Abstract
Introduction
The management of anal cancer relies on clinical and histopathological features for treatment decisions. In recent years, the field of radiomics, which involves the extraction and analysis of quantitative imaging features, has shown promise in improving management of pelvic cancers. The aim of this study was to evaluate the current application of radiomics in the management of anal cancer.
Methods
A systematic search was conducted in Medline, EMBASE, and Web of Science databases. Inclusion criteria encompassed randomized and non-randomized trials investigating the use of radiomics to predict post-operative recurrence in anal cancer. Study quality was assessed using the QUADAS-2 and Radiomics Quality Score tools.
Results
The systematic review identified a total of nine studies, with 589 patients examined. There were three main outcomes assessed in included studies: recurrence (6 studies), progression-free survival (2 studies), and prediction of human papillomavirus (HPV) status (1 study). Radiomics-based risk stratification models were found to provide valuable insights into treatment response and patient outcomes, with all developed signatures demonstrating at least modest accuracy (range: .68-1.0) in predicting their primary outcome.
Conclusion
Radiomics has emerged as a promising tool in the management of anal cancer. It offers the potential for improved risk stratification, treatment planning, and response assessment, thereby guiding personalized therapeutic approaches.
Introduction
Anal squamous cell carcinoma (ASCC) is relatively rare, accounting for approximately 2.6% of all gastrointestinal cancers.1,2 However, incidence of ASCC is increasing, particularly in high-income countries. 3 ASCC generally has a slow progression and is often responsive to loco-regional treatments due to its low rate of distant metastases at the time of diagnosis. 4 In recent decades, there has been a notable evolution in the standard of care for non-metastatic disease. The approach shifted from radical surgery to curative treatment with chemo-radiotherapy (CRT). This change in treatment strategy was based on the results of phase III trials conducted in the 1990s.5,6
Radiomics refers to the conversion of medical imaging into high-throughput, quantifiable data in order to analyze disease patterns, aid decision-making, and predict prognosis.7,8 Since the concept was introduced in 2012, data from different imaging modalities, including computerized tomography (CT), magnetic resonance imaging (MRI), and positron emission tomography (PET) scans, has been extracted and analyzed. 9 The goal is to determine patient prognosis and predict outcomes, particularly in the field of surgical oncology. 10 Applying radiomic techniques to predict CRT response in anal cancer patients enables personalized treatment, optimizing outcomes by tailoring therapy to individual characteristics. 11 Radiomic nomograms help to stratify patients, identifying those likely to respond well. 12 Predicting recurrence, survival, and disease progression aids treatment decisions, sparing high-risk patients from potentially morbid interventions. An up-to-date literature analysis on radiomics' contemporary use in anal cancer management ensures informed research and practice, harnessing the field's evolving advancements for improved patient care.
The aim of this study is to highlight the current evidence surrounding the contemporary use of radiomics in anal cancer, highlighting strengths, weaknesses, and heterogeneity between studies to guide future research within this novel field of diagnostic and predictive medicine.
Methods
Study Design and Reporting Guidelines
This study is a systematic review of non-randomized trials and follows the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guidelines. 13
Systematic Review Registration
Our systematic review was registered on PROSPERO in March 2023 (ID: CRD42023456092).
Search Strategy
The following databases were searched as part of the systematic review in July 2023: EMBASE, Medline, and Web of Science. The last date of search was 18th July 2023. The grey literature was also searched to further identify other suitable publications. The systematic search process with detailed search terms are outlined in the supplementary material S1.
Inclusion/exclusion Criteria
Inclusion Criteria
- Patient aged 18 years old and above - Studies investigating the use of radiomics to predict oncological outcomes in patients with anal cancer - Prospective or Retrospective Studies.
Exclusion criteria
- Case series/reports. - Consensus statements. - Conference abstracts. - Missing/conflicting data with no response from contacted authors.
Study Selection, Data Extraction and Critical Appraisal
A database was created using the reference managing software EndNote X9TM. Two researchers (HCT and NOS) reviewed outputs from the searches independently of each other.
Initially, duplicates were removed. Study titles were then screened and assessed for potential relevance. The abstracts of selected potential studies were then read and assessed for eligibility for inclusion, based on the inclusion/exclusion criteria detailed above. Rejected studies were grouped together in the database by their reason for exclusion. The full texts of the abstracts deemed eligible for inclusion were then further analyzed using the same criteria.
In order to extract and store data efficiently, the Cochrane Collaboration screening and data extraction tool, Covidence, was used. 14 Data was collected by two reviewers (HCT and NOS) independently, using the following headings; study details, study design, population, intervention, comparison groups, and outcomes.
A critical appraisal of the methodological quality and risk of bias of the included studies was performed. The critical appraisal was completed by two reviewers independently. Quality assessment of the included studies was performed according the Quality Assessment of Diagnostic Accuracy Studies 2 (QUADAS-2) and Radiomics Quality Score (RQS).10,15
Results
Search Results
The literature search yielded a total of 900 results. Following the removal of 450 duplicates, 450 studies were screened. After the initial screen, 237 abstracts were reviewed and assessed for eligibility, of which twelve were selected for full text review. From these twelve full texts, a total of nine studies met the inclusion criteria and were included in our analysis.16-24 See Figure 1. A PRISMA Flowchart of the selection of relevant publications included in this review.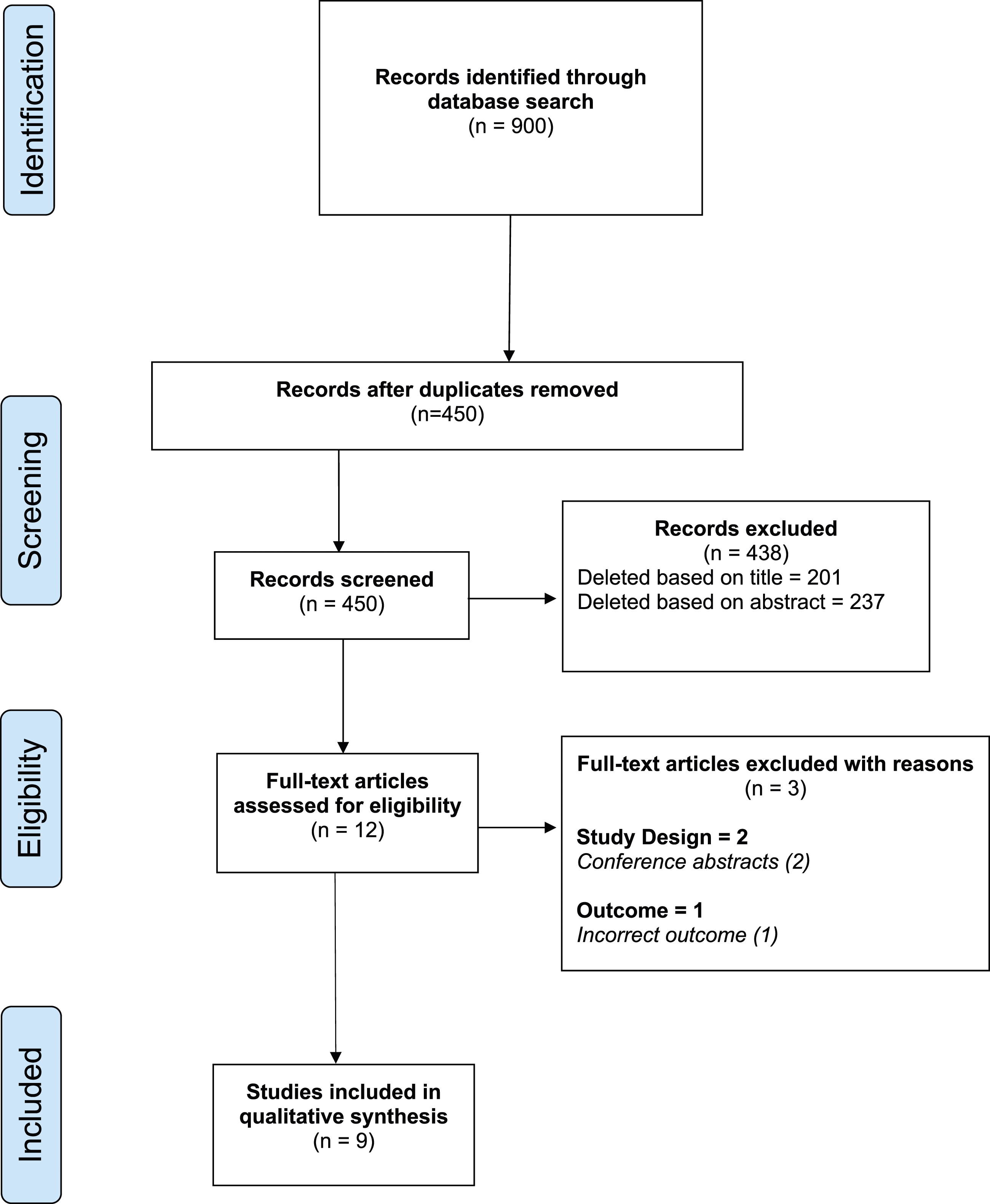
Methodological Characteristics, Quality of Studies, Patient Characteristics
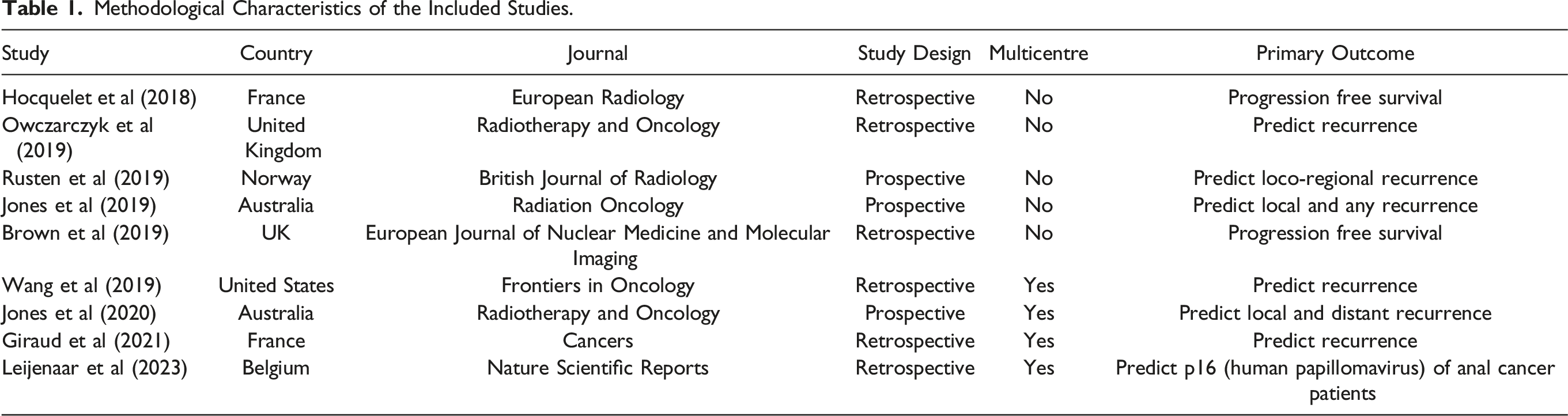
Overall, six of the included studies were retrospective in nature,16-18,21,22,24 with the remaining three being prospective.19,20,23 In total, four studies were multicentre,17,20,21,25 with the other five being single-center studies.16,18,19,22,23 The total number of participants from the nine included studies was 589.
Methodological Characteristics of the Included Studies.
Acquisition Parameters
Summary of Imaging Parameters and Radiomics for Prediction of Response to Chemo-Radiotherapy in Anal Squamous Cell Carcinomas.
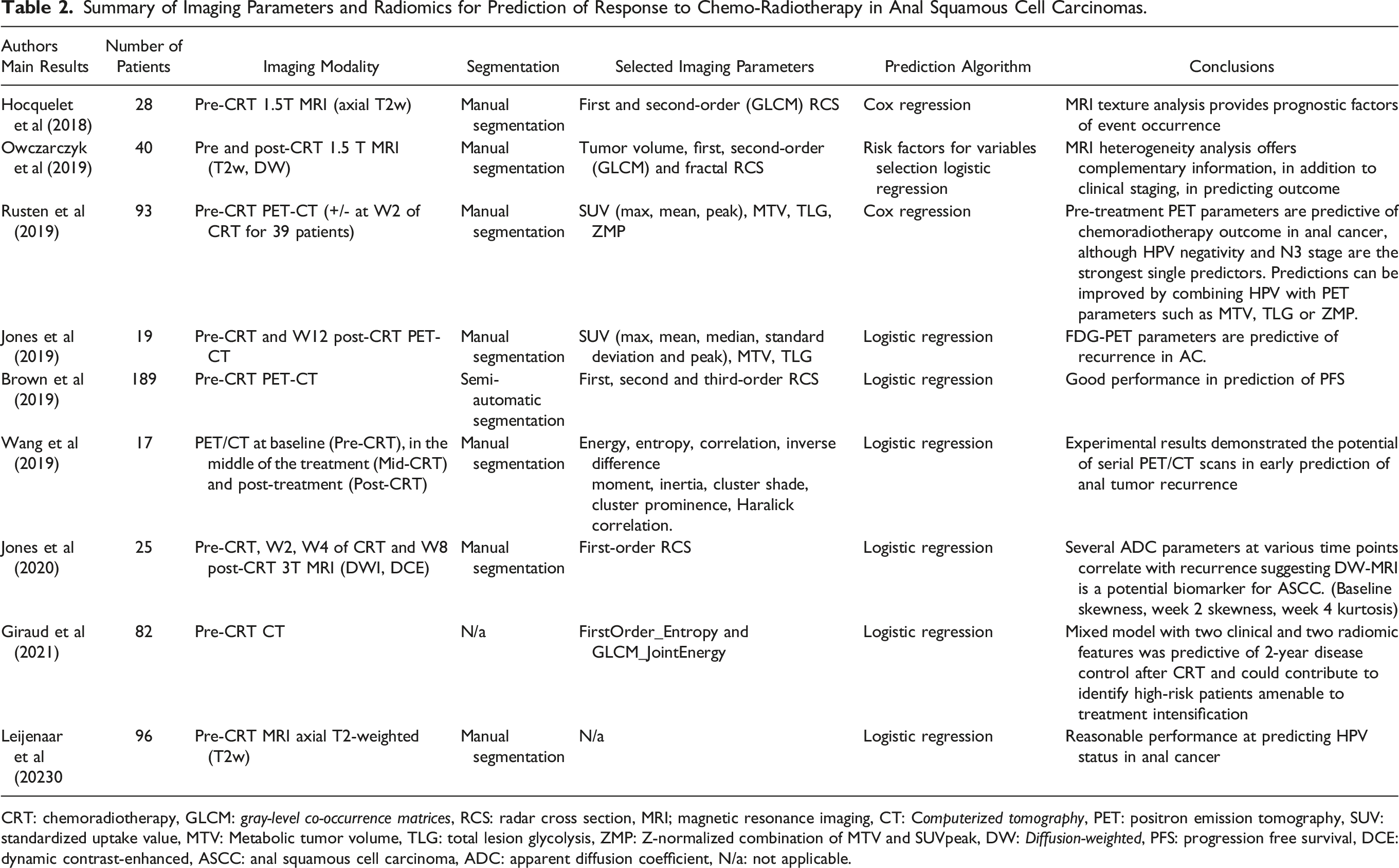
CRT: chemoradiotherapy, GLCM: gray-level co-occurrence matrices, RCS: radar cross section, MRI; magnetic resonance imaging, CT: Computerized tomography, PET: positron emission tomography, SUV: standardized uptake value, MTV: Metabolic tumor volume, TLG: total lesion glycolysis, ZMP: Z-normalized combination of MTV and SUVpeak, DW: Diffusion-weighted, PFS: progression free survival, DCE: dynamic contrast-enhanced, ASCC: anal squamous cell carcinoma, ADC: apparent diffusion coefficient, N/a: not applicable.
Development of Signatures
Exact feature extraction methods varied across studies; however, a relatively similar pathway was followed across the board. Regions of interest (ROI) were first segmented manually by experienced radiologists in all included studies. Radiomic features were then extracted from these segments using radiomics software. Specific software used for image segmentation and feature extraction is demonstrated in Table 2. Logistic and Cox regression models were subsequently applied to features to remove sub-optimal features. Remaining features were used to construct individual radiomics signatures.
Primary Outcomes
There were three main outcomes assessed in included studies (see table 1):
Disease recurrence
Giraud et al 17 aimed to identify novel prognostic factors that could effectively predict cancer recurrence within a 2-year timeframe after CRT. A pre-therapeutic MRI radiomic analysis was utilized, alongside conventional clinical variables, to construct a logistic regression model capable of forecasting the likelihood of recurrence. The study population comprised 82 patients, randomly divided into training (n = 54) and testing (n = 28) subsets. Following the selection of optimal variables, the logistic regression model incorporates two radiomic features (FirstOrder_Entropy and GLCM_JointEnergy) and two clinical features (tumor size and CRT length). Among the 82 patients included, 24 (29%) experienced disease relapse at the 2-year follow-up. Univariate analysis of the training set showed that two clinical (tumor size and CRT length) and two radiomic features (FirstOrder_Entropy and GLCM_JointEnergy) were associated with disease control and were therefore included in the model. A mixed model incorporating both clinical and radiomic features outperformed the clinical model in both the training (area under curve (AUC) .825 vs .758, accuracy 87% vs 75.9%) and testing (AUC .898 vs .714, accuracy 85.7% vs 78.6%) sets, allowing stratification of patients into high and low-risk groups for disease relapse (HR 8.60, P = .005). The combined clinical and radiomic model demonstrated predictive potential for 2-year disease control following CRT in ASCC patients.
Owczarcyk et al assessed the potential of image heterogeneity analysis of standard care MRI in predicting the outcomes of CRT in patients diagnosed with ASCC. 22 The researchers outlined the whole tumor 3D volume-of-interest (VOI) on pre- and post-treatment T2-weighted (T2w) and diffusion-weighted imaging (DWI) scans. They then identified independent imaging features that showed the highest predictive potential for disease recurrence. These imaging features were combined with the baseline clinico-pathological model. To assess the value of the extended models, the researchers used the net reclassification improvement (NRI) algorithm. The study included 40 patients who underwent 1.5 T pelvic MRI at baseline as well as post-CRT. The combination of two baseline MR heterogeneity features (baseline T2w energy and DWI coefficient of variation) demonstrated the highest predictive capability for disease recurrence, leading to a significant net reclassification index (P < .001). Cross-validation analysis further confirmed the effectiveness of these imaging variables, resulting in a 34.8% reduction in percentage error for the primary endpoint and an 18.1% reduction for the secondary endpoint when added to the baseline model.
Wang et al 24 conducted PET/CT scans at three different time points: baseline (Pre-CRT), during treatment (Mid-CRT), and post-treatment (Post-CRT). Image features were extracted from the manually delineated gross tumor volume, encompassing geometry, intensity, and texture features. The differences in feature values between two time points were also computed and analyzed. Various models, such as univariate logistic regression, multivariate models, and naïve Bayesian classifiers, were employed to identify significant predictors of tumor recurrence. The predictive accuracy was evaluated using the area under the receiver operating characteristic (ROC) curve (AUC). The study found specific geometry, intensity, and texture features that were associated with tumor recurrence in the univariate analysis. Moreover, certain features, such as “Roundness” in Post-CRT and Pre-CRT CT scans, demonstrated predictive capabilities with an AUC value of 1.00 in the multivariate logistic regression model. Additionally, early prediction ability was investigated, and combinations of features from Pre-CRT and Mid-CRT scans were identified as valuable predictors. For instance, the combination of “Orientation” in Pre-CRT standard uptake value (SUV), “Mean” in Pre-CRT CT, and “Mean of Long Run High Gray Level Emphasis” in Pre-CRT CT yielded an AUC of 1.00.
Jones et al prospectively investigated 19 patients with non-metastatic anal cancer between 2013 and 2017. 19 Each patient underwent FDG-PET/CT imaging both before CRT and 12 weeks after CRT. The researchers generated a volume of interest (VOI) around the primary tumor using six different SUV thresholds and extracted various PET parameters, including SUV max, mean, median, standard deviation, peak, metabolic tumor volume (MTV), and total lesion glycolysis. The predictive value of these parameters was analyzed using exact logistic regression and ROC AUC analyses at each time point. The results showed that certain FDG-PET parameters, particularly median SUV on the post-CRT PET scan, were significantly correlated with local recurrence (P < .01) and exhibited excellent discriminatory power (ROC AUC 1.00). Moreover, the MTV bounded by a threshold of 41% SUVmax on the pre-CRT PET scan was predictive of any recurrence (P = .03) and showed excellent discriminatory ability (ROC AUC .89).
Rusten et al 23 evaluated the predictive role FDG PET in assessing locoregional failure among 93 ASCC patients undergoing CRT. Before treatment, all patients underwent FDG-PET/CT scans, with a subgroup of 39 patients receiving additional scans two weeks into the treatment. The analysis focused on various FDG-PET parameters, such as standardized uptake values (SUVmax/peak/mean), metabolic tumor volume (MTV), total lesion glycolysis (TLG), and a Z-normalized combination of MTV and SUVpeak (ZMP). Univariate and bivariate Cox regression analysis were employed, alongside clinicopathological factors like tumor and lymph node stage, gross tumor volume (GTV), and human papillomavirus (HPV) status, to predict locoregional failure. The findings revealed that N3 lymph node stage, HPV-negative tumor status, GTV, MTV, TLG, and ZMP were significant predictors of locoregional failure (P < .01), while SUVmax/peak/mean did not exhibit significant predictive value (P > .2). In combination with N3 stage, ZMP, TLG, and MTV, HPV status emerged as the most independent predictor in bivariate analysis (P < .02). Moreover, FDG-PET parameters measured two weeks into radiotherapy showed a decrease of 30%-40% compared to the initial values; however, neither the absolute nor relative decrease in these parameters improved the prediction models for locoregional failure.
Jones et al 20 investigated 25 patients with non-metastatic ASCC in a multi-center prospective study. MP-MRIs, incorporating diffusion-weighted MRI (DW-MRI) and dynamic contrast-enhanced MRI (DCE-MRI) sequences, were conducted at baseline, during the second and fourth weeks of CRT, and 8 weeks post-treatment completion. Various histogram metrics were derived from the MP-MRI data, including maximum, mean, median, minimum, skewness, kurtosis, and standard deviation. The analysis revealed strong associations between certain apparent diffusion coefficient (ADC) metrics obtained from DW-MRI and local recurrence of ASCC, displaying excellent discriminatory capability. Notably, baseline skewness and standard deviation, as well as week 2 skewness and standard deviation, and week 4 kurtosis and standard deviation, were identified as prognostic indicators for local recurrence. Furthermore, changes in minimum ADC between baseline and week 2, and between baseline and week 4, were predictive of local recurrence. Regarding any recurrence, significant predictive factors included ADC minimum and standard deviation at baseline, as well as ADC maximum and standard deviation at week 4.
Progression-Free Survival
Brown et al 16 assessed novel imaging features extracted from pre-treatment FDG-PET/CT scans for predicting progression free survival (PFS) in ASCC patients. Radiomic feature extraction was performed using LIFEx software on baseline FDG-PET/CT images. Logistic regression models were generated using elastic net regularization and feature selection on a randomly selected training cohort, and the models were then applied to a validation cohort. Performance comparison of the models was conducted using ROC-AUC analysis, including a model with standard clinical prognostic factors (age, sex, tumor, and nodal stage—Model A), a radiomic feature model (Model B), and a combined radiomic/clinical model (Model C). The study comprised 189 patients, with 145 in the training cohort and 44 in the validation cohort. Median follow-up periods were 35.1 and 37.9 months, respectively, with 70.3% and 68.2% reaching the specified follow-up time with PFS. The prognostic model included the following radiomic features: GLCM entropy (a measure of randomness in the distribution of co-occurring pixel grey-levels), NGLDM busyness (a measure of spatial frequency of intensity changes between nearby voxels with different grey levels), minimum CT value (lowest Hounsfield Unit within the lesion), and SMTV (a standardized version of metabolic tumor volume), alongside tumor and nodal stage. The AUCs for Model A (clinical), Model B (radiomic), and Model C (radiomic/clinical) were .6355, .7403, and .7412 in the training cohort, and .6024, .6595, and .7381 in the validation cohort, respectively.
Hocquelet et al 18 aimed to investigate whether regular MRI findings and tumor texture features on pre-CRT imaging could serve as potential predictors of event-free survival (defined as disease progression or death) in patients with anal squamous cell carcinoma (ASCC) without metastasis. The study included 28 patients who underwent CRT for pathologically confirmed ASCC and had pre-CRT MRI images. Texture analysis was performed on axial T2-weighted images, using a 3D region of interest delineated around the entire tumor volume. First-order analysis quantified histogram-based features, and second-order statistical texture features were derived from the grey-level co-occurrence matrix at distances of 1 (d1), 2 (d2), and 5 (d5) pixels. Prognostic factors were evaluated using Cox regression, and model performance was assessed using the Harrell C-index.
Among the 28 patients, eight experienced tumor progression, leading to six tumor-specific deaths. After adjusting for age, gender, and tumor grade, two texture features showed associations with event occurrence: skewness (HR = .131, 95% CI = 0-.447, P = .005) and cluster shade_d1 (HR = .601, 95% CI = 0-.861, P = .027). The corresponding Harrell C-indices for these associations were .846 (95% CI = .697-.993) and .851 (95% CI = .708-.994), respectively.
Prediction of p16 (HPV) Status in Anal Cancer
Prediction of p16 (HPV) status was assessed by Leijenaar et al 21 Their paper focused on evaluating the performance of an existing CT-based radiomic signature, originally designed for oropharyngeal cancer, to predict HPV status in the context of anal cancer. Their dataset was made up of 59 patients from two different medical centers. The primary objective was to assess whether the radiomic signature could effectively predict HPV status based on p16 immunohistochemistry. After conducting predefined statistical tests, the researchers obtained an AUC value of .68 [95% CI (.32-1.00)] and an F1 score of .78 for the anal cancer dataset. The radiomic signature achieved a TRIPOD level 4 (57%) with an RQS of 61%. The study successfully demonstrates that this radiomic signature has the potential to identify the clinically relevant molecular phenotype, that is, HPV-ness, across multiple cancers, thereby serving as a promising CT imaging biomarker for p16 status.
Discussion
Factors such as male gender, N-positive stage, and tumor length > 5 cm are prognostic indicators in ASCC, correlating with less favorable clinical outcomes.25,26 Additional potential prognostic markers include: neutrophil-to-lymphocyte ratio for locoregional recurrence, age >55, skin ulceration, metastatic inguinal nodes, and a total RT dose >60 Gy.26-28 The creation of predictive nomograms has yielded promising outcomes in forecasting cancer-specific and overall survival rates, as well as aiding risk assessment; however, it is crucial to acknowledge that these nomograms have been exclusively grounded in clinical data.29-31 Notably, the promise of radiomics features extracted from medical imaging, introducing the potential for supplementary insightful data. The amalgamation of these novel parameters with pre-existing clinical factors appears to augment performance.16,17,22,23 This is precisely where the prowess of machine learning (ML) algorithms becomes evident, having already demonstrated remarkable accomplishments across various cancer categories.32-35
A proportion of ASCC patients undergoing curative CRT still require salvage abdominoperineal resection (APR) due to inadequate treatment response or local recurrence.36,37 In this context, the integration of radiomics and ML from outset imaging may play a significant role in stratifying patients who are at higher risk of requiring salvage APR. With radiomics and ML the potential to identify early indicators of treatment resistance or local recurrence, facilitating timely interventions and personalized treatment strategies.19,20,22,23 Consequently, such predictive models may not only enhance clinical decision-making, improving patient outcomes and refining therapeutic approaches in the management of anal cancer.
This review establishes the feasibility and efficacy of formulating radiomic signatures for the prediction of oncological outcomes in patients with anal cancer, based on pre/post-operative imaging data. Inclusion of various studies revealed that all developed signatures demonstrated at least modest accuracy (range: .68-1.0) in predicting their primary outcome, with one study reporting an exceptionally high AUC of 1.0. 24 However, these encouraging results are constrained by several limitations, predominantly arising from the substantial variability in imaging protocols and radiomic feature extraction methodologies.
We highlight that the use of radiomics (and radiogenomics) in the field of ASCC is still novel, but the potential is massive given the natural progression of this disease. Further refinement of radiomic methodologies will align assessment and reporting of predictive radiomics signatures.
Conclusion
The integration of radiomics into the management of anal cancer represents a potential adjunct into management, highlighting those whom have higher risk of treatment failure requiring salvage surgery. Standardized protocols, larger multicentre studies, and prospective validation are essential to underscore its role in clinical decision-making.
Supplemental Material
Supplemental Material - Radiomics; Contemporary Applications in the Management of Anal Cancer; A Systematic Review
Supplemental Material for Radiomics; Contemporary Applications in the Management of Anal Cancer; A Systematic Review by Hugo C. Temperley, Niall J. O’Sullivan, Caitlin Waters, Alison Corr, Brian J. Mehigan, Grainne O’Kane, Paul McCormick, Charles Gillham, Emanuele Rausa, John O. Larkin, James F. Meaney, Ian Brennan, and Michael E. Kelly in The American Surgeon.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by Joly Cancer Leadership Programme, Trinity St James Cancer Institute.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
