Abstract
Background
Limited evidence exists assessing whether anastomotic evaluation using indocyanine green fluorescence (IGF) during minimally invasive esophagectomy (MIE) predicts or improves outcomes. We hypothesized that IGF helps surgeons predict anastomotic complications and reduces anastomotic leaks after MIE.
Methods
In September 2019, our institution began routinely using IGF for intraoperative evaluation of anastomoses during MIE. Data were collected from patients undergoing MIE in the two years before and after this technology began being routinely used. Baseline characteristics and outcomes, including anastomotic leak, in patients who underwent indocyanine green fluorescence evaluation (ICG) and those who did not (nICG) were compared. Outcomes were also compared between ICG patients with normal versus abnormal fluorescence.
Results
Overall, 181 patients were included. Baseline demographic and clinical characteristics did not differ between the ICG and nICG groups. ICG patients experienced higher rates of anastomotic leak (10.2% vs. 1.6%, P = .015) and 90-day mortality (8.5% vs. 1.6%, P = .04) compared to nICG patients. Due to lack of equipment availability, 19 nICG patients underwent MIE after the use of IGF became routine, and none developed leaks. ICG patients with abnormal fluorescence had higher rates of anastomotic leak (71.4% vs 1.9%, P < .001) and 30-day mortality (28.6% vs 0%, P = .012) compared to those with normal fluorescence.
Discussion
Abnormal intraoperative IGF was associated with increased rate of anastomotic leak, suggesting predictive potential of IGF. However, its use was associated with an increased leak rate and higher mortality. Further studies are warranted to assess possible physiologic effects of indocyanine green on the esophageal anastomosis.
Key Takeaways
• Use of intraoperative indocyanine green fluorescence (IGF) imaging during esophagectomy is an emerging application of IGF, and its effect on patient outcomes is still under investigation. • IGF results that were judged by operating surgeons to be abnormal were associated with significantly increased risk of anastomotic leak, suggesting that IGF results could help predict complications. • Patients who underwent IGF imaging had a higher rate of anastomotic leak, so future studies should continue to evaluate the safety and physiologic effects of IGF during esophagectomy.
Introduction
Despite improving survival rates over the past several decades due to treatment innovations, esophageal cancer continues to have one of the lowest survival rates for all cancers at all stages with an overall 5 year survival of 20%.1,2 The cornerstone of curative treatment for patients with locally advanced esophageal cancer is resection, usually combined with chemoradiation depending on the clinical stage of presentation. Currently, minimally invasive esophagectomy (MIE) is a widely accepted surgical approach. 3 While esophagectomy improves overall survival, it carries its own morbidity and mortality risks. 4
Anastomotic leak is a feared complication of esophagectomy and occurs in approximately 2–25% of patients following esophageal resection and anastomosis.5‐9 Mortality associated with esophageal anastomotic leaks may be as high as 12%. 7 Risk factors for anastomotic leakage include advanced age, diabetes, heart failure, hypertension, smoking, and renal disease.4,10 Healing of the anastomotic region is related to adequate arterial perfusion and optimal tension of the anastomosis, both of which are surgically modifiable and serve to prevent leakage. 11
The use of intraoperative indocyanine green fluorescence (IGF) imaging is emerging as a means to assess the blood supply after anastomosis. 12 Indocyanine green is injected intravenously and then an infrared camera is used to visualize the blood supply in the form of fluorescence around the anastomosis. 11 While the use of IGF has been described previously in colorectal surgical cases, its use in esophagectomy cases is relatively new, and current evidence on whether anastomotic evaluation using IGF during MIE can predict esophageal anastomotic leak occurrence or improve outcomes is mixed.8,11,13,14 In this study, we aimed to assess the predictive value of IGF and whether its use improves patient outcomes. We hypothesized that its use reduces anastomotic leaks and effectively predicts anastomotic complications after MIE. We found that while IGF evaluation of the anastomosis may be helpful in predicting anastomotic complications, its use was associated with a higher rate of anastomotic leaks.
Methods
Study Population
The study was approved by the Kaiser Permanente Northern California (KPNC) Institutional Review Board with a waiver of the requirement for informed consent. We reviewed all adult patients undergoing MIE at Kaiser Permanente Oakland between September 1, 2017 and November 15, 2021. In September 2019, Kaiser Permanente Oakland began routine use of IGF for intraoperative evaluation of the anastomosis during MIE. Therefore, the study period included all cases from approximately two years before and after this change in protocol.
Study Design
Patients were identified via diagnosis and procedure codes from KPNC electronic medical records (EMR). Baseline demographic characteristics such as age, sex, and race/ethnicity as well as clinical characteristics such as body mass index (BMI), Charlson Comorbidity Index (CCI), smoking history, prior chemotherapy, prior radiotherapy, intraoperative anastomotic rating, operative duration, cancer histology, and stage were extracted from EMRs and confirmed by manual chart review. The primary outcome of interest was anastomotic leak. Secondary outcomes included anastomotic stricture, time until liquid oral tolerance, hospital length of stay, requirement for repeat interventions, readmission, 30-day mortality, and 90-day mortality.
Baseline characteristics and outcomes were compared between patients who underwent intraoperative indocyanine green fluorescence (ICG group) and patients who did not (nICG group). Among patients who underwent indocyanine green fluorescence, we also compared subgroups of patients based on whether the operating surgeon determined that they had normal or abnormal fluorescence of the anastomosis at the time of surgery. Of note, the abnormal fluorescence group included anastomoses that were considered to have borderline or abnormal fluorescence.
Technique
Each MIE was performed by one of five thoracic surgeons within our institution with minimal variability in technique. Each patient underwent a minimally invasive Ivor Lewis esophagectomy with a stapled anastomosis as we have previously described in detail.
9
Upon stapling of the intrathoracic anastomosis, indocyanine green is injected peripherally and subsequent fluorescence was visualized along the anastomosis. Upon inspection, the operating surgeon graded the anastomotic perfusion and recorded results in the operative note (Figure 1). Example of borderline indocyanine green fluorescence of the gastroesophageal anastomosis. Short arrow represents the anastomosis. Long arrow represents the gastric conduit.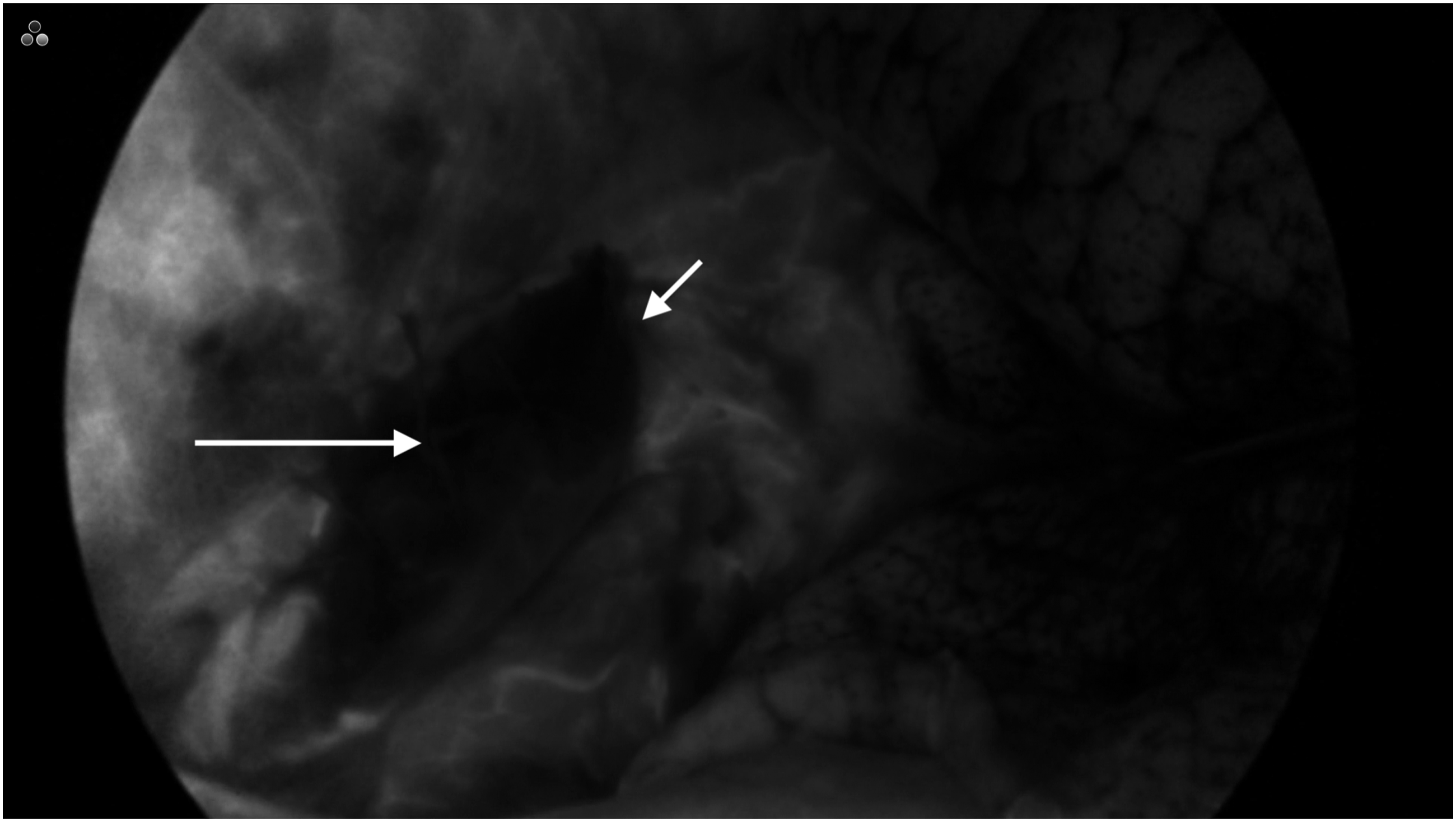
Statistical Analysis
Baseline characteristics between ICG and nICG groups were assessed in bivariate analyses using Chi-square tests and Fisher exact tests for categorical variables and t-tests for normally distributed continuous variables. Two sample Wilcoxon rank sum tests were used for non-normally distributed continuous variables. All analyses were performed using SAS v9.4 (Cary, NC, USA).
Results
Overall Cohort Demographics and Clinical Characteristics.
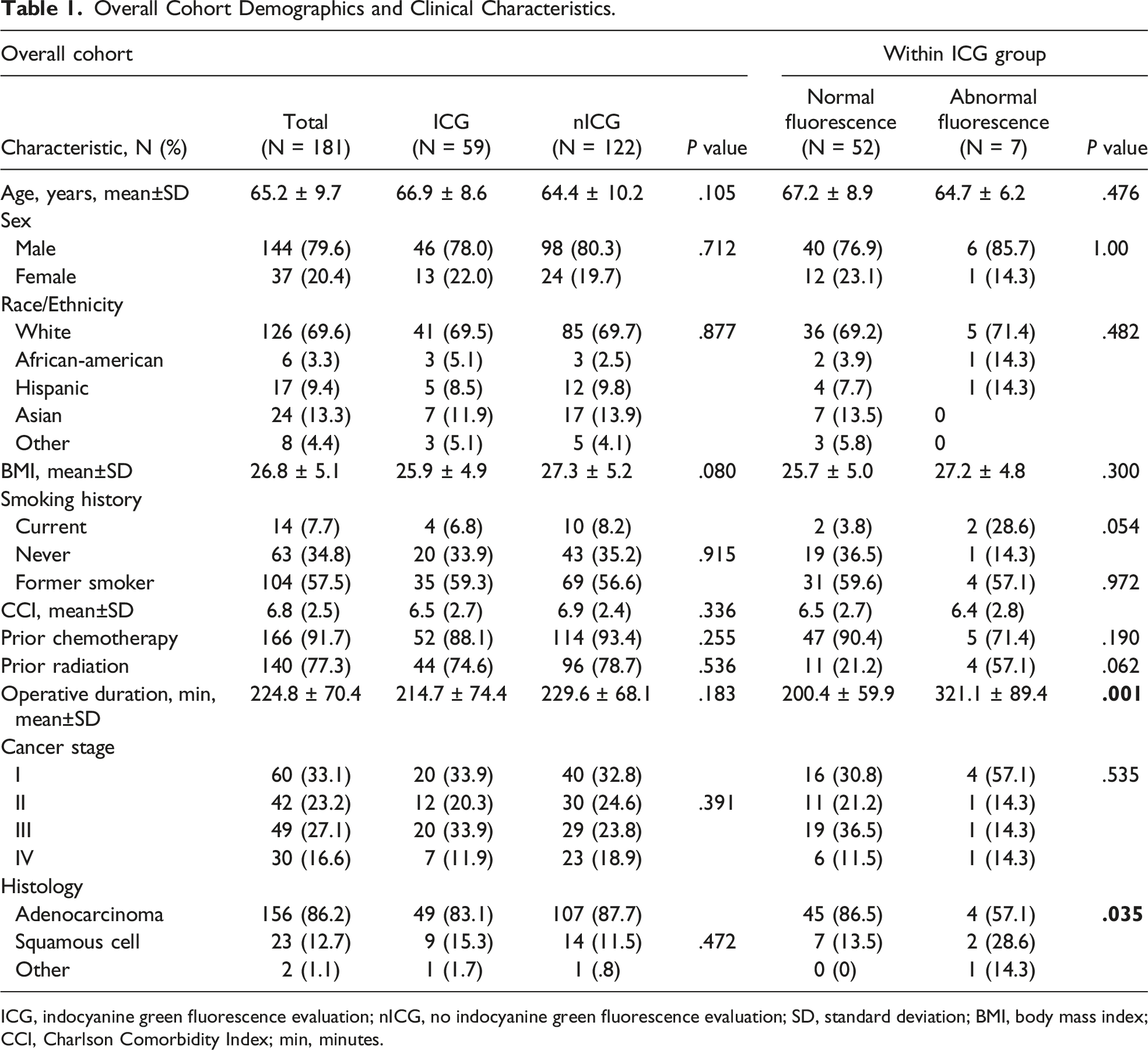
ICG, indocyanine green fluorescence evaluation; nICG, no indocyanine green fluorescence evaluation; SD, standard deviation; BMI, body mass index; CCI, Charlson Comorbidity Index; min, minutes.
ICG Fluorescence
Comparison of Minimally Invasive Esophagectomy Patient Outcomes with and without Indocyanine Green Fluorescence Evaluation.
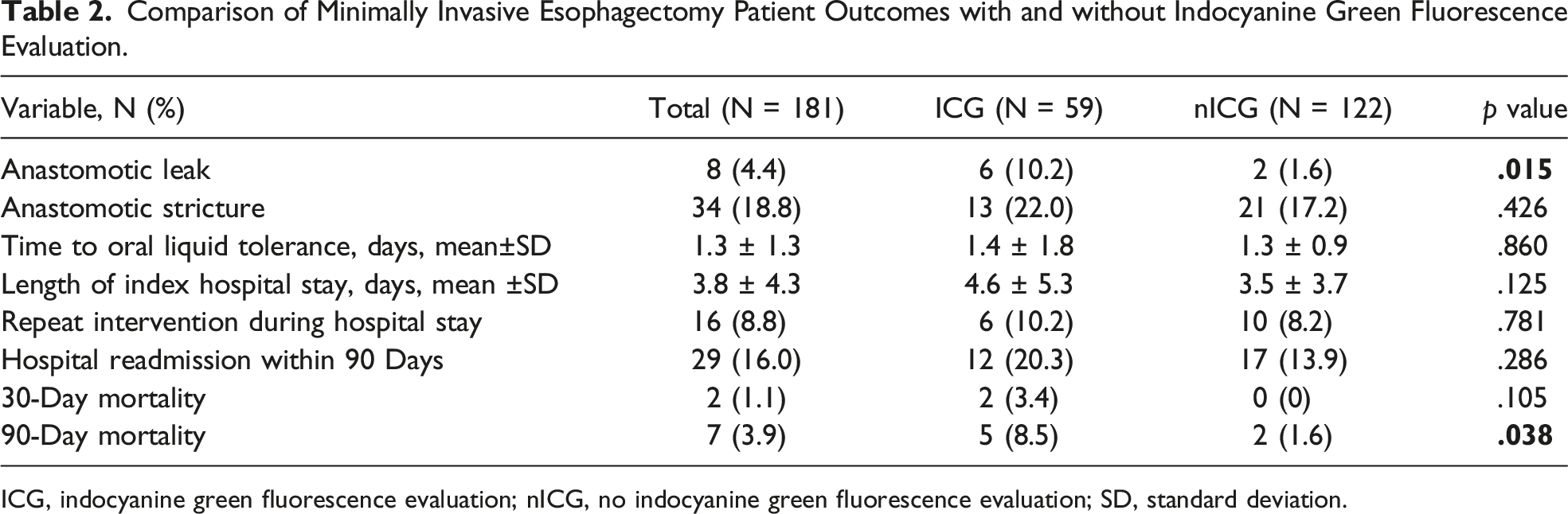
ICG, indocyanine green fluorescence evaluation; nICG, no indocyanine green fluorescence evaluation; SD, standard deviation.
Of the seven patients who died within 90 days of surgery, three had developed anastomotic leaks. Two of the patients with leaks had developed bronchoesophageal fistulas. The other four patients died from aspiration pneumonia, congestive heart failure exacerbation, diabetic ketoacidosis, and complications from metastatic disease.
Intraoperative Anastomosis Classification
Normal ICG Fluorescence vs. Abnormal ICG Fluorescence Outcomes.
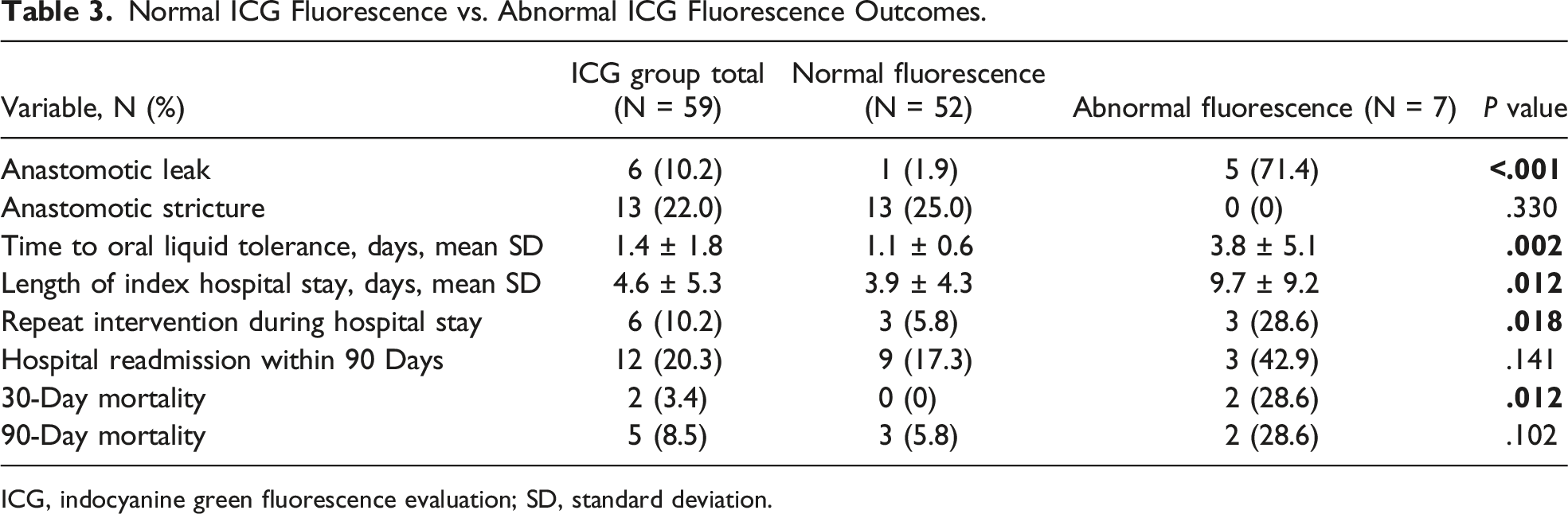
ICG, indocyanine green fluorescence evaluation; SD, standard deviation.
Discussion
Although patients who underwent IGF evaluation of the anastomosis during MIE had higher rates of anastomotic leak, the degree of fluorescence in patients who received IGF was predictive of outcomes. Though somewhat paradoxical, these results highlighted that IGF may be helpful in predicting which esophagectomy cases may result in an anastomotic leak. This study adds to the limited literature on the use of IGF in esophagectomy procedures, which is a relatively new and emerging application of indocyanine green.
Those who were deemed to have abnormal IGF by the operating surgeon had significantly increased rates of anastomotic leak, repeat interventions during hospitalization, and 30-day mortality. These patients with abnormal IGF also had longer operative times, though this correlation likely results from the fact that cases with abnormal fluorescence may be more difficult and may carry higher risks of complications, including anastomotic leak and conduit necrosis.15,16 Length of stay was not noted to be statistically different between the two groups, though, the mean length of stay in the nICG group of 3.5 days was closer to our prior data (3.6 days). 9 As previously described, our patients’ length of stay is largely facilitated by our standardized post-operative protocol. 9 This involves early oral nutrition and ambulation with eligibility for discharge on post-operative day three assuming the patient meets several discharge criteria such as absence of leak on esophagram, lack of tachycardia, absence of fever, and ability to sustain hydration and nutrition with over two liters of fluid orally per day. 9
The correlation between abnormal IGF and significantly increased rates of anastomotic leak suggested that abnormal IGF may be helpful in predicting which cases may be complicated by anastomotic leak and could potentially be managed in a different fashion than a standard esophagectomy case with normal IGF intraoperatively. Previous studies have shown that IGF results can predict increased risk of leakage in colorectal surgical cases; however, the use of IGF and its impact on outcomes after esophagectomy has not been as well described and has yielded mixed results.8,11,13,14,17 While some studies suggest that IGF can be used to predict which areas have adequate perfusion, others have found that IGF evaluation did not lead to reduced anastomotic leakage rates.11,14 Therefore, the utility of IGF in esophagectomy remains unclear.
The correlation between abnormal IGF and increased complications suggests that surgeon interpretation of IGF results may effectively assess whether anastomotic regions have “adequate” or “inadequate” perfusion. Therefore, future studies should continue to evaluate whether changes in management in response to abnormal IGF improve patient outcomes.
Pertinently, the intraoperative course for all patients in this study was unchanged in response to abnormal IGF results, and no revisions to the anastomotic site were performed in response to abnormal IGF results. This was based on surgeon discretion, as the severity of the abnormality in these patients was not deemed significant enough to cause a change in the operative course. In each of these cases, the fluorescence was considered to be borderline rather than frankly abnormal. These cases were also likely technically more difficult with higher inherent risk of anastomotic leak as evidenced by their longer durations. However, given the higher anastomotic leak rate among patients with even borderline fluorescence, these cases may be candidates for intraoperative revision if feasible.
Whether or not the anastomosis is revised, understanding which patients are at particularly high risk of anastomotic leak may still prove beneficial as it can heighten a surgeon’s degree of suspicion for anastomotic leak in the post-operative period. This can potentially lead to faster identification and intervention for patients who do develop leaks.
The higher rates of anastomotic leak among ICG patients compared to nICG patients were somewhat surprising and raise some concerns. In general, the ICG group in our study is a more recent cohort that was compared to a historical cohort, so it is possible that an unidentified confounding factor may have changed at the same time of implementation of indocyanine green use. However, 19 patients in this later period did not undergo IGF imaging simply due to an unexpected lack of equipment availability at the time of surgery. None of the patients in this unintentional subgroup of the later temporal cohort developed leaks, which raises concern regarding the use of indocyanine green. It is also worth emphasizing that age, BMI, Charlson Comorbidity Index, smoking history, operative duration, stage, and histology were not different between the ICG and nICG groups. While prior studies on IGF imaging in esophagectomy have suggested that it is generally safe, studies assessing its physiology specifically related to esophageal tissue are lacking. 8 Given the retrospective nature of our study, we are unable to definitively identify a cause for an increase in anastomotic leaks among the ICG group. Overall, the anastomotic leak rate of 10.2% in the ICG group is similar to the NSQIP data, and part of the reason we may have noticed an increase in anastomotic leak rate in our ICG group is that, historically, our leak rate has been lower at 2.1%. 9 A prospective, randomized trial may be able to shed further light on any possible detrimental effect of indocyanine green on the gastroesophageal anastomosis. Additionally, further assessment of indocyanine green in terms of vasoactivity or toxicity within esophageal tissue may help identify potential concerns with its use. Further investigation is warranted because indocyanine green could be a helpful adjunct in identifying high risk anastomoses but only if its use does not cause harm.
Our study results should be interpreted in the context of its limitations. First, the lack of randomization of our patient groups affected our ability to limit confounders. Additionally, the evaluation of IGF results was inherently subjective. However, the success among our surgeons in interpreting normal vs. abnormal fluorescence was evident in the adverse outcomes associated with patients deemed to have abnormal fluorescence at the anastomosis.
Overall, while our study showed the value of IGF imaging in terms of predicting anastomotic complications, our findings of increased risk of anastomotic leak upon its use were concerning. Further studies are warranted to further elucidate its safety and its physiologic effects in the setting of esophageal surgery.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Kaiser Permanente Northern California Graduate Medical Education Program and Kaiser Foundation Hospitals.
