Abstract
Background
Although randomized controlled trials on neoadjuvant chemotherapy for gastric cancer have included some T1-staged tumors, overall survival (OS) has not been analyzed for this subset. Due to the low negative predictive value of clinical staging and the benefits of neoadjuvant chemotherapy for locally advanced disease, identifying patient groups with early-stage gastric cancer that may benefit from neoadjuvant chemotherapy is of merit.
Aims
The objective of this study was to evaluate the relationship between OS and sequence of surgical therapy for clinical T1 gastric cancer.
Methods
The 2017 National Cancer Database was used to compare patients who had surgery-first and those who received neoadjuvant chemotherapy for T1-stage gastric cancer. OS was analyzed using a parametric regression survival-time model adjusted for covariates. The effects of these covariates on OS based on surgical sequence were examined.
Results
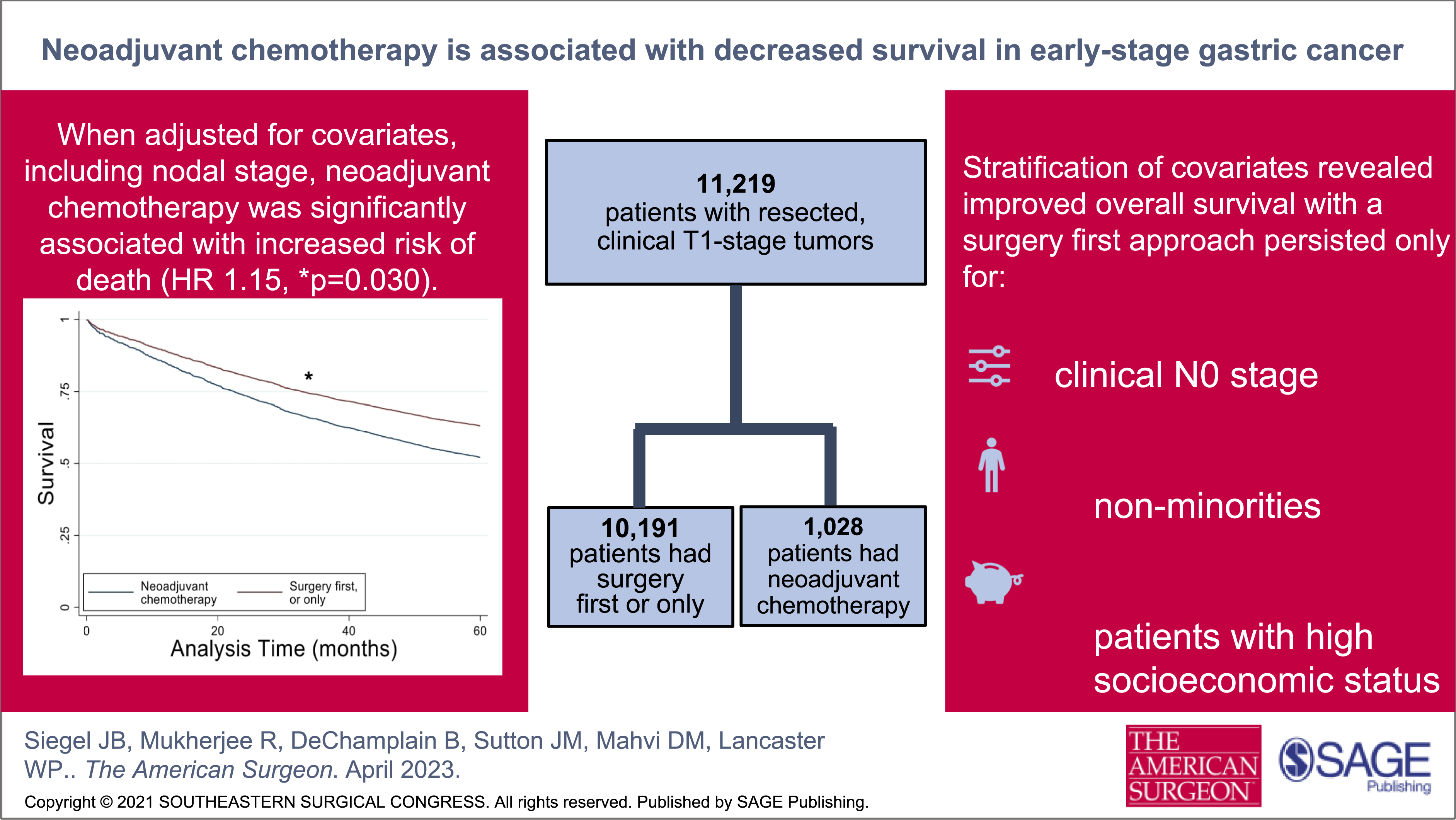
11,219 patients were included, of which 10,191 underwent surgery as their first or only treatment. When adjusted for covariates, neoadjuvant chemotherapy followed by curative-intent surgery was significantly associated with increased risk of death (HR 1.15, 95% CI 1.01-1.31, P = .030). In multivariate analysis, clinical N0 stage, non-minorities, and patients with high socioeconomic status had improved OS if they did not have neoadjuvant chemotherapy and instead had upfront surgery.
Conclusion
Neoadjuvant chemotherapy is associated with decreased OS for early-stage gastric adenocarcinoma, even for patients with clinically positive nodal disease. In addition, the lack of survival improvement with a surgery-first approach in patients with disparities deserves further study.
Key Takeaways
• Neoadjuvant chemotherapy is associated with decreased overall survival for T1 gastric cancer, even in patients with nodal disease. • A surgery-first approach for T1 gastric cancer is associated with improved survival. • There is no survival difference by therapeutic approach, surgery-first vs neoadjuvant chemotherapy, for patients with health care disparities.
Introduction
Gastric cancer is the third most common cause of cancer-related deaths worldwide. 1 Its treatment is complex, often involving multimodal regimens that can include surgery, chemotherapy, radiotherapy, and novel targeted therapies. 2 Current multidisciplinary treatment strategies are determined largely by the de novo clinical stage of the tumor; however, patient factors and multidisciplinary discussion are important modifiers. 3
The landmark Medical Research Council Adjuvant Gastric Infusional Chemotherapy (MAGIC) trial reported that patients with resectable gastric cancer ≥ Stage II treated with perioperative chemotherapy had significantly improved overall and progression free survival compared to patients treated with surgery alone, and the group that had perioperative chemotherapy had significantly smaller and less advanced tumors at resection. 4 Additional randomized controlled trials have also shown similar effects with perioperative chemotherapy; a Cochrane meta-analysis of 14 randomized controlled trials also showed relative increased survival of 19% for patients that received preoperative chemotherapy. 5 In response to these trials, current guidelines published by the National Comprehensive Cancer Network (NCCN) recommend upfront surgery for patients with T1 tumors (invasion limited to the mucosa or submucosa) without clinical nodal involvement, while patients with T2 tumors (invasion past the muscularis propria) and/or clinical nodal involvement be referred for neoadjuvant chemotherapy. 6 However, there has been little investigation into the utilization of neoadjuvant chemotherapy in early-stage gastric tumors. Most studies on neoadjuvant chemotherapy for gastric adenocarcinoma either entirely excluded or had few patients with T1 disease.3,7,8 The rationale for neoadjuvant chemotherapy for this population is reduction of tumor size, improved resection rates, inhibition of micro-metastases, and reduced tumor recurrence. 9 As tumors are occasionally upstaged on pathological diagnosis compared to their de novo clinical stage, neoadjuvant chemotherapy may be a reasonable strategy for certain groups of patients. 10 In the Cochrane meta-analysis by Ronellenfitsch et al, they found that there was an interaction with preoperative chemotherapy and age, and younger patients had a greater treatment effect. 5 Population-based cancer registries have also reported frequent omission of adjuvant therapy, and neoadjuvant chemotherapy would prevent patients from missing chemotherapy. 11 Gastric cancer is also being diagnosed in younger patients more frequently, which may be a subset that benefits from more aggressive therapy. 12 However, neoadjuvant may delay surgical resection, subject patients to side effects, and worsen malnutrition prior to curative resection.13,14
The objective of the current study was to evaluate patient survival in those with early-stage gastric cancer treated with neoadjuvant chemotherapy. The secondary objective was to determine if tumor factors, such as nodal status as well as patient factors, influence the survival outcomes in this cohort.
Methods
We performed a retrospective cohort analysis on patients who underwent curative-intent surgery for clinical stage T1 gastric adenocarcinoma to determine the association between overall survival (OS) and the sequence of surgical therapy in relation to chemotherapy. OS was compared between patients who received neoadjuvant chemotherapy vs patients who underwent surgery as their first or only treatment modality. The sequence of therapy was stratified to evaluate the effect covariates had on the treatment algorithm, and each covariate was stratified to determine the effect sequence of therapy had for specific patient and tumor factors.
Data Source and Variables
The National Cancer Database (NCDB) 2017 public use file (PUF) was queried for all data. The NCDB is estimated to include 70% of all cancer diagnoses in the US and Puerto Rico. 15 Data are de-identified and follow the privacy requirements of the Health Information Portability and Accountability Act. The Institutional Review Board at the Medical University of South Carolina determined this study exempt.
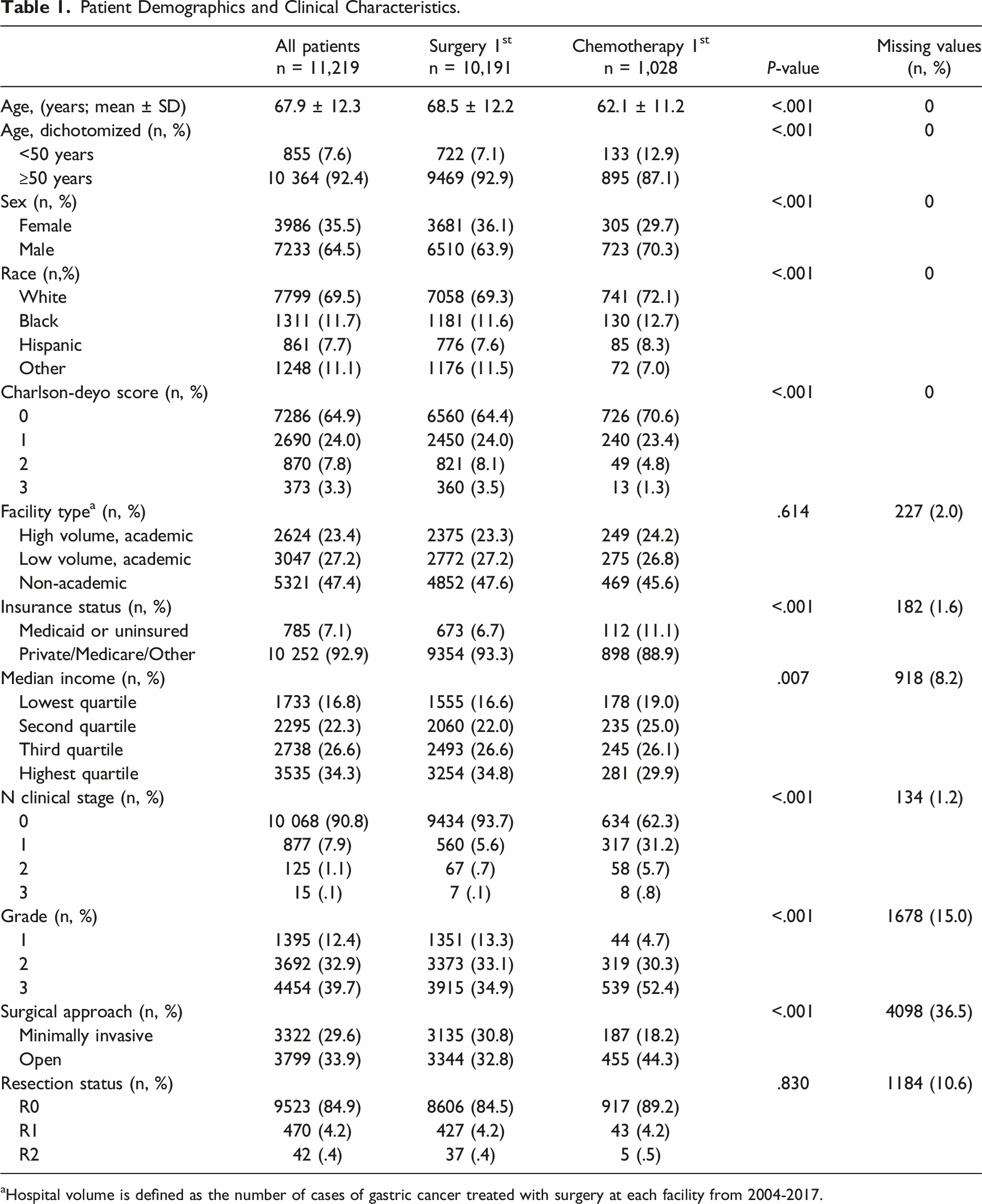
Patients were selected for the study if they had curative intent surgery and had clinical T1 stage disease. Patient- and tumor-level variables analyzed included age, sex, race, Charlson-Deyo comorbidity score, median income, insurance type, facility type, clinical T stage, clinical N stage, tumor grade, surgical approach (ie, minimally invasive vs open), resection status, and surgical therapy sequence (chemotherapy-first vs surgery-first). For analysis, age was dichotomized to <50 years and ≥50 years. Race and ethnicity were divided into four groups classified by the Commission on Cancer (CoC) as white, black, Hispanic, and other. The modified Charlson-Deyo comorbidity score as defined by the CoC was dichotomized to zero and ≥1. Median income was divided into quartiles based on patient zip code, and insurance type was dichotomized into Medicaid/uninsured vs all other insurance types to best group patients by socioeconomic status. Facility type was defined by a variable that incorporated both academic and volume status, which has been validated in previous NCDB analyses.15,16 Clinical stage was chosen for analysis instead of pathological stage as initial treatment algorithms are based off clinical staging. Adjustment for T1a, T1b, and T1c was not performed as most patients’ tumors were identified only as T1. Resection status was divided into three groups (R0 [no residual primary tumor], R1 [residual microscopic tumor], and R2 [residual macroscopic tumor]).
Population
The NCDB PUF was queried for all patients who underwent curative-intent surgery for clinical T1 stage gastric cancer between 2004-2017. Patients were excluded if they did not have adenocarcinoma histology, and if they had metastatic disease, in situ cancer only, or had missing data for clinical T stage, follow-up, survival, and/or surgery sequence in relation to chemotherapy.
Outcomes and Analysis
The primary outcome was OS in relation to treatment algorithm: surgery-first or chemotherapy-first. Survival was based on months from initial diagnosis to last contact or death up to 60 months. A parametric regression survival-time model was used to estimate survival time to compare survival between patients that had chemotherapy prior to surgery with patients that had surgery as their first or only treatment modality. We adjusted for all covariates, and since each observation in this dataset represented values from a single patient, standard errors were determined by an equation-level examination of the covariance matrix (Huber-White method). All covariates were included except surgical approach due to data missing for >30% of the subjects. The effects of each covariate on the relationship between therapy sequence and OS was examined individually for each level of the covariate with all other covariates remaining in the model. When required, corrections for multiple comparisons were performed using Sidak’s formula. Statistical analyses were performed using STATA statistical software package (version 17.0), and to plot the OS curves. A P-value <.05 was considered statistically significant.
Results
A total of 11219 patients met inclusion criteria for analysis (Figure 1); 10191 patients had surgery as their first or only treatment modality, while 1028 patients were treated with neoadjuvant chemotherapy prior to curative resection (Table 1). The mean age was 67.9 years. Significantly more men received neoadjuvant chemotherapy. Almost half of the patients were treated at non-academic centers, and most patients had private insurance. Most patients were clinically node negative, but significantly more patients who received neoadjuvant chemotherapy had clinically positive nodes. Patients who received neoadjuvant chemotherapy had significantly higher tumor grades and were less likely to have a minimally invasive resection. Most patients had an R0 resection, and resection status was not significantly different between the cohorts (Table 1). In the neoadjuvant chemotherapy group, there were 114 patients (10.1%) who were known to have a complete response with chemotherapy; 192 patients (17.0%) had unknown pathologic staging. Consort diagram depicting inclusion and exclusion criteria for our patient population. Patient Demographics and Clinical Characteristics. aHospital volume is defined as the number of cases of gastric cancer treated with surgery at each facility from 2004-2017.
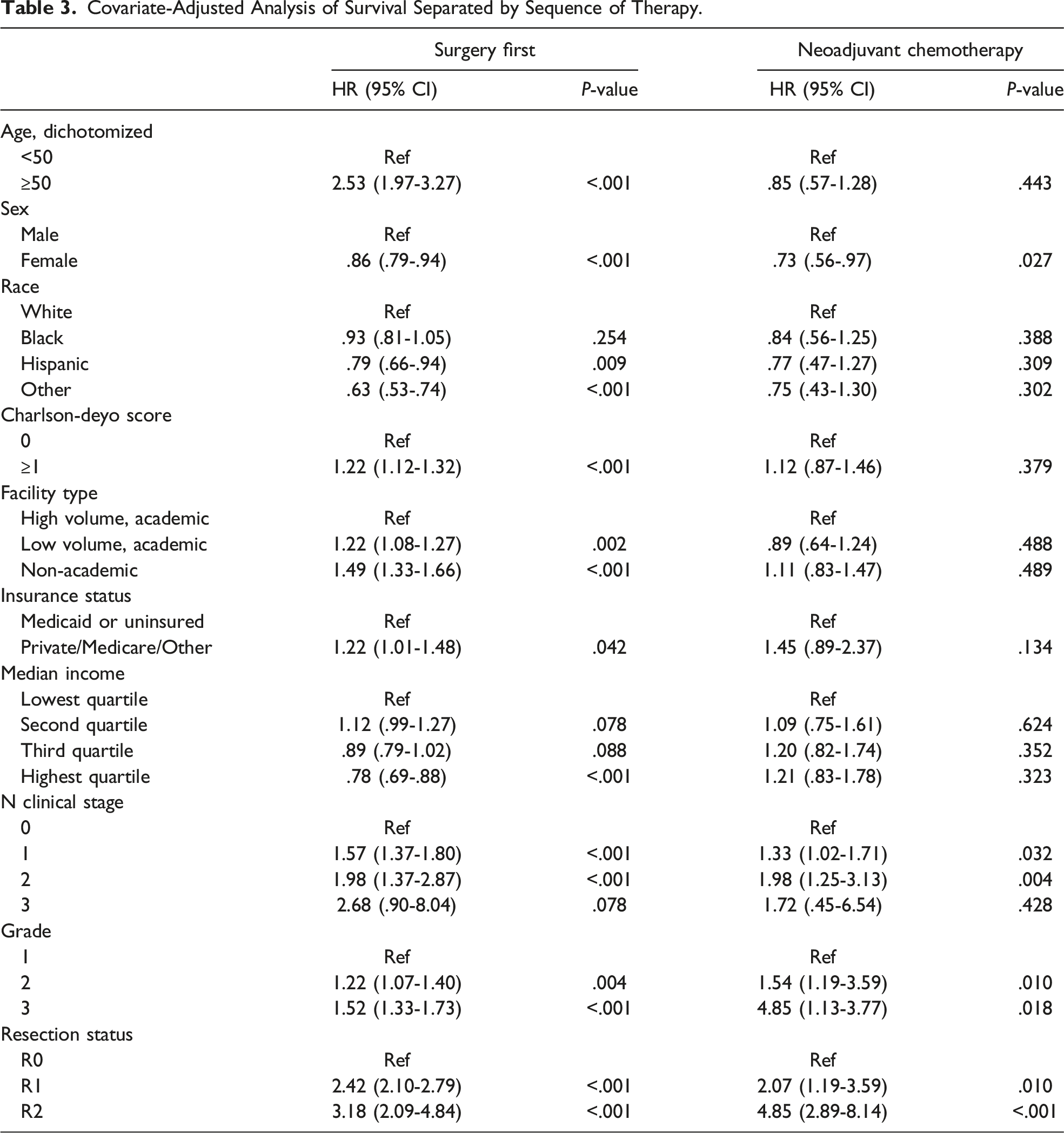
Parametric regression analysis adjusting for all covariates compared OS for patients who underwent surgery-first vs those who received neoadjuvant chemotherapy (Figure 2.). Receipt of neoadjuvant chemotherapy prior to surgical resection of a clinical T1 tumor was associated with decreased OS (HR 1.15, 95% CI 1.01-1.31, P = .030). Age ≥50, a Charlson-Deyo score ≥1, private insurance, N clinical stage ≥1, higher tumor grade, and residual tumor were all associated with significantly worse OS (Table 2). Female sex, non-white and non-black race, treatment at a high-volume academic center, and having a higher median income were associated with significantly improved OS. Parametric regression survival-time model estimating survival time following a Weibull distribution. Overall survival was significantly improved with a surgery first or only approach compared to neoadjuvant chemotherapy for patients with T1 gastric cancer adjusting for all covariates (*P = .030). Covariate-adjusted overall survival.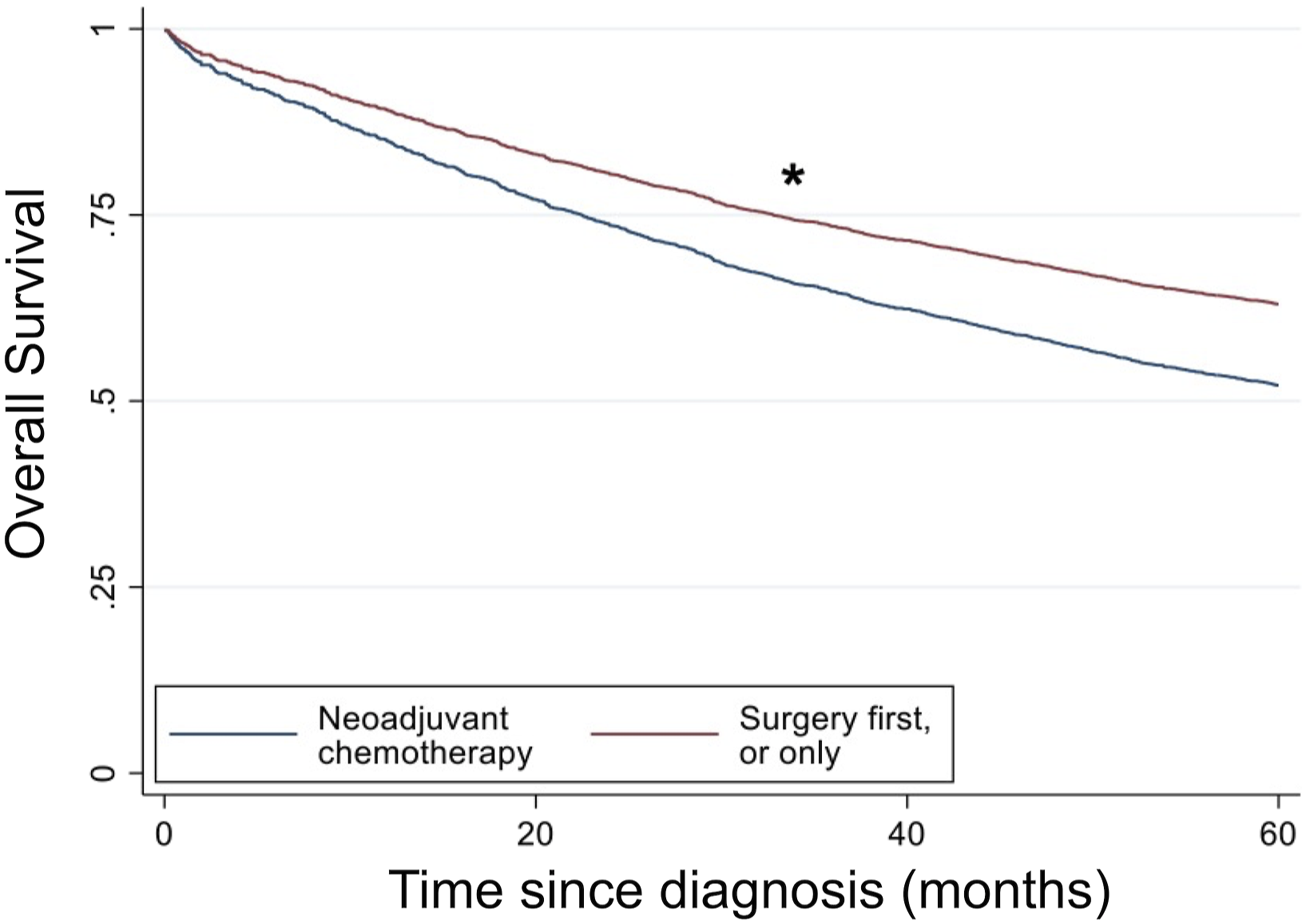

Covariate-Adjusted Analysis of Survival Separated by Sequence of Therapy.
Covariate-Adjusted Hazard Ratios (HR) for Sequence of Therapy for Level of Each Covariate.
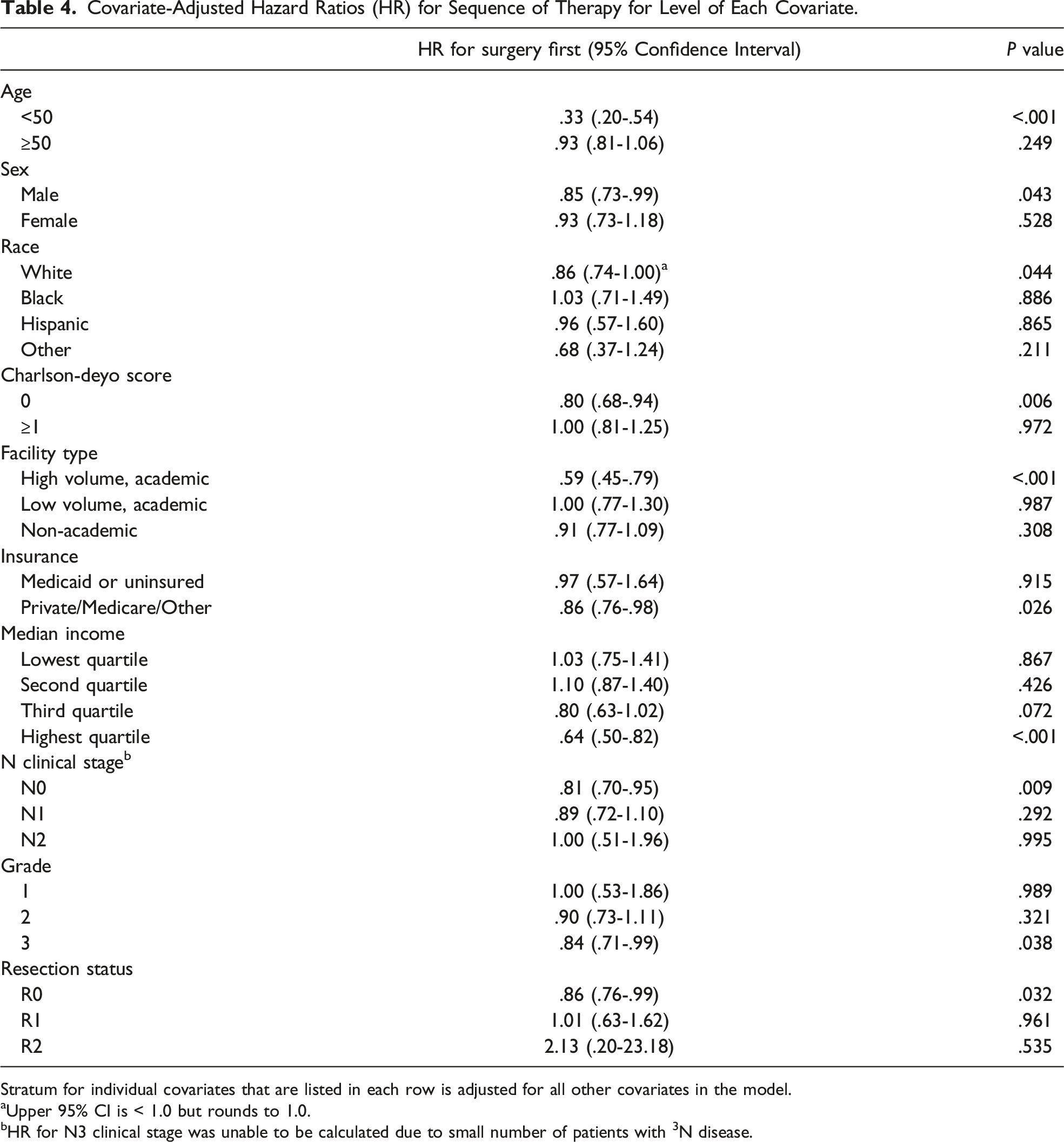
Stratum for individual covariates that are listed in each row is adjusted for all other covariates in the model.
aUpper 95% CI is < 1.0 but rounds to 1.0.
bHR for N3 clinical stage was unable to be calculated due to small number of patients with 3N disease.
Discussion
For clinical stage II or greater gastric cancer, neoadjuvant chemotherapy has been adopted as the standard of care due to improved survival seen in randomized controlled trials. However, there is a paucity of data on the impact neoadjuvant chemotherapy has on T1 gastric adenocarcinoma. In this cohort analysis, we found that neoadjuvant chemotherapy is associated with significantly decreased OS for clinical T1-stage gastric cancer. Stratification by sequence of therapy demonstrated that some patient and tumor factors influenced survival outcomes differently depending on the treatment algorithm. Further stratification of all covariates demonstrated that not all patients benefit from the improved survival with a surgery-first approach, and specific patient factors are subject to health care disparities.
Multiple randomized controlled trials have shown a significant survival advantage for a neoadjuvant chemotherapy approach in treatment of stage II gastric cancer, including patients with a T1 tumor with concurrent nodal involvement.5,17 One of the pitfalls of clinical staging is the relatively low negative predictive value, which results in node-positive patients failing to be identified as such. 18 Some patients will be incorrectly staged as stage I and miss the benefits of neoadjuvant chemotherapy. In addition, a 40%–60% of patients fail to complete the entire regimen. 11
We found that neoadjuvant chemotherapy for T1 tumors is associated with significantly decreased OS when adjusting for all covariates, including nodal stage. Our study suggests that patients with a T1 tumor--even those with nodal involvement--may not derive the same benefits from neoadjuvant chemotherapy as those with more advanced tumors. Although patients with clinical T1 tumors and clinically-involved nodes have previously shown improved survival with neoadjuvant chemotherapy in the MAGIC trial, this subgroup represented only 11.8% of the study group. 4 This subgroup was not individually analyzed and was pathologically staged as T1 and may have been downstaged if they received neoadjuvant chemotherapy. Although our surgery-first cohort included patients who would receive adjuvant chemotherapy, unlike in the MAGIC trial, almost 90% of the surgery-first group did not receive adjuvant chemotherapy, suggesting that our differing results were likely not due to receipt of adjuvant chemotherapy alone.
One rationale for administering neoadjuvant chemotherapy is to increase the likelihood of an R0 resection. Previous randomized controlled trials have shown that neoadjuvant chemotherapy significantly decreases the size of the tumor, and some patients can have a complete response with no tumor found on final pathologic staging.4,5 Although the neoadjuvant chemotherapy cohort did have significantly higher proportions of patients with more advanced disease by clinical node staging and grade, it is notable that there was not a significant difference in the resection status between the two cohorts. This suggests that for a clinical T1 tumor, preoperative chemotherapy may not be the main contributing factor to a margin-negative resection, and for a T1 tumor, chemotherapy may not provide much additional benefit. In congruence with this, a retrospective study found that patients with T1 tumors with up to N2 stage and size >30 mm had an excellent prognosis and surgery alone was sufficient therapy. 19 It may be that some patients with T1-stage cancer and clinically-involved nodes may avoid the toxicity of neoadjuvant chemotherapy.
Stratification by treatment algorithm showed age <50, Charlson-Deyo Score of 0, and surgery at a high-volume academic facility were significant predictors of improved OS in patients that had surgery-first or only. In contrast, for patients receiving neoadjuvant chemotherapy, we found OS was not impacted by these factors. It remains a possibility that surgical treatment is a modality that is more heavily influenced by the baseline health of patients compared to neoadjuvant chemotherapy. The literature has also shown that survival is improved for patients undergoing surgery if the surgery is performed at high volume academic centers. 20 Treatment at a high-volume academic center was not a predictor of improved OS in patients that received neoadjuvant chemotherapy. A possible reason may be receipt of neoadjuvant chemotherapy mitigates possible deleterious effects of treatment at a lower volume center.
When all covariates in the model were stratified, we found that some subsets of patients did not benefit from a surgery-first approach, nor did they experience any improved survival with neoadjuvant chemotherapy. Clinical N0 patients benefited from surgery first, in agreement with current NCCN guidelines, but survival in patients with clinical nodal disease was not affected by the treatment algorithm. This suggests that clinicians should consider a surgery-first approach for T1 tumors with N1 or N2 disease.
The stratification of all covariates also identified that disparities exist among patients of lower socioeconomic status and those in vulnerable population groups. These patients did not benefit from the surgery-first approach nor from neoadjuvant chemotherapy and these cohorts may be more likely to experience delays in surgical treatment. One previous study by Schultz et al. 21 Concluded that patients who were black, had Medicaid or no insurance, had shorter travel distance to the hospital, and were treated at a non-academic center were more likely to refuse resection. A population that is more likely to refuse resection would be more likely to have delays in treatment if they ultimately undergo surgery. These groups may also be prone to experience delays in treatment of surgical complications. Additional study is needed to determine what resources may be lacking for these patients.
A limitation of the present study is that the NCDB has a large amount of missing data for variables that may be confounders. Tumor location was not reported for this cohort, and surgical technique was missing for >30% of the cohort. The NCDB also does not provide information on specific comorbidities or medical events (e.g. recent myocardial infarction requiring dual anti-platelet therapy), which may have influenced the sequence of therapy and influenced the neoadjuvant chemotherapy group to have worse survival at baseline. Since patients in the neoadjuvant chemotherapy group had more advanced disease, it is likely that they underwent surgical resection rather than the a less morbid endoscopic resection of their gastric cancer. We also were not able to adjust for subclassification because the majority of patient were listed as clinical T1 stage only. This is likely another confounder since patients with T1c disease may have been more likely to have neoadjuvant chemotherapy. Furthermore, it is unclear whether the clinical staging in the NCDB was done prior to neoadjuvant chemotherapy or whether it was done after neoadjuvant chemotherapy, and if it was done after, the patients in the neoadjuvant chemotherapy group may have had more advanced disease than the surgery first group. As this is a retrospective study, further investigation is needed prior to altering therapeutic recommendations for patients.
Conclusion
We investigated the impact of neoadjuvant chemotherapy in patients with T1 gastric adenocarcinoma. Neoadjuvant chemotherapy was associated with decreased OS for these patients. Sub-group analysis demonstrated patients of higher socioeconomic status or with greater access to care had improved OS with a surgery-first approach, and in contrast, patients of lower socioeconomic status and those with more access to care issues did not see any differences in OS whether they had surgery or chemotherapy first. We speculated that delays in care and thus the selection of a particularly indolent cancer phenotype may be responsible for the inferior outcome in patients with disparities. Our data support other retrospective reviews and suggests neoadjuvant chemotherapy may not confer additional survival benefit for T1 tumors even with clinical nodal disease. We propose this should be investigated in a prospective manner to potentially spare patients the side effects of systemic chemotherapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclaimer
The NCDB is a joint project of the Commission on Cancer of the American College of Surgeons and the American Cancer Society. The data used in the study are derived from a de-identified NCDB file. The American College of Surgeons and the Commission on Cancer have not verified and are not responsible for the analytic or statistical methodology employed, or the conclusions drawn from these data by the investigator.
Data Availability Statement
All the data used in this study was accessed from the National Cancer Database 2017 Public Use File, which is available through application from the American College of Surgeons.
