Abstract
In recent years, isolated non-operative management of penetrating liver injuries has become the standard of care for the hemodynamically stable patient. However, when the patient becomes hemodynamically unstable, adjuncts such as resuscitative endovascular balloon occlusion of the aorta (REBOA) deployed in Zone 1 can be used to achieve complete aortic occlusion from the celiac axis down. Unfortunately, hemorrhage control through REBOA comes at the risk of deadly intra-abdominal ischemia. Partial REBOA (pREBOA) introduces the opportunity to make targeted changes in volume and thus titrate the amount of aortic occlusion in real-time to adequately manage hemorrhage while allowing some distal blood flow. This is a novel approach and one which may give providers more time to gain definitive hemorrhage control while minimizing the morbidity of ischemia. Here, we present a case of life-threatening penetrating liver injury that was successfully managed non-operatively with the assistance of p-REBOA.
Keywords
Trauma to the liver spans a wide spectrum of severity, and patient outcomes vary greatly based on the mechanism of injury, location of injury, and patient-specific clinical factors. Destruction of the liver parenchyma can lead to severe hemorrhage, independent of the American Association for the Surgery of Trauma (AAST) classification of severity, resulting in significant morbidity and mortality. Penetrating liver injuries pose additional challenges due to the trajectory and complexity of injury. In addition to parenchymal injuries, a penetrating injury may further damage major hepatic vasculature including hepatic arteries, hepatic veins, portal venous system, or inferior vena cava, leading to life-threatening hemorrhage.
While standard of care has previously been operative intervention, there is growing literature to support non-operative management of penetrating liver injuries in the hemodynamically stable patient. 1 When massive intra-abdominal hemorrhage causes instability, adjuncts such as resuscitative endovascular balloon occlusion of the aorta (REBOA) can provide complete aortic occlusion (CBAO) to achieve relative proximal hemodynamic stability. Resuscitative endovascular balloon occlusion of the aorta allows the operative team time to mobilize and gain definitive hemostasis but also poses the risk of potentially deadly intra-abdominal ischemia. Utilizing REBOA for partial aortic occlusion (PBAO) has gained attention over the past several years because PBAO affords improvement in central blood pressure while minimizing the amount and duration of distal ischemia.2,3 Partial aortic occlusion allows the provider to make targeted changes in volume and thus titrate the amount of aortic occlusion, and thereby the amount of blood flow distally. This may provide more time to mobilize resources for definitive hemorrhage control while minimizing the morbidity of ischemia. While multiple studies have demonstrated extended survival time in swine models, the literature in human subjects remains scarce. 2 Gomez et al leveraged the first REBOA catheter FDA approved for both CBAO and PBAO (pREBOA-PRO™, Prytime). They demonstrated the safety of Zone 1 partial occlusion for extended periods, but the majority of patients from this study suffered blunt injuries, and injury patterns were not included. 3
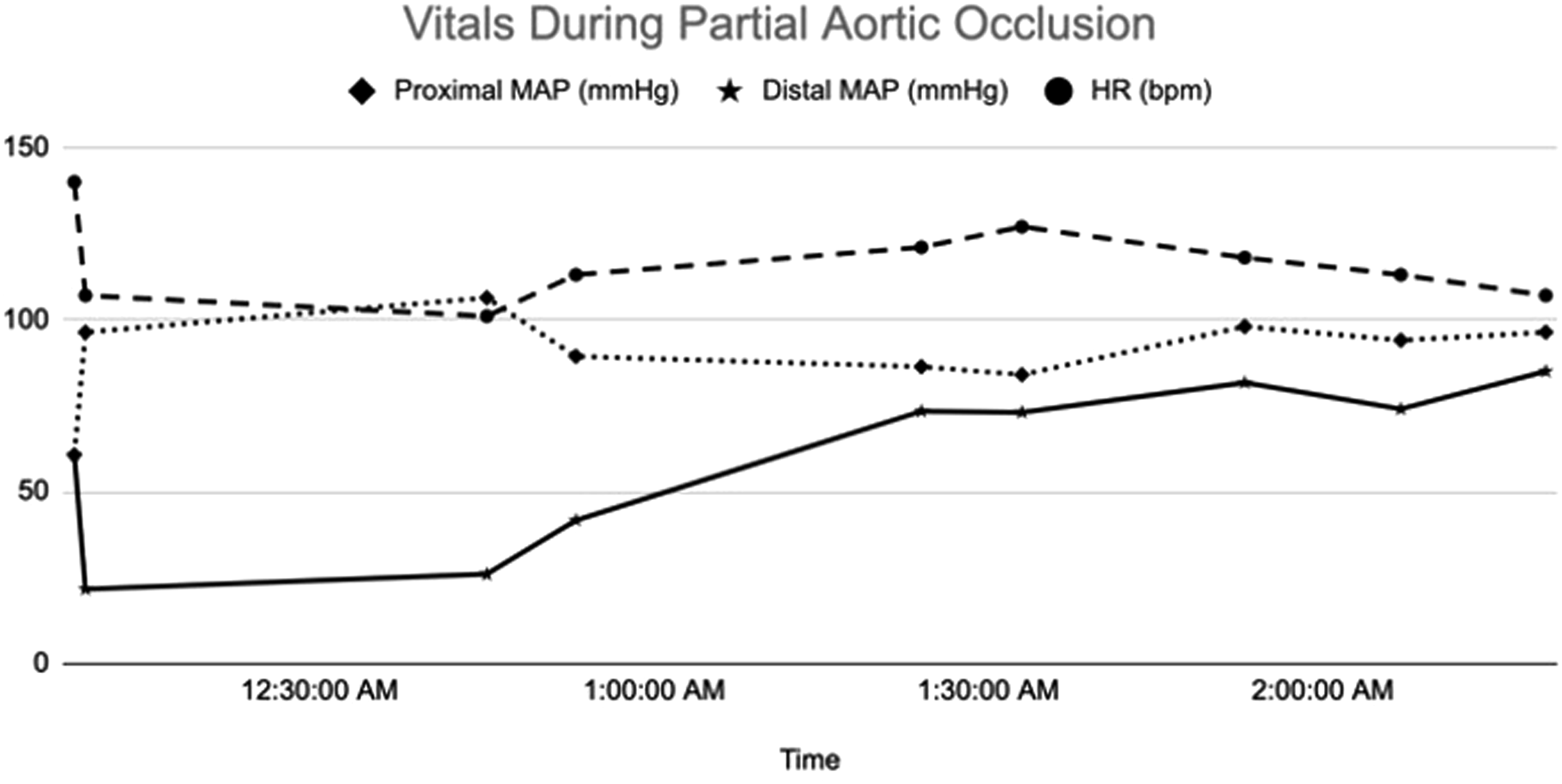
In our case, we present a patient with a life-threatening penetrating liver injury that was successfully managed non-operatively by leveraging PBAO. A 46-year-old male suffered gunshot wounds (GSW) to the epigastrium, right flank, right shoulder, and back. On presentation, he was hemodynamically stable and sent for computed tomography (CT), which revealed a large, right-sided hemothorax and AAST Grade IV liver laceration with multiple pseudoaneurysms and contrast extravasation (Figure 1(a)). Immediate interventions included placement of a right chest tube with one-liter of bloody output and an early common femoral arterial line; interventional radiology (IR) was also consulted. While mobilizing IR, the patient’s blood pressure dropped to 70/56 mmHg. Continuous real-time monitoring of his blood pressure allowed immediate identification of his clinical decline, and PBAO was deployed in Zone 1. The REBOA catheter was inflated with 10cc of normal saline that achieved a proximal pressure of 126/88 mmHg and distal pressure of 35/31 mmHg. PBAO placement stabilized the patient for transport to IR, where he underwent complex angio-embolization of the liver over the next 2 hours. Coil embolization was performed to address a large focus of active extravasation communicating with the pleural space from the lateral branch of segment VIII (Figure 1(b)). During that time, the patient was further resuscitated and PBAO was slowly deflated as hemostasis was achieved. Deflation began 40 minutes after initiation of PBAO and the device was completely deflated at 123 minutes. Throughout the procedure, an experienced provider adjusted deflation of PBAO to meet the goals of systolic blood pressure >90 mmHg and mean arterial pressure >65 mmHg (Figure 2). In total, the patient received two-liters of autotransfusion, four whole blood (WB), two packed red blood cells (pRBC), two fresh frozen plasma (FFP), and one unit of platelets. His post-procedure course was complicated by a retained hemothorax, requiring a video-assisted thoracoscopy (VATS) and a bile leak, requiring endoscopic retrograde cholangiopancreatography (ERCP) with stent placement. At the time of VATS, a small hole was visualized in the right side of the diaphragm abutting the liver; repair of the diaphragm injury was not required. He did not suffer from rhabdomyolysis or acute kidney injury and was discharged to home in three weeks. Radiographic imaging of the patient’s injury. (A) Computed Tomography of the abdomen demonstrating a Grade V liver laceration with active extravasation (white arrow), pseudoaneurysm (white triangle), and significant destruction of liver parenchyma (dark gray areas of liver). (B) Angiogram of the liver demonstrating active extravasation of IV contrast from segment VIII (white star). Graphical depiction of the patient’s (1) heart rate in beats per minute, (2) proximal mean arterial pressure (MAP) in mmHg, and (3) distal MAP in mmHg during partial resuscitative endovascular balloon occlusion of the aorta. Distal MAP equilibrates with the proximal MAP as occlusion is relaxed while maintaining hemodynamic stability.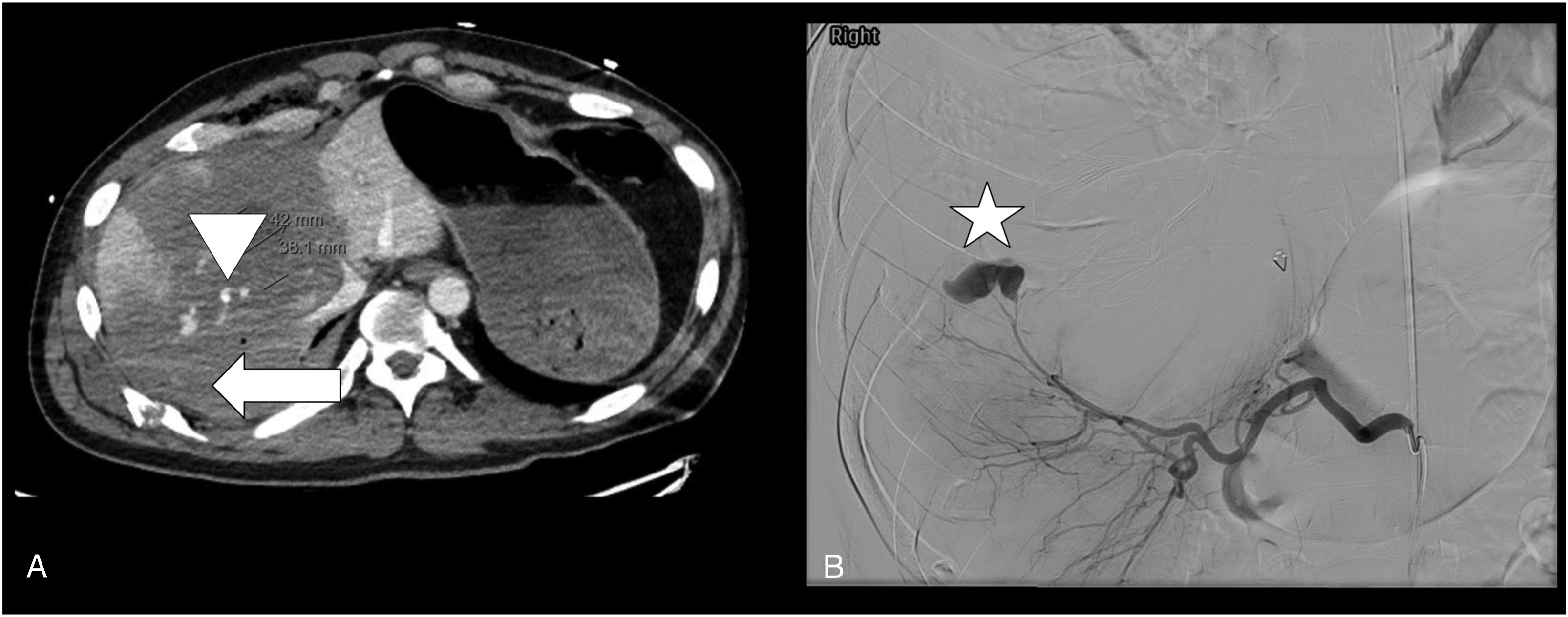
Traditional management of this injury would have obligated the patient to the operating room for emergent laparotomy because of sustained hypotension without response to ongoing transfusion. Due to the intrahepatic arterial damage, location of the liver injury, and >50% parenchymal destruction, there were concerns that while operative intervention may be warranted, it may at best result in a right hepatectomy and at worst, death. However, because of the decision to implement PBAO, IR was able to mobilize and ultimately gain major hemorrhage control while still allowing perfusion of the other intra-abdominal organs and distal extremities. Hemorrhage control and resuscitation was achieved endovascularly and with likely less blood product administration than a traditional laparotomy would have required. All of this was accomplished while maintaining adequate cerebral and cardiac perfusion, minimizing complete distal ischemia, and without large shifts in blood pressure that commonly occur in total occlusion of the descending aorta with tradition REBOA. It is plausible to suggest the complication of bile hemothorax is expected given the severity of his injury, whether he was managed operatively or non-operatively.
Non-operative management of severe liver trauma has always been predicated on a hemodynamically stable patient. The management of severe liver trauma has continued to progress over the course of the past several decades. With adjuncts such as angio-embolization and delayed laparoscopy for bilomas, complication rates, including failed non-operative rate (<10%), have dropped significantly. 4 In select instances of isolated liver injury, PBAO may serve as an adjunct to restrict the blood flow long enough to gain hemostasis by non-operative means. However, PBAO for severe liver trauma has only been described in swine models to date. In these studies, swine that underwent PBAO tolerated extended occlusion times, had lower lactates, and higher pH. 2 Here, we were able to demonstrate successful non-operative management of an unstable patient with a liver injury encompassing more than half of his right lobe with the aid of PBAO. This case study is a strong example of how REBOA can be used to create hemodynamic stability and thus bridge a patient who would otherwise require surgical intervention to endovascular repair. It is important to note that if the patient had become unstable, the surgical team would have proceeded to the OR. In some institutions, a hybrid room may be a good alternative.
Though PBAO offers the promise of additional time to address complex and severe injury, the optimal pressures above and below the balloon and the maximum duration of occlusion have yet to be established. More robust studies are required to fully elucidate how this technology and technique can best play a role in the management of trauma patients. Additionally, PBAO should be performed by an experienced, well-trained provider who is familiar with the technology and knows when and how to use it appropriately. Moreover, PBAO is not meant to serve as definitive hemorrhage control; it is another tool in the physician’s armamentarium that may buy time to organize resources and enhance options available to halt hemorrhage. More studies are required to develop an optimal algorithm for deploying and managing PBAO in the setting of hypotensive trauma patients, but our case demonstrates an instance where it can be employed safely and effectively in patients with penetrating liver injuries.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J Nguyen, DO receives honoraria from Zimmer-Biomet, Teleflex, and Pry-time Medical for educational lectures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: C Meyer is supported by NIH T32 Training Grant in Critical Care, NIGMS (5T32GM095442-11).
