Abstract
A new magnetic P. harmala biochar/Mn-ZnFe2O4 material was prepared by the one-step pyrolysis approach and characterized by FT-IR, SEM, XRD, and BET techniques. The efficacy of removing rhodamine B (RhB) via adsorption and catalytic degradation was examined. Adsorption removal attained 82% of 10 mg·L-1 RhB at pH 4.0, 90 min of agitation, and 3.2 g·L-1 of adsorbent. The adsorption kinetics indicated a diffusion-limited mechanism and matched the second-order model. The equilibrium data are better suited by the Langmuir isotherm, and the maximum monolayer adsorption capacity (
1. Introduction
The reuse of water after pollutant removal is one of the most serious environmental concerns. When a toxic substance enters the water, its quality deteriorates. Dyes are a major source of pollution, particularly in water treatment. Several synthetic organic dyes are widely used and essential in a variety of industries. Dye-contaminated effluent disposal can seriously pollute surface and groundwater [1]. This is because most dyes are toxic and endanger human health. The level of contamination by a specific dye distinguishes its true application. Rhodamine B (RhB) dye, for example, is widely used in the textile, food, cosmetics, and pharmaceutical industries [2, 3]. Huang et al. [4] disclosed the high toxicity in a body of water as a result of RhB disposal. Congenital diseases and cancer are among the health risks posed by RhB in water [5, 6]. The removal of RhB dye from water can be accomplished through catalytic degradation and/or adsorption. Several materials have been developed for the catalytic and adsorption removal of RhB. Among these catalysts are Ti/Ru0.3Ti0.7O2 [3], iron-doped mesoporous silica [7], nanozero valent iron (nZVI)@biochar [8], and TiO2-AuNP/polydimethylsiloxane sponge [9], which were reported. RhB adsorbent materials include zinc ferrite-polyaniline [1], activated pine cone [10], stalk corn-activated carbon [11], Argemone mexicana [12], bentonite-titanium dioxide composites [13], graphene oxide/silicalite-1 composites [14], carbon xerogels [15], and modified alpha-alumina [16]. When biomass is heated to a point of thermal breakdown without oxygen, a material called biochar is created that is rich in carbon and has fine grains. The solid biochar itself, the liquid biooil, and the syngas are the three byproducts of pyrolysis. Temperature, heating rate, and residence duration all affect the biochar’s characteristics [17]. A variety of biochars were made for RhB adsorption using a variety of materials, including macroalgae [18], olive waste biomass [19], goat manure [20], and cocoa pod husk [21]. Due to their high surface-to-volume ratio, superparamagnetic characteristics, and nanoscale size, several ferrite nanoparticles have been applied to the filtration of water. However, one of the fundamental issues with these nanomaterials’ actual usage as adsorbents is their tendency to aggregate. As a result, they are frequently combined with polymers to create nanocomposites that can be utilized as adsorbents. The spinel ferrites have the chemical formula MeFe2O4, where Me stands for many metals that can be included in the ferrite lattice, including Mg, Ni, Cu, Mn, Co, and Zn. These metal-based ferrites are frequently employed for adsorption research because of their strong magnetic permeability and chemical stability [22]. In environmental purification processes, magnetic biochar has the potential to be used as an adsorbent, catalyst, or soil remediation agent. This is due to the fact that its magnetic separation capabilities make it possible to easily recover pollutants and/or adsorbents from water, rendering it an environmentally safe material. Additionally, the combination of magnetic nanomaterial and biochar results in high adsorption efficiency. Many magnetic biochars have been described as adsorbents, including banana peel biochar/Fe3O4 [23], ferric and biological sludge biochar [24], and Eichhornia crassipes/Fe3O4 [25].
In this study, we sought to prepare a new magnetic biochar/Mn-ZnFe2O4 of the Peganum harmala plant for the adsorption removal and catalytic degradation of RhB dye in water. The adsorption performance and removal efficiency of RhB dye from aqueous solutions will be evaluated. Also, the application of the new adsorbent in real water will be studied.
2. Experimental
2.1. Instrumentation
The spectrophotometric measurements of RhB dye were made using a Cintra 1010 double-beam UV-Vis spectrophotometer and Cintra 2.4 software (GBC Scientific Equipment, Braeside, Australia) at a fixed wavelength of 554 nm. For the pyrolysis process, a Nabertherm vacuum muffle furnace model B180 (Lilienthal, Germany) with a protective gas connection and a programmed furnace controller with a temperature range of 30-3000°C was used. The FT-IR spectra of the P. harmala biochar/Mn-ZnFe2O4 were recorded in the wavenumber range of 400–4000 cm-1 using a Fourier transform infrared spectrometer (Perkin Elmer 1750, MA, USA) using the KBr discs technique.
On the Bruker D8 Advance diffractometer (Berlin, Germany), an X-ray diffraction (XRD) pattern was captured using Cu-K1 anode radiation at 1.54058 and a secondary monochromator. To determine the phase composition and average crystallite size of the magnetic nanoparticles, the scanning speed was set at 1° min-1. The surface area and porosity were determined using the fully automated Micromeritics® TriStar II Plus analyzer model 2390t V2.03 (GA, USA). At 77 K and a relative pressure (P/Po) in the range of 0.0586–0.996, N2 was adsorbed and then desorption. Before measurement, the samples were degassed at 120°C for two hours. The specific surface area and total pore size were determined using the Brunauer-Emmett-Teller (BET) and the Barrett-Joyner-Halenda (BJH) models. The SEM micrograph of the surface morphology of the prepared magnetic biochar was performed on JEOL scanning electron microscope (MA, USA). A Jenway bench pH meter model 4510 (Keison International Ltd., Chelmsford, UK) was used to test the pH of the samples, and it was calibrated using buffer solutions of pH 4 and 9. Stuart orbital mechanical shaker model S1500 (London, UK), with a speed of 30-300 rpm, was used to agitate the samples. Hamilton water purification system (Hamilton Glass Ltd., Margate, UK) provided double-distilled water for the duration of the study. For all filtering purposes, qualitative filter paper No. 101 (Dorsan Filtration SL Co., Barcelona, Spain) was used. Glass bottles with Teflon caps were used for the adsorption study.
2.2. Reagents
All reagents used were of analytical reagent grade, and the standard solutions were prepared in DDW. The standard RhB solution was prepared using rhodamine B dye purchased from Suvachem Co. (Maharashtra, India). Manganese chloride (MnCl2), zinc chloride (ZnCl2), and ferric chloride nonahydrate FeCl3·9H2O, from Alpha Chemika™ (Mumbai, India), and sodium hydroxide (NaOH), from Sigma-Aldrich, were used to prepare the magnetic nanoparticles. By taking the appropriate amount of the reagent, a stock solution of 1000 mg·L-1 RhB was prepared. The working RhB concentration of 10 mg·L-1 was obtained by diluting the stock solution with double-distilled water.
2.3. Preparation of P. harmala Biochar/Mn-ZnFe2O4
It was decided to employ Mn and Zn in the magnetic biochar’s ferrite since Mn-ZnFe2O4 is one of the most prevalent soft ferrites. These ferrites are favored because of their good stability, high saturation magnetization, and high permeability. This establishes them as well-known and popular materials for catalysis and wastewater treatment. The one-step impregnation pyrolysis method was applied to prepare the P. harmala biochar/Mn-ZnFe2O4 adsorbent. According to the modified method [26], the P. harmala powder was dried at 105°C for 24 h and sieved to 0.1 mm. A 100 mL chemical solution of the nanoparticle precursor was prepared by mixing 30 mL of 0.25 mol·L-1 MnCl2, 20 mL of 0.25 mol·L-1 ZnCl2, and 50 mL of 0.5 mol·L-1 FeCl3 solution to get molar ratios of 0.6 : 0.4 : 2.0 from Mn2+, Zn2+, and Fe3+, respectively. Then, 10 g of the plant powder was added to the above solution and mechanically stirred for 2 h. Thereafter, 50 mL of 10 mol·L-1 sodium hydroxide was added dropwise, and the slurry was heated to 90°C for one hour till a viscous gelatinous solution was formed. The resulting gel material was placed in a tightly sealed and cleaned porcelain crucible and pyrolyzed under an N2 atmosphere in the NABER/B180 furnace for one hour at a setting temperature of 600°C and a heating rate of 10°C·min-1 from 100 to 600°C. Further, the magnetic biochar was cooled at room temperature, immersed in 0.1 mol·L-1 HCl solution under stirring for one hour, washed with double-distilled water to effluent that was barely neutral, and then dried, milled, and stored for further use. A ferrite with the chemical formula Mn0.6Zn0.4Fe2O4 is produced using these molar ratios of the metal ions, which is referred to as Mn-ZnFe2O4 throughout this study. A schematic diagram of the synthesis protocol and adsorption mechanisms is shown in Figure 1. Similarly, the P. harmala biochar was produced by direct pyrolysis of plant powder at a temperature of 600°C for 1 hour.

One-step impregnation synthesis protocol of P. harmala biochar/Mn-ZnFe2O4.
The point of zero-electric charge (
2.4. Adsorption Procedure
The removal of RhB dye was investigated using a 10 mL solution containing 10 mg·L-1 and 50 mg adsorbent and shaking for time intervals ranging from 2 to 100 minutes at 250 rpm at room temperature. The effect of pH on dye removal was studied from 1.0 to 9.0 using 0.1 mol·L-1 solution of HCl or NaOH for adjustment. The adsorbent dose was investigated from 0.2 to 5.0 g·L-1. The initial concentration of RhB was examined from 5.0 to 100 mg·L-1. After the adsorption process, the adsorbent was collected by the magnet, and the remaining dye was measured. For dye quantification, a calibration curve was created by plotting the absorbance (A) of RhB standard solutions at concentrations (
With
Also, the adsorbent RhB amount at equilibrium (
2.5. Catalytic Degradation Procedure
For the catalytic degradation of RhB, the heterogeneous Fenton-like degradation approach was examined. Four trials on RhB removal were conducted in accordance with the reported method [23] using the following materials: P. harmala biochar alone, P. harmala biochar and H2O2, biochar/Mn-ZnFe2O4 alone, and biochar/Mn-ZnFe2O4 and H2O2. When utilizing the biochar alone or the biochar/Mn-ZnFe2O4, 50 mg of the substance was added to 20 mL of a 10 mg·L-1 RhB solution. The removal efficiency was measured following a pH 4.0 adjustment and 90 min of shaking. The mixture for the P. harmala biochar/H2O2 or P. harmala biochar/Mn-ZnFe2O4/H2O2 combinations was made by combining 50 mg of the material with 20 mL of 10 mg·L-1 RhB solution, followed by the addition of a 0.5 mL aliquot of 1.2% (
3. Results and Discussion
3.1. Characterization of Biochar/Mn-ZnFe2O4
Figure 2 depicts the obtained FT-IR spectra of the biochar alone and the biochar/Mn-ZnFe2O4. A characteristic absorption band at 551 cm-1 corresponded to the interaction between metal and oxygen ions that occurred in the tetrahedral and octahedral positions in the spinel crystal lattice of ferrite [28]. A weak absorption peak at 1702, 2334, and 2941 cm-1 were assigned for the C=O, C-N, and C-H stretching vibrations of the aromatic compounds in the biochar. Also, the broad absorption band ranged from 3200 to 3640 cm-1 corresponding to the O-H stretching vibration of metal-OH. As a result, this confirmed that both characteristics of the P. harmala biochar and the impregnated Mn-ZnFe2O4 nanoparticles are depicted in the FT-IR spectrum. Characteristic bands for the negatively charged metal oxide groups of Fe-O, Zn-O, and Mn-O appeared at 543 and 591 cm-1.

FT-IR spectra of untreated the P. harmala biochar (a) and biochar/Mn-ZnFe2O4 (b).
The morphological microstructure of the prepared P. harmala biochar/Mn-ZnFe2O4 was investigated using SEM analysis. The SEM analysis for determining the morphological surface structure is shown in Figure 3. The prepared magnetic biochar has an amorphous microstructure, and the stuffing Mn-ZnFe2O4 nanoparticles were randomly dispersed over the carbon of the biochar surface as white spots. The rough and irregular microstructure of the surface also indicates surface heterogeneity.

SEM micrographs for P. harmala biochar alone (a), biochar/Mn-ZnFe2O4 at 500x (b), and biochar/Mn-ZnFe2O4 at 10000x (c).


Figure 4 shows the XRD pattern of the developed P. harmala biochar/Mn-ZnFe2O4. The profile of the material contains a broad peak around 10-30° corresponding to the cellulose crystal. Also, a small peak was observed at 43° which is typical of graphite [29]. Moreover, the characteristic peaks for Fe3O4 nanoparticles were clearly distinguished by peaks at 2Ɵ of 29.71°, 34.99°, 36.60°, 42.51°, 52.73°, 56.20°, 61.70°, and 72.94°, which correspond to the (220), (311), (222), (400), (422), (511), (440), and (533) planes. This indicates that the prepared magnetic biochar has the same crystal planes as both ZnFe2O4 and MnFe2O4 according to the XRD JCPDS (Joint Committee on Powder Diffraction Standards) data files No. 22-1012 and No. 74-2403, respectively [30].
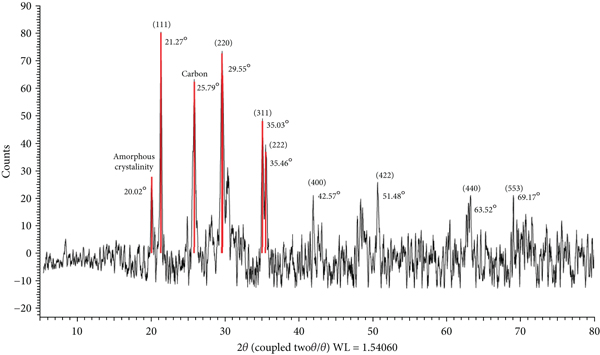
XRD pattern of the prepared P. harmala biochar/Mn-ZnFe2O4.
The surface area and porosity of magnetic biochars derived from a plant that is open to interaction with RhB are determined by the plant’s initial composition and structure, the activation method used, and the agents used. The isotherm profile of N2 adsorption-desorption on the developed adsorbent is shown in Figure 5. The multipoint BET model at a relative pressure (P/Po) from 0.058 to 0.274 was used to determine the surface area of the prepared biochar/Mn-ZnFe2O4. The specific surface area (

The N2 adsorption-desorption isotherm of P. harmala biochar/Mn-ZnFe2O4 (a) and multipoint BET isotherm model (b).

Variation of pore volume and incremental pore volume with pore width (a). The BJH model of pore area (b) and pore volume (c) with pore width of P. harmala biochar/Mn-ZnFe2O4.
3.2. Adsorption Removal
3.2.1. Effect of pH
The effect of pH on the adsorption of RhB dye is a significant parameter since it can alter the ionic form of the dye as well as the surface charge of the adsorbent. As can be seen in Figure 7(a), the removal was higher than 82% at pH 2-5 and then rapidly decreased from 80 to 15% at pH 6-10. As a result, pH 4 was chosen as the appropriate value. As shown in Figure 7(b), the estimated value of

Effect of pH on RhB removal by P. harmala biochar/Mn-ZnFe2O4 (a) and determination of the point of zero-electric charge (b): RhB (10 mg·L-1), sample volume of 20 mL, adsorbent weight 100 mg, and shaking time of 100 min.

Interpretation of higher removal at lower pH values can be due to the adsorbent surface being positively charged and the dye existing in the cationic and the zwitterions forms. At pH less than 6.3, electrostatic attraction and the hydrophobic-hydrophobic (
3.2.2. Adsorbent Dose
The impact of adsorbent dosage on removal effectiveness and adsorbed amount (
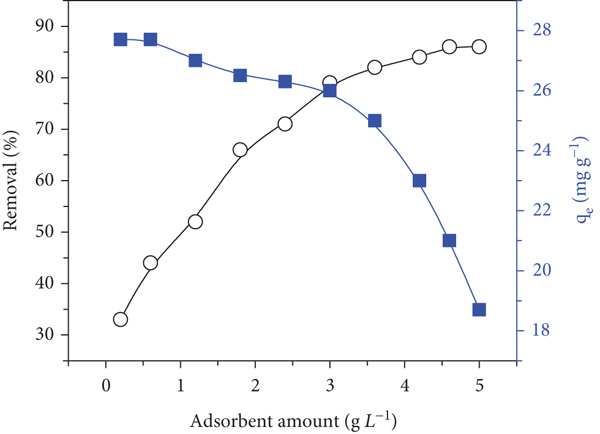
Effect of P. harmala biochar/Mn-ZnFe2O4 dosage on the adsorption of RhB (10 mg·L-1), pH 4, sample volume of 20 mL, and shaking time of 100 min.
3.2.3. Initial RhB Concentration
The influence of initial dye concentration on removal efficiency and the adsorbed amount was examined at dye concentrations ranging from 5 to 100 mg·L-1 to compute the experimental adsorption capacity (
3.2.4. Adsorption Removal Kinetics
The time effect on removal justifies the use of this magnetic biochar in terms of the rate at which the dye can transfer from the aqueous phase to the solid adsorbent. The agitation period was examined over a while of 5 to 110 min. Figure 9 depicts the results obtained for the variation of the adsorbed amount

Effect of shaking time on the P. harmala biochar/Mn-ZnFe2O4 adsorption of RhB (10 mg·L-1), pH 4, sample volume of 20 mL, and adsorbent weight 50 mg.
By comparing the fit of experimental results to the pseudo-first-order, pseudo-second-order, and intraparticle diffusion models, the mechanism that controls adsorption kinetics was investigated. Given that
Plotting
The pseudo-second-order suggested by Ho et al. [37] is expressed by
Figures 10(a)–10(c) display the plotting of these models. Additionally, Table 1 includes the collected kinetic data. A higher linear correlation coefficient (
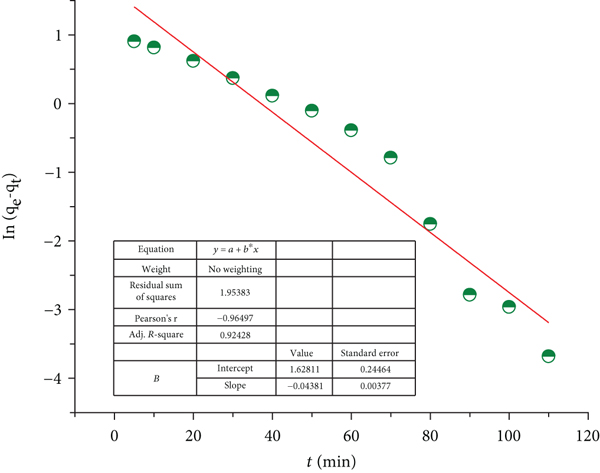
Pseudo-first-order (a), pseudo-second-order kinetic (b), and intraparticle diffusion (c) kinetic models.


Adsorption kinetic data for RhB dye adsorption onto P. harmala biochar/Mn-ZnFe2O4.
A multistep adsorption mechanism was implied by a linear relationship with a steeper slope at beginning intervals, followed by a second linear range with a lower slope, as depicted in Figure 10(c). It was also proposed that a film diffusion phase occurred during the initial adsorption stages, which were indicated by straight lines that did not pass through the origin. Thus, intraparticle diffusion contributes to the rate-limiting step. With an
3.2.5. Adsorption Equilibrium
The equilibrium study allows for the evaluation of the developed biochar’s adsorption capacity as well as the dye-adsorbent binding strength. Equilibrium experiments were carried out at pH 4.0 with a shaking time of 90 minutes to reach equilibrium, and the initial concentration of RhB ranged from 5 to 100 mg·L-1. A comparison of the experimental data and fitting to the Langmuir and Freundlich isotherms is presented in Figure 11. The experimental data followed a profile very close to the Langmuir isotherms.

Experimental equilibrium data and the fitting to the Langmuir isotherm and Freundlich isotherm models: RhB concentration 5-100 mg·L-1, pH 4, sample volume of 20 mL, P. harmala biochar/Mn-ZnFe2O4 adsorbent weight 50 mg, and shaking time 90 min.
To obtain the isotherm parameters, the linear forms of the Langmuir, Freundlich, Tempkin, and Dubinin-Radushkevich (D-R) isotherm models were tested to fit the experimental results. The Langmuir isotherm [39] is expressed by
The Freundlich isotherm [40], which is an empirical model of heterogeneous surface adsorption, is given by
The adsorbent-adsorbate interaction and heat of adsorption could be evaluated using the Tempkin isotherm model [41] as described by
The homogeneous surface or a constant adsorption potential is not assumptions made by the Dubinin-Radushkevich (D-R) isotherm model [42] as represented by
The slope (
Adsorption free energy (
The tested isotherm models are plotted in Figures 12(a)–12(d). Also, the calculated isotherm parameters are shown in Table 2. The RhB adsorption was best fitted to the Langmuir model (

The linear plots of the Langmuir (a), Freundlich (b), Tempkin (c), and Dubinin-Radushkevich (d) isotherm models.



Adsorption isotherm parameters for RhB dye adsorption onto P. harmala biochar/Mn-ZnFe2O4.
Figure 12(c) shows the linearized Tempkin adsorption isotherm applied to the experimental results. Good correlation (
A high correlation coefficient (
3.3. Catalytic Removal of RhB
Using the Fenton-like method and hydrogen peroxide (H2O2) as a catalyst for degradation, the degradation of RhB was investigated. The results were compared to the adsorption removal using the adsorbent biochar/Mn-ZnFe2O4 in the absence of H2O2. The results obtained are depicted in Figure 13.
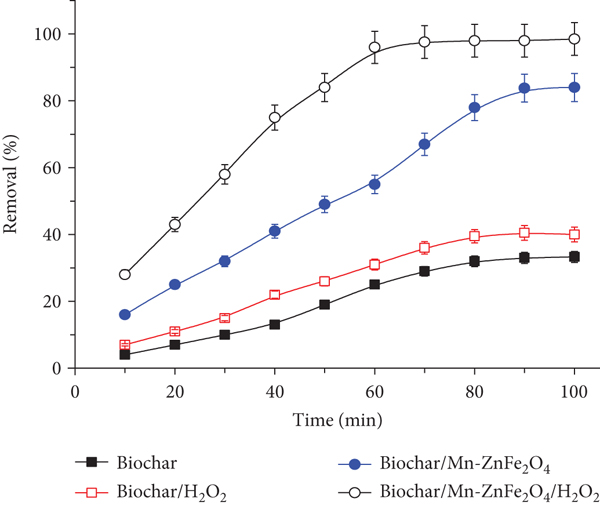
Catalytic degradation and adsorption removal of RhB using P. harmala biochar and biochar/Mn-ZnFe2O4: 20 mL of 10 mg·L-1 RhB solution, pH 4.0, shaking time 10-100 min, adsorbent weight 50 mg, and H2O2 of 0.03% (
In the absence of H2O2, the results indicated no degradation effect on RhB when using the biochar and magnetic biochar/Fe3O4. This was evidenced by measuring the absorbance of the eluted RhB after adsorption. The removal was ≥33 and 84% for biochar and biochar/Mn-ZnFe2O4, respectively.
In the presence of H2O2, the removal reached 40 and 99%, with an increase in the removal by 7 and 15%, respectively. Further, by testing the adsorption in light-exposed and light-protected samples, there was a significant difference in the adsorbed amount
In biochar alone, the proposed degradation reaction is a homogeneous catalytic degradation as can be represented by Equations (14)-(16).
The formed hydroxyl radicals are strong oxidizing agents which can easily oxidize RhB dye to produce an intermediate RhB• which decolorizes the parent dye.
Reactions (14) and (15) can occur with biochar in the absence of ferrite nanoparticles and lead to a slight enhancement for dye removal.
The physicochemical properties of the prepared biochar are primarily dependent on the pyrolysis temperature, heating rate, retention time, and chemical pretreatment. The pyrolysis of agricultural waste at 600°C leads to the thermal degradation of hemicellulose, cellulose, and lignin into biosolid, syngas, and biooil. The main superficial functional groups on the surface of biochar (biosolid) are COOH, C=O, OH, and C-O which affect both the adsorption and catalytic degradation of RhB.
When using the biochar/Mn-ZnFe2O4 heterogeneous Fenton degradation method, the biochar support is deemed to increase the reactivity of the loaded ferrite nanoparticles. The ferrite nanoparticles had the intrinsic peroxidase-like activity that enables Mn-ZnFe2O4 to react with H2O2 to afford the hydroxyl radicals (HO•) [49]. The sunlight illumination of the ferrite portion in Mn-ZnFe2O4 permits the UV radiation to react with nanoparticles and producing of electron- (
The generated
Moreover, the Fe3+ ions on the surface of Mn-ZnFe3O4 nanoparticles can react with H2O2 to initiate the Fenton reaction to produce additional HO• radicals as shown by
The enormous amount of HO• produced in the case of biochar/Mn-ZnFe2O4 heterogeneous Fenton degradation is expected to reveal a higher degree of dye degradation and thereby efficient and fast removal than adsorption removal as compared to the biochar alone which had limited sources of radical generation. Worth mentioning, as can be seen in Figure 13, the rate of dye removal was linear in the case of the biochar and biochar/Mn-ZnFe2O4 which indicates that the concentration of H2O2 was suitable for RhB decoloration and the self-quenching of the hydroxyl radicals (HO•) was negligible.
3.4. Removal of RhB from Tap and Industrial Wastewater
The developed magnetic biochar was applied to the removal of RhB from industrial wastewater using 50 mg adsorbent, 20 mL spiked wastewater of RhB at concentrations of 5 and 10 mg·L-1, and 0.03% (
Removal data of RhB dye from industrial wastewater using by adsorption onto P. harmala biochar/Mn-ZnFe2O4 (
4. Comparison to Other RhB Removal Adsorbents
The adsorption capacity of P. harmala biochar/Mn-ZnFe2O4 was compared to other reported adsorbents in Table 4. The developed magnetic biochar had a higher capacity than other reported biochars [44, 46, 50] and other adsorbents [43, 45, 47] for the removal of RhB. Despite this, some other plant biomass such as Gmelina arborea leaf powder [51] showed extremely higher RhB adsorption capacity than the present material. The obtained capacity is primarily dependent on the method of preparation, pyrolysis temperature, heating rate, and retention time, so it can vary to some extent at other preparation conditions. Despite this, the capacity obtained is sufficient for RhB removal from natural waters. Table 5 compares the developed magnetic biochar’s catalytic degradation removal effectiveness to that of other catalysts. The removal was superior to Fe-bentonite/H2O2 [52] in terms of heterogeneous Fenton catalytic degradation and comparable other listed materials. Thus, the developed magnetic biochar proved to be an alternative and suitable for the adsorption and catalytic removal of RhB from a water sample.
Adsorption capacity of RhB using different various reported materials.
Catalytic degradation removal of RhB using P. harmala biochar/Mn-ZnFe2O4 compared to other materials.
5. Conclusions
The magnetic biochar was prepared by a one-step impregnation pyrolysis under nitrogen at 600°C of P. harmala plant powder pretreated with MnCl2, ZnCl2, and FeCl3. The FT-IR analysis showed that the characteristic bands for metal oxide appeared at 543 and 591 cm-1. The XRD pattern showed the crystalline structure of Mn-ZnFe2O4 embedded in the biochar matrix. The microstructure of the prepared magnetic biochar had higher porosity. The adsorption isotherm was best fitted with the Langmuir model (
Footnotes
Data Availability
The authors can provide the available data on request after the publication of this article, which will be considered by the corresponding author.
Conflicts of Interest
The authors declare no potential conflict of interest.
Authors’ Contributions
Abdulrahman F. Alharbi was responsible for the material characterization and reviewed and edited the manuscript. Abdullah A. Alotaibi was responsible for the conceptualization, resources, and supervision and reviewed and edited the manuscript. Hassan E. M. Gomaa was responsible for the data curation and resources and reviewed and edited the manuscript. Abdulaziz A. M. Abahussain reviewed and edited the manuscript. Sami M. Abdel Azeem was responsible for the conceptualization, data curation, project administration, and funding acquisition and wrote, reviewed, and edited the manuscript.
Acknowledgments
The authors extend their appreciation to the Deanship of Scientific Research at Shaqra University for funding this research work through the project number SU-ANN-202222.
