Abstract
Background
The efficacy of microwave ablation in treating hepatic tumors requires advanced ultrasound skills. Failure of proper technique has the potential for either under- or over-treatment and possible harm to the patient. Emprint SX™ navigation provides surgeons with intra-operative, real-time navigation through augmented reality localization of the ablation antenna and the expected ablation zone. We hypothesize that incorporating this technology leads to improved targeting and optimizes ablation coverage. This study utilizes a simulated model to evaluate ablation outcomes using Emprint SX™ navigation vs standard ultrasound.
Methods
Surgical residents and faculty were recruited from a single institution. Using a novel tumor ablation simulator, participants performed ablations via 2 modes: standard ultrasound guidance (STD) and Emprint SX™ navigation (NAV). Primary outcome was the percentage of under-ablation. Secondary outcomes included percentage of over-ablation, time to complete trial, and number of attempts to position antenna.
Results
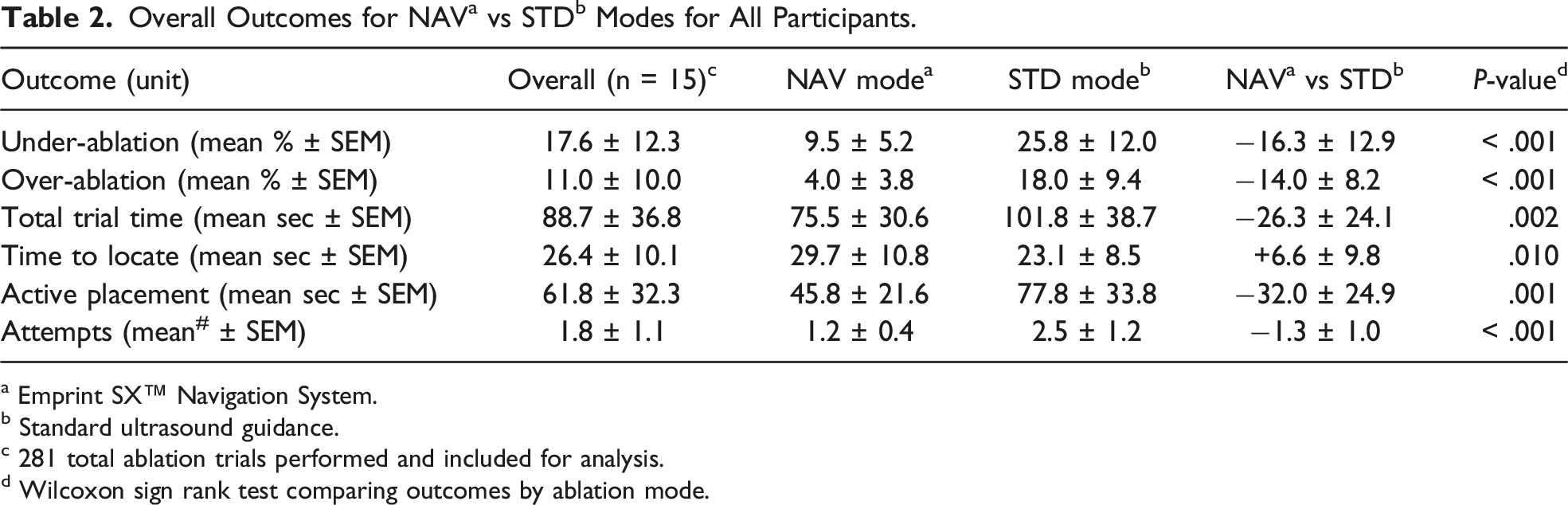
281 trials were performed by fifteen participants, with 47% female and 60% novice ablationists. Under-ablation volume decreased by a mean of 16.3% (SEM ±12.9, P < .001) with NAV compared to STD. Over-ablation volume decreased by a mean of 14.0% (±8.2, P < .001). NAV time was faster by a mean of 32 seconds (±24.9, P < .001) and involved fewer antenna placement attempts by a mean of 1.3 (±1.0, P < .001). For novice ablationists, all outcomes were improved with NAV and novices saw larger improvements compared to experienced ablationists (P = .018).
Discussion
In a simulated model, NAV improves ablation efficacy and efficiency, with novices gaining the greatest benefit over standard ultrasound.
Key Takeaways
• A novel tumor ablation simulator provides pre-clinical training for surgeons. • Navigational guidance improves efficacy and efficiency of simulated ablations. • Novices demonstrate the greatest improvements when utilizing navigational guidance.
Introduction
Microwave ablation (MWA) has been repeatedly demonstrated to be an effective and safe alternative or adjunct therapy in treating select patients with hepatocellular carcinoma.1,2 It has similar, if not improved, outcomes as compared to radiofrequency ablation of liver tumors, with faster ablation times and lower local tumor progression rates.3,4 While the technology of thermal ablation has undergone significant improvements over the past 2 decades, the effectiveness of MWA is highly dependent upon the acquired skillsets of the surgeon, 5 with evidence that at least 100 procedures are needed to achieve acceptable proficiency and local recurrence rates.6,7
One of the MWA systems available for surgeons is Emprint™, which utilizes Thermosphere™ technology for performing ablations, which has been shown to provide spherical and predictable ablation zones with comparable recurrence rates.8-10 The Emprint SX™ Ablation System is an integrated microwave ablation and image-guidance navigational system, which uses Thermosphere™ ablation technology while providing surgeons additional intra-operative targeting and projected ablation size information prior to ablation activation. 11 The system utilizes electromagnetic (EM) sensors to track the real-time positions of the ultrasound probe and ablation antenna and produces a three-dimensional (3D) augmented reality graphical visualization of these tools to aide in antenna navigation and placement. While long-term follow-up experience has not been published, early investigations utilizing Emprint SX™ for laparoscopic antenna placement and ablation have shown a statistically significant improvement in targeting accuracy and efficiency.12,13
Whereas previous studies of Emprint SX™ have primarily focused on improving antenna targeting and placement, there is no data evaluating its effectiveness in ensuring appropriate ablative tumor margins, nor its use in novice ablationists. This study utilizes a pre-clinical simulator to evaluate the ablation efficacy and targeting efficiency outcomes of hepatic tumor ablations using the Emprint SX™ navigation system (NAV), compared to using Emprint™ with the standard ultrasound guidance technique (STD). Our hypothesis is that surgeons using the NAV system will obtain a lower rate of under-ablating tumors (Figure 1), and that this difference will be greater for less experienced ablationists. Schematic of a bilobed tumor (gray) with surrounding 5 mm (light blue) and 10 mm (dark blue) margins. At a given spherical ablation zone (red), under-ablation (yellow) and over-ablation (green) areas are demonstrated.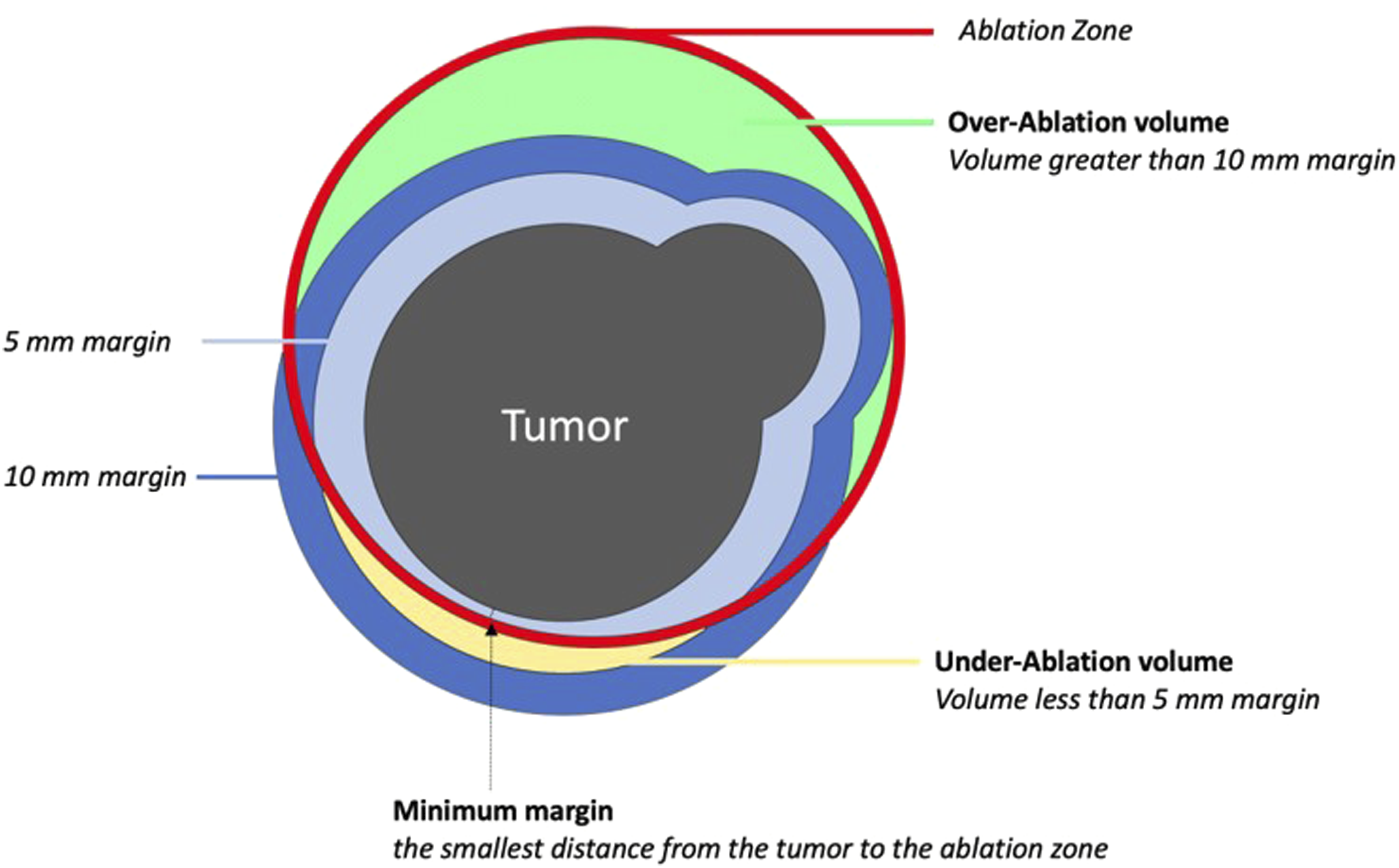
Methods
Overview
After obtaining Institutional Review Board approval, residents and faculty within the Department of Surgery at a single academic institution were recruited to participate and offered a token financial incentive for their participation. Each participant provided informed consent. Demographic data was collected prior to participation. Participants performed up to 20 trials of simulated hepatic tumor ablation using the Emprint SX™ simulator, which was designed by Medtronic Inc. (Boulder, CO) and utilized software from InnerOptic Technology Inc. (Hillsborough, NC) that was previously validated to ensure accuracy of output measurements and reflect use of the clinical version of the Emprint SX™ Ablation System.
Description of Simulator
This simulator displays a virtual tumor within simulated liver parenchyma. It utilizes electromagnetic (EM) tracking to localize a mock ultrasound probe and ablation antenna and the user can place the antenna into the model for the ablation (Figure 2A). This simulates an open surgical procedure, rather than laparoscopic surgery, to focus the user’s experience on developing both ultrasound and ablation expertise. The simulator has 2 distinct modes: the first mode simulates standard of care ultrasound guidance (STD), emulating the two-dimensional ultrasound image (Figure 2B); the second mode simulates Emprint SX™ guidance (NAV), providing an augmented reality view of the ultrasound scan and antenna positions in 3-dimensional space (Figure 2C). In this mode, the ultrasound probe and antenna are displayed in space and the surgeon uses these images to select placement of the microwave antenna. Navigation information includes the projected trajectory of the antenna and the three-dimensional projected ablation zone, which is based on the selected power and time for ablation. (A) Screenshot of the Emprint SX™ ablation simulator display. Labels in gray boxes added for clarification. Targeting obround indicates center of ablation zone based on antenna trajectory. (B) Simulator during standard ultrasound-guided ablation (STD). (C) Simulator during navigational-guided ablation (NAV).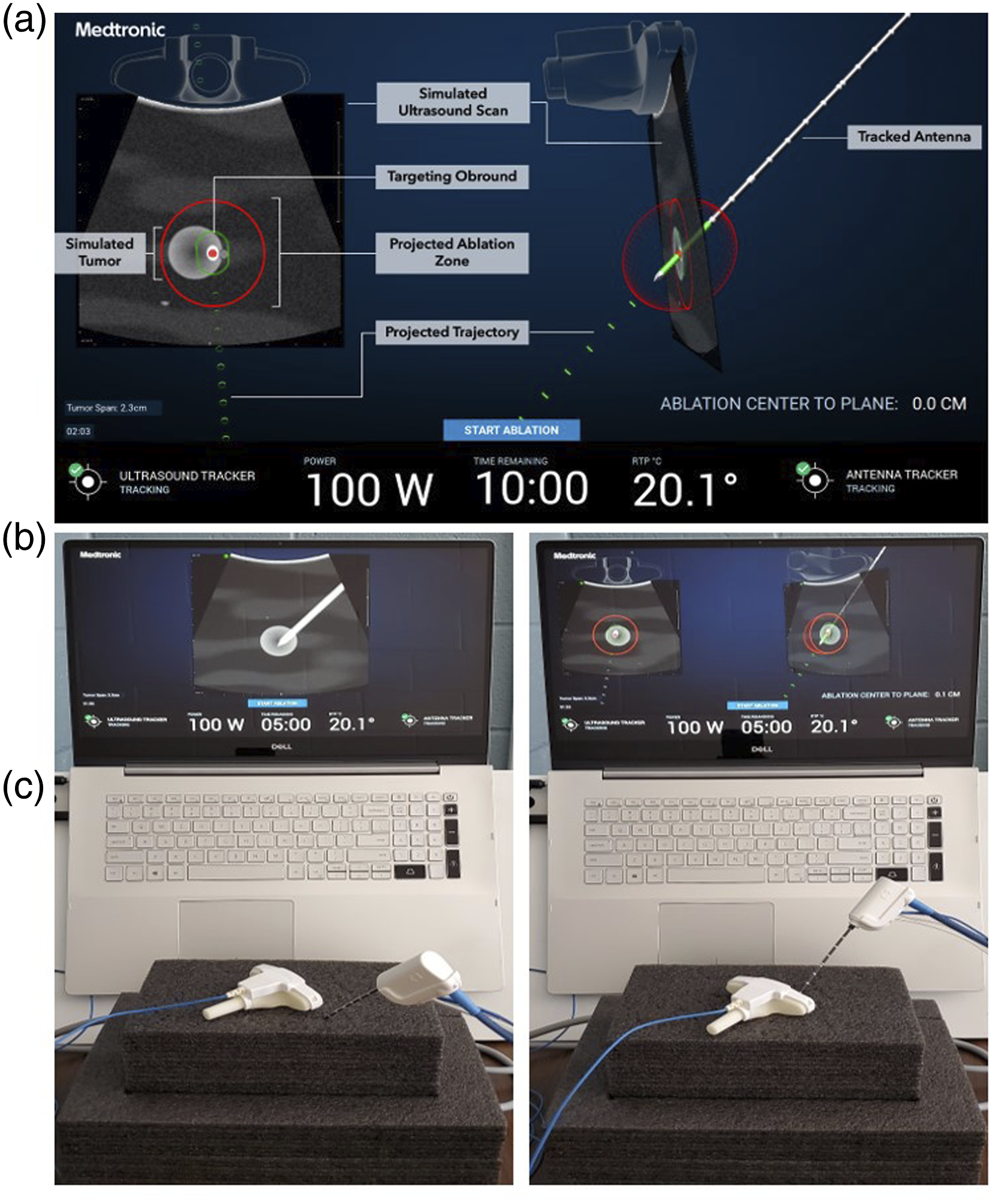
Intervention
Ablations were performed using a mock BK Medical® 8816 ultrasound probe and a 15 cm Emprint™ antenna on a 20 × 13 × 9 cm foam block, representing the liver. Each trial consisted of a tumor ranging from 1.5 cm to 2.5 cm in diameter, and morphology is either single or bilobed to simulate non-uniform shapes. Tumors were randomized to different locations and depths within the simulated liver. The monitor mirrored the real-world intra-operative display of the BK ultrasound screen or the Emprint SX™ screen and includes depth markers, tumor size, ablation power (in watts), and ablation time (Figure 2A).
Prior to data acquisition, 20 tumors were randomly simulated so that the characteristics (position, size, depth, and appearance) of each tumor were different. Using a block randomization scheme, 10 of the tumors were assigned to the STD trials and 10 tumors were assigned to the NAV trials. While the order of each NAV vs STD trial was randomly generated for the 20 trials, each participant completed the trials in the same order on the same tumors, for a total of 20 trials, with 10 ablations using each mode. To simplify ablation planning, power was kept constant at 100 W and the time could be adjusted by the participant in 30-second intervals from 1 minute to 10 minutes. Unlike real-world ablations, simulated ablations occurred instantaneously during the simulation, so ablation time was not factored into trial time. Each participant was given a single ablation attempt per tumor but were allowed as much time and as many attempts at antenna placement prior to ablation.
Data Collection
Each participant was read a standard script of study purpose and instructions for using the simulator in both STD and NAV modes. Complete ablation of a tumor was defined as ablation of total tumor with 5 mm margins circumferentially to generally reflect clinical ablation standards. Participants were given 1 practice trial using each mode prior to beginning. The primary outcome was percent of under-ablation of tumors, which is reflective of the surgeon’s intent when performing this procedure. Secondary outcomes included over-ablation of tumors, total trial time (defined as time from beginning of each simulated tumor to ablation), time to locate tumors within liver parenchyma (defined as time from beginning of each simulated tumor to first placement of antenna within foam block), and number of attempts at antenna placement prior to ablation of each tumor. Ablation results (e.g., under-ablation volume/percent, over-ablation volume/percent, and trial time) were recorded automatically using the Emprint SX™ software. Time to locate tumor and number of placement attempts were recorded by the experimenters observing the trials. Active placement time is calculated as total trial time minus time to locate tumor.
Statistics
Mean outcomes of all participants were compared between ablation modes (NAV vs STD) using a Wilcoxon sign rank rest. Outcomes by self-defined expertise (novices vs intermediate and experts) were calculated using a two-sample t-test. Outcomes between ablation modes by demographic data were compared using a Kruskal-Wallis test. All analysis was performed using SAS (SAS Institute Inc., Version 9.4, Cary, NC). Statistical significance was considered at P < .05. Prior to recruitment, a pilot study was performed using 6 novice participants to determine study workflow as well as to estimate standard deviation, effect size, and, subsequently, the number of required subjects and trials. Results from this power calculation determined that a minimum of 180 total trials were needed with a minimum of 9 participants for sufficient statistical significance using a 2-sided test with 80% power.
Results
Participant Demographics.
a Emprint SX™ Navigation System.
b Standard ultrasound guidance.
c 281 total ablation trials performed and included for analysis.
d Wilcoxon sign rank test comparing outcomes by ablation mode.
Outcomes by Ablation Mode
Comparing treated tumor volume using the 2 modalities (Table 2), we demonstrate that mean under-ablation volume is 16.3 (SEM ± 12.9) percent less with NAV vs STD mode (NAV = 9.5 vs STD = 25.8, P < .001). Mean over-ablation volume is 14.0 (±8.2) percent higher with STD mode (NAV = 4.0 vs STD = 18.0, P < .001). NAV mode had a mean total trial time that is 26.3 (±24.1) seconds faster than STD (NAV = 75.5 vs STD = 101.8, P = .002). Conversely, during NAV trials, participants took more time to locate the tumor by 6.6 (±9.8) seconds compared with STD trials (NAV = 29.7 vs STD = 23.1, P = .010). NAV trials had a mean of 1.3 (±1.0) fewer attempts compared to STD (NAV = 1.2 vs STD = 2.5, P < .001).
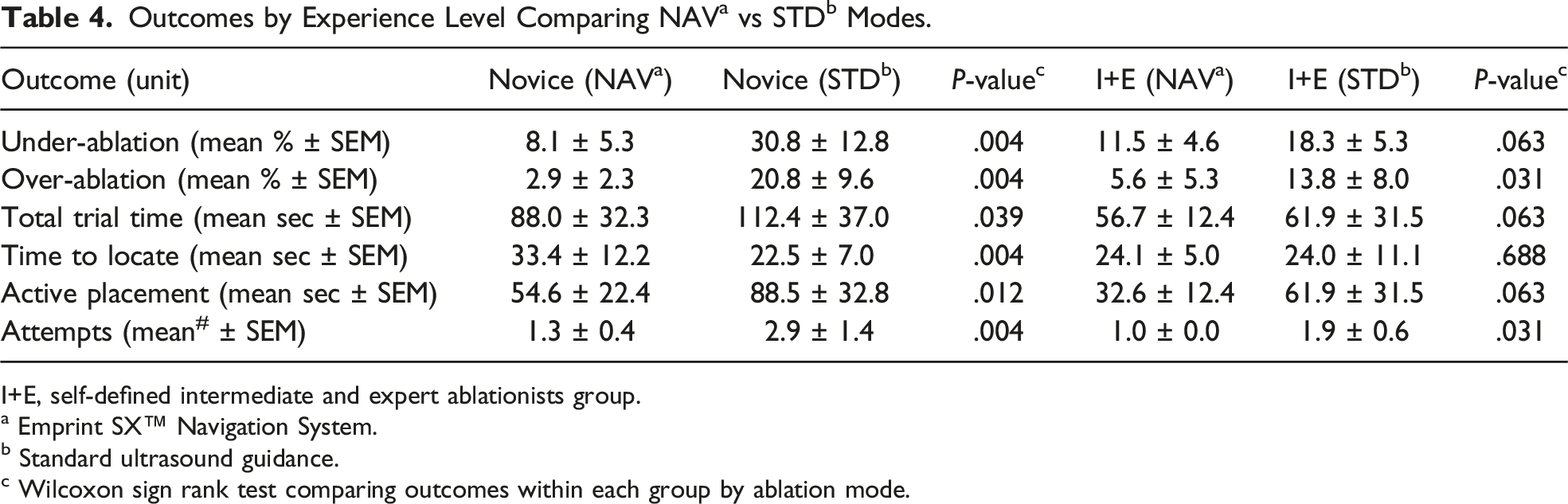
Outcomes by Experience Level
Outcomes by Experience Level.
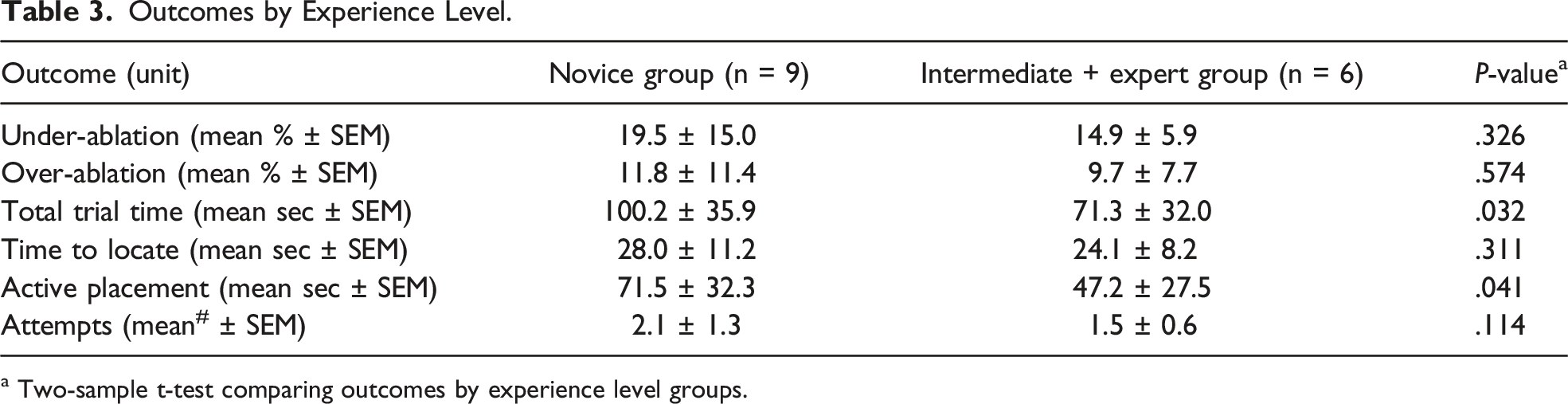
a Two-sample t-test comparing outcomes by experience level groups.
I+E, self-defined intermediate and expert ablationists group.
a Emprint SX™ Navigation System.
b Standard ultrasound guidance.
c Wilcoxon sign rank test comparing outcomes within each group by ablation mode.
When comparing the magnitude of mean improvement in performance when using NAV vs STD mode by experience levels, novices realize a larger improvement compared to the intermediate and expert group, as seen in both a decrease in under-ablation percentage (P = .018) and over-ablation percentage (P = .018, Figure 3A). Time to locate the tumor, however, was increased for novices using NAV mode by a mean difference of 10.9 (±8.8) seconds vs intermediate and experts by a mean difference of 0.1 (±7.8) seconds (P = .034, Figure 3B). There were no statistically significant differences comparing total trial time, active placement time, or number of attempts between the 2 groups. Mean differences when using navigation (NAV) vs standard (STD) ablation modes for novice ablationists vs intermediate and expert ablationists. (A) Under- and over-ablation results. (B) Time results. (*P = .018, ‡P = .034).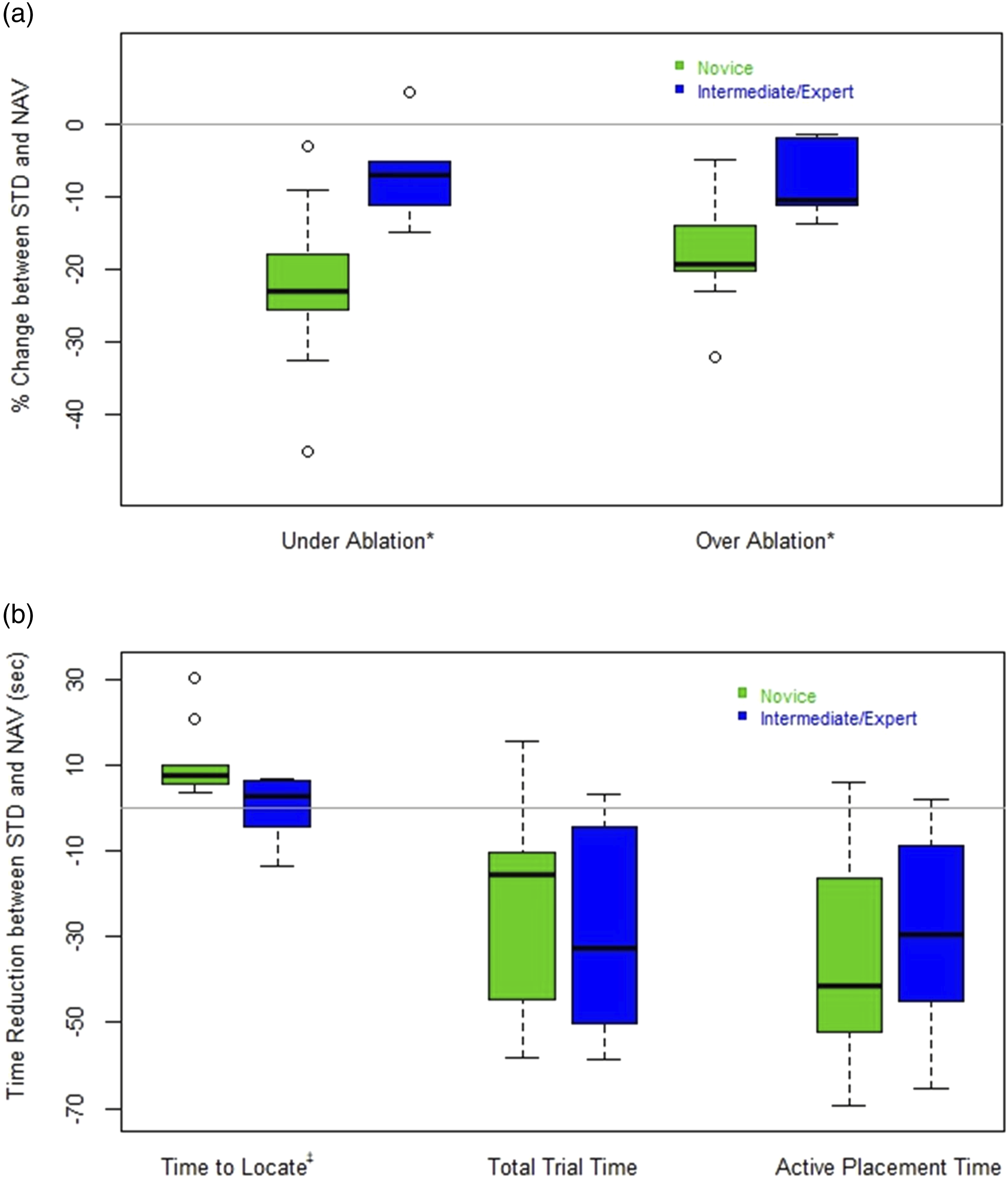
a Emprint SX™ Navigation System.
b Standard ultrasound guidance.
c Kruskal-Wallis test comparing outcomes by experience level groups.
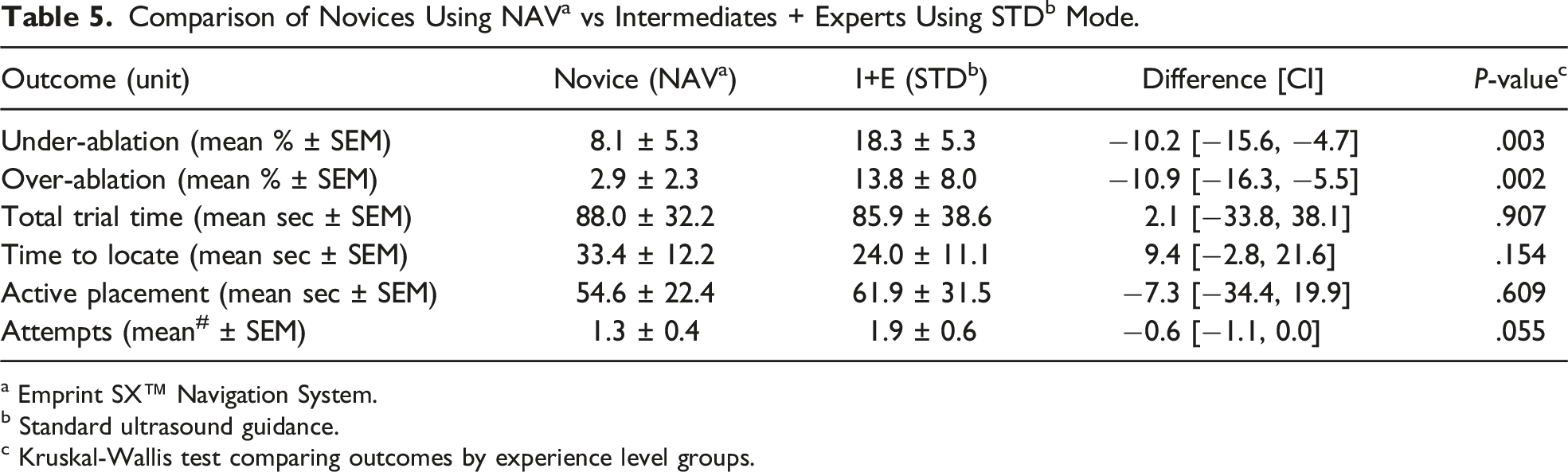
Outcomes by Demographics
When evaluating self-defined level of ultrasound experience, comparing self-defined somewhat (S) vs mostly and completely (M+C) competent groups, the S group saw a non-significant improvement in performance using NAV over STD than the M+C group: M+C group under-ablation improvement using NAV vs STD = 13.5% vs S group = 21.9%, P = .142; M+C group over-ablation improvement using NAV vs STD = 12.0% vs S group = 18.0%, P = .142.
Similarly, self-defined previous video game experience is not associated with statistically significant improvements in performance despite a minor detected difference when comparing over-ablation improvement using NAV vs STD. Those who defined their video game usage as frequent or occasional (F+O group) as compared to those stating rare or no use (R+N group) saw a slight improvement in over-ablation percentage: F+O = 18.1% vs R+N group = 12.0%, P = .111.
Discussion
Previously published preliminary evidence has suggested that using a tumor ablation navigation system may substantially improve overall targeting and placement proficiency, a critical skill necessary to overcome the steep learning curve associated with the procedure.6,7,12 In this study, participants perform better when using NAV vs STD ablation modes by decreasing the volume of under-ablation of the tumor, the volume of over-ablation of healthy liver parenchyma, the time to perform ablations, and the number of antenna placement attempts prior to each ablation. Taken together, these findings suggest that utilizing Emprint SX™ navigation and visualization improves tumor ablation outcomes compared to standard ultrasound guidance in a simulated benchtop environment. Performance improvement is likely due to the additional information the navigation system provides by visualizing the ablation zone in three-dimensions overlayed with the tumor location, allowing for better planning to optimize the ablation zone positioning and dose delivered.
When looking at surgeon experience level with MWA, it is not surprising that this correlates with performance, 14 and more specifically laparoscopic performance.15,16 Our study demonstrates that both novice and more experienced ablationists perform better, such as decreasing over-ablation percentage, when using NAV. Furthermore, Emprint SX™ navigation provides additional information, leading to improvements in proficiency gaps that exist based on experience. In fact, novices using navigation perform better than the more experienced ablationists utilizing standard ultrasound navigation in the endpoints of under- and over-ablation. Such a finding reinforces the notion that navigation likely assists with the learning curve of the procedure, dramatically improving proficiency of novices compared with more experienced ablationists when using standard of care ultrasound navigation.
This speaks to the potential benefits of this EM simulator for pre-clinical training of surgeons focusing on a variety of skills. Previous work supports the use and effectiveness of EM navigation simulators in surgical training. 17 To our knowledge, this is the first study to compare surgical ablation outcomes using an entirely simulated ablative environment with EM tracking and software. While there are limitations, the use of such a simulator allows for rapid iteration in the development of surgical procedures and devices, while potentially allowing for the studies to be conducted with less effort and cost. Expansion of this simulated model to other surgical tasks, such as robotics, may demonstrate similar improvements in performance and many other potential research questions could be explored via simulation.
While this prospective study demonstrates significant improvements in surgeon performance, the simulator did not model the adjustment of ablation power, ability to ablate single tumors multiple times, avoidance of vascular structures, and differences in textural makeup between the foam block and actual livers. As such, future studies should evaluate the translation of simulated skills to the clinical setting. The proprietary nature of the simulator, however, limits its potential larger use. This study also did not include any analysis of the financial costs, compared to the benefits, of Emprint SX™ over standard ablation systems without navigation.
Footnotes
Acknowledgments
The authors would like to acknowledge the work from InnerOptic Technology Inc. in developing the simulator software.
Author Contributions
Study design was performed by IMK, RSG, SR, PLS, KMN, and DAG.
Data collection was performed by IMK, RSG, PLS, and DAG.
Data analysis and interpretation was performed by IMK, RSG, SR, AS, and DAG.
Manuscript preparation and editing was performed by IMK, RSG, SR, AS, and DAG.
Decision to submit for publication was performed by IMK and DAG.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DAG is the principal investigator of this trial sponsored by Medtronic Inc. RSG, SR, and AS are affiliated with Medtronic Inc., and assisted in study design, data collection, analysis and interpretation, as well as in writing this report. Medtronic designed and provided the simulator for use during this study. IMK, PLS, and KMN declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Medtronic Inc., Boulder, CO [Grant Number 19-2087].