Abstract
Background
Hispanic individuals have a disproportionately higher incidence and mortality for stomach, cervix, and liver cancers compared to Non-Hispanic White people. Since disparities in cancer incidence are influenced by multiple factors including immigration, elucidating the effect of birthplace and exposure to risk factors on the prevalence of these cancers is crucial for identifying high-risk populations and target risk reduction interventions.
Methods
The National Institutes of Health All of Us Research Program is a prospective, multidimensional biomedical data resource of underrepresented, minoritized people. The Registered Tier Dataset v5 was utilized to evaluate the prevalence and risk of stomach, cervix, and liver cancers among United States (US) born and non-US born Hispanic participants.
Results
Of over 434 000 current participants, 60 540 are Hispanic; 30 803 (50.9%) reported being US born and 29 294 (48.4%) non-US born. Non-US born Hispanic participants had significantly higher prevalence (.39% vs .21%, P < .001) and associated risk (OR 1.84, 95% CI 1.29-2.64, P < .001) of liver cancer, and trend towards higher prevalence of stomach (.14% vs .09%, P = .076) and cervix cancers (.27% vs .20%, P = .083) compared to US born counterparts. US born Hispanic patients with these 3 cancers were significantly younger than non-US born cohort (mean age 56.8 vs 61.7 years, P < .001).
Discussion
This is the first report using All of Us data to show that non-US born Hispanic participants have a higher risk of liver cancer compared to US born participants. Further analyses, including genomic studies, are necessary to understand these differences and identify targets for risk reduction interventions.
Key Takeaways
Hispanic individuals born outside of the US had significantly higher prevalence and risk of primary liver cancer compared to US born counterparts. US born Hispanic patients with stomach, cervix, and liver cancers were significantly younger than non-US born Hispanic patients.
Introduction
The Hispanic population is the second largest group in the United States (US) after non-Hispanic White (NHW) people. 1 Hispanic individuals accounted for 18.5% of the US population in 2019 and are predicted to reach 28% or 111 million people by 2060 according to US Census Bureau 2 data. Hence, health disparities that affect this group are a major public health issue.
Worldwide, cancer is the leading cause of death in Hispanic people, even though the leading cause of death in the US and among NHW people is heart disease. 3 Although the incidence and mortality rates of common cancers such as colorectal, lung, and breast are lower among Hispanic individuals, they have a higher incidence of infection-related cancers, namely, stomach, cervix, and liver compared to NHW individuals. 3 Several risk factors such as lifestyle, socioeconomic status, access to health care, and exposures have been associated with higher incidence. However, there is paucity of studies that evaluate the interplay of sociocultural, socioeconomic, behavioral, and biologic risk factors on the overall incidence and outcomes of these cancers. Thus far, information regarding cancer risk among the Hispanic population and ethnic subgroups have been mostly derived from large retrospective, cancer registry studies which typically describe factors associated with risk rather than causation. Genomic studies utilizing large datasets such as The Cancer Genome Atlas (TCGA) lack representation of minoritized groups, particularly Hispanic patients.4,5 Nevertheless, to design meaningful interventions to reduce cancer risk, a multidimensional risk assessment based on a comprehensive database along with genomic data that encompasses diverse, underrepresented groups is essential.
The All of Us Research Program, funded by the National Institutes of Health (NIH), is a rich biomedical database that includes persons from underrepresented demographic categories in biomedical research. The program collects data on race, ethnicity, age, sex, gender, sexual orientation, disability status, access to care, income, educational attainment, and geographic location. Thus, this database captures medical data related to diseases, as well as environmental, social, economic, and genomic components of health. Adults (≥18 years old) can enroll, including healthy participants. Data from surveys, electronic health records (EHR), physical measurements, biosamples, and wearable devices are collected from the participants and stored on a common cloud environment online for researchers to access. Enrollment opened in May 2018 with plans to enroll at least 1 million people. 6 Currently, the program has over 434 000 participants and is an excellent resource to study multidimensional risk in minoritized people.
Since Hispanic people in the US represent US and non-US born individuals, the influence of birthplace on the multidimensional risk of these cancers needs to be further studied. Therefore, we aimed to evaluate the impact of birthplace and exposure to known risk factors among the Hispanic population using the All of Us database. This study provides an initial step to understand the multidimensional risk of highly prevalent cancers among Hispanic people and identify a specific “at-risk” group to target screening efforts and interventions.
Methods
We performed a retrospective study using the All of Us Registered Tier Dataset v5 (May 2018 to November 2021). Hispanic participants ≥18 years old with primary stomach, uterine cervix, or liver cancers were included using Systematized Nomenclature of Medicine (SNOMED) diagnosis codes (Appendix A). Participants with secondary malignancies of the liver, neuroendocrine tumors and lymphomas were excluded (Appendix A). The primary outcomes were the prevalence and risk of stomach, cervix, and liver cancers among Hispanic participants based on birthplace.
Demographic variables including age, gender, education level, socioeconomic status, insurance status, and comorbidities were collected. Survey data from the following questions “what is the highest grade or year of school you completed,” “what is your current employment status,” “what is your annual household income from all sources,” and “are you covered by health insurance or some other kind of health care plan” were used to understand level of formal education completed, socioeconomic status, and insurance, respectively. Smoking was also determined from the survey question “do you now smoke cigarettes every day, some days or not at all?” Participants who answered yes to smoking every day and some days were included in the smoking variable. Alcohol use was similarly determined by the survey question “on a typical day when you drink, how many drinks do you have?” Participants who answered to drinking 3-4 drinks/day or more were counted in the alcohol use variable. Comorbidities including obesity (BMI ≥ 30), diabetes (SNOMED 73211009), hypertension (SNOMED 38341003), cirrhosis (SNOMED 19943007), Helicobacter pylori (H. pylori) (SNOMED 721730009), human papillomavirus (HPV) (SNOMED 720005005), and hepatitis (SNOMED 235862008) were also analyzed.
Univariate logistic regression analyses were performed for stomach, cervix and liver cancers separately with the following variables for all 3 cancers: age (1 year increase in age), gender, birthplace, obesity, income level, unemployment, education, and insurance. Additionally, cirrhosis and hepatitis for liver cancer, and HPV and HIV for cervix cancer were included. Multivariable logistic regression analysis was performed after excluding the factors that were not significant on univariate analysis (insurance, education, income, and employment). Smoking and alcohol use were also not included in the analysis due to a large number of missing data.
Categorical data was reported as percentages and analyzed using chi-square test. Continuous variables were reported as means with standard deviations and analyzed with two-sample t-test. Results from the multivariable logistic regression analysis were reported as odds ratio (OR) with 95% confidence interval (CI). Statistical significance was set at P-value < .05. All analysis was completed in the Jupyter notebook using R.
Results
Demographics of US Born and Non-US Born Hispanic Participants
Demographics of US Born and Non-US Born Hispanic Participants.
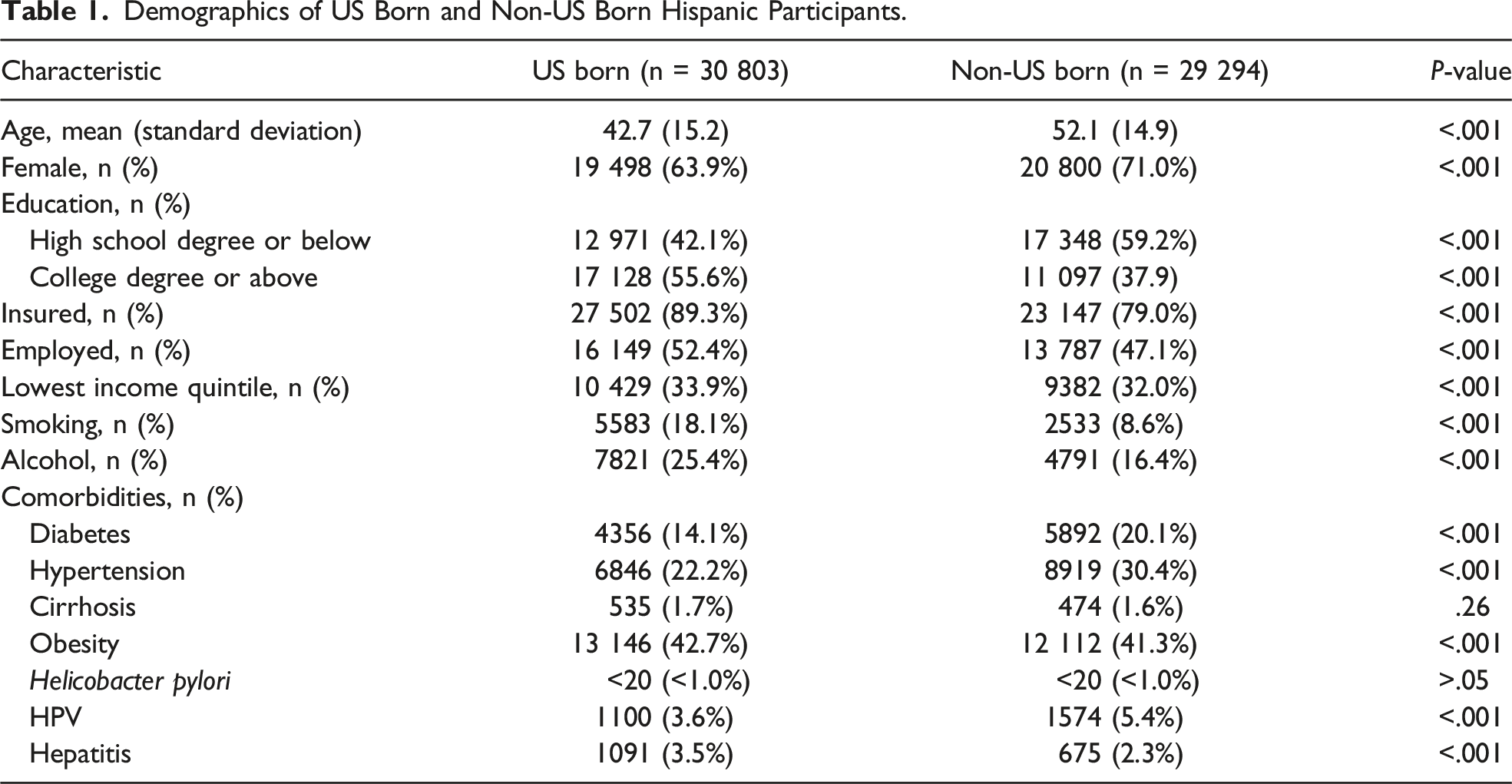
When we analyzed comorbidities among Hispanic participants, non-US born participants had significantly higher rates of diabetes (20.1% vs 14.1%, P < .001) and hypertension (30.4% vs 22.2%, P < .001) but lower rates of obesity (41.3 vs 42.7%, P < .001) compared to the US born group. There was no significant difference in the rate of cirrhosis (1.6% vs 1.7%, P = .26) between the 2 groups (Table 1).
As stomach, cervix, and liver cancers are related to H. pylori, HPV, and hepatitis, respectively, we also analyzed the rates of these infections among Hispanic participants. There were less than 20 participants with H. pylori reported in both US born and non-US born groups. Non-US born Hispanic participants had significantly higher rates of HPV (5.4 vs 3.6%, P < .001) but lower rates of hepatitis (2.3% vs 3.5%, P < .001) than US born Hispanic participants (Table 1).
Prevalence and Risk of Stomach, Cervix and Liver Cancer Among US Born and Non-US Born Hispanic Patients
Next, we investigated the prevalence of stomach, cervix and liver cancers among Hispanic participants. The prevalence of liver cancer in non-US born Hispanic participants was almost 2-fold higher than in US born participants (.39% vs .21%, P < .001). The prevalence of stomach (.14% vs .09%, P = .076) and cervix (.27% vs .20%, P = .083) also trended higher in Hispanic participants born outside of the US compared to those born in the US but was not statistically significant (Supplemental Table).
Multivariable Logistic Regression Analysis of Risk Factors for Stomach Cancer Among Hispanic Participants.
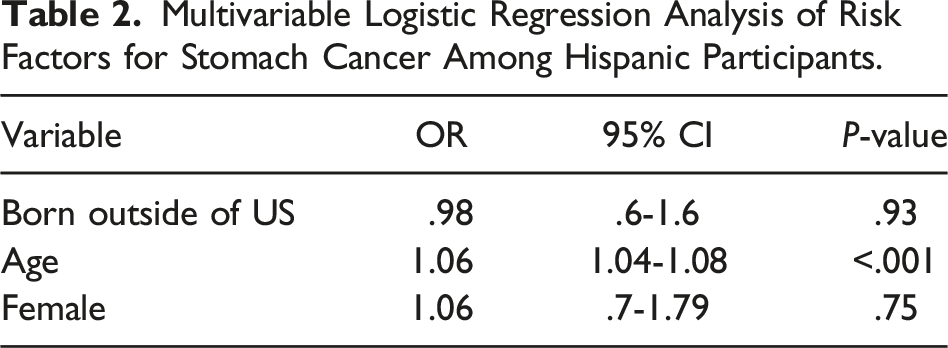
Multivariable Logistic Regression Analysis of Risk Factors for Cervix Cancer Among Hispanic Participants.

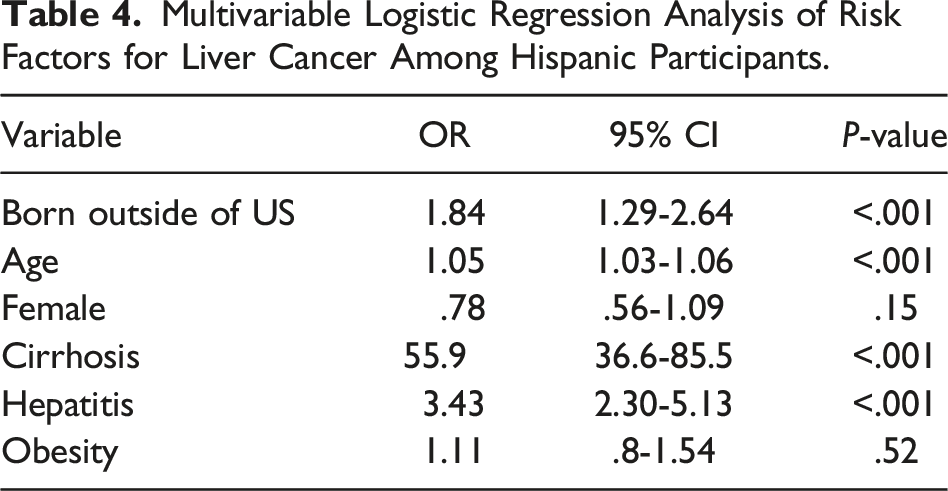
Multivariable Logistic Regression Analysis of Risk Factors for Liver Cancer Among Hispanic Participants.
Risk Factors for Liver Cancer Among US Born and Non-US Born Hispanic Patients
Analysis of rates of risk factors (diabetes, cirrhosis, smoking, alcohol, hepatitis, and obesity) for Hispanic patients with liver cancer showed a significantly higher rate of obesity in the US born group compared to the non-US born group (54.7% vs 38.9%, P = .043). In addition to birthplace, cirrhosis (OR 55.9, 95% CI 36.6-85.5, P < .001) and hepatitis (OR 3.43, 95% CI 2.30-5.13, P < .001) were independent risk factors for liver cancer among Hispanic participants based on multivariate logistic regression analysis (Table 4).
Discussion
Although Hispanic men and women have a lower incidence and mortality rates for all cancers combined, they have a disproportionately higher incidence of infection-related cancers with 2-fold higher incidence of stomach and liver cancers compared to NHW people and 38% higher incidence of cervix cancers compared to NHW women.3,7 Risk of stomach, cervix, and liver cancers, is greatly influenced by factors such as lifestyle, socioeconomic status, exposure to infectious agents, access to health care, and screening. Studies to evaluate this disparate incidence should focus on the interplay of multiple interdependent factors that ultimately influence genetic and epigenetic changes and cancer risk.
Our study investigating the prevalence and risk of stomach, cervix, and liver cancers among Hispanic participants in the All of Us Research Program based on birthplace is an attempt to understand the prevalence and causality of risk factors. We observed that non-US born Hispanic participants have a significantly higher risk of liver cancer (OR 1.84, 95% CI 1.29-2.64) compared to US born Hispanic participants. This observation differs from some of the previously published reports that have reported higher incidence of hepatocellular carcinoma (HCC) in US born Hispanic men.8-10 A California-based, multi-ethnic cohort study of nearly 37 000 Hispanic individuals and 189 incident cases of HCC, reported the incidence rate of HCC was twice as high in US born vs non-US born Hispanic men (44.7 vs 23.1), but comparable in women (14.5 vs 13.4). 10 We did not analyze prevalence by sex in our study because two-thirds of the Hispanic participants are women. Our data may also differ from observations from specific geographic regions because the All of Us database represents participants from the entire US. Birthplace did not significantly influence the prevalence of stomach and cervix cancers, perhaps due to low numbers of patients with these cancers in this study.
We observed US born Hispanic patients with stomach, cervix, and liver cancers were significantly younger than non-US born patients. Younger age at diagnosis for these 3 cancers in Hispanic patients compared to NHW patients have been reported previously.8,11,12 Studies have suggested that interplay between environmental influences and biologic susceptibility may contribute to the earlier onset of cancers among Mexican Americans.13,14 However, we do recognize our initial cohort of US born Hispanic participants was younger than non-US born Hispanic participants, which could also partially explain this age difference we observed among Hispanic patients.
It is also important to note that non-US born Hispanic participants in our study are more disadvantaged than US born Hispanic participants as evidenced by lower rates of post-secondary education, insurance and employment. Unfortunately, lower socioeconomic status has been reported to result in poor access to high-quality care, lower screening rates, delays in treatment, and low treatment adherence. 13 These socioeconomic differences may be contributing to the higher prevalence rates of stomach, cervix and liver cancer we observed among non-US born Hispanic participants compared to US born.
Since the most common etiologies of HCC are metabolic diseases (non-alcoholic fatty liver disease, obesity, hyperlipidemia, and metabolic syndrome), alcoholic liver disease, and Hepatitis B (HBV) and C virus (HCV) infections, the incidence of these risk factors was further evaluated. Similar to previously published reports, we observed higher rates of obesity in US born Hispanic patients. Hispanic people from different geographic regions such as Puerto Rico, Mexico, and Cuba have different liver cancer risks. 15 In a study using the Florida cancer registry that disaggregated immigrant Hispanic people using a Hispanic origin identification algorithm, Puerto Rican people had the highest rate of liver cancer compared to Hispanic individuals from Mexico and Cuba. 16 A subsequent study that described the association between HCC etiology and race and ethnicity reported that Puerto Rican men had high rates of alcohol liver disease HCC, while Afro-Caribbean and continental Hispanic men had high rates of HBV-HCC and metabolic HCC, respectively. 17 The exact birthplace for non-US born Hispanic participants was not available in the All of Us database, and hence disaggregation of Hispanic participants into subgroups was not possible and is a major weakness in the dataset.
Development of HCC is a multistep process with the accumulation of genetic and epigenetic changes that result in activation of oncogenes and inactivation of tumor suppressor genes. Particularly, epigenetic silencing of tumor suppressor genes by promoter hypermethylation or histone deacetylation plays an important role in carcinogenesis. 18 The most common somatic mutations in HCC occur in TERT promoter and CNNB1 and TP53 genes. 19 A genome wide association study using TCGA showed a higher rate of TP53 mutations (70%) in Black/African American patients compared to Caucasian patients (22.6%) and harboring deleterious TP53 mutations was associated with decreased overall and disease-free survival. 5 However, of the 373 patients included in this TCGA study, Asian and Caucasian patients represented 95% of the study cohort and there were no Hispanic patients.
For our future studies, we plan to use the upcoming genomic data in the All of Us database to study genetic changes associated with cancer risk. Although we found an increased prevalence and risk of liver cancers in non-US born Hispanic participants, they actually had significantly lower rates of certain risk factors for liver cancer including smoking, alcohol, obesity, and hepatitis compared to US born Hispanic participants. This suggests genetic predisposition and epigenetic modifications are also contributing factors to the prevalence of liver cancers and genomic studies need to be completed.
There are several limitation to our study, including missing, miscoding, or underreporting of data. Additionally, we cannot study Hispanic participants based on country-of-origin because this information is not available. Furthermore, over two-thirds of the participants were female; hence, the impact of sex on stomach and liver cancer risk could not be analyzed. Despite these limitations, our study is strengthened by the representation of Hispanic participants in the All of Us Research Program.
In summary, we have started a multidimensional risk assessment of highly prevalent cancers among Hispanic participants in the All of Us Research Program and have identified Hispanic people born outside of the US as a high-risk population for liver cancer. Further work aimed at investigating genetic and epigenetic contributions to cancer risk in Hispanic people needs to be completed to better understand the differences between those born in the US and outside of the US, with the ultimate goal to focus screening efforts and target risk reduction interventions to those at highest risk.
Supplemental Material
Supplemental Material - Prevalence of Primary Liver Cancer is Affected by Place of Birth in Hispanic People Residing in the United States: All of Us Research Program Report
Supplemental Material for Prevalence of Primary Liver Cancer is Affected by Place of Birth in Hispanic People Residing in the United States: All of Us Research Program Report by Jingjing Yu, MD, Brittany G. Sullivan, MD, Girish N. Senthil, Amber Gonda, PhD, Farideh Dehkordi-Vakil, PhD, Belinda Campos, PhD, Farshid Dayyani, MD, and Maheswari Senthil, MD in The American Surgeon
Footnotes
Acknowledgments
The authors would like to thank all the participants of the All of Us Research Program. We would also like to thank Dr Hoda Anton-Culver and others who are a part of the All of Us Research Program at the University of California, Irvine. The All of Us Research Program is supported by the National Institutes of Health.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Appendix
SNOMED Code
Diagnosis
Gastric Cancer
Included:
363349007
Malignant tumor of stomach
408647009
Adenocarcinoma of stomach
276809004
Early gastric cancer
447785000
Leiomyosarcoma of stomach
187742008
Malignant tumor of body of stomach
93717002
Primary malignant neoplasm of body of stomach
187732006
Malignant tumor of cardia
187734007
Malignant neoplasm of cardio-esophageal junction of stomach
93738008
Primary malignant neoplasm of cardia of stomach
254553001
Carcinoma of cardia
187741001
Malignant tumor of fundus of stomach
93809003
Primary malignant neoplasm of fundus of stomach
269460009
Malignant tumor of greater curve of stomach
93818001
Primary malignant neoplasm of greater curvature of stomach
269459004
Malignant tumor of lesser curve of stomach
93867004
Primary malignant neoplasm of lesser curvature of stomach
187740000
Malignant tumor of pyloric antrum
93976007
Primary malignant neoplasm of pyloric antrum
187736009
Malignant tumor of pylorus
93977003
Primary malignant neoplasm of pylorus
372014001
Primary malignant neoplasm of stomach
372143007
Carcinoma of stomach
Excluded:
721634009
Primary malignant neuroendocrine neoplasm of stomach
709830006
Malignant carcinoid tumor of stomach
276811008
Gastric lymphoma
Cervical Cancer
Included:
363354003
Malignant tumor of cervix
254887002
Adenocarcinoma of cervix
184781000119102
Priamry adenocarcinoma of cervix uteri
254888007
Adenosquamous carcinoma of cervix
188176007
Malignant neoplasm of endocervical canal
372097009
Malignant neoplasm of endocervix
123842006
Endocervical adenocarcinoma
93779009
Primary malignant neoplasm of endocervix
37298004
Carcinoma of endocervix
254886006
Squamous cell carcinoma of cervix
372099007
Malignant neoplasm of exocervix
93789008
Primary malignant neoplasm of exocervix
372024009
Primary malignant neoplasm of uterine cervix
285432005
Carcinoma of cervix
372098004
Carcinoma of endocervix
Liver Cancer
Included:
93870000
Malignant neoplasm of liver
408646000
Adenocarcinoma of liver
109841003
Liver cell carcinoma
274902006
Combined hepatocellular carcinoma and cholangiocarcinoma
187777008
Malignant neoplasm of intrahepatic gall duct
447109003
Primary malignant neoplasm of intrahepatic bile duct
109842005
Intrahepatic bile duct carcinoma
187767006
Malignant neoplasm of liver and intrahepatic bile ducts
95214007
Primary malignant neoplasm of liver
109843000
Hepatoblastoma
234601002
Sarcoma of liver
126852003
Neoplasm of intrahepatic bile ducts
Excluded:
94381002
Secondary malignant neoplasm of liver
1691000119104
Metastasis to liver from adenocarcinoma
813671000000107
Secondary malignant neoplasm of liver and intrahepatic bile duct
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
