Abstract

A 23-year-old male was involved in a motor vehicle accident as the restrained driver with airbag deployment. He presented as a level 1 trauma alert due to the severity of the accident. He was normotensive on arrival but complained of diffuse abdominal, chest, and head pain. FAST was performed and was positive for fluid in the abdomen. Since the patient maintained his blood pressure in the normotensive range, he was taken to the CT scanner where he was found to have a large hematoma in the suprahepatic region of the IVC (Figure 1). He was also found to have pulmonary and mesentery contusions. Due to the concern for IVC injury, the patient was taken to the OR for an exploratory laparotomy. Minimal free fluid was found in the abdomen; however, there was a large hematoma above the liver contained by the coronary ligaments. This was consistent with a zone I hematoma. Locations for total vascular occlusion of the liver. Image shown is after IVC repair.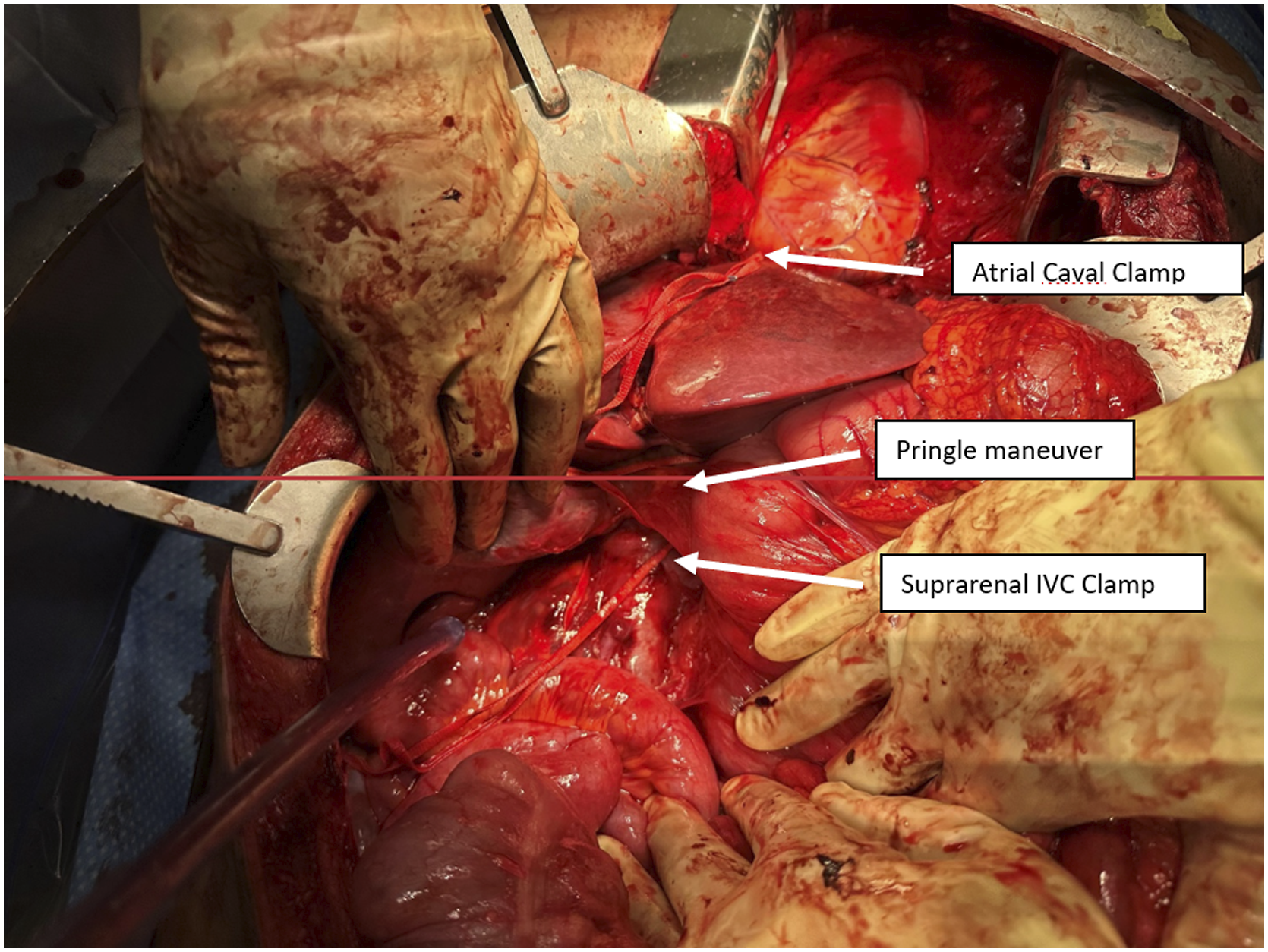
In preparation for total isolation of the liver, the patient was given a 1:1:1 ratio of packed red blood cells (PRBC), fresh frozen plasma (FFP), and platelets (PLT) of 6 units each to account for the decrease in preload. Total isolation of the hepatic inflow and outflow was obtained in the following manner (Figure 1): A Pringle maneuver was performed to control both portal venous and hepatic arterial inflow to the liver. The infrahepatic suprarenal IVC was exposed through an extended Cattell-Braasch and a Kocher maneuver. This included mobilizing the midgut retroperitoneal attachments. Once the suprarenal IVC was exposed circumferentially, it was occluded with both Rummel tourniquet technique and vascular clamps. A sternotomy was then performed to access the atrial caval IVC. The pericardial sac was entered and a vascular clamp was placed on the atrial caval IVC as it entered into the right atrium.
After total hepatic isolation was achieved, the suprahepatic hematoma was entered. After removal of the blood, a grade V liver injury was identified with complete transection of the suprahepatic IVC. The injury was repaired with 2-0 Prolene suture, taking bites of both IVC and diaphragm circumferentially for additional strength. The left hepatic vein was traumatically macerated and was not salvageable; thus, it was ligated. Great care was taken to reimplant the middle and right hepatic veins without occluding their orifices. Before the conclusion of the anastomosis, both the infrahepatic IVC and Pringle clamps were relaxed to allow for venous back bleeding to remove any potential air in the portal venous system. Venous flow was restored without evidence of bleeding from the suture line of the IVC repair. Before releasing the atrial caval clamp, the patient was given 1 mg of calcium chloride as well as 4 amps of bicarbonate to anticipate the lactic acid back wash. Time from incision to repair of the suprahepatic IVC was 1 hour and 56 minutes, with 56 minutes of total clamp time of the liver.
Due to the damage control nature of the operation and the possibility of abdominal compartment syndrome, the patient was left open and a negative pressure vacuum dressing was placed in both the abdomen and the chest cavities. On POD #1, the patient returned to the OR with no significant bleeding and was closed without complications. A mediastinal chest tube was left in place, and a right upper quadrant JP drain was also placed. Repeat CT imaging on POD #3 demonstrated an intact inferior vena cava with no thrombus or bleeding (Figure 2). There was no evidence of acidosis, renal or significant hepatic dysfunction. A liver duplex exam was performed to assess the portohepatic circulation on POD #5. There were no signs of inflow obstruction with hepatopetal flow of the portal vein and no outflow obstruction of the hepatic veins. The patient developed an acalculous cholecystitis confirmed by PIPIDA scan and was subsequently treated with US guided drainage. He was discharged on post-operative day 12 with normal liver function tests and a cholecystostomy tube. CT imaging demonstrating appearance of the IVC before and after repair. (A),(B) and (C) demonstrate coronal, sagittal and axial CT imaging, respectively, of the patient demonstrating hematoma around the IVC pre-operatively. (D), (E) and (F) demonstrate the same imaging post-operatively demonstrating restoration of flow through the IVC. 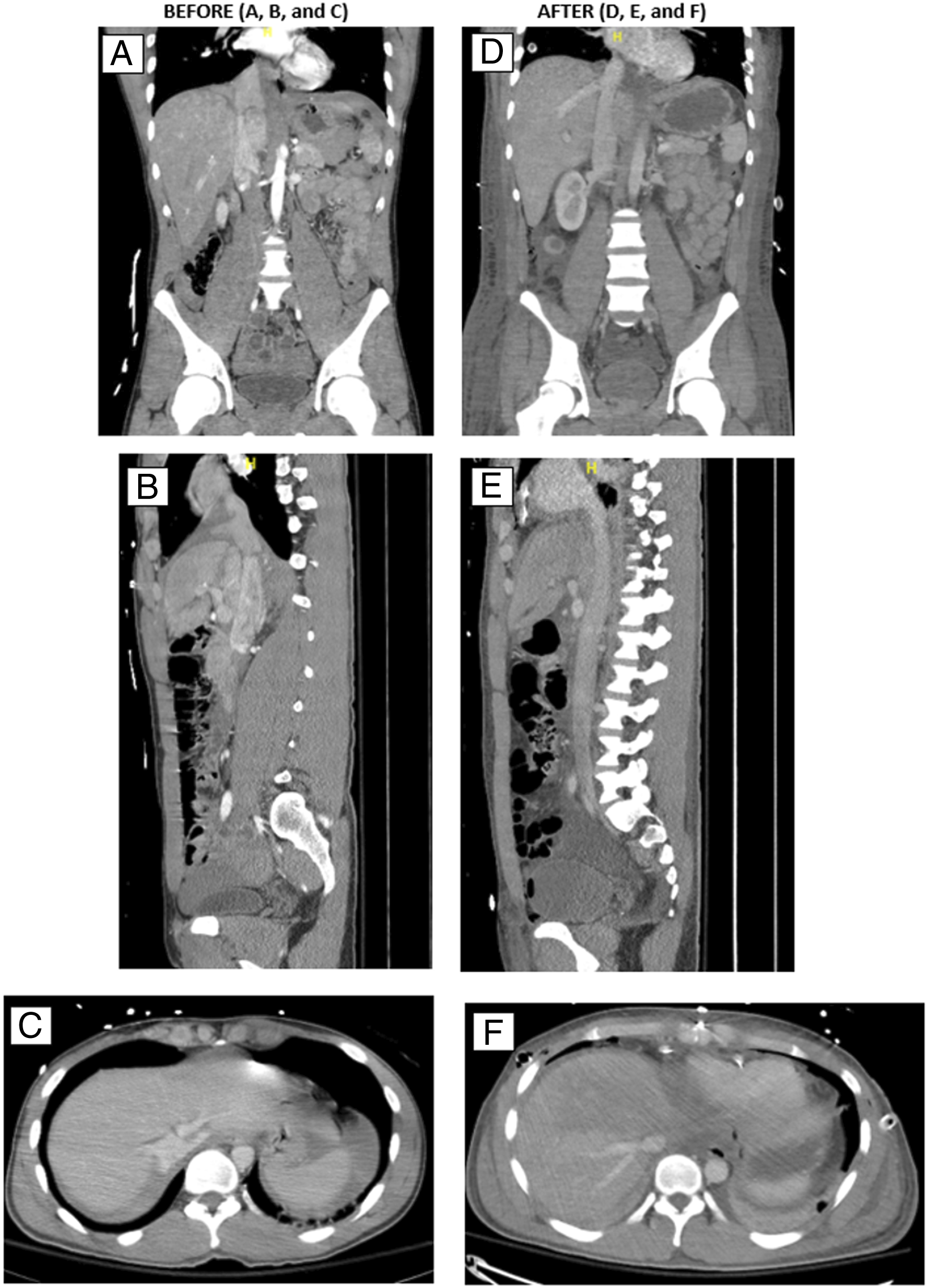
The inferior vena cava (IVC) is well protected by many visceral organs, and as a result, injuries to this structure are rare. However, when injuries do occur to any location of the IVC, they are associated with a high mortality of approximately 40-80%. 1,2 More than 30% of patients who survive to be evaluated at a trauma center will eventually die within 24 hours and 30-50% will die in the field before arrival. While the exact mechanism is not known, suprahepatic IVC injuries are likely a result of a direct blow to the space between the liver and heart at the level of the xiphoid. This would push the liver caudally and lead to an anterior to posterior tear of the suprahepatic IVC.
Total transections of the suprahepatic IVC are exceedingly rare and patients are unlikely to survive. There are only 5 reported cases. Of these, only 2 survived.3,4 Our case represents the first time that this injury has been repaired without bypass or shunt (Schrock) with full recovery. This method takes less time than other reported techniques and has a low risk for air embolus, and it is well tolerated by the patient with shorter ICU and hospital length of stay. Although the patient’s age was advantageous to his recovery, we believe that this technique would still work for an older patient as long as they are given adequate preload and have good functional reserve otherwise.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported (in whole or in part) by HCA Healthcare and/or an HCA Healthcare affiliated entity. The views expressed in this publication represent those of the author(s) and do not necessarily represent the official views of HCA Healthcare or any of its affiliated entities.
