Abstract
Background
Cancer care guidelines based on clinical trial data in homogenous populations may not be applicable to all rectal cancer patients. The aim of this study was to evaluate whether patients enrolled in rectal cancer clinical trials (CTs) are representative of United States (U.S.) rectal cancer patients.
Methods
Prospective rectal cancer CTs from 2010 to 2019 in the United States were systematically reviewed. In trials with multiple arms reporting separate demographic variables, each arm was considered a separate CT group in the analysis. Demographic variables considered in the analysis were age, sex, race/ethnicity, facility location throughout the United States, rural vs urban geography, and facility type. Participant demographics from trial and the National Cancer Database (NCDB) participants were compared using chi-squared goodness of fit and one-sample t-test where applicable.
Results
Of 50 CT groups identified, 42 (82%) studies reported mean or median age. Trial participants were younger compared to NCDB patients (P < .001 all studies). All but three trials had fewer female patients than NCDB (48.2% female, P < .001). Less than half the CT groups reported on race or ethnicity. Eighteen out of 22 trials (82%) had a smaller percentage of Black patients and 4 out of 8 (50%) trials had fewer Hispanic or Spanish origin patients than the NCDB. No CTs reported comorbidities, socioeconomic factors, or education. CT primary sites were largely at academic centers and in urban areas.
Conclusion
The present study supports the need for improved demographic representation and transparency in rectal cancer clinical trials.
Key Takeaways
• Rectal cancer patients enrolled in clinical trials are more likely to be younger, male, and White compared to the patients in the NCDB. • Lack of transparency in reporting participant comorbidities, socioeconomic factors, and health literacy limits an evaluation of inclusion and representation of rectal cancer patients in clinical trials. • Clinical trials are primarily at academic institutions in urban areas.
Introduction
It has been well established that there are significant disparities in rectal cancer prevention, treatment and survival by race, ethnicity, and gender.1-3 Randomized controlled trials represent high quality evidence and are often the basis of clinical decision-making and guideline development. 4 Underrepresentation of women and minorities in clinical trials has raised concerns about trial generalizability. 5 In response, the U.S. Department of Health and Human Services introduced a Strategic Framework on Multiple Chronic Conditions (MCCs), which identified objectives to increase the external validity of clinical trials for patients with several comorbidities. 6 However, it is not clear how broadly this framework has been implemented. 5
Previous evaluations of cancer clinical trials have demonstrated that they are not reflective of the cancer population. An analysis of therapeutic, nonsurgical clinical trials from 1996 to 2002 conducted by the National Cancer Institute showed that disparities in age, gender, race, and ethnicity were present in a variety of studies in breast, lung, colorectal, and prostate cancer. 7 Since the National Institutes of Health Revitalization Act was enacted in 1993 to promote inclusion of women in clinical trials, the number of women in trials has increased, but has not reached parity. 8 Furthermore, although 61% of cancers occur among the elderly, previous reports show that patients over 65 comprise only 25% of participants in cancer clinical trials, particularly in early-stage cancer trials.9,10 Though prior studies report this underrepresentation and limited demographic information, the degree of difference between patients enrolled in clinical trials for rectal cancer and the general population has not been analyzed.
The aim of this study is to compare the patients included in US rectal cancer clinical trials to the patient population in the National Cancer Database (NCDB). A secondary aim was to identify demographics that could be reported in clinical trials to enable more robust comparisons.
Methods
National Cancer Database Data
The NCDB rectal cancer patient population was used as a proxy for US rectal cancer patients, as the NCDB includes 70% of all new rectal cancer diagnoses from 1500 US cancer centers. 11 The NCDB data dictionary was used to create a list of possible variables for inclusion. 12 Demographic information, facility type, and facility location were extracted.
Clinical Trial Data and Systematic Review
The guidelines set forth by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) were followed. Prospective clinical trials published between 2010 and 2019 were identified using a PubMed search conducted with the terms “rectal cancer” and “rectal adenocarcinoma.” The references of the National Comprehensive Cancer Network (NCCN) guidelines for rectal cancer and the bibliography of each included trial were also reviewed to identify additional studies.
13
All resulting records were reviewed by a single reviewer (R.G.), and all included studies were reviewed by a second reviewer (A.O.) with reconciliation. Inclusion criteria were phase II and III clinical trials focusing on rectal cancer treatment and conducted in the United States. Studies examining only quality of life or those unrelated to rectal cancer treatment were excluded (Figure 1). Flow diagram outlining study selection. A total of 31 clinical trials met inclusion criteria. For studies with multiple clinical trial arms, separate demographic information was reported for each arm. Therefore, each arm was analyzed separately, resulting in 50 total clinical trial groups for analysis. Abbreviations: National Comprehensive Cancer Network (NCCN).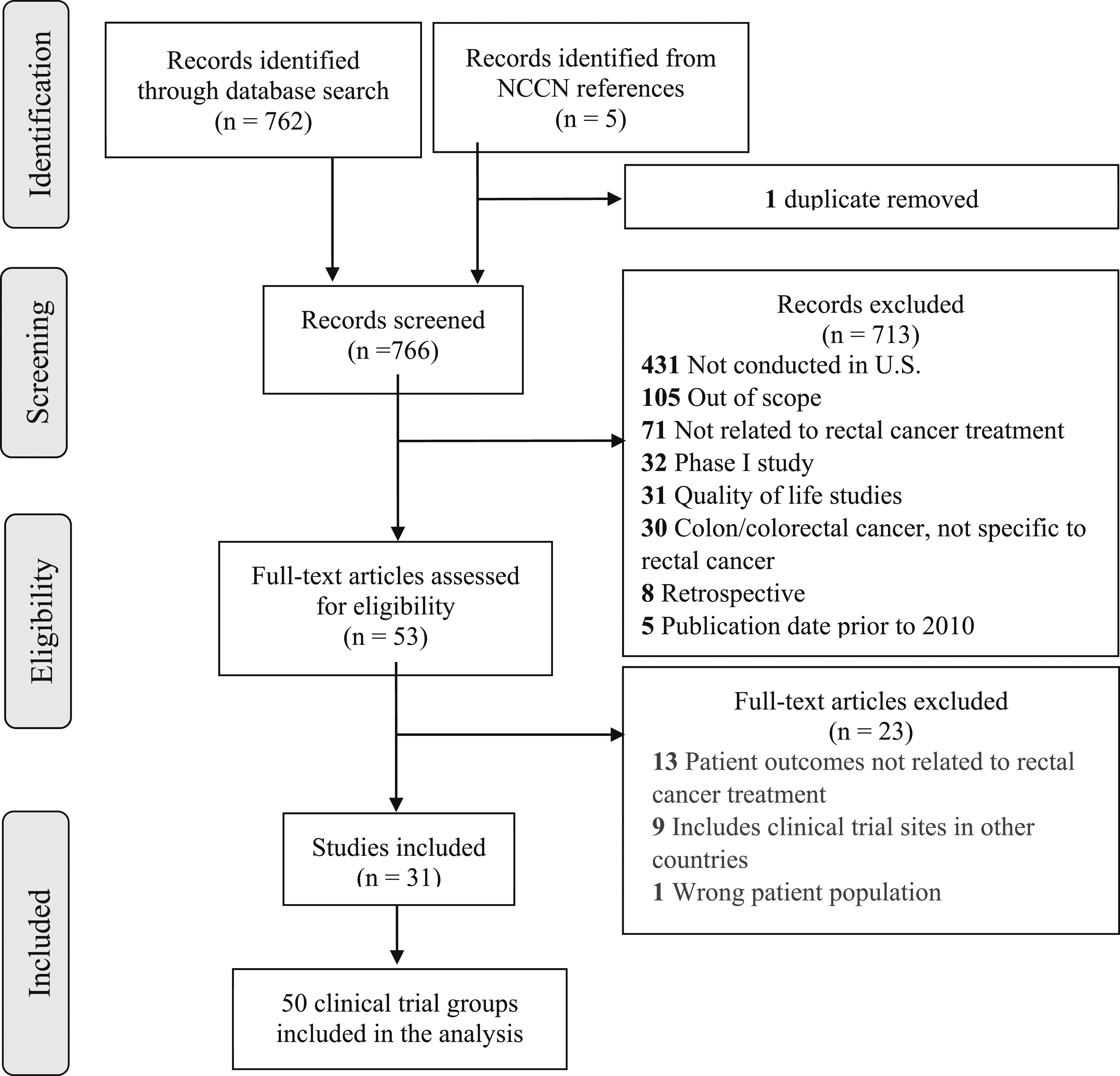
Demographics were extracted from trials by analyzing the text of the paper, as well as tables and figures. In cases where a clinical trial had multiple arms, each arm was analyzed separately and included as a separate “clinical trial group” for the purpose of data analysis. Demographics were coded in the same format as the NCDB data dictionary where possible. 12 Demographic information captured in the NCDB but not reported in a clinical trial was noted.
Primary facility location in the NCDB represents where a patient is from and is categorized by state. The primary facility location of a trial was determined using the associated clinicaltrials.gov page, or the location of the principal investigator listed in the publication. Most clinical trials included multiple sites, and all sites listed in the publication and the clinicaltrials.gov page were recorded.
Clinical trial sites were characterized as either academic, comprehensive cancer centers, or community hospitals, in accordance with NCDB data dictionary. Clinical trial sites were also classified as metropolitan, urban, or rural based on the population size of the clinical site, which reflected the coding in the NCDB dictionary. 12 If facility locations were not clear, site information was excluded from the analysis.
Statistical Analysis
Trial demographics were compared to NCDB data using individual one-sample t-tests and chi-square analysis for each known variable. Statistical significance was established with alpha-threshold < .05. All statistical analysis was conducted using SAS 9.3 (Cary, NC).
Results
Demographic Characteristics of Rectal Cancer patients in the National Cancer Database.
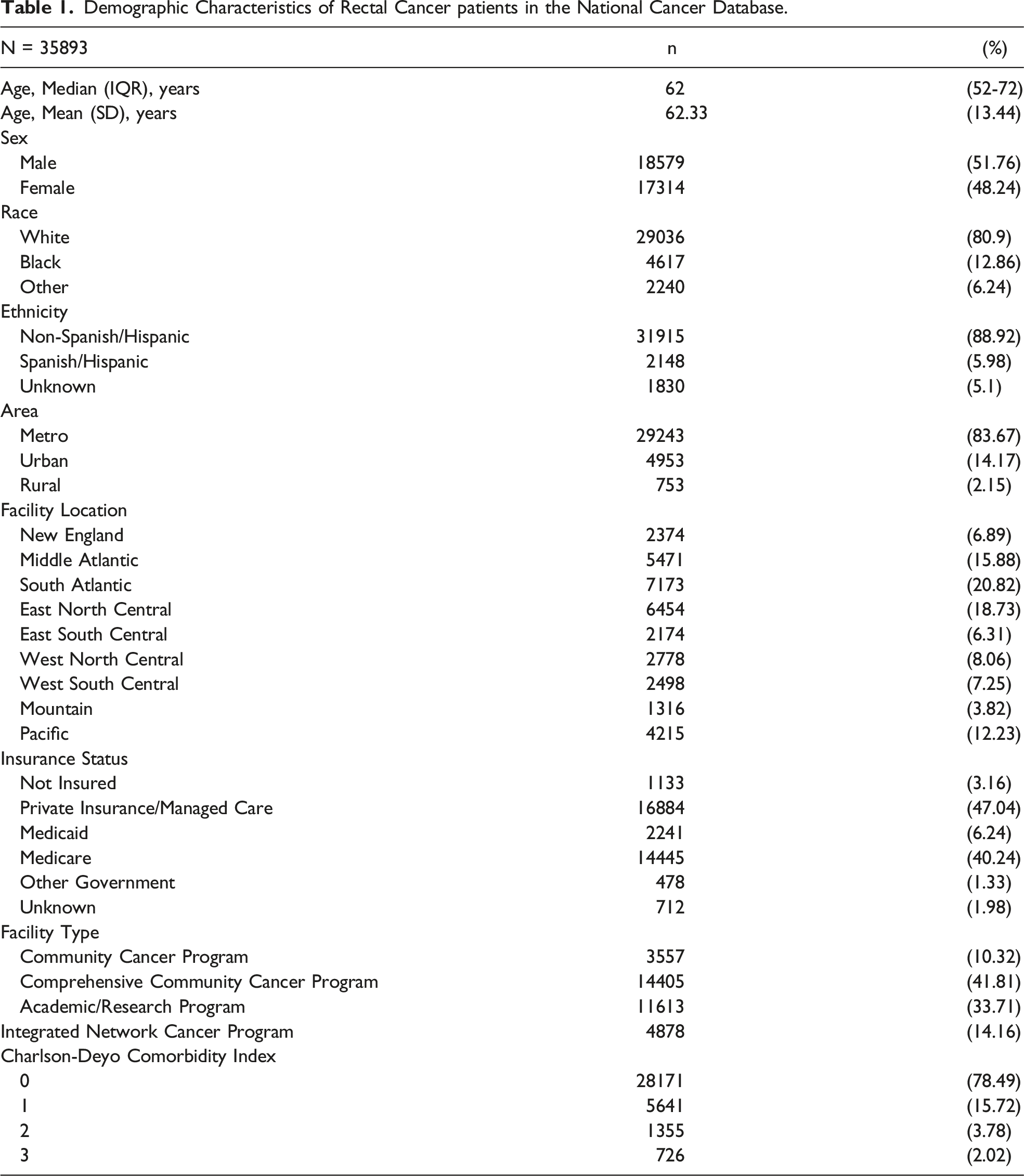
The majority of trials included patients with local or locoregionally advanced rectal adenocarcinoma without distant metastasis, as 13 trials (32.3%) included stage II-III cancers and 14 trials specified T3-T4 cancers (45.2%) (Supplemental Table 1). The remaining trials included patients with T2 or T2-T3 cancers (9.7%), and one trial included stage I-IV disease. Of the 31 trials, the majority assessed neoadjuvant or adjuvant chemoradiotherapy regimens, with or without the addition of biologics. Five trials evaluated a combination of induction, consolidation or adjuvant chemotherapy alone in addition to neoadjuvant chemoradiotherapy.18-20,27,32 Only one trial investigated the effect of neoadjuvant chemotherapy alone without chemoradiotherapy. 16 In two studies, the primary intervention assessed was surgical, and in three studies, the use of neoadjuvant chemoradiotherapy impacted the surgical treatment (surgical timing or local excision).14,15,24,25,35 Finally, one trial investigated the use sentinel lymph node mapping in patients undergoing surgery with or without neoadjuvant chemoradiotherapy. 39
Clinical Trials Demographics Results
Of the 50 included trial groups, 42 (82%) reported mean or median age, and all but 3 were significantly younger than the mean or median age of NCDB patients.11,14,15,24 Twenty-seven trial groups reported median age of participants.14,16-18,20-23,27-29,32,34,37,38,41-44 In 21 of these 27 trial groups, the median age difference was at least 5 years younger than the NCDB median age of 62 (IQR 52 - 72). The median age of participants in each trial was significantly different from the median age in the NCDB on one-sample median test (P < .001). Likewise, fifteen (29%) clinical trial groups reported mean age of participants (range 42 - 64 years).15,19,24,25,30,35,36,39,40 Both individual clinical trial groups and the collective mean for all groups (57.84 years ± 5.19) differed significantly in mean age of participants compared to the mean age of NCDB patients on one-sample t-test (62.33 years ± 13.44, P < .01).
Forty-eight of 50 trial groups (94%) reported sex. The proportion of female trial participation was between 19 - 53%, while NCDB patients were 48.2% female. Forty-seven of the 50 trial groups had a statistically significant difference in the proportion of female patients compared to NCDB (P < .001), and all but 3 trials had fewer female patients than the percentage listed in NCDB.16-18 Seventy-three percent of trials had fewer than 40% females enrolled.
Twenty-one trial groups (42%) reported race,14,17,18,20-22,24,25,28,31,34,35,37,38,42-44 and 8 trial groups (16%) reported patient ethnicities.20,21,31,32,34,37 Eighteen out of 22 trials (82%) had a smaller percentage of Black patients and 4 out of 8 (50%) trials had fewer Hispanic or Spanish origin patients than the NCDB, which has 12.9% Black patients and 6% Hispanic or Spanish origin patients (Figure 2, P < .001). In addition, only 14 clinical trial groups (28%) reported on Asian race among participants, and only 6 trial groups (12%) reported on Native Hawaiian/Pacific Islander or American Indian/Alaska Native race. Clinical trial group 6, which had the largest proportion of Hispanic or Spanish origin patients (%), was conducted at the University of New Mexico.
21
Race (a) and ethnicity (b) distribution in clinical trials and NCDB. Twenty-one clinical trial groups reported on race, and 7 reported on ethnicity. Chi-square goodness of fit demonstrated significant differences in race and ethnicity of clinical trial participants compared to that of NCDB patients (P < .001).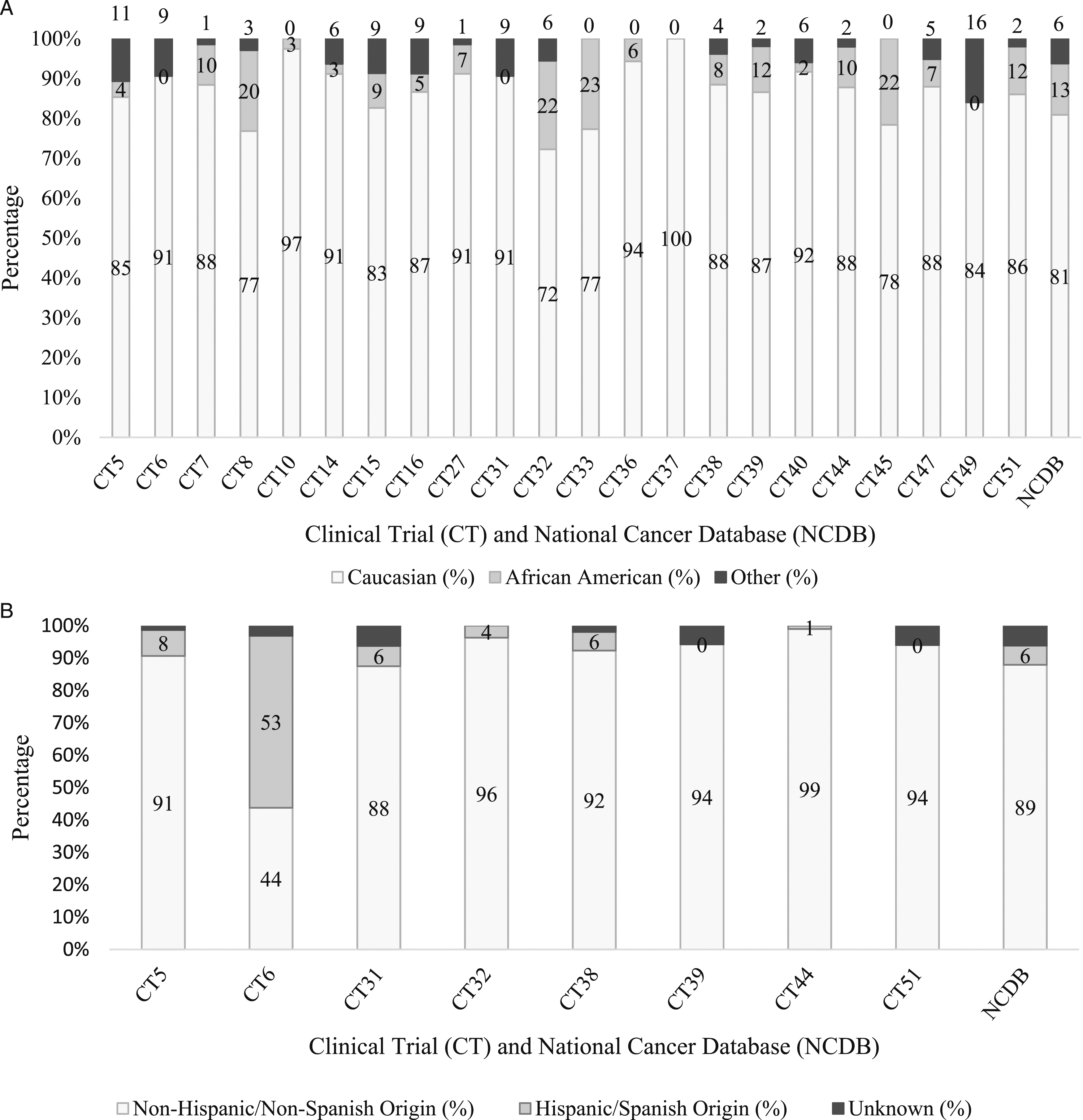
Demographics Limited by Data Availability
None of the included trials reported patient comorbidities or Charlson-Deyo scores. Several studies included Eastern Cooperative Oncology Group (ECOG) performance statuses (ranging 0-2), or included ECOG performance status of 0-2 as part of their inclusion criteria.19-21,23,25-28,34,38,40,41,43 Patients in NCDB had median Charlson-Deyo scores of 0, with 78.4% of patients having a score of 0 and 15.7% having a score of 1. Only four of 50 (8%) trial groups reported BMI and one (2%) reported smoking status.20,23,25 No trials reported insurance status, education, or income.
Clinical Trial Locations
Facility Type of all CT Sites Compared to the NCDB Sites.

Twenty-five clinical trials reported the primary facility type.
aChi-squared goodness of fit test could not be performed as the integrated facility type cell value was 0.
Abbreviations: clinical trial (CT); National Cancer Database (NCDB).
Regional Facility Locations and Urban/Rural Classification of CTs and NCDB.
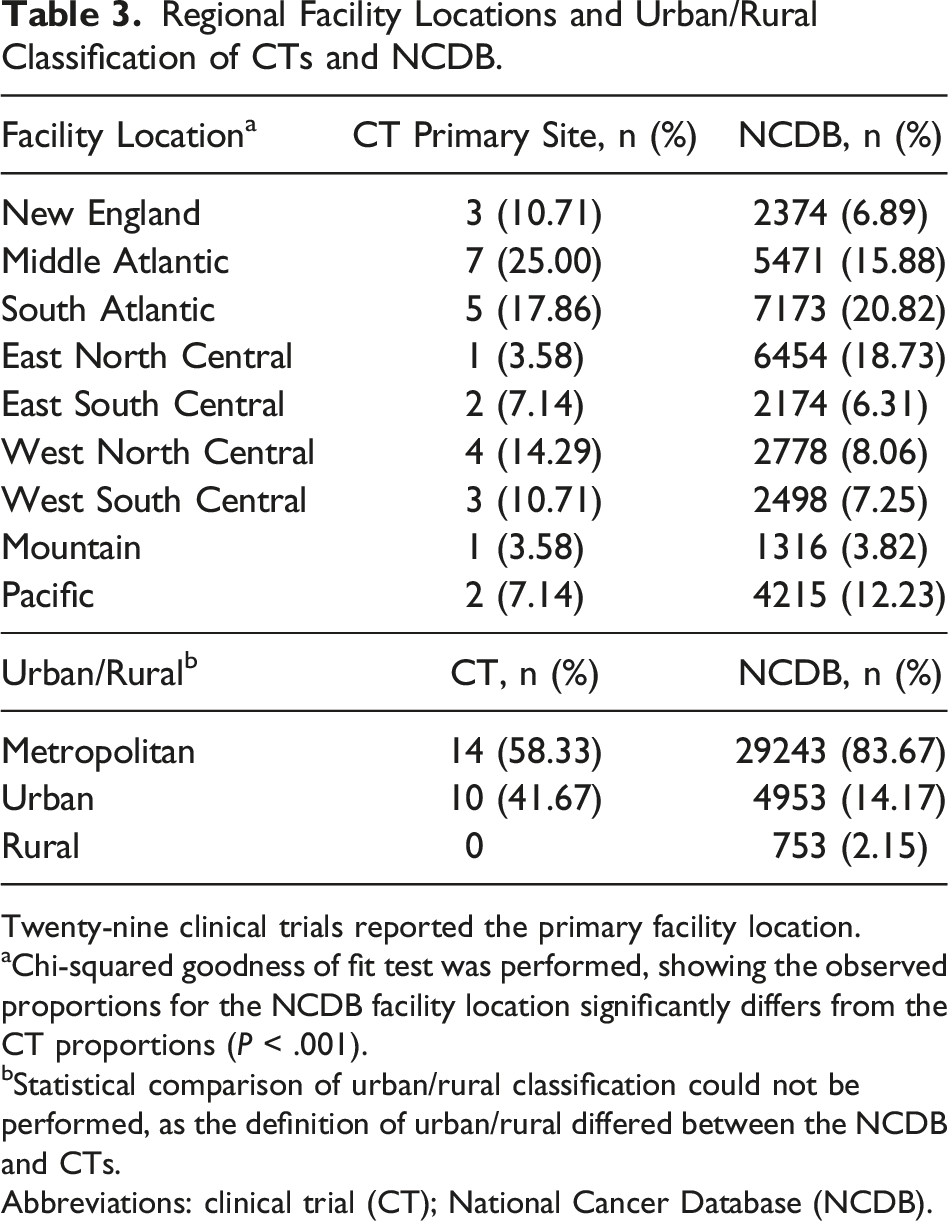
Twenty-nine clinical trials reported the primary facility location.
aChi-squared goodness of fit test was performed, showing the observed proportions for the NCDB facility location significantly differs from the CT proportions (P < .001).
bStatistical comparison of urban/rural classification could not be performed, as the definition of urban/rural differed between the NCDB and CTs.
Abbreviations: clinical trial (CT); National Cancer Database (NCDB).
Discussion
Rectal cancer clinical trials are used to inform clinical decision-making and practice guidelines that delineate the standard of care. However, this study found that patients included in clinical trials may represent a significantly different population than the average cancer patient, as defined by patient demographics in the NCDB. The data demonstrate that patients in prospective rectal cancer trials are more likely to be younger, White, metropolitan and male compared to patients reported in the NCDB. In addition, heterogeneity in reporting demographics and comorbidities inhibits more robust comparisons of study findings. This raises a concern that clinical trial results may not be generalizable to all rectal cancer patients.
These present findings are consistent with previous studies on disparities in cancer clinical trials. A large retrospective cohort study of 19,340 cancer patients at MD Anderson found that clinical trial participants were more likely to be younger White males with fewer comorbidities relative to trial non-participants. 45 In order to increase minority representation, understanding the reasons for these disparities is important. Many explanations have been proposed for the underrepresentation of minorities, women, and older adults in clinical trials. These include patient bias towards participation, provider bias towards eligibility, and differences in access to care.46,47 The impact of systemic racism on healthcare at the institutional level, and its contribution to factors such as access to care and comorbidities, is difficult to measure.2,48-50 That said, several studies have shown disparities in survival outcomes, as well as neoadjuvant, primary surgical resection, and adjuvant treatments offered to Black patients. The basis for disparate management of these patients remains unclear. These findings are particularly concerning in the setting of steady incidence in rectal cancer among younger Black patients. 51 Additionally, differences in perceptions of medical research among minorities affect participation in clinical trials.52,53 While this study focused on representation of Non-Hispanic Black and Hispanic participants, our findings also highlight a lack of explicit reporting on Asian and Native American race in most rectal cancer trials. In order to enroll more inclusive populations in clinical trials, the availability of trials in underserved areas, as well as institutional efforts to rebuild trust in these trials, should be improved.
Likewise, the findings of this study support the existing literature on sex and gender disparities in clinical trial enrollment. 54 A systematic review in 2016 found that sex bias continues to exist in human surgical clinical research, reflected by differences in enrollment and fewer than one-third of studies performing data analysis by sex. 55 Additionally, studies often do not distinguish between biological sex and gender in demographic reporting. Gender disparities in rectal cancer research are of particular concern in the setting of rising global incidence of rectal cancer among women younger than 50 years of age. 56 Though there is limited data on intersectionality between race and gender and how it impacts trial enrollment, women of color and elderly minority groups are particularly underrepresented. 7 It should be noted that none of the trials in our study provided any data on nonbinary gender, which parallels previous studies highlighting the lack of reporting of LGBTQ + patients in cancer clinical trials. 57
One important result of this study is that very few trials provided sufficient demographic information about their participants to enable a robust comparison to the NCDB. Few trials reported important information such as BMI, smoking status, alcohol use, or comorbidities, and the NCDB does not include this information either. Almost no studies included detailed information on insurance status, income, education, and health literacy levels, which would better characterize the patient populations studied. These factors are particularly relevant to the elderly cancer population, and are compounded by the fact that patients older than 65 were found to be underrepresented in clinical trials. Despite making up more than 60% of cancer patients, older adults only represent 32% of patients in phase II and III clinical trials. 58 Older adults often have several barriers to care and trial participation, including provider bias, underinsurance, lower electronic health literacy, economic constraints, hearing or visual impairments, higher rates of comorbid conditions that might prompt exclusion, and transportation issues that likely hinder enrollment. 59
This study also demonstrates a gap in access to clinical trials in rural areas. While approximately 15-19% of the US population lives in a rural area, these communities have significant shortages in both surgeons and oncology providers, which are only expected to worsen in the future.60-63 Patients in rural areas are also less likely to receive colorectal cancer screening, are more likely to present at advanced stages, and are at increased risk of experience adjuvant radiation delays.63-66 Future efforts to increase equity in access to cancer care should emphasize transparent and standardized reporting of these variables.
In an attempt to address this issue of clinical trial diversity and inclusion, the US Department of Health and Human Services released a detailed guide on how to expand the diversity of clinical trial populations in 2020. 67 These guidelines recommend adopting enrollment practices that enhance inclusivity, such as including trial locations in areas with large populations of minority patients and increasing transparency around financial reimbursements for expenses incurred by trial participation. 67 The American Society of Clinical Oncology also suggested that cancer clinical trials broaden their eligibility criteria when examining comorbidities (e.g., brain metastases, kidney, and cardiac function). 68 The current study demonstrates that these disparities persist in rectal cancer trials despite efforts towards equity in enrollment. Due to the possible lag time in adopting these guidelines, future studies should reassess whether progress has been made toward broader inclusion in response to these guidelines.
Limitations
This study was limited by the lack of detailed demographic information reported in rectal cancer clinical trials as well as heterogeneity in demographic reporting, which limited comparison to the demographics of NCDB rectal cancer patients. The heterogeneity in race and ethnicity classifications between trials and the NCDB also presented a challenge. NCDB uses a complex system of sub-categorization, with more than 30 specific categories for race, while the majority of trials restrict race classification to White, Black, or Other, and ethnicity to Hispanic or Non-Hispanic. Future trials should use more inclusive race and ethnicity classification where possible to balance inclusivity and feasible statistical comparison. Furthermore, some clinical trial sites were difficult to identify if not listed on the clinicaltrials.gov page or in the publication. Ensuring the accuracy of facility type and location per the NCDB data dictionary was also challenging in some cases, resulting in exclusion of this information for these trials in the analysis. Lastly, because the comparison group was the NCDB, only clinical trials conducted in the United States were included. This led to the exclusion of many otherwise relevant studies.
Conclusions
Clinical trials are considered the gold standard of medical research. Though they offer tremendous value in evaluating efficacy of interventions, the present study highlights gaps in representation and transparency. Future clinical trials in rectal cancer should aim to increase inclusive demographic and geographic representation, and to standardize reporting of these patient factors.
Supplemental Material
Supplemental Material - Disparities in Clinical Trial Enrollment and Reporting in Rectal Cancer: A Systematic Review and Demographic Comparison to the National Cancer Database
Supplemental Material for Disparities in Clinical Trial Enrollment and Reporting in Rectal Cancer: A Systematic Review and Demographic Comparison to the National Cancer Database by Tuleen Sawaf, Rachana Gudipudi, Asya Ofshteyn, Anuja L. Sarode, Katherine Bingmer, Jonathan Bliggenstorfer, Sharon L. Stein, and Emily Steinhagen in Journal of The American Surgeon™
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author’s Note
Meeting Presentation: Presented at American College of Surgeons Virtual Clinical Congress 2020 as an oral presentation, October 2020.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
