Abstract
Background
Acute kidney injury (AKI) after endovascular aortic aneurysm repair (EVAR) is uncommon though carries significant morbidity. Procedural risk factors are not well established for acute renal failure (ARF) that requires initiation of dialysis. The goal of this study was to examine the impact of ARF on patients undergoing EVAR and identify risk factors for ARF using a large, national dataset.
Methods
Patients undergoing EVAR were identified from the National Surgical Quality Improvement Program (NSQIP) database over 9 years, ending in 2019. Demographics, indication for repair, comorbidities, procedural details, complications, hospital and ICU LOS, and mortality were recorded. Patients were stratified by presence of ARF and compared. Patients were further stratified by indication for EVAR and presence of ARF. Multivariable logistic regression (MLR) analysis was performed to determine the independent predictors of ARF.
Results
18 347 patients were identified. Of these 234 (1.3%) developed ARF requiring dialysis. Mortality (40 vs 1.8%, P < .0001), ICU LOS (5 vs 0 days, P < .0001), and hospital LOS (11 vs 2 days, P < .0001) were all significantly increased in patients with ARF. Multivariable logistic regression identified increasing diameter, creatinine, operative time, preoperative transfusions, ASA class, emergent repair, female gender, and juxtarenal/suprarenal proximal landing zone as predictors of ARF.
Conclusions
ARF after EVAR causes significant morbidity, prolongs hospitalizations, and increases mortality rates. Those patients at risk of ARF after EVAR should be closely monitored to reduce both morbidity and mortality.
• Of 18 467 patients undergoing EVAR for abdominal aortic aneurysms, 234 (1.3%) developed acute renal failure requiring dialysis (ARF). • ARF led to significant increases in mortality (40% vs 1.8%), longer ICU stays (5 vs 0 days), and prolonged hospital stays (11 vs 2 days).Key Takeaways
Introduction
Abdominal aortic aneurysms (AAAs) are associated with significant morbidity and mortality, particularly untreated. 1 The advent of endovascular aortic aneurysm repair (EVAR) has led to significant reductions in the morbidity associated with AAA repair and improvements in short-term mortality. 1 One devastating complication associated with AAA repair is acute kidney injury (AKI).2,3 EVAR has led to a significant reduction in rates of AKI compared to open repair, though AKI remains a significant cause of morbidity in this patient population.4,5
Unfortunately, risk factors for AKI after EVAR are not often agreed upon, and most studies exclude patients undergoing EVAR for rupture.6-8 In addition, there are variable definitions of AKI after EVAR, ranging from small increases in serum creatinine to decreased postoperative urine output, and change in glomerular filtration rates.8-13 While some of these definitions are based on established criteria such as the RIFLE, AKIN, or KDIGO staging systems, there is still variability in how this affects clinical outcomes.2,10,12,13 Previous studies have shown decreased renal function to negatively impact both short- and long-term survival after EVAR.11,12,14 In fact, Zarkowsky’s 2016 study found that of 56 patients requiring postprocedural dialysis less than twenty-five percent had three-year survival after elective endovascular aneurysm repair, which may mitigate the mortality benefit associated with elective endovascular AAA repair. 7 Therefore, the goal of this study was to determine clinical risk factors for acute renal failure (ARF) that required either temporary or permanent postoperative dialysis after EVAR both for elective aneurysm repair and emergent repair of ruptured aneurysms.
Methods
Identification of Patients
The National Surgical Quality Improvement Program (NSQIP) database was searched for patients undergoing EVAR by procedure codes from 2011 to 2019, using the vascular targeted procedure files. All patients within the EVAR-specific dataset, including infrarenal, juxtarenal, suprarenal, and type IV abdominal aneurysms, were included. Patient demographics (age, gender, and race), comorbidities, indication for repair (size, rupture, symptomatic aneurysm, and dissection), proximal and distal aneurysm extent, postoperative complications (ARF requiring dialysis, pneumonia, prolonged mechanical ventilation, pulmonary embolism, deep vein thrombosis, and sepsis), LOS, and mortality were extracted and recorded. Patients were then stratified based on presence of ARF (patients who required new initiation of temporary or permanent dialysis after EVAR) and compared. This study was exempt from review by the University of Tennessee Health Science Center’s institutional review board given the deidentified nature of the dataset.
Comparisons
Using SAS version 9.4 (SAS Institute, Cary, NC), data was compared. Chi-square analysis was utilized for categorical variables and Wilcoxon-Rank sum test for continuous variables. Multivariable logistic regression (MLR) was used to identify independent predictors of ARF after EVAR. A P-value <.2 was used for inclusion in the MLR model. Using backwards stepwise elimination, a model was constructed to identify predictors of ARF. P < .05 was considered significant. The area under the receiver operating curve (AUROC) was reported with 95% confidence intervals for the final model.
Those undergoing EVAR for rupture and for size of aneurysm were then considered separately. Both patient groups were compared based on the presence of ARF. Unique MLR models were created to identify the predictors of ARF for these two patient populations.
Results
Patient Characteristics
Comparison of Baseline Characteristics and Adverse Outcomes Amongst All EVAR, EVAR With ARF, and EVAR Without ARF.
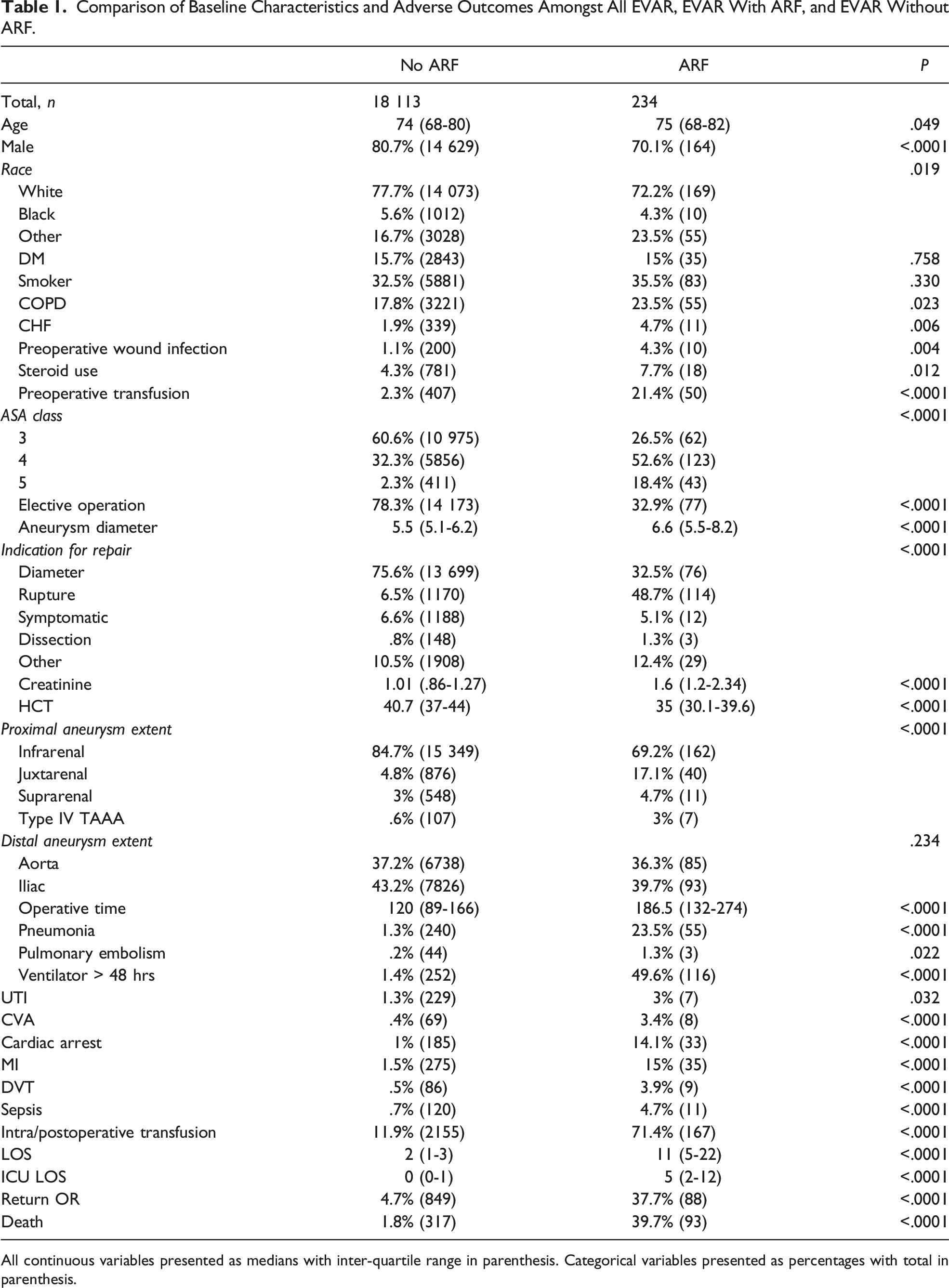
All continuous variables presented as medians with inter-quartile range in parenthesis. Categorical variables presented as percentages with total in parenthesis.
Comparison—ARF—All EVAR Patients
Endovascular aortic aneurysm repair patients were then compared based on the development of postoperative ARF. A total of 234 patients developed ARF (1.3%) (Table 1). Acute renal failure was more common in females (30% vs 19%, P < .0001), older patients (75 vs 74 years old, P = .049), patients with CHF (4.7% vs 1.9%, P = .006), COPD (23.5% vs 17.8%, P = .023), those with ongoing steroid use (7.7% vs 4.3%, P = .012), and those with ongoing wound infections (4.3% vs 1.1%, P = .04). Those requiring preoperative transfusions, patients with higher ASA, and patients undergoing EVAR for rupture were also at an increased risk of ARF (P < .0001). Patients with suprarenal and juxtarenal aneurysms had higher incidence of ARF (P < .0001). Those that developed ARF had significantly higher rates of all complications including a 10-fold increase in MI and stroke, and half of ARF patients requiring mechanical ventilation for greater than 48 hours. While only 4.7% of ARF patients developed postoperative sepsis, the mortality rate was nearly 40% for these patients.
Comparison—ARF—EVAR for Aneurysm Diameter
Acute Renal Failure in Patients Undergoing EVAR for Aneurysm Size and for Rupture.
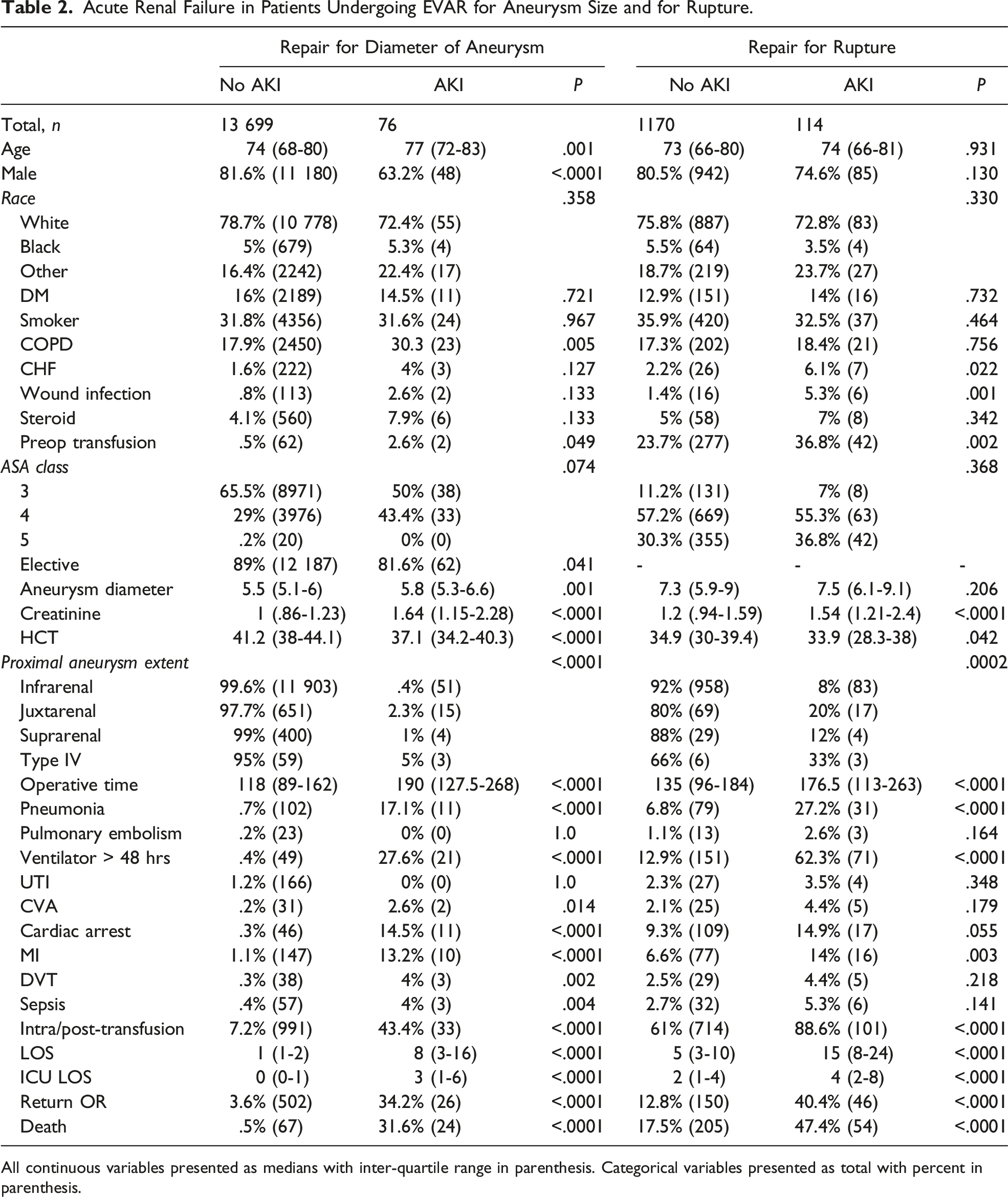
All continuous variables presented as medians with inter-quartile range in parenthesis. Categorical variables presented as total with percent in parenthesis.
Comparison—ARF—EVAR for Rupture
For those patients undergoing EVAR for rupture, those that developed ARF had no differences in age, gender, race, or comorbidities other than CHF (6.1% vs 2.2%, P = .022) (Table 2). Similarly, there were no differences in ASA class nor diameter of aneurysm. Patients that developed ARF had higher baseline creatinine (1.5 vs 1.2, P < .0001) and lower hematocrit (33.9 vs 34.9, P = .042) with significantly longer operative times (177 vs 135 minutes, P < .0001). Patients with juxtarenal and suprarenal aneurysms were at an increased risk of ARF (P = .002). Those patients that required intraoperative and postoperative transfusions and those requiring return trips to the operating room were also at an increased risk of developing ARF (P < .0001). Acute renal failure patients had significantly longer ICU LOS (4 vs 2 days, P < .0001) and hospital LOS (15 vs 5 days, P < .0001). For patients who developed ARF, there was no significant difference in postoperative sepsis, UTI, PE, stroke, or DVT. However, there was a significant increase in rates of pneumonia (27% vs 7%, P < .0001), prolonged mechanical ventilation (62% vs 13%, P < .0001), MI (14% vs 6.6%, P = .003), and death (47.4% vs 17.5%, P < .0001).
Multivariable Logistic Regression
Multivariable Logistic Regressions for Predictors of Acute Renal Failure.
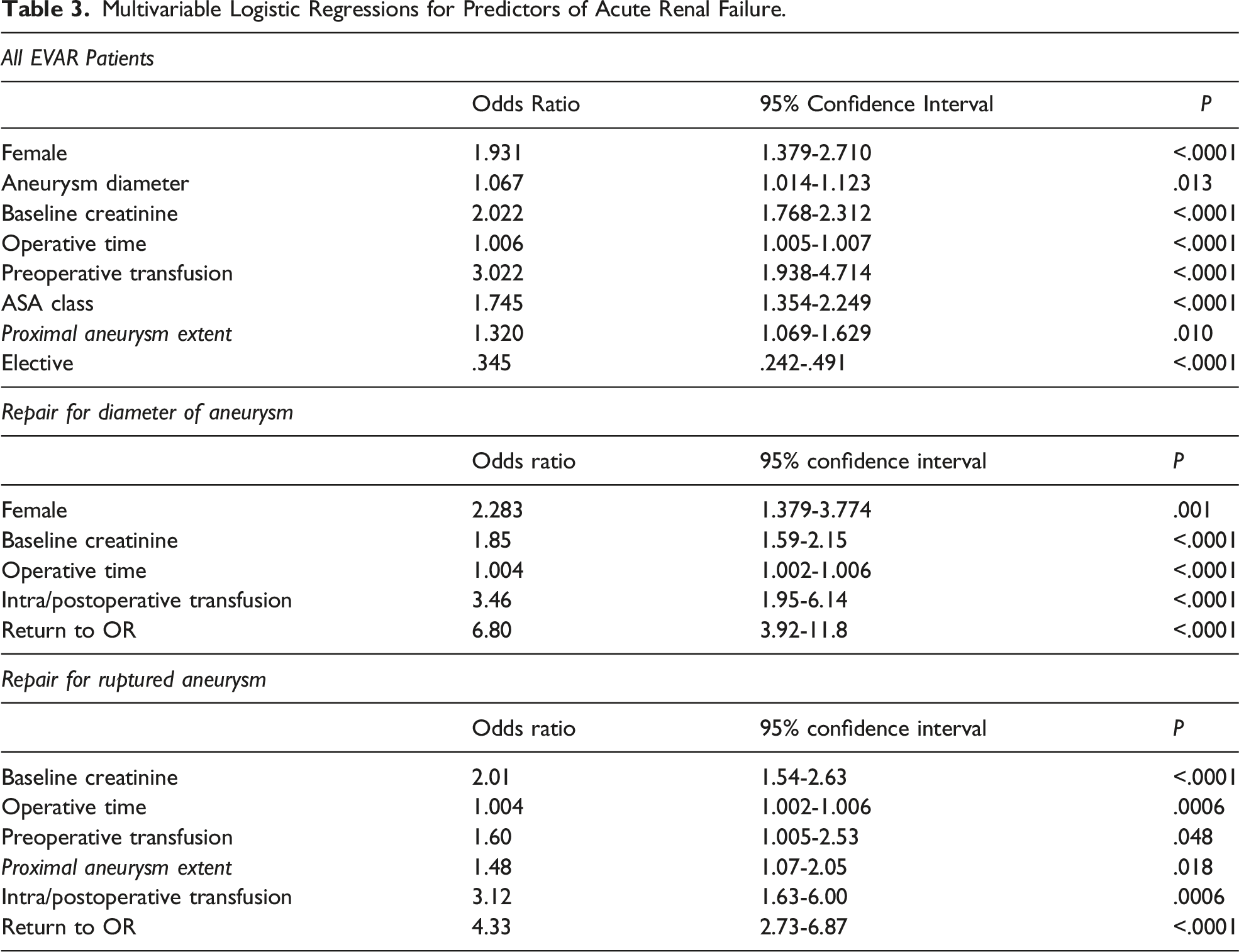
For rupture patients, MLR identified no protective variables. Higher admission creatinine, need for preoperative as well as intra/postoperative transfusions, longer operative times, higher proximal aneurysm extent, and requiring return to the operating room were all independently associated with ARF (Table 3).
Patients undergoing EVAR for diameter of aneurysm similarly had no protective features. However, higher admission creatinine, female gender, longer operative times, need for intra/postoperative transfusions, and requiring return to the operating room all increased the risk of developing ARF (Table 3).
Discussion
The need for dialysis after EVAR is relatively uncommon, occurring in only 1.3% of patients in the NSQIP database, though it is associated with significant morbidity and mortality. In the current study, patients that developed ARF had prolonged ICU (5 days) and hospital (11 days) LOS with a 39.7% mortality rate. While the mortality rate with ARF after EVAR is more pronounced in patients who present with rupture (47%), patients who had repair for aneurysm diameter and developed ARF also carried a significant mortality risk with a 31.6% rate. For all patients undergoing EVAR, independent risk factors for developing ARF included gender (females at increased risk), baseline health status (ASA class and admission creatinine level), aneurysm characteristics (larger diameter and more proximal aneurysm extent), and difficulty of repair (longer operative times and urgent/emergent repair). When patients undergoing repair for diameter were analyzed separately, females continued to experience higher risk, as did those with elevated baseline creatinine and with longer operative times. In addition to these factors, ongoing transfusion requirements and need for return to the operating room predisposed these patients to ARF. For those undergoing repair after rupture, gender no longer impacted the incidence of ARF. Instead, risk factors for ARF included hemodynamics (transfusion requirements), baseline renal function (creatinine), complexity of repair (longer OR time and more proximal aneurysm extent), and need for return to the operating room. While there are similarities between the risk factors for these patients, this study has established independent risk factors for both rupture patients and those undergoing repair for aneurysm size alone. Though ARF after EVAR is relatively uncommon, the morbidity and mortality associated with it are significant; thus, these patients at risk for ARF should be carefully monitored postoperatively.
The advent of EVAR leads to a significant reduction in the incidence of ARF compared to open repair. 4 However, AKI continues to be a frequent issue for patients after EVAR with 10%-30% of patients experiencing some decline in renal function.3,12,15 Any level of AKI is associated with worse long-term renal function and mortality compared to those who did not develop AKI.6,7,11,14 Statius van Eps et al found that, for patients undergoing elective EVAR, development of AKI leads to a 24% rate of decreased renal function two years postrepair. 3 In addition, ARF requiring dialysis after EVAR has previously been reported between .3% and 1%.6,7,15 The higher incidence in this study is likely secondary to the inclusion of rupture patients, which many previous studies have excluded. Similar to previous studies, it is unclear what impact this has on long-term renal function, or which patients have eventual renal recovery. The increased risk for ARF in females is of particular concern, though the reason for this is unclear. Our finding of a 2-fold increased risk has been seen in other studies though the mechanism for this has yet to be determined.8,11 Clearly, future studies will be needed to both help delineate the risk factors for temporary vs permanent dialysis and mitigate this gender disparity.
By using ARF requiring dialysis, we sought to capture the most clinically significant renal dysfunction for short-term outcomes. In this study, ARF led to a 40% 30-day mortality rate. This substantial increase in mortality is similar to that reported by Zarkowsky who found a 5-fold increase in mortality for patients requiring dialysis after elective EVAR, with less than 25% overall survival at three years for patients who developed ARF. 7 The significant impact on morbidity and mortality associated with AKI/ARF after EVAR cannot be understated.
The impact of the proximal aneurysm extent on renal outcomes has been widely debated. Multiple studies have found that patients undergoing complex EVAR requiring pararenal fixation to be at an increased risk of AKI.5,10,11 This study confirms the impact of proximal aneurysm extent on ARF in patients undergoing EVAR for rupture; however, proximal aneurysm extent did not impact renal function after elective repair. This difference for elective repairs has been found in previous studies with Pisimisis finding proximal fixation to impact neither AKI nor CKD. 16 Similarly, a recent meta-analysis found no difference in change in GFR at one year with suprarenal fixation. 17 Contrary to this, Banno found a 20% reduction in GFR for suprarenal fixation at 3 year follow-up, which was significantly higher than those undergoing infrarenal EVAR. 13 Some of the variability in these reports may be secondary to different definitions of AKI used.
Similar to previous studies, for all patients undergoing EVAR, regardless of indication, baseline creatinine was found to be predictive of developing ARF. 15 This finding is not surprising and has been well reported with multiple studies finding preoperative creatinine or eGFR to be associated with the development of AKI.2,6,9,11 Many surgeons try to decrease contrast volume used in these patients or try preoperative hydration to decrease the risk of renal injury. Unfortunately, neither contrast volume nor hydration strategies can be ascertained from the NSQIP database. There have been variable reports in the literature on the impact of contrast volume on development of AKI.7,10,14,15,18 It appears unclear at this point if AKI after EVAR is mainly secondary to contrast-induced nephropathy or has more to do with micro emboli, injury from wire manipulation, coverage of accessory renal arteries, or some combination of all of these factors. 2 A recent study found exercise tolerance and cardiovascular reserve to have a significant impact on renal dysfunction after EVAR. 19 How all of these factors work in concert for the development of AKI requires ongoing research. In addition, studies are ongoing as to the role of various hydration strategies to prevent AKI, including a recent pilot trial for the use of preoperative sodium bicarbonate which shows promising results. 20 Hopefully, future institutional-based studies can further show the impact of contrast and hydration on postoperative renal dysfunction, particularly for patients that develop ARF.
Rupture patients have typically been excluded from previous studies likely because of the variability in the cause of renal dysfunction in this patient population.3,5,13,18 This study has found that similar to previous studies, patients who develop AKI or ARF after rupture have significantly worse outcomes.4,14 Some of the risks associated with ARF in this patient population are related to hemodynamics (ongoing transfusion requirements) as one might expect. This confirms Novack and others findings that lower baseline hemoglobin/hematocrit and ongoing transfusion requirements are associated with a higher incidence of AKI. 11 While this is not modifiable, the significant impact on renal dysfunction and the high mortality associated with ARF may help guide family and patient discussions about goals of care. Similarly, as would be expected, more difficult repairs (longer operative times more proximal aneurysm extent, and need for multiple operations) increase the risk of developing ARF. While the etiology may be different for cause of ARF in rupture patients, these patients do have similarities to those undergoing elective repair. Interestingly, gender and aneurysm diameter did not impact ARF for these patients, unlike the overall and the repair for diameter cohorts.
Limitations
There are several limitations to this study. While risk factors for ARF have been established, there is no information within the NSQIP on renal recovery or whether these patients required a short cycle of temporary dialysis or permanent dialysis. Similarly, outcomes are limited to 30 days, so the morbidity and mortality associated with ARF may be understated. In addition, 10% of patients underwent aneurysm repair for unknown reason (listed as other), which may decrease the generalizability of this study. Like all large databases, the NSQIP has inherent limitations such as coding errors and lack of clinical context for each patient. Though the vascular-specific files do provide some information on aneurysm morphology, data is lacking regarding specifics of the aneurysm and how the repair was performed. Future studies with more granular aneurysm-specific details may help identify modifiable risk factors associated with ARF after EVAR
Conclusions
Patients requiring initiation of dialysis after EVAR are at a significant risk for morbidity and death, regardless of indication for repair. Risk factors for ARF hinge primarily on baseline renal function, complexity of repair, need for blood transfusions, and those requiring re-operation. In addition to this, women are at a 2-fold risk of ARF when repair was performed for diameter. Future studies will be needed to elucidate this disparity. While no modifiable risk factor was identified, patients at an increased risk of ARF should be closely monitored to hopefully mitigate some of these risks.
Footnotes
Author’s Note
The study was presented in part as a poster presentation at the Southern Association for Vascular Surgery, January 19-22, 2022 Manalapan, Florida.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
