Abstract
We present a case of a premature infant who had an initial diagnosis of an innominate artery compression syndrome. This was approached by a median sternotomy for an aortopexy. However, the patient was found to have a distal tracheal stenosis due to a tracheal cartilage deficiency and was treated by a tracheal resection and primary anastamosis.
A one-month-old female infant born prematurely at 28 weeks of gestation was transferred from a referring NICU for failure of recurrent attempts at extubation. At flexible bronchoscopy there, a distal pulsatile anterior tracheal compression was visualized, concerning for an extramural vascular compression. A CT scan did not show a vascular ring. However, the innominate artery was in close apposition to the anterior tracheal wall but with the endotracheal tube in situ (Figure 1A). (A) CT chest showing close apposition of the innominate artery to the trachea with an endotracheal tube in-situ and a large thymus. (B) Flexible bronchoscopy showing a tracheal stenosis which is easily traversed to the carina (inset showing a close-up “gastric cardiac” like feature of the stenosis). (C) Dynamic CT chest showing a distal tracheal stenosis during the expiratory phase. (D) Operative external appearance of the stenosis before and after the repair. (E) Flexible bronchoscopy showing a widely patent trachea post resection and anastomosis.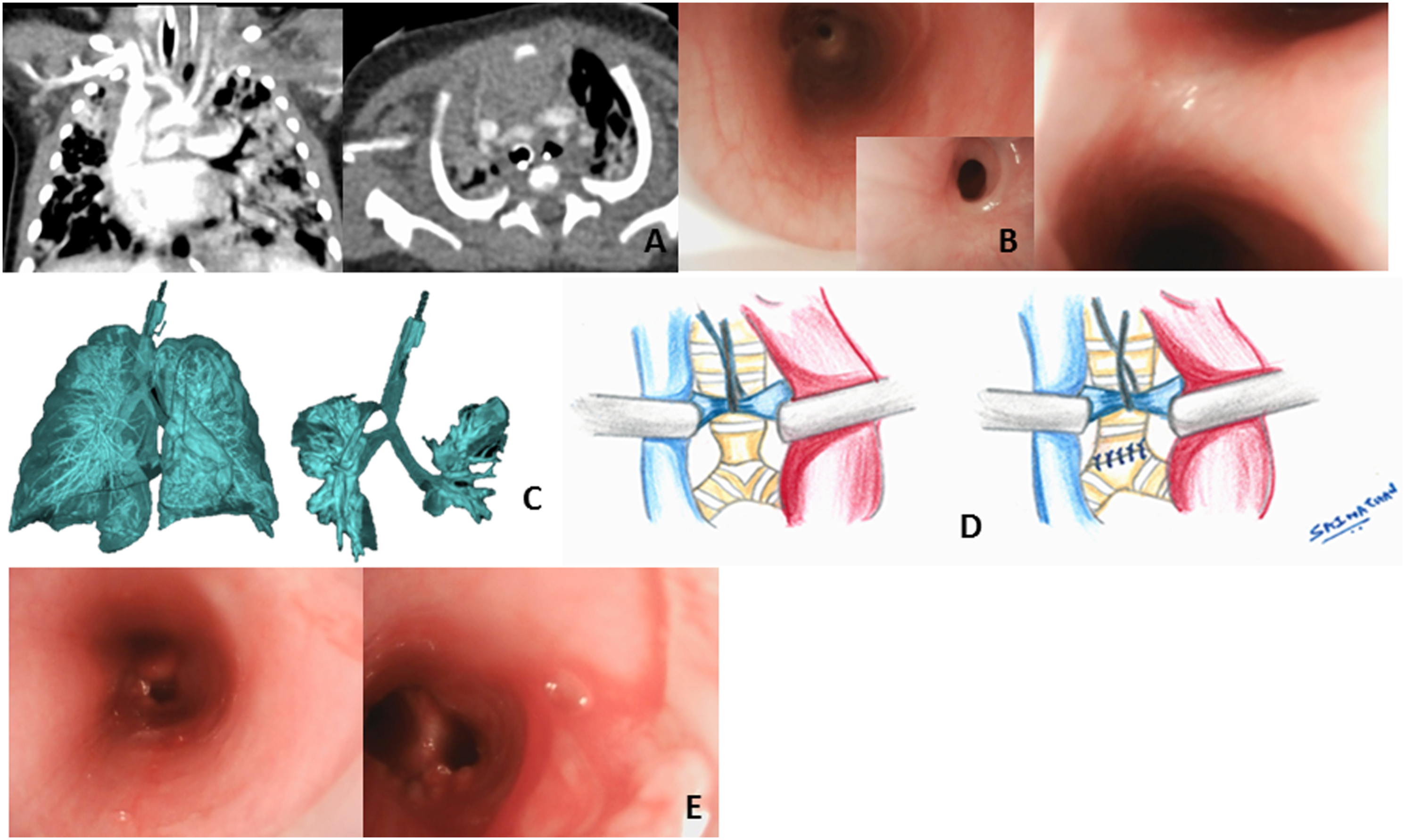
Given the clinical presentation of recurrent airway obstruction, the patient was taken to the operating room with a working diagnosis of an innominate artery compression syndrome. A repeat flexible bronchoscopy in the operating room revealed an 80% distal tracheal stenosis which could be passed beyond easily to the carina suggesting an extramural pathology (Figure 1B).
We proceeded with a midline approach to an aortopexy to address the innominate artery compression by an upper partial median sternotomy. A subtotal thymectomy was performed followed by an aortopexy of the anterior aspect of the aortic arch and the base and mid portion of the innominate artery to the posterior aspect of the left half of the sternotomy. On aortopexy, however a suboptimal result was seen. While there was some resolution of the anterior aspect of the stenosis, the posterior part formed by the membranous trachea showed an intrusion causing a residual 50% stenosis. Hence, a decision was made to incorporate mid ascending aorta in the aortopexy. There was no further improvement in the tracheal stenosis. The incision was converted to a full sternotomy in order to inspect the trachea externally. The intrathoracic aorta was exposed between the ascending aorta and the superior vena cava. External inspection showed a wasting of the trachea .5 cm above the carina, which correlated with the stenosis by bronchoscopy (Figure 1D). In order to further characterize the stenosis and since the patient was not prepared for cardiopulmonary bypass to facilitate a resection of the tracheal stenosis, the chest was closed, and a dynamic chest CT was performed. It showed a dynamic distal segmental tracheal stenosis (Figure 1C).
The patient was taken the next day for a tracheal resection with cardiopulmonary bypass. Close inspection of the distal trachea just above the carina showed stenosis due to absence of a single tracheal ring. That portion of the trachea was entirely membranous. The area was primarily resected and anastomosed with 6-0 Prolene suture in an interrupted manner. Part of the anastomosis was taken into the right proximal bronchus (Figure 1D). Repeat bronchoscopy showed no further stenosis and a widely patent anastomosis (Figure 1E). Pathology confirmed absence of cartilage in the resected tracheal segment. The patient was successfully extubated on the third postoperative day and had an uneventful postoperative course.
Respiratory distress in an infant from airway obstruction can be due to an intrinsic abnormality of the airway or an extrinsic compression. Absence of tracheal cartilage is an intrinsic airway abnormality which is a very rare and often a missed cause for such an airway obstruction. 1
The tracheal ring has a rigid anterior portion formed by a U-shaped cartilage and a soft flat posterior portion formed by the membranous trachea. The anterior cartilaginous portion can rarely be hypoplastic or absent. When the defect involves multiple rings, it causes a long-segment abnormality which is clinically mistaken for tracheobronchomalcia and is often associated with other anomalies such as a tracheo-esophageal fistula and abnormal carinal branching patterns. 2
On the other hand, a single tracheal cartilage ring absence causes a short segment dynamic tracheal stenosis such as in our case. The short segment stenosis tends to occur in the distal trachea just above the carina and presentation at an early age generally requires a tracheal airway device to mitigate the respiratory distress as evident from our case. A CT scan performed in the expiratory phase may be required to diagnose such a dynamic stenosis. 2 Bronchoscopy can be deceiving as it is easy to pass through the stenosis unlike a fixed cicatricial stenosis. It may be mistaken for complete tracheal rings. The stenosis has been described as “gastric cardia” (Figure 1B- inset) like in appearance on bronchoscopy. 1 Optical coherence tomography2,3 can help in clarifying the diagnosis during bronchoscopy by demonstrating the absence of the tracheal cartilages; particularly for the long segment variant. Given the rarity of the diagnosis, it is usually diagnosed incidentally postoperatively by pathological exam of the resected trachea. 1 The treatment is resection and end to end anastomosis in case of a short segment abnormality. When the defect straddles the carina, the anastomosis may have to be taken to the bronchus as in our case.
This case also illustrates the versatility of a midline sternotomy approach to an aortopexy as compared to an anterior thoracotomy one. 4 As described earlier, the initial working diagnosis was an innominate artery compression syndrome causing the tracheal obstruction; which is treated by an anterior aortopexy. By providing a wider exposure, the midline approach affords the ability to perform a more complete thymectomy, to revise the extent of the aortopexy to include the distal ascending aorta in case of a suboptimal outcome with an aortopexy limited to the innominate artery, facilitates the reimplantation of an abnormally leftward originating innominate artery, helps address residual tracheomalacia by performing a posterior membranopexy and provides the ability to perform direct tracheal surgery. If we had used an anterior thoracotomy approach, adequate external examination of the trachea would have not been possible, missing the true diagnosis and the possibility of subjecting the patient to a tracheostomy given the suboptimal outcome from an aortopexy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
