Abstract
This study investigates the structural, optical, dielectric, and electromagnetic absorption properties of spinel‐structured Mg–ZnAl2O4 synthesized through a sol–gel technique. The structural analysis confirmed the presence of various phases, including MgO, ZnO, ZnAl2O4, and MgAl2O4, with an average crystallite size of approximately 88 nm. The material demonstrated a reflection loss of −71 dB at 14.02 GHz, indicating its potential for effective microwave absorption, with an effective absorption bandwidth of 3.66 GHz at a thickness of 5 mm. The optical band gap was determined to be 3.19 eV, lower than that of ZnAl2O4 and MgAl2O4, suggesting enhanced microwave absorption capabilities. Dielectric characterization revealed a range of dielectric constants from 3.09 to 6.31 across the frequency range of 8 to 18 GHz. These findings highlight the promising application of Mg–ZnAl2O4 in microwave absorption technologies due to its favorable structural and electromagnetic properties.
Keywords
1. Introduction
Spinel‐structured zinc and magnesium aluminate–based materials have garnered significant attention because of their unique properties and potential applications [1–4]. Due to their structural characteristics of optical behavior, dielectric properties, and even electromagnetic microwave absorption capabilities, spinel‐structured aluminates hold immense promise in various fields [5–9]. For example, Jamal et al. explored the structural, optical, and dielectric properties of zinc aluminate nanoparticles using the sol–gel combustion method [9]. The material was characterized, and its dielectric properties were evaluated in a frequency range from 100 kHz to 8 MHz. It can be observed that the dielectric permittivity of zinc aluminate is smaller than some of the other members in the spinel family [10, 11]. However, zinc ferrite and manganese ferrite have larger values of dielectric permittivity [1, 11, 12]. Moreover, the dielectric permittivity of the material exhibits dispersion in the low‐frequency region. The interfacial polarization of the Maxwell–Wagner type could be the reason for this phenomenon. Additionally, the AC conductivity of the material increases with frequency. The frequency exponent factor was found to be nearly unity, and it did not show any appreciable variation with temperature. This is attributed to AC conduction through the hopping of electrons between sites using quantum mechanical tunneling.
Furthermore, Jagadeeshwaran et al. investigated the structural, optical, and electrical properties of sintered Mg–Zn aluminate systems [4]. Using the chemical coprecipitation method, they combined the insulating MgAl2O4 and semiconducting ZnAl2O4 to create the Mg–Zn aluminate compound. The dominance of Mg2+ ions over Zn2+ ions in the compound was attributed to their difference in ionic radii. The UV absorbance spectra showed a blue shift in the optical bandgap, which was explained by the Burstein–Moss effect and linked to the increase in sintering temperature. The real and imaginary parts of the dielectric constant and tan δ displayed a dispersive nature, suggesting a non‐Debye‐type relaxation behavior in all the Mg–Zn aluminate samples. This behavior was believed to be influenced by the electrode interface effect, grain boundaries, and grain interior. These findings suggested that the spinel aluminate sample is more suitable for various applications [1, 13–16].
Ferrites are commonly used to absorb microwaves in order to counteract electromagnetic pollution [12, 17]. However, ferrites’ limited ability to lose dielectric energy restricts their applications. The permeability and permittivity of the absorbers are vital factors for microwave attenuation, and extensive research has been conducted to harness these properties [18, 19]. Improving the permittivity of microwave absorbers requires addressing conductive loss, electron hopping, dipole polarization, and interfacial polarization [20]. Recent studies have shown that modifying the morphology of nanostructures can regulate the energy band gap, striking a balance between conductivity and dielectric characteristics [21, 22]. Research on spinel aluminates has shown outstanding microwave dielectric properties [23]. For instance, Hongyao Jia et al. demonstrated the enhanced high‐temperature dielectric polarization of composites made of barium titanate and magnesium aluminum spinel, highlighting their potential for microwave absorption [24]. Additionally, Yuan Wang investigated the improved dielectric properties and high‐temperature microwave absorption performance of Zn‐doped Al2O3 ceramics [25]. The 3% Zn‐doped Al2O3 ceramic exhibited the strongest absorption performance, with a maximum peak of −12.1 dB and a wide effective absorption bandwidth (EAB) (reflection loss [RL] below −10 dB from 9.3 to 12.3 GHz). These findings suggest that Zn‐doped Al2O3 ceramic has great potential as a high‐temperature microwave absorption material.
Microwave‐absorbing materials are widely recognized for their capacity to absorb microwaves [26–28]. These materials have a unique structure combining a low permittivity phase with a high electrical conductivity phase. A medium permittivity phase is introduced at the nanoscale to enhance their microwave absorption capabilities across a broad range. For instance, submicrometer‐sized ZnO particles are employed as the electrically lossy phase, whereas ZnAl2O4 nanograins serve as the medium permittivity phase [29]. The heterojunction of the ZnO/ZnAl2O4 composite benefits from a high carrier concentration and mobility, which enhances interfacial polarization capability and dielectric loss. Consequently, this composite material becomes an excellent choice for various microwave absorption applications. Additionally, Reza Peymanfar conducted a study on the microwave absorption performance of ZnAl2O4 [20]. The research focused on the impact of interfacial interactions at grain boundaries on microwave absorption. To analyze this, polystyrene and polyvinylidene fluoride media were used. The combination of ZnAl2O4 nanostructure resembling blueberries and polyvinylidene fluoride nanocomposite showed impressive RL of −95.63 dB at a frequency of 9.63 GHz, with a thickness of 3.00 mm. Additionally, it exhibited a wide bandwidth of 2.80 GHz (RL < −20 dB) at a thickness of 2.50 mm. Notably, the ZnAl2O4/polystyrene nanocomposite displayed a broad efficient bandwidth of 9.14 GHz (RL < −10 dB). Furthermore, the energy band gap of nanoparticles can be reduced by expanded carbon microspheres, as demonstrated in the study of the expanded carbon microspheres of ZnAl2O4 nanocomposite [30]. In the case of the ZnAl2O4/polyvinylidene fluoride nanocomposite, a RL of −84.95 dB at 13.63 GHz was achieved with a thickness of 2.00 mm. Similarly, the expanded carbon microspheres/ZnAl2O4/polystyrene exhibited a wide efficient bandwidth of 7.47 GHz (RL 10 dB) at a thickness of 2.00 mm. Notably, the expanded carbon microspheres/ZnAl2O4/polyvinylidene fluoride nanocomposite demonstrated an impressive, efficient bandwidth of 3.10 GHz (RL ∼ 20 dB). In addition, the rGO/ZnO/ZnAl2O4 composites have excellent microwave absorption properties [31]. It demonstrates a minimum RL of −44.5 dB at 10.1 GHz and an EAB of 3.8 GHz. The composite shows synergistic effects that contribute to its strong absorption and broad bandwidth, making it a potential candidate for microwave‐absorbing materials.
The spinel Mg/Zn aluminates have demonstrated favorable properties that make them promising for applications in microwave absorption and dielectric and optical fields. This study focuses on investigating the structural, optical, dielectric, and electromagnetic microwave absorption properties of a spinel‐structured Mg–ZnAl2O4. The goal of our research is to gain a better understanding of the interplay between these properties and how they influence the behavior of Mg–ZnAl2O4 in microwave absorption. We use techniques such as X‐ray diffraction (XRD), scanning electron microscopy (SEM), and atomic force microscopy (AFM) to analyze the crystal structure and morphology of Mg–ZnAl2O4. This helps to identify any deviations or deformations from the ideal spinel lattice arrangement. Subsequently, we use UV–vis spectroscopy to conduct optical characterization, analyzing the absorption spectrum and unveiling the bandgap energy of Mg–ZnAl2O4. Additionally, we employ Fourier transform infrared spectroscopy (FTIR) to examine structural changes. Moreover, our research extends to investigating the electromagnetic absorption properties of Mg–ZnAl2O4 by conducting a detailed examination of its microwave absorption capacity across a wide frequency range. Through the use of CST simulation software, we evaluate key parameters, including RL at different thicknesses, to gain valuable insights into the electromagnetic response capabilities of Mg–ZnAl2O4.
2. Experimental Section
Mg–ZnAl2O4 powder was prepared using a sol–gel technique. Analytical reagent grades (above 99% purity) of zinc nitrate hexahydrate (Sigma‐Aldrich), magnesium nitrate hexahydrate (Loba Chemie), aluminum nitrate nonahydrate (SRL Chemicals), and citric acid anhydrous (SRL Chemicals) were used. Initially, the zinc nitrate hexahydrate, magnesium nitrate hexahydrate, and aluminum nitrate nonahydrate were dissolved in distilled water in a 500‐mL beaker. Then, citric acid was added with a metal‐to‐citric acid ratio of 1:1. The mixture was stirred at 250 rpm and maintained at 80°C to form a gel. The gel was collected and heated overnight at 120°C to obtain a dry powder. The collected powder was manually ground and sintered at 650°C for 6 h to obtain Mg–ZnAl2O4 with a spinel structure. Figure 1 illustrates the experimental procedure followed for the preparation of spinel Mg–ZnAl2O4 in this work. The resulting Mg–ZnAl2O4 powder performed detailed physical, structural, and EM wave absorption properties and was further tested using advanced analytical techniques. XRD was conducted using a Bruker D8 ADVANCE, with a 2θ range from 20° to 80° and a step size of 0.02° and Cu–Kα radiation (λ ∼ 1.54 Å). SEM (Quanta FEG 250) was used to observe the microstructure and surface morphology of the prepared samples. The surface roughness and grain boundary width of Mg–ZnAl2O4 were observed using a Bruker AFM Probes–RTESPA‐300 atomic force microscope from the United States. The examination was conducted in tapping mode with a frequency of 0.5 Hz. FTIR (Shimadzu IRAffinity‐1) was used to identify the functional groups present in the powder sample by blending it with spectrally pure KBr captured in the 4000–400 cm−1 range with a resolution of 2 cm−1. The optical properties were also measured in the ultraviolet–visible range (200–700 nm) using a Jasco V‐770 UV–vis spectrophotometer. A Rohde & Schwarz (ZNB 20) VNA was used with the DAK 3.5 kit for the dielectric property study. Further microwave absorption study for different compositions and thicknesses was examined with the CST software.

Experimental procedure for preparation of spinel Mg–ZnAl2O4.
3. Results and Discussion
Figure 2 presents the XRD pattern of Mg–ZnAl2O4 after being heat treated at 650°C for 6 h via the sol–gel synthesis technique. The XRD profile exhibits a highly crystalline nature, with most peaks appearing in the range of 30°–70°. To analyze the XRD profile, we employed the X’Pert HighScore Plus software to identify the crystalline phases present in the sample. The diffraction peaks in the XRD profile correspond to MgO (PDF No.: 98‐001‐2412), ZnO (PDF No.: 98‐001‐1316), ZnAl2O4 (PDF No.: 98‐000‐5499), and MgAl2O4 (PDF No.: 98‐000‐8471). Among these phases, MgO, ZnAl2O4, and MgAl2O4 exhibit a cubic structure, whereas ZnO displays a hexagonal structure [32]. Additionally, we conducted line profile analysis to determine the crystallite size from the XRD data, yielding an average crystallite size of 88 Å [33]. The XRD quantification analysis demonstrated that ZnAl2O4 had the highest phase quantification at 34.2%, followed by ZnO at 26.8%, MgO at 24.4%, and MgAl2O4 at 14.6% (Table 1). Notably, ZnAl2O4, ZnO, and MgO comprised a significant portion (85.4%) of the prepared samples, indicating their direct impact on the mechanical and electrical properties, as well as their band structure and polarization behavior [31]. Zinc aluminate spinel (ZnAl2O4) has gained significant attention for various applications due to its wide band gap (∼3.8 eV), high fluorescence efficiency, excellent chemical and thermal stability, and low surface acidity [34]. The influence of interfacial interactions at grain boundaries on microwave absorption is well recognized. Previous studies have reported noteworthy microwave absorption properties of Zn and Al‐containing structures [20, 25, 31]. Moreover, we believe that the Mg–ZnAl2O4 interfacial interactions at grain boundaries can be assessed by the presence of ZnO, MgO, ZnAl2O4, and MgAl2O4 phases, which show synergistic effects that contribute to its strong absorption and broad bandwidth, making it a potential candidate for microwave absorbing material [20, 29, 31, 35, 36]. These interactions play a vital role in understanding the nature and strength of the connections between Mg–ZnAl2O4 and its adjacent materials. By analyzing the behavior of these phases, we can unravel the complex mechanisms underlying grain boundary interactions in Mg–ZnAl2O4. In particular, identifying ZnO is a significant indicator for assessing the interfacial bonds at grain boundaries. This compound’s presence suggests a potential interaction between zinc (Zn) atoms from ZnAl2O4 and oxygen (O) atoms from an external source. Similarly, detecting MgO signifies a possible coupling between magnesium (Mg) atoms found in either ZnAl2O4 or another compound close to it and O atoms originating from an external entity. Furthermore, observing the formation of the Mg–ZnAl2O4 phase allows us to infer potential reactions between Mg atoms in either ZnAl2O4 or neighboring compounds with aluminum (Al) entities present at grain boundaries. The examination of the interactions between crystalline phases at grain boundaries and microstructure was used and explained in subsequent sections through the use of FTIR, FE‐SEM, and AFM studies. These analytical tools provide valuable information on the molecular vibrations, elemental composition, topographical features, and surface forces associated with the aforementioned interfaces.

XRD profile of Mg–ZnAl2O4.
XRD analysis of crystal structure parameters.
The FTIR spectra of Mg–ZnAl2O4 show distinct bands at specific wave numbers, as depicted in Figure 3. These bands are observed at 3522, 3479, 2305, 2017, 1632, 1493, 1404, 740, 698, and 642 cm−1. The presence of bands at higher wave numbers, such as 3522 and 3479 cm−1, suggests the existence of hydrogen bonding or O–H stretching vibrations within the compound [37, 38]. Conversely, lower wave number peaks (740, 698, and 642 cm−1) may indicate lattice vibrations or metal–oxygen bonding interactions [39]. In particular, the band at 740 cm−1 is attributed to Al–O vibrations [40]. Vibrations of M–O and M–OH (where M is a metal ion—Al or Mg) in the octahedral metal layer are associated with the region around 600–700 cm−1. The band at 698 cm−1 represents AlO6 groups, which make up the MgAl2O4 spinel and confirm the formation of MgAl2O4 spinels in all the as‐synthesized samples [41]. The bands at 1632 and 648 cm−1 correspond to Zn–OH translation modes [42]. The FTIR analysis validates the findings from the XRD analysis and also indicates the presence of the Mg–ZnAl2O4‐like phase in the samples under study.

FTIR spectra of Mg–ZnAl2O4.
Figure 4 displays FE‐SEM images of nanoparticles at different magnifications, ranging from 10 μm to 500 nm, demonstrating consistent morphologies. The synthesized nanostructures had an average size below 90 nm, as shown in the bottom right image of Figure 4 at 500‐nm resolution. The sol–gel method resulted in hierarchical microspheres and hollow particles as the morphology. The surface area‐to‐volume ratio is known to be important in modifying the properties of nanoparticles. By controlling the size of the nanostructures, the grain interfaces can be increased, leading to enhanced dipole and interfacial polarizations. However, the size of the nanostructures also affects the energy band gap, polarizability, and conductive loss, which were examined extensively. The study revealed that the agglomerated morphology of the sol–gel microspheres and hollow particles contributed to a reduction in the energy band gap [39]. The aforementioned observation entails that there exists a tendency for nanoparticles to aggregate, leading to the formation of a cohesive structure that exhibits a constructive attribute in relation to the absorption of microwave radiation. This finding implies that when these nanoparticles come together and form clusters, they demonstrate an enhanced capacity for absorbing electromagnetic waves within the microwave frequency range. This phenomenon highlights the significance of interparticle interactions and their influence on the overall behavior and performance of nanomaterials in terms of microwave absorption. The agglomeration of nanoparticles observed here can be interpreted as a collective phenomenon driven by intermolecular forces, such as van der Waals interactions or electrostatic attractions. As a result, these attractive forces act to bring individual nanoparticles into close proximity, enabling them to interact more effectively with incoming microwave radiation. This assembly process leads to an overall increase in the effective surface area available for interaction with microwaves, thereby enhancing the absorption capacity of the constructed structure. The constructive nature of this aggregation lies in its ability to facilitate cooperative interactions between adjacent nanoparticles and maximize their collective response toward microwave irradiation [39].

FE‐SEM images of Mg–ZnAl2O4.
Grain boundaries, which are regions where different crystalline grains meet, play a crucial role in determining material properties. In the case of Mg–ZnAl2O4, it has been observed that interfacial interactions at these grain boundaries result in enhanced microwave absorption capabilities [43]. Figure 5 presents the AFM images of the 2D and 3D structures of Mg–ZnAl2O4, showing the width of the grain boundaries. The grain boundary width is calculated as the distance between successive inflection points on the AFM curve (refer to Figure 5(a)). The highest average grain boundary width recorded is 952 nm, whereas the lowest width is 635 nm. Grain boundaries are interfaces between adjoining crystalline regions within a material, characterized by structural discontinuities and distinct electrical properties. These boundaries often act as sites for enhanced microwave absorption due to factors such as localized strain, charge accumulation, or increased defect density. The presence of grain boundaries can lead to polarization effects and the creation of localized electromagnetic hotspots, facilitating efficient microwave energy dissipation. The widest segments of grain boundaries are also found in the sheared samples, which also contain several pores along the grain boundary network.

AFM images of Mg–ZnAl2O4: (a) 2D image with grain boundary width and (b) 3D image.
(b)

Furthermore, in Figure 5(b), it can be observed that the average surface roughness (R z ) is measured at 118 nm, whereas the roughness Rmax shows a value of 1369 nm. The maximum peak height (R p ) is also found to be 225 nm. Surface roughness is a crucial factor that affects the interaction between microwaves and materials. When microwaves hit a rough surface, they encounter more scattering events due to the surface irregularities (Figure 5(b)). This increased scattering results in higher absorption of microwave energy by the material. Moreover, it has been observed that as the average surface roughness increases (Rz = 118 nm), more energy is absorbed by the material through microwave irradiation. This correlation arises from two main factors. Firstly, increased roughness enhances the effective interfacial area between the material and its surroundings (Rmax 1369 nm), enabling more efficient energy transfer. Secondly, heightened roughness (R p = 225 nm) creates additional microscopic defects or nanostructures on the surface, which act as localized energy sinks and further improve microwave absorption capabilities.
The optical performance of Mg–ZnAl2O4 is depicted in Figure 6 using UV–vis spectra data. The absorption of Mg–ZnAl2O4 was found to increase from 200 to 335 nm, followed by a decrease from 335 to 425 nm. Beyond 425 nm, the absorption remained relatively constant. To determine the direct band gap of spinel‐structured Mg–ZnAl2O4, Tauc’s plots were used based on the absorption data (Figure 6(b)). By extrapolating the linear portion of the plots of (αhυ)2 versus hυ to the energy axis, the direct (allowed) energy gap (E g ) of the sample was determined [44, 45]. Generally, polycrystalline zinc aluminate (ZnAl2O4) has an optical band gap of 3.8 to 3.9 eV [46], whereas polycrystalline magnesium aluminate (MgAl2O4) has an optical band gap of 4.13 to 4.62 eV prepared by using the sol–gel synthesis [47]. In the present work, the absorption spectra yielded a band gap value of 3.19 eV for spinel‐structured Mg–ZnAl2O4, which is lower than ZnAl2O4 and MgAl2O4. The morphology of Mg–ZnAl2O4 nanostructures can regulate the energy band gap, striking a balance between conductivity and dielectric characteristics (Figure 4). It is also well known that optical energy bandgaps play crucial roles in determining their dielectric properties and microwave absorption properties. A lower optical band gap leads to higher microwave absorption properties [37, 38, 48]. The phenomenon under examination postulates that a reduced optical band gap, or the minimum energy required for an electron to jump from the valence band to the conduction band, results in augmented characteristics of microwave absorption. The optical band gap usually pertains to electronic transitions related to photons in the visible light spectrum. However, this study inquiry focuses on the implications of lowering this specific band gap on microwave absorption properties. By decreasing the magnitude of the optical band gap, it is posited that electromagnetic radiation in the microwave range exhibits increased propensities for absorption by a material. Microwave absorption refers to the ability of a given substance to capture and convert microwave energy into internal energy or heat. The rationale behind this assertion rests upon the fundamental principle that a lower optical band gap enables a more facile movement of electrons within a material’s lattice structure when subjected to microwaves. Consequently, this heightened electron mobility allows for enhanced interaction with incident microwaves, leading to an elevated propensity for absorption by the material. Therefore, based on these theoretical considerations, it can be inferred that manipulating and reducing the optical band gap could yield materials with improved microwave absorption properties. The addition of Mg to ZnAl2O4 in this study caused structural changes that resulted in a lower energy band gap of 3.19 eV, showing enhanced microwave absorption properties.

(a) UV–vis spectra and (b) energy band gap of Mg–ZnAl2O4.
(b)

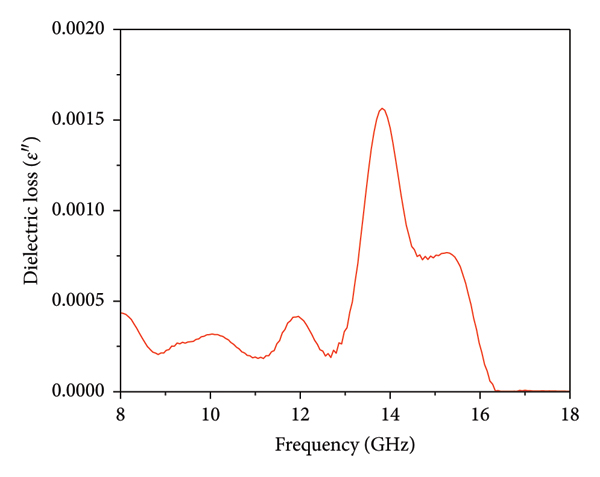
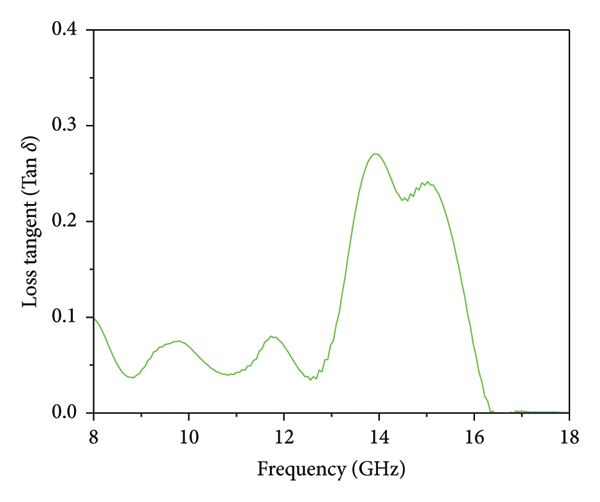
The measurement of dielectric properties provides valuable information about a material’s relative permittivity and permeability. Complex permittivity and permeability are crucial factors in studying a material’s microwave absorption properties [45]. The dielectric measurement was performed for this study using the open‐ended coaxial cable method. This method involves using a dielectric probe to make contact with the material and analyzing the reflected wave to extract important parameters such as dielectric constant (ε׳), dielectric loss (ε״), conductivity (σ), and loss tangent (tan (δ)) [45]. This method is suitable for measuring the dielectric properties of solids, semisolids, liquids, and powders. However, it may not be as accurate for measuring the complex permeability of magnetic materials. Nevertheless, relevant information on the dielectric properties of magnetic materials can be found in the literature [49–51]. The Mg/ZnAl2O4 structure is commonly regarded as a nonmagnetic material. However, the presence of notches in its permeability curves suggests the existence of extensive exchange and natural resonances [29]. These resonances are believed to arise from dislocations and defects present in the crystal structure. The complex permittivity is expressed as ε r = ε׳−j ε״, where ε׳ represents energy storage and ε״ represents energy loss. Figure 7 showcases the dielectric constant, dielectric loss, conductivity, and loss tangent in the frequency range of 8 to 18 GHz, providing a visual representation of these properties. The Mg–ZnAl2O4 material has a range of ε׳ values from 3.09 to 6.31, with an average of 4.52, in the frequency range of 8 to 18 GHz, as depicted in Figure 7(a). It is observed that the dielectric constant increases as the frequency increases, reaching a maximum of 12.26 GHz. Afterward, the dielectric constant decreases with increasing frequency until it reaches a minimum of 16.52 GHz. Beyond 16.52 GHz, there is a slight increase in the dielectric constant up to 18 GHz. This behavior is attributed to the increased interfacial polarization process, which increases the dielectric constant with higher frequencies. Additionally, the reduction in the space charge polarization effect causes a decrease in the dielectric constant as the frequency increases [45]. Excessive permittivity can negatively affect impedance match and lead to significant reflection and weak absorption. The ability of a material to attenuate electromagnetic waves depends on both the real and imaginary components of permittivity. Therefore, it is more suitable to examine these properties using dielectric loss. When the permittivity satisfies the impedance match criteria, a higher dielectric loss indicates better electromagnetic absorption capabilities. As an electromagnetic absorbent, higher values of the imaginary component of complex permittivity and greater dielectric loss are desired. Figure 7(b) illustrates a significant increase in dielectric loss within the frequency range of 12.26 GHz to 16.52 GHz, peaking at 13.82 GHz. Additionally, Figure 7(c) demonstrates the conductive properties of the Mg–ZnAl2O4 material, showing that the conductivity (σ) reached its peak at 13.82 GHz with a value of 1.2 S/cm. Moreover, Figure 7(d) depicts the behavior of the loss tangent in the Mg–ZnAl2O4 material, which exhibited a similar trend to the dielectric loss and conductivity. The loss tangent (tan δ) is defined as the ratio of the imaginary component to the real component of the complex permittivity. The real and imaginary parts of the dielectric constant and tan δ displayed a dispersive nature, suggesting a Debye‐type relaxation behavior in the entire Mg–ZnAl2O4 sample. This behavior was believed to be influenced by the Mg/ZnAl2O4/MgO/ZnO phase interface effect, grain boundaries, and grain interior. Based on these findings, it was suggested that the spinel aluminate sample is more suitable for microwave absorption applications.

Dielectric parameters of spinel Mg–ZnAl2O4: (a) dielectric constant, (b) loss factor, (c) conductivity, and (d) loss tangent.
(b)
(c)

(d)
Further study of the dielectric loss mechanism of the heterostructure Debye dipolar relaxation study can be conducted. The permittivity (ε
r
) of the material can be expressed [45, 52] as follows:
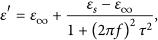

Finally, from the above two equations, the relationship between ε′ and ε″ can be expressed as follows [45, 53, 54]:
The equation mentioned above suggests that a plot of ε′ versus ε″ will form a semicircle. This semicircle is referred to as the Cole–Cole plot, which explains the Debye relaxation mechanism in the material. Each semicircle represents a Debye relaxation process, whereas a distorted semicircle represents another relaxation process in the sample [45, 53, 54]. Additionally, a straight line in the plot indicates conduction loss. In Figure 8(a), the Cole–Cole plot of the spinel Mg–ZnAl2O4 is shown. The graph clearly demonstrates the presence of multiple relaxation processes in the spinel and conduction loss indicated by the straight line [55]. Therefore, both relaxation loss and conduction loss contribute to the dielectric loss of the Mg–ZnAl2O4.

(a) Cole–Cole plot. (b) Reflection loss profile of spinel Mg–ZnAl2O4 at various thicknesses.
(b)

The performance of microwave‐absorbing material can be evaluated by measuring the RL, which indicates the amount of energy that is reflected back from the material’s target [38, 45]. A lower RL value signifies less reflected energy, and RL values below −10 dB indicate that the material absorbs over 90% of the electromagnetic wave. The frequency range where the RL value is below −10 dB is considered the EAB and is a crucial parameter in the design of microwave‐absorbing materials [45]. The RL value should be maximized to achieve optimal microwave absorption, and the absorption bandwidth should be as wide as possible within the desired frequency range. The RL of spinel Mg–ZnAl2O4 can be calculated using appropriate formulas based on transmission line theory and the material’s electromagnetic parameters [45, 56]:





Microwave absorption properties of spinel Mg–ZnAl2O4.
Furthermore, the magnetic loss of synthesized Mg–ZnAl2O4 can be significant, primarily arising from hysteresis and eddy current losses, which depend on the material’s composition, crystalline structure, and external magnetic field frequency [62–64]. Typically, as Mg–ZnAl2O4 possesses a spinel structure, its magnetic loss characteristics can be tailored through the ratios of magnesium and zinc, influencing the size and orientation of magnetic domains. Higher magnetic losses in this material often translate to improved wave absorption performance, as the dissipated energy leads to effective attenuation of incident electromagnetic waves. However, excessive magnetic loss might result in overheating, potentially degrading the material’s structural integrity and overall efficacy over long‐term applications. Thus, although magnetic losses can enhance wave absorption capabilities, careful consideration must be given to optimizing the material’s properties to avoid any adverse effects associated with thermal management.
Figure 9 displays a schematic diagram illustrating the microwave absorption mechanisms of Mg–ZnAl2O4 composites, when subjected to an alternating electric field, interfacial polarization occurs at the boundaries of the ZnAl2O4/MgAl2O4 grain boundaries. The polar ZnO molecule acts as the site for dipole polarization, along with defects and residual groups in Mg–ZnAl2O4. Increasing the content of MgO and/or ZnO enhances both interfacial polarization and dipole polarization, resulting in increased absorption and attenuation of electromagnetic energy. MgO/ZnO/Mg–ZnAl2O4 benefits from a high carrier concentration and mobility, which enhances interfacial polarization capability and dielectric loss [65]. Additionally, the conductance loss caused by Al2O3 and ZnO absorbents plays a crucial role in attenuating electromagnetic waves, and this effect is further strengthened by increasing the content of conductive components. The nanolayered structure of ZnO, ZnAl2O4, MgO, and MgAl2O4 provides numerous interfaces, which cause multiple reflections and scattering of electromagnetic waves, leading to the dissipation and attenuation of a greater amount of electromagnetic energy within the composites. As a result, the combined effects of interface polarization, dipole polarization, conductive loss, and multiple scattering contribute to the exceptional microwave absorption properties of the prepared Mg–ZnAl2O4 composite, allowing for broad bandwidth and strong absorption.

Schematic illustration of microwave absorption mechanisms of spinel Mg–ZnAl2O4.
4. Conclusion
In conclusion, this study successfully synthesized spinel‐structured Mg–ZnAl2O4 using a sol–gel technique. Structural analysis confirmed the presence of various phases, including ZnO, MgO, ZnAl2O4, and MgAl2O4, with an average crystallite size of 88 nm. The material exhibited a lower band gap of 3.19 eV compared with ZnAl2O4 and MgAl2O4, indicating enhanced microwave absorption capabilities. Dielectric characterization revealed a dielectric constant range of 3.09 to 6.31 across 8–18 GHz, with a maximum RL of −71 dB at 14.02 GHz and an EAB of 3.66 GHz at 5 mm thickness. The high carrier concentration and mobility of Mg–ZnAl2O4 were found to enhance its interfacial polarization and dielectric loss, contributing to the material’s strong microwave absorption performance. The synergistic effects of interfacial interactions at grain boundaries, involving the various phases present, played a crucial role in the material’s exceptional microwave absorption properties. These findings establish Mg–ZnAl2O4 as a promising candidate for microwave absorption applications.
Data Availability Statement
All data generated or analyzed during this study are included in this article.
Conflicts of Interest
The authors declare no conflicts of interest.
Author Contributions
All of the authors mentioned contributed significantly to the development and writing of the paper.
Funding
The authors received no financial support for the research.
Footnotes
Acknowledgments
The authors would like to acknowledge and thank the Central Sophisticated Instrumentation Facility, BITS Goa, and Dr. Swagatam Sahoo, Department of Electronics and Communication Engineering, National Institute of Technology Jamshedpur, Jharkhand, for their support in dielectric studies and the DAK 3.5 facility. We also thank Dr. Anirban Chatterjee for providing CST simulation software facilities.
