Abstract
Introduction
Patients with cirrhosis have an increased risk of complications after trauma, including bleeding, unplanned operations, and death. The benefit of venous thromboembolism (VTE) chemoprophylaxis in trauma patients with cirrhosis (CTPs) is not clear, especially since cirrhotic patients are hypercoagulable. We hypothesized that CTPs receiving VTE chemoprophylaxis (vCP) have a lower risk of death with no increased risk for unplanned operations compared to patients with cirrhosis not receiving vCP.
Methods
The 2017-2019 TQIP database was queried for patients with cirrhosis. Patients on outpatient anticoagulant therapy or with a history of bleeding diathesis, interhospital transfers, severe head injury, deaths < 72 hours, and hospitalization < 2 days were excluded. A multivariable logistic regression analysis was performed.
Results
From 10,011 CTPs, 6,350 (63.4%) received vCP. Compared to patients without vCP, the vCP group had decreased mortality (4.5% vs. 5.5%, P = 0.03) but a similar rate of unplanned operations (1% vs. 0.6%, P = 0.07). This persisted on multivariable analysis, with a decreased associated risk of mortality (OR 0.54, CI 0.42-0.69, P < 0.001), and a similar risk of unplanned operation (P = 0.85).
Conclusion
CTPs received VTE chemoprophylaxis in under two-thirds of cases. On multivariable analysis, vCP was associated with a decreased risk of mortality and a similar risk of unplanned operations. These findings suggest that vCP appears safe. Further investigation is needed to confirm this finding.
Introduction
Less than .3% of the United States adult population suffers from cirrhosis. In contrast, more than 1% of trauma patients present with cirrhosis, and the wide-ranging systemic abnormalities in these patients impact outcomes after trauma. 1 Historically, patients with cirrhosis were thought to have an increased risk of bleeding and reduced risk of venous thromboembolism (VTE). 2 However, recent data demonstrate that cirrhosis may lead to a hypercoagulable state by affecting the production of anticoagulant factors, such as antithrombin and protein C and S.3-7 In support, Subhani. et al. found that patients with chronic liver disease (CLD) have double the risk of venous thromboembolism (VTE) compared to those without CLD. 8
Traumatic injury is a major risk factor for VTE by completing Virchow’s triad with the addition of endothelial injury and/or venous stasis secondary to immobility. 9 As such, the use of VTE chemoprophylaxis (vCP) in trauma patients is crucial.10,11 However, there are no specific recommendations for vCP initiation for trauma patients with cirrhosis, and practice patterns are highly variable, with some centers utilizing laboratory values such as platelet count, international normalized ratio (INR), and partial thromboplastin time (PTT) to determine if and when to initiate vCP.7,12,13 However, studies on non-trauma populations have suggested that the use of vCP does not increase the risk of major bleeding.4,12-15 Although, there are no large high-quality studies examining this relationship in only trauma patients.
Thus, we sought to determine the incidence of vCP in trauma patients with cirrhosis and analyze the association between the use of vCP and the incidence of mortality and unplanned operations. We hypothesize that trauma patients with cirrhosis benefit from vCP by having a lower risk of mortality but similar risk of an unplanned return to the operating room (OR) compared to patients who do not receive vCP.
Methods
This study was deemed exempt by the institutional review board at our institution, and a waiver of informed consent was granted due to the use of a deidentified national database. A retrospective analysis of the Trauma Quality Improvement Program (TQIP) was performed between 2017 and 2019. All trauma patients ≥ 18 years of age with a diagnosis of cirrhosis were included. Those on outpatient anticoagulant therapy, with a history of bleeding diathesis, interhospital transfers, serious traumatic brain injury (TBI), deaths < 72 hours, and patients hospitalized less than two days were excluded. Serious TBI was defined by an abbreviate injury scale (AIS) grade 2 or higher for the head.
Demographic variables collected included age, sex, and comorbidities such as hypertension, diabetes, smoking status, previous cerebrovascular accident (CVA), chronic obstructive pulmonary disease (COPD), chronic kidney disease (CKD), and previous myocardial infarction (MI). The injury profile data collected included the mechanism of injury, injury severity score (ISS), and AIS for the thorax, abdomen, and spine.
The primary outcome was in-hospital mortality. The secondary outcomes were VTE and unplanned return to the OR. Other outcomes measured included packed red blood cells (PRBC) transfusion within the first four hours of arrival, total hospital length of stay (LOS), intensive care unit (ICU) LOS, ventilator days, and in-hospital complications, including CVA, MI, cardiac arrest, acute respiratory distress syndrome (ARDS), pneumonia, acute kidney injury (AKI), superficial and deep incisional surgical site infection (SSI), organ space SSI, and sepsis.
Descriptive statistics were performed for all variables and stratified by the use of vCP. Categorical data were compared using chi-square and reported as percentages. Continuous variables were reported as medians with an interquartile range.
A multivariable logistic regression model was also used to evaluate the risk of mortality and VTE after adjusting for sex, age, ISS, vital signs on admission (hypotension (systolic blood pressure <90 mmHg), tachypnea (respiratory rate >22 breaths per minute), and tachycardia (heart rate >120 beats per minute)), Glasgow coma scale (GCS) on admission, smoking status, obesity, pelvic fracture, long bone fracture, spine fracture, and requirement of an operative procedure. These were chosen after coauthors' discussion and a literature review.12,16 The adjusted likelihood for mortality and complications were reported with an odds ratio (OR) and 95% confidence intervals (CI) after stratification by patients who received vCP and patients who did not receive vCP. A Hosmer–Lemeshow goodness-of-fit test was performed for the model for risk of death and found a chi-square value of 17.6 with a P-value of .20, which suggests that the model fits the data well. For the model on the risk of unplanned return to the OR a Hosmer–Lemeshow goodness-of-fit test was performed and found a chi-square value of 7.22 with a P-value of .512, which suggests that the model fits the data well.
To account for possible survival bias, the model analyzing risk of complication only included a subset of patients who survived the entire index hospitalization. We also performed a subset analysis on patients with severe TBI. All P-values were two-sided, with a statistical significance level of < .05. All statistical analyses were performed with IBM SPSS Statistics for Windows, Version 28. (Armonk, NY: IBM Corp).
Results
Demographics, Comorbidities, and Injury Profile for trauma patients with cirrhosis who received vs not received vCP.
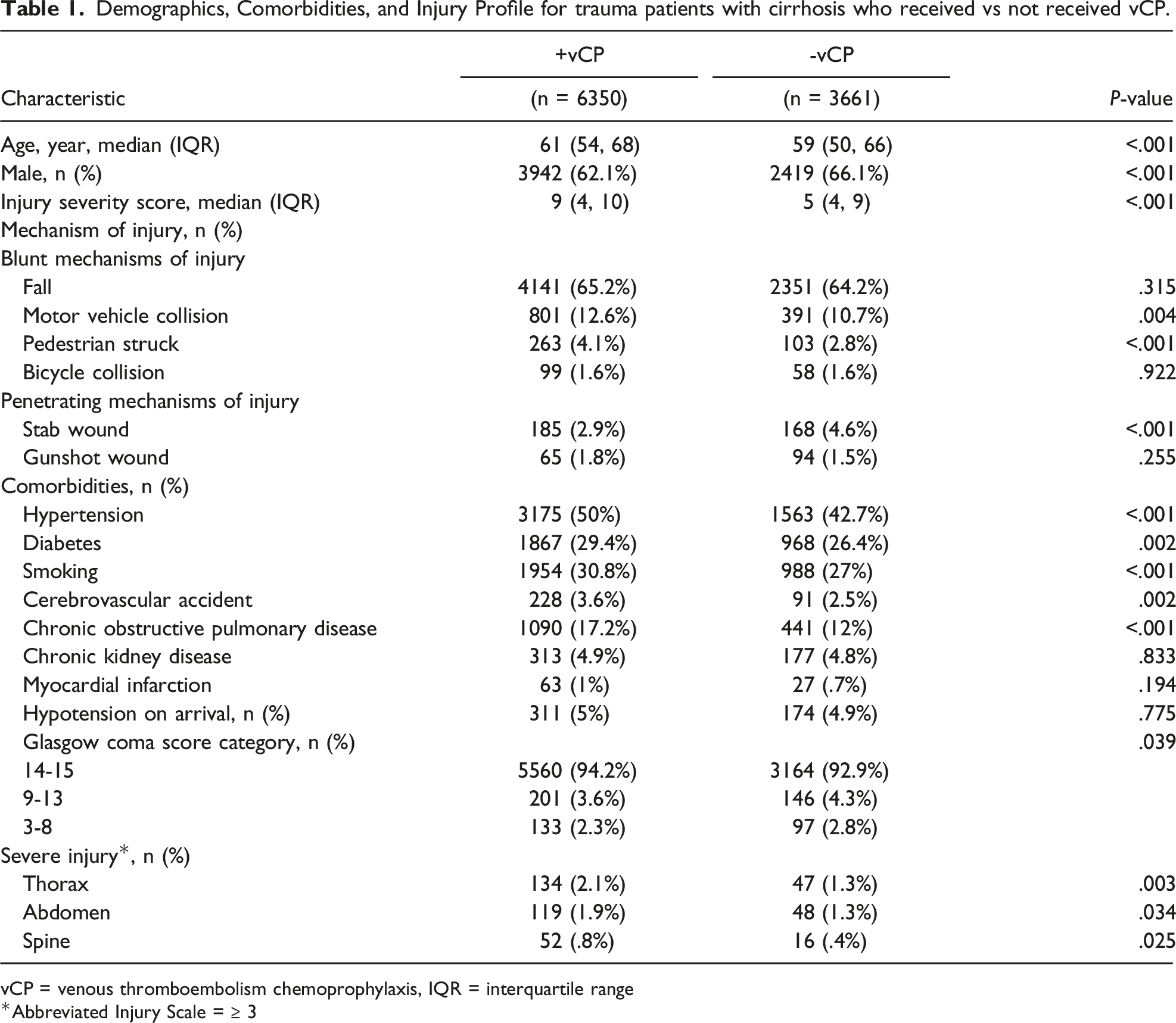
vCP = venous thromboembolism chemoprophylaxis, IQR = interquartile range
Abbreviated Injury Scale = ≥ 3
Clinical Outcomes For Trauma Patients With Cirrhosis Who Received Versus Not Received vCP.
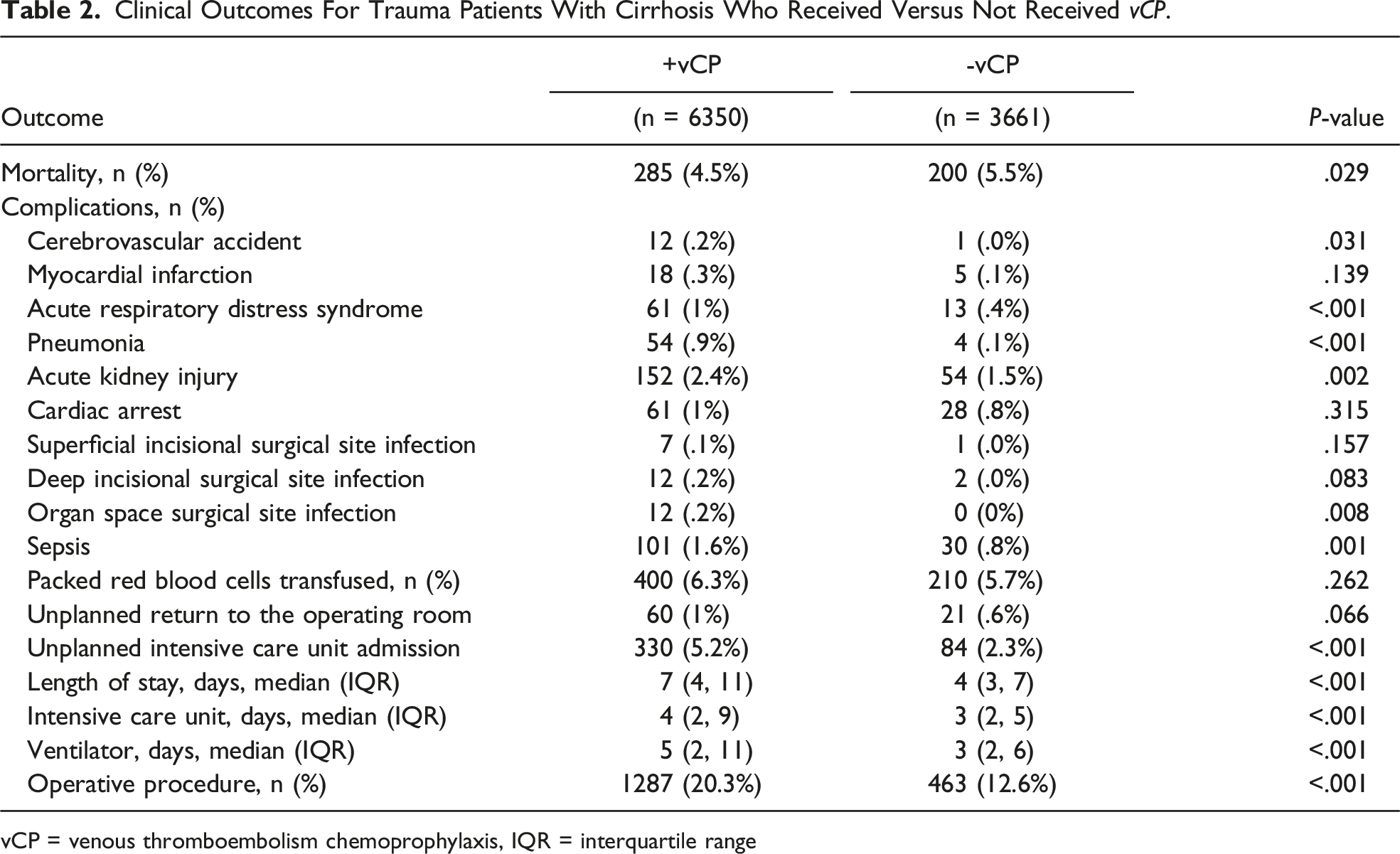
vCP = venous thromboembolism chemoprophylaxis, IQR = interquartile range
Multivariable Logistic Regression Analyses* for Risk of Mortality and Unplanned Return to the OR in Adult Trauma Patients With Cirrhosis Who Received Vcp Vs Did Not Receive vCP.
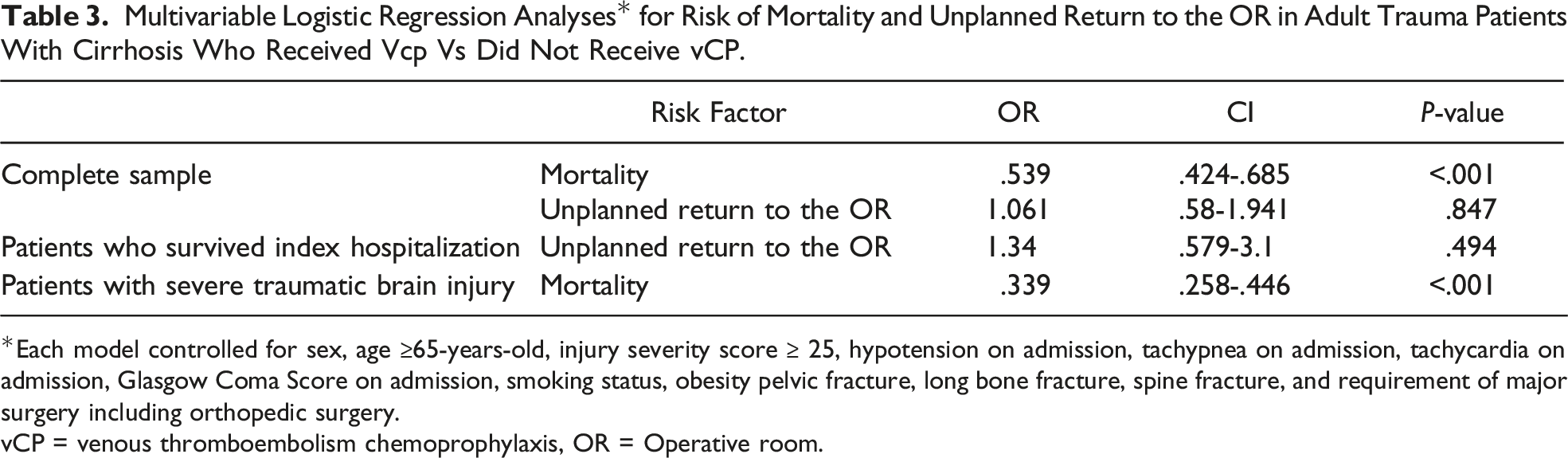
Each model controlled for sex, age ≥65-years-old, injury severity score ≥ 25, hypotension on admission, tachypnea on admission, tachycardia on admission, Glasgow Coma Score on admission, smoking status, obesity pelvic fracture, long bone fracture, spine fracture, and requirement of major surgery including orthopedic surgery.
vCP = venous thromboembolism chemoprophylaxis, OR = Operative room.
On subgroup analysis of patients who survived index hospitalization, there was a similar risk of unplanned return to the OR (OR = .968, CI 0.490-1.911, P = .925) between cohorts (Table 3).
In a subset analysis of trauma patients with cirrhosis and a serious head injury, patients receiving vCP still had a decreased associated risk of mortality (OR = .339, CI 0.258-.446, P < .001) compared to patients not receiving vCP (Table 3).
Discussion
The decision for the use of vCP in trauma patients with cirrhosis is difficult and lacks significant evidence. The results of this national analysis show that vCP is administered to less than two-thirds of trauma patients with cirrhosis staying in the hospital for more than two days. Confirming our hypothesis, vCP was associated with a decreased associated risk of mortality but a similar risk of unplanned return to the OR, suggesting vCP is safe at least in select patients determined by practicing trauma surgeons.
The use of vCP in trauma patients with cirrhosis is still controversial.17,18 Available studies have reported vCP practices in cirrhotic patients hospitalized for medical conditions to be very low. A recent meta-analysis including 19 million patients reported that just over 25% of patients with cirrhosis hospitalized for medical conditions received vCP. 8 This current study focused only on trauma patients and found that nearly 65% of patients with cirrhosis hospitalized for longer than two days received vCP. The significantly higher rate of vCP in trauma patients compared to medical patients can be explained by the fact that trauma patients are inherently at higher risk for VTE, and vCP is almost always part of an ICU and trauma checklist during rounds. Additionally, trauma surgeons may be more comfortable identifying and managing bleeding, whereas VTE and specifically a pulmonary embolism represents a potentially preventable cause of death.
Patients with cirrhosis represent a high-risk population for hemorrhage and, thus, unplanned return to the operating room or death. Therefore, the safety of vCP in patients with cirrhosis is of significant concern. A single-center retrospective cohort study of non-trauma patients with cirrhosis demonstrated that patients receiving vCP had a lower risk of bleeding compared to those receiving mechanical or no vCP. 4 Yerke, J. et al reported a similar finding in a single-center retrospective study, where vCP administration to hospitalized non-trauma patients with cirrhosis was associated with decreased risk of major bleeding. 14 In our study, vCP administration in trauma patients did not increase the risk of unplanned operations or transfusions and was associated with decreased risk of mortality. These results also remained true in a subset of patients with severe TBI. This suggests that vCP in trauma patients with cirrhosis is likely safe and beneficial. However, the decision to start vCP needs to be individualized, including assessment of the degree of cirrhosis as determined by the Child-Pugh classification or Model for End-Stage Liver Disease (MELD) scores and other laboratory abnormalities (e.g., thrombocytopenia and viscoelastic assays), which may impact decisions and outcomes.19,20 Future research using a more granular dataset may help elucidate the relative impact of these variables on the overall risk of morbidity and mortality in trauma patients with cirrhosis and thereby inform guidelines on exactly when to initiate vCP.
This study has multiple limitations inherent to its retrospective database design, such as coding errors, missing information, and reporting bias. Given the retrospective design, the findings cannot be interpreted as causation, and there is likely some degree of selection bias between the cohorts despite a robust attempt to control for potential confounders. Also, this study lacks pertinent variables, including the dosage and timing of vCP and the rate of missed dosing, which have been demonstrated to be associated with VTE. 11 In addition, TQIP lacks data regarding therapeutic levels (e.g., anti-Xa) of vCP and other factors associated with VTE, such as the use of subcutaneous compression devices and ambulation. Furthermore, we did not account for differences in cirrhosis severity (e.g., Child-Pugh classification or MELD scores), including thrombocytopenia and international normalized ratio. Despite these limitations, this study is strengthened by its large national population.
Conclusion
Current guidelines for vCP in trauma lack specific recommendations for trauma patients with cirrhosis. Misinterpretation of hemostatic alterations can predispose clinicians to avoid the use of vCP for fear of increased bleeding risk, as seen by the fact that over one-third of cirrhotic trauma patients staying in the hospital for more than two days did not receive vCP. In addition, vCP was found to be associated with a decreased risk of mortality and a similar rate of unplanned return to the OR. In conjunction, these results suggest that the use of vCP may be safe in trauma patients with cirrhosis. However, further prospective research is needed to validate these findings and develop guidelines to help standardize care for this high-risk subset of trauma patients with cirrhosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
