken to the post-anesthesia care unit and admitted for observation postoperatively. She was given a clear liquid diet immediately after surgery to reduce the risk of post-ERCP pancreatitis and advanced the following day. Her pathology ultimately returned with pancreatic ductal adenocarcinoma, and she was referred to Surgical Oncology. She underwent a pancreaticoduodenectomy followed by adjuvant chemotherapy. As demonstrated by this case, laparoscopic-assisted ERCP is straightforward procedure and provides a valuable additional tool beyond MRCP and percutaneous transhepatic cholangiography for both diagnostic and therapeutic interventions on the pancreaticobiliary system after RYGB.
Footnotes
Acknowledgments
Figure made with BioRender.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
