Abstract
Unpleasant breathing can occur due to bad odors from wearing a medical mask. Electrospinning has been used to incorporate Eucalyptus extract into polyvinyl alcohol (PVA) to overcome the problem of medical masks and as an alternative, as part of an antiflu breathing face mask. This research aims to extend the shelf life of the pleasant odor by encapsulating Eucalyptus essential oil (EO) in nanofibers by coaxial electrospinning. Initially, nanofibers were produced using a blend structure of Eucalyptus aqueous extract (EA) and PVA, along with a core–shell structure of EO and PVA by electrospinning. The morphology of the nanofibers was investigated by scanning electron microscopy (SEM) and transmission electron microscopy (TEM), showing average diameters of 270 ± 47 nm for the blend nanofibers and 343 ± 126 nm for the core–shell nanofibers. TEM confirmed a core–shell structure presumably caused by the nonmiscibility of EO and PVA, as well as the high viscosity of the PVA solution. The loading capacity (LC) and encapsulation efficiency (EE) were calculated for the core–shell nanofibers to be 16.43 ± 3.29 and 55.20 ± 2.31, respectively. The presence of EO in the nanofibers was shown through FT‐IR spectroscopy. At the same time, variations in peak intensity allowed tracing the displacement of EO and the aging of the nanofibers. A released study showed that after 7 hours of release in a conditioning room with 30°C and 70% humidity, the percentages of remaining Eucalyptol in the nanofibers were 68.36% and 83.63% for the blend and the core–shell structure, respectively. The size of the pores in these nanofibers on a layer ranged from 50 nm to 200 nm.
1. Introduction
Medicinal plants have met the requirements to cure or prevent many diseases since ancient times. Among these diseases are colds and flu, which are contagious diseases of the upper respiratory tract (larynx, ears, and nose) and mainly affect the nose [1]. Eucalyptus has a long history of being used to treat colds and as a deodorizing agent. An essential medicinal agent of this plant is called cineol, which makes the secretions flow, and the mucus is removed more easily. Due to its antiseptic effect, it prevents the growth of bacteria in the respiratory system [2]. Incense is a 6000‐year‐old aromatherapy technique that is used as a complementary therapy. This treatment method has received much attention due to the use of natural and biocompatible materials in the 21st century [3]. Eucalyptus incense regulates and activates various body systems such as the nervous system, relieves headaches and weakness, and increases the body’s immunity against infection and flu [4]. Also, this incense’s effectiveness has been proven in treating infectious diseases, such as throat infections, colds, bronchitis, asthma, sinusitis, and respiratory care [5]. Domingues et al. [6] showed that eucalyptus oil in addition to a deodorizing agent can be highly efficient against Escherichia virus MS2, a surrogate of SARS‐CoV‐2, increasing the multifunctionality of medical masks.
An electrospinning system produces fibers whose diameter is between less than one micron and several nanometers, which can be used in various fields, including health, therapy, energy, and environment. One of the most important uses of electrospun nanofibers is their use in health and medical applications. Electrospun nanofibers and their structure filled with plant extracts, and essential oils (EOs) have received much attention in recent years [7]. Due to their antimicrobial and anti‐inflammatory properties, these materials are used for food packaging, wound care against bacteria, wound healing, and other applications [8]. Lou et al. [9] demonstrated that nanofibrous membranes are effective at repelling Forcipomyia taiwana when Eucalyptus globulus oil is incorporated with polyvinyl alcohol (PVA) in a 90/10 ratio. In another work, Benas et al. [10] have encapsulated Eucalyptus EO through mixing and a layer‐by‐layer technique in polyvinyl butyral using electrospinning. They showed that adding 0.2 g of eucalyptus into polyvinyl butyral fabric as an intermediate layer in a medical mask through mixing results in a deodorization rate of 80% after 2 h. They successfully proved that this type of mask’s middle layer not only effectively protects against coronavirus, but also provides better scents and makes it more comfortable for consumers. Core–shell nanofibers are one of the one‐dimensional nanostructures that have unique properties and many applications. The electrospinning process is more important among the various methods of producing two‐component core–shell nanofibers [11]. One of the important applications of core–shell nanofibers is the encapsulation of an unstable compound from an active medium. In coaxial electrospinning, it is also possible to produce core–shell nanofibers with a liquid core. In some biological applications, the core must never solidify and remain in liquid form [12]. Zhang et al. [13] demonstrated that thymol was slowly released as an antibacterial agent when encapsulated in poly(lactide‐co‐glycolide) using coaxial electrospinning.
PVA is a water‐soluble, semicrystalline, biocompatible, hydrophilic polymer with thermal and chemical stability, good mechanical properties, and is nontoxic. Also, this polymer has good processability, and its best solvent is water. It has a hydroxyl group in its repeating units, which causes this polymer to get cross‐linked. Today, polymers are used increasingly in a variety of industrial applications. In addition, PVA in the form of fibers is a good choice for use in industries, such as filtration, tissue engineering, optics, membranes, and drug release [14]. Khanzada et al. [15] demonstrated that electrospun nanofibers made from aloe vera and PVA exhibited significant antibacterial activity against both Gram‐positive (Staphylococcus aureus) and Gram‐negative (Escherichia coli) bacteria, making them suitable for use in face masks and surgical gowns.
The filter is a porous structure with a large volume of air. The use of nanofiber technology in filtration has received much attention due to the creation of engineered porosity, flexibility, and the use of fibers with different subtleties. The fabric’s surface pores enable Van der Waals interactions, facilitating the adsorption of external agents [16]. Using nanofibrous membranes has many advantages. Still, a significant weakness of the nanofilter is that the nanofibers alone do not have the necessary strength, so to increase the strength, the nanofibers are placed on a suitable substrate. Whenever the target application is a breathing mask, a three‐layer spunbond–melt blown–spunbond is used as a backing layer [17]. Wang et al. [18] demonstrated that PVA‐based medical masks exhibit high humidity resistance and self‐charging performance, which enhances their electrostatic adsorption efficiency.
This study aims to develop a core–shell nanofiber structure that will gradually release a pleasant odor and extend its shelf life. This study involved encapsulating EO in PVA nanofibers using coaxial electrospinning and comparing its properties with those of a blend structure. We utilized PVA nanofibers on a spunbond–melt blown–melt blown–spunbond (SMMS) layer due to their biocompatibility, surface pores, and self‐charging properties, which were reviewed in the articles. These characteristics enhance van der Waals interactions and electrostatic adsorption efficiency, which improves the adsorption of external agents. The size of the pores in these nanofibers on a layer ranged from 50 nm to 200 nm. The mechanism behind the release of eucalyptus from the core–shell nanofibers in this structure involves the process of hydrolysis. When a person inhales and exhales, the water vapor causes both surface and mass hydrolysis of the nanofibers. This process results in the gradual release of eucalyptus. A released study showed that after 7 hours of release in a conditioning room with 30°C and 70% humidity (human breathing conditions), the percentages of remaining Eucalyptol in the nanofibers were 68.36% and 83.63% for the blend and the core–shell structure, respectively. This report shows that the fabricated structure in this work can alleviate unpleasant breathing caused by bad odors from wearing medical masks, serving as an alternative for part of an anti‐flu breathing face mask.
2. Materials and Methods
PVA in powder form with a molecular weight of 72,000 Da, aqueous extract (EA) (prepared using the steam distillation method with water) with a concentration of 0.67 g/mL, and EO with a concentration of 0.19 g/mL were purchased from Merck Co. (Germany), Dineh Co. (Tehran, Iran), and Narmak Co. (Tabriz, Iran), respectively. The 100 mL phosphate‐buffered saline (PBS) tablets were purchased from Pharmed Co. (Tehran, Iran).
2.1. PVA Electrospinning
4%, 6%, 8%, and 10% W/V PVA solutions were prepared by dissolving 0.16, 0.24, 0.32, and 0.4 g PVA powder in 4 mL distilled water, respectively, under magnetic stirring (Heidolph Co., Germany) at 80°C, 900 rpm, and 2 hours [19]. A volume of 1 mL of solution was picked up by syringe and electrospinning operation (setup consisted of voltage source 0–30, digital syringe pump MS20‐20 model Daiwha Co. made in Korea, aluminum foil collector plate, and steel needle with 0.7 mm outer diameter) was performed with voltage 20 kV, a distance of 18 cm, and a feed rate of 0.2 mL/h. Table 1 and Figure 1 show the electrospinning conditions at different concentrations and their schematic.
Electrospinning conditions at different concentrations.

Schematic of PVA electrospinning.
2.2. PVA/EA Electrospinning (Blend Electrospinning)
After optimizing the electrospinning of PVA, 10%, 20%, 30%, and 40% in grams of EA relative to the weight of the polymer were added to the 8% w/v solution of PVA. Then, the solutions were placed on a magnetic stirrer for 1 h at 900 rpm and room temperature. The solutions were changed to yellow in color and uniform. Then, electrospinning was done with a voltage of 20 kV, a distance of 18 cm, and a feed rate of 0.2 mL/h. Table 2 and Figure 2 show the electrospinning conditions at different concentrations of EA and its schematic.
Blend electrospinning conditions at different concentrations of EA.

Schematic of PVA and EA for blend electrospinning.
2.3. Coaxial Electrospinning
First, an 8% w/v solution of PVA in distilled water was prepared to prepare a nanofibrous layer with a core–shell structure of PVA and EO. Then, 1 mL of the polymer solution and 1 mL of EO were picked up by two different syringes. Electrospinning was performed with a coaxial needle with a 2.4 mm outer diameter and 1.05 mm inner diameter, a shell feed rate of 0.5 mL/h, a core feed rate of 0.15 mL/h (feed rate optimization achieved with trial and error), a voltage of 20 kV, and a distance of 20 cm. In this structure, the polymer solution was placed in the shell and EO in the core. Table 3 and Figure 3 show the coaxial electrospinning parameters and its schematic.
Coaxial electrospinning parameters.

Schematic of PVA and EO for coaxial electrospinning.
2.4. Fabrication of Nanofibrous Filter Media
To prepare the nanofibrous filter media, electrospinning was performed on a four‐layer nonwoven (SMMS, weight of one square meter: 21.5 g, thickness: 0.111 mm, fiber diameter taken from scanning electron microscopy (SEM) images: 14 ± 3.67 μm, Baftineh Co., Iran). To do this, aluminum foil was wrapped around the rotating roller. Then, the SMMS layer was placed on it, and nanofibers were collected on the SMMS layer (Figure 4). Two types of nanofibrous filter media were prepared: (1) blend nanofibers on the SMMS layer (BNSM) and (2) core–shell nanofibers on the SMMS layer (CNSM).

Schematic of the fabrication of nanofibrous filter media.
2.5. SEM and Transmission Electron Microscopy (TEM)
A SEM (AIS2100 Seron technology, Korea) was used to analyze the nanofibers’ morphology after gold sputter coating. An image analysis software (Digimizer, MedCalc Software, Belgium, Version 5.4.6) was used to calculate the diameter and porosity of the nanofibers and nanofibers on a SMMS layer from the SEM images. A TEM (Ara‐80 Ara Pajuhesh, Iran) was used to examine the volume of nanofibers (core–shell structure). Before analysis, the electrospun fiber was placed on the mesh copper TEM grid.
2.6. Statistical Analysis
Analysis of variance (ANOVA) was done with a 95% confidence interval to compare the average diameter of PVA: (PVA and EA) nanofibers as a blend, and PVA and EO as a core–shell by Minitab software. The Tukey test was also performed to compare the means one by one.
2.7. Fourier Transform Infrared Spectroscopy (FT‐IR)
FT‐IR spectroscopy using the ATR setup (Thermo‐Nicolet‐Nexus, USA) with a spectral range (400–4000 cm−1) was used to investigate the functional groups. Five spectra related to PVA, EO, EA, and nanofiber (blend and core–shell) were compared using Origin software (OriginLab Corporation, USA).
2.8. Loading Capacity (LC) and Encapsulation Efficiency (EE)
According to equations (1) and (2), the LC and EE of nanocarriers were calculated, respectively [20, 21]. Spectrophotometer UV‐visible absorption (Photonix, Tayf Sanj Tajhiz Pishraft, Iran) was used to determine EA and EO concentration on the surface nanofibers at wavelengths of 206 nm:

2.9. Eucalyptus Release Study
To compare the release rate of EA and EO from nanofiber samples, a spectrophotometer UV‐visible absorption (Photonix, Tayf Sanj Tajhiz Pishraft, Iran) was used. First, standard curves were prepared for concentrations of 10, 100, 200, and 400 μg/mL EA and EO in PBS (the EO emulsion in PBS was prepared by stirring rapidly at 1200 rpm for 1 h, followed by immediate placement of the emulsion in the spectrophotometer), and the diagram was plotted at the maximum wavelength (206 nm) in terms of concentration relative to adsorption. Since the release environment in the application of the breathing mask is humid air, to simulate the actual application environment as a breathing mask, a condition room (Rex Co., Iran) was used. For this purpose, the conditioned room was set with a temperature of 30°C and a humidity of 70% (inhalation and exhalation conditions). Eight samples were prepared with 30 mg weight. One sample was considered as a control sample, and the other was placed in the conditioned room. Each of the samples was taken out of the conditioning room at an hour interval time. Then, the concentration of EA and EO was measured after complete release in 5 mL PBS.
2.10. Porosity Measurement
Porosity is defined as the percentage of free space volume to the total volume of material bulk. The percentage of open area is determined by equation (3), in which A0 is the share of air and A
t
is the total area. SEM images were used to calculate the percentage of open area and measure the porosity and pore size distribution of nanofibrous filter media using ImageJ software. It is assumed that porosity and percent open area are equivalent for two‐dimensional textiles, such as woven fabrics and thin nonwovens [22]:
2.11. Air Permeability Rate
Several samples from different parts of the nanofibrous filter media were selected to test the air passage through the nanofibrous filter media. The 5‐cm square surface of the sample was tested by an air permeability measuring device (SDL Co, United Kingdom). A standard pressure of 100 Pascale was applied to each sample, and the air permeability of the nanofilter media was measured [23]. Also, the thickness of the nanofilter samples was measured by a thickness gauge (OSK Co., Japan).
3. Results and Discussion
EE and EO were loaded in nanofibers using electrospinning (in blend and core–shell states), and different physicochemical aspects of nanofibers and release rate were investigated.
3.1. Morphology of Nanofibers
The nanofibers’ morphology was analyzed using SEM and TEM. In the electrospinning process, the concentration of the polymer solution, the applied voltage, the feed rate of the polymer solution, and the electrospinning distance are important factors in the formation of stable jets and nanofibers [24]. In the first stage, PVA solutions at different concentrations 4%, 6%, 8%, and 10% W/V were prepared. At 4% concentration, the electrospinning jet was unstable and nanofiber formation failed. Nanofibers were formed after stable electrospinning with increasing PVA concentrations as reported in a previous work [25]. Figures 5 and 6 show the impact of different concentrations of PVA on morphology and diameter. Nanofiber diameter increased with an increase in PVA concentration from 6% to 10%, which can be correlated with the increase in viscosity.

SEM image of nanofibers produced under different PVA concentrations: (a) 6%, (b) 8%, and (c) 10%.
(b)

(c)


The effect of PVA concentrations on the nanofiber average diameter.
Similar trends were reported by Lou et al. [9], where diameters grew from ∼300 to 360 nm as PVA concentration increased from 12 to 16 wt.% at an applied electrospinning voltage of 20 kV. In this regard, in another work, Supaphol and Chuangchote reported that PVA solution viscosity and nanofiber diameter increased with increasing PVA concentration [26]. As the concentration increases, the viscoelastic force increases, which prevents the jet from being pulled by the Coulomb force, resulting in a larger diameter fiber in accordance with the literature [27].
The concentration of 8% resulted in finer nanofibers with a low standard deviation. This concentration was used in the blend and core–shell electrospinning.
In the next step, the blend electrospinning technique was performed by adding 10%, 20%, 30%, and 40% EA in grams relative to the weight of the polymer in an 8% PVA solution. By adding a high percentage of EA (40% by weight of the polymer), a stable jet was not formed due to the decrease in optimum viscosity, while nanofibers were formed in other percentages of EA (10, 20, 30%). Since a high percentage of Eucalyptus extract is desirable for effectiveness as a drug, the addition of 30% of EA to the PVA solution was evaluated [28].
Core–shell nanofibers were also formed (by trial and error in feed rate and electrospinning jet appearance) on the collector, in which PVA was the shell and EO the core. Figure 7 shows the SEM image of blend nanofibers and core–shell nanofibers with 270 ± 47 nm and 343 ± 126 nm average diameters, respectively. ANOVA was performed with a 95% confidence interval to compare the mean diameter of PVA, blend, and core–shell nanofibers, which indicated a significant difference.

SEM image of blend and core–shell nanofibers. (a) Blend nanofibers with 30% EA by weight of the polymer, with 270 ± 47 nm average diameter. (b) Core–shell nanofibers EO as the core and PVA as the shell, with 343 ± 126 nm average diameter.
(b)

The Tukey test was also performed to compare the means. Table 4 and Figure 8 show the mean diameter of nanofibers and the results of the Tukey test, which shows the significant difference in the mean diameter of the core–shell structure with the other structures, which can be related to the increase in feed rate [29].
The results of the Tukey test, which shows the significant difference in the mean diameter of various nanofibers.
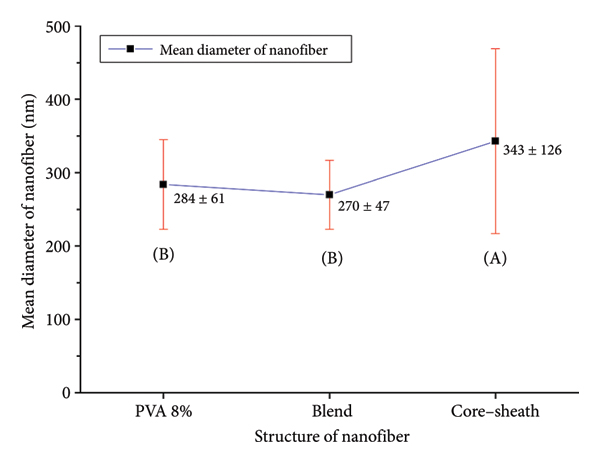
Mean diameter of nanofibers and the grouping results of the Tukey test.
TEM was used to confirm the core–shell nanofibers structure (Figure 9). Differences in dark and light regions proved that EO was encapsulated in PVA, and the core–shell structure was formed, even if it was not formed continuously and uniformly, and in some parts was discontinuous. More optimization is needed to achieve a uniform core–shell structure, which is affected by the nonmiscibility of EO and proper viscosity of PVA solution, as reported in the literature [30]. The mean diameter of nanofibers in the TEM image matched the mean diameter in the SEM images.

TEM image of EO loaded in the PVA nanofiber.
3.2. FT‐IR Spectroscopy
The FT‐IR spectra of EA, EO, PVA, blend, and core–shell nanofibers are shown in Figure 10. The spectrum of PVA exhibited absorption bands of –OH stretching at 3400 cm−1, ‐C=O stretching related to unhydrolyzed groups at 1720 cm−1, ‐CH2 at 1440 cm−1, and ‐CH3 at 1337 cm−1. Four absorbance peaks were observed for EA, a broad band at 3400 cm−1 related to –OH stretching, 2938 cm−1 related to ‐CH stretching, 1375 cm−1 related to ‐CH2, and 1252 cm−1 related to C‐O [10]. The spectrum of EO exhibited absorption bands of ‐CH stretching at 2800 cm−1 and ‐CH2 at 1375 cm−1. According to Figure 9, the characteristic spectrum of the blend nanofibers is similar to the characteristic spectrum of PVA nanofibers, which can be due to the released EA before the experiment.

FT‐IR spectra of (a) EO, (b) EA, (c) PVA nanofiber, (d) blend nanofiber, and (e) core–shell nanofiber.
The interaction between polymers and active compounds often results in detectable FT‐IR peak shifts, as reported in similar encapsulation systems [13]. In our study, the observed peak displacements and intensity changes in the core–shell nanofibers (Figure 10) align with the characteristic spectra of both PVA and EO, confirming the successful incorporation of EO within the PVA matrix. These shifts further suggest molecular‐level interactions between EO and PVA, consistent with encapsulation behavior observed in other polymer‐based systems.
3.3. LC and EE
The electrospinning technique is under focus for producing core–shell structures with high EE [31]. LC and EE for the blend nanofiber were 20.63 ± 0.87 and 69.13 ± 1.07, respectively (Table 5). For core–shell nanofibers, the LC and EE were 16.43 ± 3.29 and 55.20 ± 2.31, respectively. LC and EE were expected to be higher in the core–shell structure and were not comparable with the blend structure one due to nonuniformity and continuity in encapsulating EO by coaxial electrospinning, and the amount of Eucalyptol in EO and EA was not the same. The amount of Eucalyptus in EO was less than that in EA.
LC and EE for blend and core–shell nanofibers.
3.4. Eucalyptus Release Study
The release of Eucalyptus in a simulated environment for a respirator mask was investigated in a conditioning room with a temperature of 30°C and a humidity of 70%. Figure 11 shows the percentage of remaining Eucalyptol in the nanofibers after putting them in the conditioned room for 7 h.

Percentage of remaining Eucalyptol in the nanofibers after putting them in the conditioned room for 7 h.
The controlled samples (0 h) had the highest concentration of Eucalyptol in BNSM and CNSM nanofibrous filter media. The concentration of Eucalyptol in the nanofibrous filter media decreased when the samples were exposed to temperature and humidity. The percentage of remaining Eucalyptol in the BNSM and CNSM nanofibrous filter media by the end of the seventh hour was 68.36% and 83.63%, respectively. Because PVA is a biodegradable polymer, the polymer chains are degraded by hydrolysis in a humid environment and converted into more minor compounds [32]. Hydrolysis occurs on the surface and mass of PVA nanofibers. That resulted in the release of Eucalyptol from the nanofibrous filter media due to the difference in concentration of Eucalyptol within the nanofibers and the environment [33]. The release rate of Eucalyptol in the blend and core–shell structure is not comparable due to not having the same concentration of Eucalyptol. However, the release rate in the core–shell structure is expected to be more limited than the blend structure [34]. According to linear regression, it can be predicted that the percentage of remaining Eucalyptol concentration in BNSM and CNSM nanofibrous filter media finishes at 25 and 44 h, respectively, by destruction and diffusion mechanisms [35]. Encapsulated Eucalyptol in nanofibers can be used in a respirator mask to increase release time and provide more protection.
3.5. Porosity of Layers
As previously mentioned, porosity is a morphological attribute that is characterized by the presence of empty spaces between the components of nanofibers [22]. The porosity and pore size distribution in the SMMS layer, BNSM, and CNSM nanofibrous filter media were analyzed using SEM images. The SMMS, BNSM, and CNSM nanofibrous filter media had thicknesses of 0.111 mm, 0.135 mm, and 0.128 mm, respectively. Figures 12 and 13 show the porosity percentage and pore size distribution in the SMMS layer, BNSM, and CNSM nanofibrous filter media, respectively. The results indicated that the porosity in the SMMS layer was more than that of CNSM and BNSM, respectively. In addition, the pore size of the SMMS, BNSM, and CNSM nanofibrous filter media was computed for 100 pores; the results were 75.10 ± 30.11 μm, 62.01 ± 30.71 nm, and 180.6 ± 65.69 nm, respectively. Based on the SEM evidence and the assumption that the layer was thin (two‐dimensional), it can be concluded that an increase in fiber diameter leads to an increase in porosity and pore size in consistence with Soliman et al.’s work [36]. Since the size of the pores in BNSM and CNSM was between 50 nm and 200 nm, the produced nanofibrous filter media were in the macropores category [37].

Porosity percentage in various layers.

Pore size distribution of (a) SMMS layer, (b) BNSM nanofibrous filter media, and (c) CNSM nanofibrous filter media.
(b)
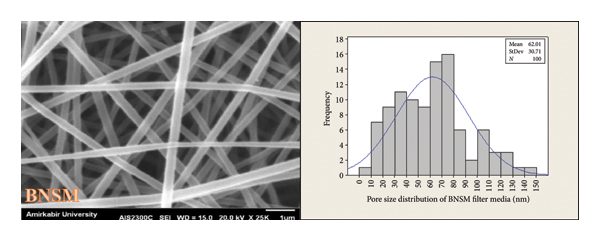
(c)
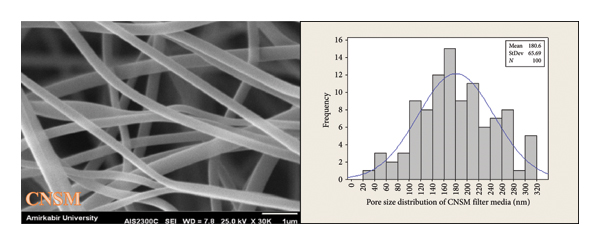
3.6. Air Permeability Rate
Air permeability is an important feature of any filter media and depends on its structural parameters [38]. By conducting air permeability tests, an investigation was carried out on the air permeability of the two produced nanofibrous filter media, namely, BNSM and CNSM. The value of air permeability for the SMMS, BNSM, and CNSM nanofibrous filter media was 66.28 ± 3.5 (mL/s·cm2), 5.2 ± 1.9 (mL/s·cm2), and 5.5 ± 1.5 (mL/s·cm2), respectively. Figure 14 displays the ratio of air permeability, nanofiber diameter, porosity, and thickness for both BNSM and CNSM nanofibrous filter media. Results showed that the air permeability in CNSM nanofibrous filter media was higher than in BNSM nanofibrous filter media due to lower filter thickness, higher fibers’ mean diameter, and porosity.

Comparison of the air permeability, nanofiber diameter, porosity, and thickness in two samples of BNSM and CNSM.
4. Conclusions
Due to the growth of public health awareness, the use of medical masks has increased. Unpleasant odors can sometimes arise from the long‐term use of medical masks, leading to discomfort while breathing. To address this issue and prolong the durability of a pleasant scent, coaxial electrospinning has been employed to encapsulate Eucalyptus EO within PVA. Initially, we fabricated core–shell nanofibers using coaxial electrospinning of EO and PVA, and then, we compared their characteristics with the blend structure nanofibers of Eucalyptus EA and PVA. The average diameter of the core–shell nanofibers was 343 ± 126 nm. The presence of EO in the nanofibers was demonstrated using FT‐IR spectroscopy. TEM images revealed a core–shell structure likely formed due to the immiscibility of EO and PVA, along with the high viscosity of the PVA solution. LC and EE were calculated for the core–shell nanofibers to 16.43 ± 3.29 and 55.20 ± 2.31, respectively. After exposing core–shell nanofibers for 7 hours in a conditioning room at 30°C and 70% humidity (simulating human breathing conditions), we found that they retained approximately 83.63% Eucalyptol. Additionally, eucalyptol can remain stable in these conditions for up to 44 h. For the application in medical masks, we utilized an SMMS layer as the backing layer for nanofibers. The size of the pores in the nanofibers on the SMMS layer ranged from 50 nm to 200 nm, which falls into the macropore category. This report indicates that the fabricated structure developed in this work can help alleviate unpleasant breathing caused by bad odors from long‐term use of medical masks. It achieves this by extending the release time of Eucalyptol and offering enhanced protection time, potentially serving as an alternative for certain antiflu breathing face masks.
Data Availability Statement
The data used to support the findings of this study are included within the article.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
The authors declare that no contractual funding was received for this research. The authors gratefully acknowledge the financial support provided by the Research Affairs of Amirkabir University of Technology, which enabled this work as part of a postgraduate project.
Footnotes
Acknowledgments
The authors declare that no contractual funding was received for this research. The authors gratefully acknowledge the financial support provided by the Research Affairs of Amirkabir University of Technology, which enabled this work as part of a postgraduate project. They express their deep appreciation for the thesis authored by Farzaneh Karampour Shahvali which was published in the Amirkabir University Library in the field of research on PVA/Eucalyptus nanofiber.
