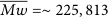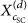Abstract
The role of eumelanin as a natural pigment in protecting human skin from ultraviolet (UV) light has drawn vast interest in the research and industrial community. Encapsulation of the compound by various shell materials has been extensively studied to optimize and prolong its shielding efficiency from UV penetration through the skin. Polylactide (PLA)‐based copolymers have been widely used in the encapsulation of various active compounds due to their biocompatibility and biodegradability that facilitate sustained release of the active compounds. In this work, stereocomplex PLA (sc‐PLA) derived from mixtures of poly(D‐lactide‐caprolactone‐D‐lactide), P(DLA‐b‐CL‐b‐DLA), a triblock copolymer with linear poly(L‐lactide), and PLLA are employed to encapsulate eumelanin by an oil‐in‐water emulsion (O/W) technique. The effect of eumelanin distribution in PLA’s enantiomers and ultrasonication on the physicochemical properties, encapsulation efficiency, and release behavior of the nano/microparticles were evaluated. The potential application of the resulting particles for sunscreen products was assessed in terms of UV absorbance and in vitro sun protection factor (SPF). The nano/microparticles show a hollow spherical structure, whose size can be controlled by ultrasonication. The distribution of eumelanin and the ultrasonication process play a key role in the growth of sc‐PLA and the crystalline structure of the particles. The highest encapsulation efficiency of 46.6% was achieved for sc‐PLA2U particles. The high content of eumelanin and the hollow structure with a large surface area lead to improvement in the UV absorbance and sunscreen performance of the particles, as revealed by the increase in the SPF value from 9.7 to 16.5. The materials show high potential for various applications, especially in cosmetic and pharmaceutical fields, as UV‐shielding products.
1. Introduction
Prolonged overexposure to ultraviolet (UV) radiation has become a critical problem for human skin. Degenerative changes are caused by the high‐energy UV light that disrupts the covalent bonds in the structures of organic substances potentially result in skin cancers, such as melanoma and carcinoma [1, 2]. Although the majority of UV radiation consisting of UVA (320–400 nm), UVB (280–320 nm), and UVC (220–280 nm) is captivated by the stratospheric ozone layer and gases in the atmosphere, some UVA radiation can reach the earth’s surface [3]. Furthermore, UVA exposure generates reactive oxygen‐containing species, including hydrogen peroxide, superoxide anions, and singlet oxygen, which can cause damage to the structures of DNA, lipids, and proteins in skin cells. This is caused by deep penetration of UVA to the dermis extent, which causes skin damage [2].
Natural pigments have been extensively utilized as UV‐shielding materials to protect the living cells of the human skin from damage. The aromatic component in their structure shows photoprotection ability by absorbing most UV light [2, 4]. Melanin, a natural pigment found in most living organisms, is a biomacromolecule with various prominent functional properties besides its excellent UV protection, such as antioxidant, free‐radical scavenging, anti‐inflammatory, photothermal therapy, chelating ability to metal ions, and thermoregulation [4–6]. Melanin is mainly classified into two types according to its colors and precursors: eumelanin and pheomelanin. Eumelanin is a pigment responsible for black to brown color on human skin and eyes, whereas pheomelanin is responsible for yellow to red‐haired color [7]. Eumelanin shows higher photoprotection due to its broadband absorption emanating from the chromophores of 5,6‐dihydroxyindole (DHI) and 5,6‐dihydroxyindole‐2‐carboxylic acid (DHICA) units. These provide large π‐electron clouds, which enable the absorption of UV light [8].
Recently, numerous studies reported the UV‐shielding property of various nanocomposites of eumelanin. Wang et al. developed polymeric nanocomposites based on polyvinyl alcohol (PVA) and eumelanin to enhance the photostability of the films for outdoor applications [1]. The composite films showed improved photostability and UV‐shielding performance while maintaining high transparency. In a separate study, embedded eumelanin mixed with titanium dioxide (TiO2) nano/microparticles in polyurethane (PU) films exhibited the value of the ultraviolet protection factor (UPF) four times higher than that without eumelanin. This indicated enhanced UV‐shielding ability and photostability due to the synergistic effects of eumelanin and TiO2 [7].
Despite their promising features, most organic UV absorbers, including eumelanin, have several limitations, such as direct photodegradation and free radical leaching. This directly affects the lifetime of eumelanin in protecting the skin cells [9]. To overcome these drawbacks, several approaches have been performed to extend the lifetime of organic UV filters and to control their uptake into the cells. Encapsulation has been widely used to prolong chemical stability and control the release of active compounds to the target sites [10, 11]. It is reported that the solubility and quantity limitations of UV filters in sunscreen products that affect their photostability can be improved by encapsulation [12]. Various polymeric materials have been employed as the shell structure to effectively protect the core materials from severe environments that extend their lifetime through the reduction of their fast leakage [13].
Polylactide (PLA) and its copolymers have been widely used as a shell material due to their biocompatibility and degradability that facilitate the sustained release of various active compounds [14, 15]. However, applying PLA in drug entrapments requires optimized properties, such as particle size, stability, and degradation profile. These can be achieved by modifications of PLA‐based copolymers. PLA has a chiral center in its repeat unit, leading to the formation of 2 enantiomeric forms, i.e., poly(L‐lactide) (PLLA) and poly(D‐lactide) (PDLA) [16, 17]. Stereocomplex (sc‐PLA) is formed by the cocrystallization and assembly of the two enantiomers in a closely packed 31‐helical crystal lattice, which results in denser chain packing under the influence of strong hydrogen bonding [18]. This leads to enhanced hydrolysis resistance with higher thermal and mechanical properties than its homocrystallite PLLA or PDLA counterparts, which are beneficial for utilization in practical applications [19]. Sc‐PLA can also facilitate the confinement and release of various drugs and vitamins, such as insulin, rifampicin, triamcinolone, fluorouracil, ascorbic acid, folic acid, and vitamin E [19, 20]. Several methods have been developed for preparing nano/microparticles based on sc‐PLA, including precipitation, solvent evaporation, layer‐by‐layer assembly, double emulsion, microfluidics, and spray drying [20–23]. The size of particles can be controlled by different fabrication techniques. Im et al. proposed a method for formulating sc‐PLA through oil‐in‐water (O/W) emulsion blending of PLLA and PDLA [24]. The technique showed nearly 99% of the stereocomplexation degree and high efficiency to load fluorouracil (5‐FU), as the hydrophobic nano/microparticles formed by the spontaneous stereocomplexation enabled the entrapment of this hydrophilic drug. The materials are also compatible, which is beneficial for cosmetic and biomedical applications.
Hollow nanoparticles from PLA copolymers have been developed as UV‐shielding materials in personal care products due to their excellent light scattering characteristics and low density [25, 26]. The material’s UV shielding capabilities, toxicity, structural morphology, and formation mechanism were investigated. However, the encapsulation of UV absorbers, such as eumelanin, using sc‐PLA as the shell material, has rarely been studied utilizing the emulsification method. The key properties of the emulsions, such as stability, rheology, and physical appearance, are strongly influenced by the droplet size and particle size distribution inherited by the preparation methods. High‐energy emulsification has been used extensively to produce emulsions with smaller droplet sizes and higher stability. Colloid mills, high‐pressure or high‐speed homogenizers, and ultrasonic homogenizers are commonly employed in this procedure. Among these, ultrasonication is promising compared to other methods, as the high‐intensity ultrasound waves induce cavitation due to the intense shear pressures, high pressure, and temperatures, generating emulsions with greater stability, low polydispersity, and smaller droplet sizes [27]. Although this technique is well established for emulsion preparation in various applications, its effects on the encapsulation of eumelanin and the formation of sc‐PLA through oil‐in‐water emulsion have not been extensively studied. It is expected that ultrasonication treatment during the preparation of nano/microparticles will result in fine particles and improve the emulsion stability, which is advantageous for UV protection in cosmetic applications.
This study aims to develop a process for preparing eumelanin‐loaded nano/microparticles using sc‐PLA as the shell by employing poly(D‐lactide‐caprolactone‐D‐lactide), P(DLA‐b‐CL‐b‐DLA), a triblock copolymer, and linear PLLA as the precursors. The encapsulation process was performed by an oil‐in‐water emulsion (O/W) technique, followed by solvent evaporation. The effect of eumelanin distribution in different PLA’s enantiomeric phases and an ultrasonication treatment on the encapsulation efficiency and physicochemical properties of the resulting nano/microparticles are investigated. Potential applications of the nano/microparticles in UV shielding cosmetic products are examined by determining the in vitro sun protection factor (SPF) of sunscreen products containing the materials in the UVA‐UVB regions. In addition, the cytotoxicity of the resulting materials is evaluated.
2. Experimental
2.1. Materials
The P(DLA‐b‐CL‐b‐DLA) block copolymer and linear PLLA were synthesized in our laboratory. Eumelanin production and purification processes were carried out as described by Bayram et al. [28]. Dichloromethane (DCM) was purchased from Thermo Fisher Scientific. Dimethyl sulfoxide (DMSO) and sodium hydroxide (NaOH) used in the nano/microparticle preparation and encapsulation efficiency determination were obtained from Carlo Erba Chemicals. Polysorbate 80 or Tween 80 and phosphate buffer saline (PBS) pH 7.4 were purchased from Acros Organics. A 3M™ Transpore™ surgical tape was used to examine the SPF value of sunscreen products containing the prepared particles. Nivea Creme (60 mL, Beiersdorf, Thailand) was used as a base cream for sunscreen application, whose ingredients are listed as follows: aqua, paraffinum liquidum, cera microcristallina, glycerin, lanolin alcohol (eucerit®), paraffin, panthenol, magnesium sulfate, decyl oleate, octyldodecanol, aluminum stearates, citric acid, magnesium stearate, sodium anisate, limonene, geraniol, hydroxycitronellal, linalool, citronellol, benzyl benzoate, cinnamyl alcohol, and parfum. All materials were used without further purification.
2.2. Synthesis of the P(DLA‐b‐CL‐b‐DLA) Block Copolymer and Linear PLLA
The P(DLA‐b‐CL‐b‐DLA) block copolymer (
2.3. Eumelanin Production and Purification
Eumelanin was extracted from Streptomyces strains isolated from the soil. First, the air‐dried soil was mixed with CaCO3 at a ratio of 10 : 1 (w/w), followed by incubation at 37°C for 4–7 days in a humid environment. The diluted pretreatment soil samples were then seeded into the ISP 2 medium (Difco, USA) and incubated at 26°C for 14 days. The colonies of Streptomyces strains were then transferred to new ISP 2 media to isolate them. This pure Streptomyces strain was stored at −80°C in 1/2 nutrient broth containing 20% glycerol before further purification. Eumelanin‐producing Streptomyces strains were then transferred to sterile liquid media and incubated in an orbital shaker at 35°C and 200 rpm for 7 days. The samples were centrifuged at 10,000 rpm for 10 min, and the process was repeated twice. The supernatant was then adjusted to pH 2 with 6 M HCl and left at room temperature for 24 h. This suspension was then centrifuged at 10,000 rpm for 10 min, whose supernatant and precipitates were separated. Eumelanin in the precipitate was washed thrice with DI water at 10,000 rpm for 10 min. The purified eumelanin was lyophilized and stored at −20°C until further use.
2.4. Preparation of Nano/Microparticles
The nano/microparticles were prepared by an oil‐in‐water emulsion method, as summarized in Figure 1. For eumelanin‐loaded particles, 2 mg of eumelanin was dissolved in 0.5 mL DMSO with sonication (∼1 min). To determine the effect of eumelanin distribution in different PLA’s enantiomeric matrices, the eumelanin solution was added to the following solutions: (i) the P(DLA‐b‐CL‐b‐DLA) copolymer in DCM; (ii) PLLA in DCM; and (iii) a mixture of copolymer and PLLA in DCM. The solutions (i) and (ii) were then mixed with their enantiomeric counterpart, i.e., PLLA in DCM and the P(DLA‐b‐CL‐b‐DLA) copolymer in DCM, respectively, as the precursors for stereocomplexation. These mixtures are called the oil (O) phase with the copolymer to PLLA weight ratio of 2 : 1 (5% w/v, in 10 mL DCM). The water phase (W) was prepared from DI water containing polysorbate 80 (1% v/v). The same procedures were performed to prepare the blank particles, except for adding a eumelanin solution.

The schematic diagram of the preparation procedures of eumelanin‐loaded nano/microparticles.
To investigate the effect of ultrasonication, the emulsions (O/W) were separately prepared by mechanical stirring and ultrasonication. The first‐mentioned method was conducted by adding the dropwise oil phase to the water phase (1 : 2 v/v) with continuous stirring at 800 rpm. The latter approach was performed by adding the oil phase dropwise to the water phase (1 : 2 v/v) with continuous stirring at 800 rpm, and then, ultrasonication was performed using a probe ultrasonic (Branson Digital Sonifier® SFX 550, BRANSON Ultrasonics Corp.) with 50% amplitude for 5 min. The emulsions from both methods were then continuously stirred at 800 rpm at room temperature for 3 h to evaporate the solvent until the expected volume was observed and the liquid level was no longer changing. After the solvent evaporation, the suspension was centrifuged at 6000 rpm for 2 h. The precipitate was separated from the supernatant. The precipitate was collected and washed 3 times with DI water, while the supernatant was stored for further encapsulation efficiency analyses. After that, the precipitates were dried overnight at 60°C. The dried products were stored in a desiccator for further characterization. The final products are coded as sc‐PLA1 (blank particles), sc‐PLA2 (particles with eumelanin distributed in the copolymer), sc‐PLA3 (particles with eumelanin dispersed in PLLA), and sc‐PLA4 (particles with eumelanin distributed in a copolymer/PLLA mixture) to distinguish them according to the stirring method and eumelanin distribution. The letter “U” after the sample labels indicates the use of the ultrasonication process during the emulsification, e.g., sc‐PLA1U.
2.5. Characterization of Nano/Microparticles
The chemical structures, interactions, and properties of the resulting nano/microparticles were examined by Fourier transform infrared (FTIR) spectroscopy. The spectra were recorded on a Thermo Scientific Nicolet iS5 spectrometer in an attenuated total reflectance mode by coadding 32 scans at a resolution of 2 cm−1. The morphological characteristics of the nano/microparticles were observed by a field emission‐scanning electron microscope (FE‐SEM Hitachi SU‐8030, Tokyo, Japan) and a transmission electron microscope (TEM JEOL JEM‐2100Plus, Tokyo, Japan) with an accelerating voltage of 200 kV. The materials were first dispersed in DI water, followed by sonication for 30 min. For SEM measurements, the dispersed nano/microparticles were dropped onto a silicon wafer, followed by air‐drying for 24 h. Prior to analysis, the specimens were coated with a thin Pt layer for enhanced conductivity.
The particle size distribution was measured by a particle size analyzer (the laser diffraction method, Malvern Mastersizer 3000, Malvern, United Kingdom). Zeta potential was measured by a Malvern Zetasizer Nano‐ZS (Malvern, United Kingdom). Synchrotron FTIR spectra were recorded at the BL4.1 IR spectroscopy and imaging beamline at the Synchrotron Light Research Institute (SLR, Thailand). The samples were analyzed in an attenuated total reflectance (ATR) mode, using a Bruker Vertex 70 spectrometer coupled with a Bruker Hyperion 2000 microscope (Bruker Optics, Ettlingen, Germany). The nano/microparticles were placed on IR‐transparent barium fluoride windows, and the measurements were conducted at 64 scans and 4 cm−1 resolution, with 20 kHz velocity. The X‐ray diffraction (XRD) spectra were measured with an X‐ray diffractometer (D8 Advance, Bruker, Germany) using CuKα radiation (1.5406 Å) at 40 kV and 40 mA. Thermal properties of the nano/microparticles were examined on a differential scanning calorimeter (DSC 3+, Mettler Toledo, Switzerland) under a nitrogen atmosphere (a flow rate of 10 mL/min). The samples weighing 1‐2 mg were examined at a heating and cooling rate of 20°C/min from 0 to 200°C. The heat of melting of the homocrystallite (∆H
hc) and the stereocomplex crystallite (∆H
sc) were used to determine the homocrystallinity (X
hc) and stereocomplex crystallinity (X
sc) of the materials by using the following equations:
The heat of melting for 100% homocrystallinity and stereocomplex crystallinity is 93 J/g and 142 J/g, respectively [29].
2.6. Encapsulation Efficiency of Nano/Microparticles
The encapsulation efficiency was determined by direct and indirect methods. The direct method was performed by dissolving the nano/microparticles in DCM. A certain volume of NaOH solution was subsequently added to the mixture to extract eumelanin into the aqueous phase. The DCM and aqueous phases were separated for UV‐Vis measurements (Thermo Scientific, GENESYS™ 180). The indirect method was conducted by measuring the absorbance of the unentrapped eumelanin in the supernatant after centrifugation of the particles after the encapsulation process using a UV‐Vis spectrophotometer. A standard solution of eumelanin was prepared and diluted in 0.01 M NaOH with various concentrations between 2.5 and 40 ppm to construct the calibration curve. The absorbance of these solutions was observed and used to determine the concentration of eumelanin in the resulting particles. The encapsulation efficiency was then calculated as follows:
2.7. In Vitro Release of Eumelanin from the Nano/Microparticles
The in vitro release of eumelanin from the nano/microparticles was determined using a dialysis method. The nano/microparticles were added to PBS, pH 7.4 (5 mg/mL) and incubated in a reciprocal shaking thermostatic waterbath (Biobase SWB‐100X12, Shandong, China) with a speed of 120 rpm at 37°C. At specific time intervals (0.5, 1, 2, 4, 6, 24, 48, 72, and 144 h), an aliquot of the release medium was collected. An equal volume of fresh PBS was added to keep the equilibrium of the release medium. The absorbance of the withdrawn aliquot was measured by a UV‐Vis spectrometer, and the cumulative release of eumelanin was calculated according to the method described by Park et al. [30]
2.8. UV Absorbance and In Vitro SPF Measurements of Sunscreen Products
UV absorbance and in vitro SPF of sunscreen products containing the nano/microparticles were determined according to the procedure proposed by Widsten et al. [31]. Briefly, 1% of the nano/microparticles (w/w) were added to a base cream (1 g) in a 10 mL beaker and homogenized at ambient temperature. The blended sunscreen products were then applied (2.0 mg/cm2) to a two‐layer 3M Transpore™ tape attached to a quartz cuvette to simulate the skin. A two‐layer tape without sunscreen attached to a quartz cuvette was used as the blank. The UV absorbance of the sunscreen products was recorded in a range of 290–400 nm. Three replicates of each sample were measured, whose average values were reported. The SPF values were calculated according to Mansur’s equation [32] as follows:
2.9. Cytotoxicity Analysis
The cytotoxicity of the prepared particles was examined. Essentially, HaCaT, in vitro spontaneously transformed keratinocytes from the histologically normal skin, was cultured in a DMEM medium supplemented with 4.5 g/L glucose, 2 mM/L glutamine, and 10% fetal bovine serum (Gibco, NY, USA). The cells were grown at 37°C and 5% CO2 atmosphere until reaching 80% confluent. The cells were then trypsinized with 0.25% trypsin‐EDTA (Gibco, NY, USA) and inactivated enzyme activity by completed media. The cell viability was examined under an optical microscope after staining with 0.4% trypan blue (Gibco, NY, USA).
The HaCaT cells were seeded onto a 96‐well plate at a density of 1.2 × 105 cells/well and incubated overnight at 37°C in a 5% CO2 atmosphere. The cells were exposed to the tested particles for 48 h, and the cell viability was examined using an MTT assay (3, [4, 5‐dimethylthiazol‐2‐yl]‐2,5‐diphenyltetrazolium bromide (Invitrogen, CA, USA)). DMSO (100 μl, Chem Cruz, CA, USA) was added to each well to dissolve the generated formazan from the reaction. The color intensity was measured by a Varioskan Flash microplate reader (Thermo, MA, USA) at 590 nm. The percentage of cell viability was calculated as follows:
3. Results and Discussion
3.1. Physicochemical Properties of Nano/Microparticles
3.1.1. Morphological Analysis of Nano/Microparticles
The morphology of the eumelanin‐loaded nano/microparticles was examined by FE‐SEM and TEM. SEM images, as shown in Figure 2, reflect a hollow raspberry‐like structure consisting of separate plates/flakes forming a spherical shell structure with a high degree of porosity. The smaller particle size with a more regular spherical shape was observed when ultrasonication was employed in the eumelanin distribution. The hollow polymeric structures were reported to have excellent UV scattering properties in our recent study [26]. SEM images of cryo‐fractured particles, as shown in Figure 3, confirm the hollow structure with a relatively thick shell and hollow center. This structure also plays a key role in increasing UV light blocking and boosting the SPF value by increasing the path length of the UV light through scattering [33, 34]. The UV light that penetrates through the sc‐PLA shell is absorbed and travels through the air void core, which prevents the light from being directly reflected. This results in the highly efficient confinement of UV radiation [35–37]. However, the UV absorbability of hollow particles also depends on their particle size and size distribution. The particles with a hollow structure and a small particle size can effectively act as an SPF booster. It is noted that particles with less regular spherical structures and particle agglomeration were still observed even after ultrasonication, likely due to van der Waals forces between the particles during the sample preparation step [38]. TEM images of the material are compared in Figure 4. The results clearly show separate droplets, which confirm the structural regularity of the nano/microparticles as observed in SEM images. Moreover, the decrease in the particle size of the ultrasonication‐assisted particles is confirmed. The results reveal that the eumelanin distribution procedures and the ultrasonic process strongly affect the structure and the size of nano/microparticles, in which the ultrasonication effectively decreases the particle size with a more regular spherical shape.

SEM images of (a) sc‐PLA1U, (b) sc‐PLA1, (c) sc‐PLA2U, (d) sc‐PLA2, (e) sc‐PLA3U, (f) sc‐PLA3, (g) sc‐PLA4U, and (h) sc‐PLA4.

SEM images of cryo‐fractured particles of (a) sc‐PLA1U, (b) sc‐PLA1, (c) sc‐PLA2U, (d) sc‐PLA2, (e) sc‐PLA3U, (f) sc‐PLA3, (g) sc‐PLA4U, and (h) sc‐PLA4.

TEM images of (a) sc‐PLA1U, (b) sc‐PLA2U, (c) sc‐PLA3U, (d) sc‐PLA4U, (e) sc‐PLA1, (f) sc‐PLA2, (g) sc‐PLA3, and (h) sc‐PLA4.
(b)

(c)

(d)

(e)

(f)

(g)

(h)

The particle size distribution of the resulting nano/microparticles obtained from the DLS method is compared in Figure 5. The particle size was in a range of 0.38–135 μm for the ultrasonication‐assisted particles (Figure 5(a)). A broader distribution ranging from 0.38 to 290 μm was observed for those prepared without ultrasonication (Figure 5(b)). The volumetric average particle size (D v ) and mean diameter (D), as summarized in Table 1, reflect a smaller size for ultrasonication‐assisted particles than those without ultrasonication. The results indicate that ultrasonication treatment during emulsification plays an important role in reducing the particle size. The intense energy provided by ultrasonication allows the droplet disintegration during emulsification, resulting in a decrease in the final particle size. Meanwhile, the eumelanin distribution step has no significant effect on the size of the resulting particles. It is noted that a decrease in the particle size may also result from slight degradation of the copolymers chains. A decrease in the hydrodynamic size of particles due to the ultrasonication treatment has been reported [39, 40]. The reduction in polymer molecular weight occurs following the vigorous mechanical forces caused by the propagation of acoustic energy through the liquid. The degradation rate due to this cavitation mainly depends on several factors, such as the duration and intensity of ultrasonication treatment, solution concentration, and the nature of the polymer and the solvent [41].

Particle size distribution of nano/microparticles (a) with ultrasonication and (b) without ultrasonication.
(b)

Summary of particle size distribution, zeta potential, and encapsulation efficiency of nano/microparticles.
3.1.2. FTIR Analysis
FTIR analysis was performed to identify the functional groups and interactions among the constituents of the eumelanin‐loaded nano/microparticles. The spectra of pure eumelanin and the particles prepared from different eumelanin distribution steps and ultrasonication processes are compared in Figure 6(a). All spectra show characteristic bands of PLAs, i.e., C‐H stretching at 2996–2849 cm−1, C=O stretching at 1755 and 1748 cm−1, CH3 rocking, and C‐COO stretching at 954 cm−1. The β‐form crystal with a 31 helical conformation of sc‐PLA was observed at 908 cm−1 [16, 17, 42–44]. However, the characteristic bands of eumelanin, especially the N‐H bending mode at around 1600 cm−1, cannot be detected in the spectra of both sc‐PLAU and sc‐PLA series. The absence of these bands is likely because of the low eumelanin loading contents within the nano/microparticles or the complete coverage of the compound inside the particles, as ATR‐FTIR spectra are largely dominated by the bands of functional groups located at the sample’s surfaces.

(a) FTIR spectra of (A) eumelanin, (B) sc‐PLA1U, (C) sc‐PLA2U, (D) sc‐PLA3U, (E) sc‐PLA4U, (F) sc‐PLA1, (G) sc‐PLA2, (H) sc‐PLA3, and (I) sc‐PLA4 nano/microparticles and (b) ratiometric analysis of FTIR spectra of the nano/microparticles in the C‐H stretching modes.
(b)

A ratiometric approximation was performed to evaluate the intensity changes of the characteristic bands after eumelanin encapsulation by normalization of the spectra with a reference band at 2992 cm−1 (C‐H stretching mode). The band was selected as its intensity was significantly unchanged. The normalized spectra, therefore, can be compared across different samples, as shown in Figure 6(b). Interestingly, the normalized intensities of the bands at 2925 and 2855 cm−1, associated with Tween 80, varied with the eumelanin distribution steps and the ultrasonication treatments. When eumelanin was distributed in the P(DLA‐b‐CL‐b‐DLA) copolymer (sc‐PLA2U and sc‐PLA2), the normalized intensities of the two modes were the highest. The samples prepared with ultrasonication treatments showed higher normalized intensities than those from mechanical stirring, as summarized in Table 2. The results reflect the relative concentration of Tween 80 attached to the surface of the nano/microparticles. The sequence of mixing eumelanin in PLLA or the copolymer affected the adsorption of Tween 80 on the particle’s surfaces. The materials prepared from the eumelanin/copolymer showed the highest Tween 80 residues, which play a key role in the particle’s stabilization and homogenization during emulsification. The main objective of changing the sequence of mixing eumelanin is to investigate the effect of stereocomplex formation on the encapsulation mechanism of eumelanin. Eumelanin inclusion either before or simultaneously with the formation of the stereocomplex structure involving the PDLA copolymer and linear PLLA may result in varied encapsulation efficiency and eumelanin placements in the particle due to the ease of the two components undergoing stereocomplexation. The difference in the content of the Tween 80 on the surface of the particles prepared from different sequences was unexpectedly observed. The results indicate that eumelanin was uniformly distributed in the stereocomplex shell and at the surface when the compound was introduced to the single‐component enantiomers. This leads to higher encapsulation efficiency and a higher releasing rate (discussed later). In addition, the polar functional groups in the eumelanin structure at the surface may induce the deposition of Tween 80 from the water phase. In contrast, when eumelanin is introduced with the two enantiomer mixture, stereocomplex is readily formed and entraps the eumelanin in the core, leading to a lower release rate and also a lower content of Tween 80 deposited at the particle’s surfaces. The schematic representation of the particles obtained from these mechanisms is illustrated in Figure 7. Attia Shafie and Mohammed Fayek [45] reported that the adsorption of Tween 80 on the particle’s surface could decrease the surface tension and hence increase the encapsulation efficiency. The authors also found that the adsorption of Tween 80 generated a thicker coating layer, resulting in higher zeta potential. To verify this, the zeta potential and encapsulation efficiency of the resulting particles were examined and will be discussed in the next section. Similar results were also observed for the 2941 cm−1 band, corresponding to the C‐H stretching of PLA, suggesting that the preparation method, i.e., the sequence of eumelanin distribution in different PLA matrices, can control the stereocomplexation mechanisms.
Band intensities of 2941, 2925, and 2855 cm−1 modes after normalization by a reference band (2992 cm−1).

Schematic representation of nano/microparticles obtained from different sequences of mixing eumelanin.
3.1.3. Zeta Potential of Nano/Microparticles
The zeta potential of the nano/microparticles showed negative values of −31 to −42 mV. The negatively charged nature might be due to the functional end groups of the sc‐PLA shell and eumelanin, such as carboxylic acid and hydroxyl groups. The highly negative values increase the stability of the resulting particles in emulsion. The particle’s surface that became negatively charged allows the electrostatic repulsion, which subsequently enhances the stability of the formed emulsion. The nonionic surfactant, Tween 80, could also assist in stabilizing the emulsion as it can lower the interfacial tension between the oil and water phases through interactions with its polyoxyethylene head and hydroxyl tail groups. The ultrasonication‐assisted particles showed slightly more negative values than those without ultrasonication due to the high shear forces from ultrasonication that can disrupt the emulsion droplets to a finer size. As mentioned above, the strong mechanical forces provided by ultrasonication may also induce the depolymerization of PLA chains. The shorter polymer chains may provide more functional groups on the surface of the fine particles, which leads to a higher repulsion between the particles and stabilizes the emulsion [40]. Although when eumelanin was distributed in the copolymer/PLLA mixture, sc‐PLA4U showed the highest zeta potential of −42.5 mV, and the results indicated that eumelanin distribution had a minor effect on the stability of the resulting particles.
3.1.4. Thermal Analysis of Nano/Microparticles
The thermal properties of the resulting particles were examined by DSC analysis, whose thermograms are compared in Figure 8. The first heating thermograms (Figure 8(a)) show a peak at 60°C, corresponding to the melting peak of PCL domains from the copolymer that overlapped with the melting peak of eumelanin [46, 47]. Two endothermic peaks at around 130 and 200°C, associated with the melting of homocrystallite and sc‐crystallite, were observed, regardless of the eumelanin distribution processes and ultrasonication. Although an equimolar blend is employed, an incomplete formation of sc‐crystals occurred as the melting peak of homocrystallite PDLA was still observed in the first heating thermograms. This might be due to the mismatch in molecular weights of the two PLA components, in which much larger DLA sequences in the copolymer may have higher chain mobility restrictions. The observed T m of all samples, as summarized in Table 3, are slightly lower than 200°C of typical sc‐crystals [24]. This indicates a shorter chain‐folding length of the crystals due to the mismatch in the chain lengths and the presence of PCL segments. In addition, the inclusion of eumelanin in the oil phase containing the PLA enantiomeric components might interrupt the formation of the stereocomplex structure during emulsification, resulting in lower melting temperatures. Interestingly, ultrasonication‐assisted particles showed relatively similar T m , irrespective of the eumelanin distribution processes, indicating similar stereocomplex structures. In contrast, the particles prepared by mechanical stirring showed slight differences in T m as a result of different eumelanin distribution procedures. The ultrasonication‐assisted particles showed narrower sc‐crystallite melting peaks, implying higher regularity in their crystalline arrangements [48].

DSC thermograms of (a) first heating, (b) cooling, and (c) second heating of (A) eumelanin, (B) sc‐PLA1, (C) sc‐PLA2, (D) sc‐PLA3, (E) sc‐PLA4, (F) sc‐PLA1U, (G) sc‐PLA2U, (H) sc‐PLA3U, and (I) sc‐PLA4U nano/microparticles.
(b)

(c)

Thermal properties of eumelanin and nano/microparticles obtained from DSC thermograms.
The ultrasonication‐assisted particles showed higher melting enthalpy of both homocrystallite (∆H hc) and sc‐crystallite (∆H sc), as summarized in Table 3. This is because the ultrasonication facilitates the intermolecular hydrogen bonding between PLLA and PDLA segments in the copolymer, leading to an increase in the seeding and growth rate of sc‐crystals and hence crystallinity during emulsification [49]. The short ultrasonication treatment time during emulsification could facilitate the growth of sc‐crystals. However, it is insufficient to disrupt the crystalline structure of the resulting crystals. It was reported that ultrasonication treatment could effectively remove the amorphous region and increase the crystalline value at an optimum ultrasonication time (5 min). However, the crystalline value is decreased due to nonselective elimination over a longer period that eliminates both crystalline and amorphous regions [50]. The eumelanin distribution steps also affect the crystallinity, where sc‐PLA2U showed the highest crystallinity of both homo‐ and sc‐crystallites. Moreover, the absence of a cold crystallization peak in the first heating thermogram confirms the improvement of chain arrangements [21]. The ultrasonication‐assisted particles exhibited higher crystallization temperatures (T c ) in the cooling thermograms, as shown in Figure 8(b). This indicates faster crystallization and more regular chain arrangements that enhanced the sc‐crystallite formation, likely due to the larger surface area provided by a smaller particle size [51, 52]. The decrease in polymer chain length due to the collapsing bubbles caused by cavitation improves the microscale mixing and turbulence during crystallization treated by ultrasonication. The faster diffusion of these short polymer chains increases the nucleation rate. As the nucleation rate increases, the induction time will decrease, and the crystallization rate can be accelerated. Therefore, the number of formed crystals increases while the particle size decreases [53]. The second heating thermograms of all samples also showed two endothermic peaks similar to the first heating counterparts (Figure 8(c)). The peak at about 130°C for ultrasonication‐assisted particles is associated with the melting of the incomplete crystallizable domains during the previous cooling process [54]. The results indicate that orderly arrangements of the crystalline structures of the resulting particles can be obtained by employing ultrasonication and distributing eumelanin in the copolymer phase during emulsification.
3.1.5. XRD Analysis of Nano/Microparticles
XRD spectroscopy is employed to examine the crystal structures of homocrystallite and stereocomplex of sc‐PLA particles, as shown in Figure 9. Three intense diffraction peaks located at 2θ = 11.9, 20.7, and 24.0° correspond to the (110), (300)/(030), and (220) planes of sc‐crystallites. These agree with previous reports that the complexation of PLLA/PDLA (β‐conformation) consists of two helical chains (32 of PLLA and 31 of PDLA) with the triclinic unit cell of a = 0.916, b = 0.916, and c = 0.870 nm (α = 109.2, β = 109.2, and γ = 109.8°) [55, 56]. Some studies also reported that the unit cell of sc‐crystallite is trigonal, comprising six helical chains (three 32 helix of PLLA and three 31 helices of PDLA), with unit cell dimensions of a = b = 1.498 nm and c = 0.870 nm, where a = b = 90° and γ = 120° [57, 58]. The existence of lactate homocrystallite was confirmed by the signals at 2θ = 14.8, 16.7, and 19.2°, corresponding to the (010), (200/110), and (203) planes. This α‐form comprises an orthorhombic unit cell with two 103 helix chains (a = 1.059, b = 0.598, and c = 2.869 nm). The observed unit cell is similar to that of neat PLA (a = 1.07, b = 0.595, and c = 2.78 nm) [55, 59, 60]. The ultrasonication‐assisted particles showed higher peak intensity of sc‐crystallite than those without ultrasonication, reflecting higher crystallinity. In addition, the eumelanin distribution procedures also affect the diffraction peak intensity, where the most intense peak was observed in sc‐PLA2U particles. Scherrer’s equation was used to estimate the crystallite size of the resulting particles by using full‐width half maximum (FWHM) as follows: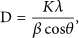

XRD patterns of (a) sc‐PLA1U, (b) sc‐PLA2U, (c) sc‐PLA3U, (d) sc‐PLA4U, (e) sc‐PLA1, (f) sc‐PLA2, (g) sc‐PLA3, and (h) sc‐PLA4 nano/microparticles.
The data on FWHM, crystallite size, and crystallinity derived from XRD patterns are summarized in Table 4. The peak position and peak width of all particles were slightly different. The ultrasonicated particles showed narrower peak width at 2θ = 20.8° than those without ultrasonication, indicating that the ultrasonication‐assisted process produces sc‐crystals with larger domain sizes or higher quality. The difference in the peak width is also affected by the eumelanin distribution step. The materials prepared from eumelanin distribution in the copolymer (sc‐PLA2U and sc‐PLA2) showed narrower peaks than their counterparts. This confirms that the sequence of eumelanin addition in the oil phase before emulsification plays a key role in the crystal quality of the resulting particles. In contrast, the peak width of homocrystallite remained unchanged for the ultrasonicated particles. The differences in the peak width imply the average crystallite sizes (



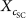
Summary of crystalline characteristics of nano/microparticles.
(a)d‐spacing; (b)crystallite size; (c)average crystallite size; (d)crystallinity of sc‐crystallites, homocrystallites, and sum of hc and sc‐crystallites Xc.
3.1.6. FTIR and S‐FTIR Analyses of UV‐Irradiated Nano/Microparticles
The structural changes of eumelanin and nano/microparticles under UV exposure are examined by FTIR and synchrotron‐based FTIR spectroscopy. sc‐PLA2U was selected to represent the resulting particles due to its superiority in crystallinity and thermal stability compared to its counterparts. Figure 10 shows the FTIR spectra of eumelanin before and after UV exposure. A broad band centered at 3400 cm−1 is attributed to the O‐H stretching of its hydroxyl groups. The C=O stretching modes at 1758, 1715, and 1675 cm−1 are assigned to ketone/aldehyde, carboxylic acid, and amide groups, respectively. After UV exposure, the intensity of the 1758 cm−1 band decreased rapidly, reflected by a sharp negative band in the difference spectrum after subtracting the original sample. This indicates a conversion of ketone/aldehyde groups caused by photodegradation [1]. An increase in the intensity of the 1600 cm−1 band, corresponding to the N‐H and aromatic rings, reflects the deformation of eumelanin’s aromatic/conjugate structures. It was reported that UVA radiation could induce oxidative modifications of dihydroxyindole in the eumelanin structure, generating indolequinone moiety. This moiety occurred simultaneously with the free pyrrole‐2,3,5‐tricarboxylic acid (PTCA), leading to the disintegration of the eumelanin structure into its building units, as summarized in Figure 11 [61].

FTIR spectra of eumelanin: (a) before UV exposure, (b) after 8 h of UV exposure, and (c) the difference spectrum.
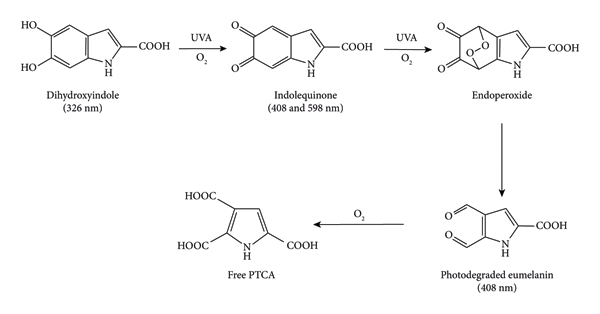
A proposed mechanism of photo‐induced structural modifications of eumelanin [61].
The corresponding difference FTIR spectra of eumelanin and the resulting eumelanin‐loaded particles after 8 h of UV exposure are compared in Figure 12(a). Changes in the intensities of the bands associated with eumelanin were observed after UV irradiation, i.e., C‐H stretching at 2995 cm−1, C=O stretching at 1758 cm−1, and C‐O stretching at 1200 cm−1. A slight decrease in intensity of the 1750 cm−1 band of the C=O stretching of PLA, which overlapped with that of eumelanin, was observed, indicating the ester bond breaking [62]. However, the absence of an anhydride C=O band at around 1845 cm−1, typically formed after a high degree of photodegradation indicates that the photodegradation of the sc‐PLA shell is negligible [62, 63].
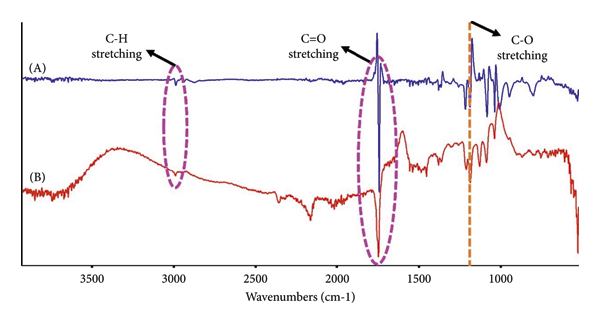
The effect of UV exposure on chemical structures of eumelanin and nano/microparticles examined by FTIR and synchrotron‐based FTIR spectroscopy: (a) difference FTIR spectra of (A) sc‐PLA2U particles and (B) eumelanin after 8 h of UV exposure, (b) normalized FTIR spectra of nano/microparticles after 8 h of UV exposure, (c) normalized FTIR spectra of sc‐PLA2U particles as a function of UV exposure time, and (d) synchrotron‐based FTIR spectra of sc‐PLA2U particles before and after 8 h of UV exposure.
(b)

(c)

(d)

The ratiometric approximation was used to evaluate the intensities of significant bands after UV irradiation by normalizing the spectra with a reference band at 2992 cm−1. The normalized intensities of the 2925 and 2855 cm−1 bands, corresponding to the C‐H stretching mode of Tween 80, are dependent on the eumelanin distribution process and ultrasonication treatments (Figure 12(b)). As shown in Table 5, the distribution of eumelanin in the copolymer (sc‐PLA2U and sc‐PLA2) yielded the highest values for both bands. In contrast, the samples with ultrasonication treatment showed higher values than those with mechanical stirring. The results suggest that the excess Tween 80 attached to the surface of the nano/microparticles changed after UV radiation. This also implies that the distribution of eumelanin in the copolymer phase and the ultrasonication treatments in the preparation steps could prevent the degradation of Tween 80 on the particle’s surfaces. Similar results were also observed on the 2941 cm−1 band, associated with the C‐H stretching of PLA. This indicates that the preparation method can be optimized to prevent degradation of the sc‐PLA shells. FTIR spectra of the particles as a function of UV exposure time are further examined, as shown in Figure 12(c). The intensity of the bands at 2925 and 2855 cm−1 decreased after a certain time of UV irradiation, indicating the degradation of Tween 80 attached to the particle’s surface. Similar results were also observed for the C‐H stretching band of PLA at 2941 cm−1, reflecting the degradation of sc‐PLA shells after UV exposure.
Normalized peak intensities of 2941, 2925, and 2855 cm−1 modes by an internal standard band (2992 cm−1) after UV exposure.
Synchrotron‐based FTIR spectra are further examined to obtain additional information. It was confirmed that the crystalline regions of sc‐PLA shells are affected by UV irradiation, as the peak intensity at 873 cm−1 of the amorphous phase increased after UV exposure (Figure 12(d)). This indicates the destruction of the sc‐PLA crystalline phase. Teixeira et al. [63] reported that PLA chains could undergo photodegradation both in the crystalline and amorphous regions, where the crystalline regions showed lower photodegradability. The decomposition of PLA by UV radiation mainly occurred at the C‐O bonds of ester groups. This involves a photolysis reaction that results in the breakdown of the C‐O backbone and promotes the cleavage of PLA chains. In contrast, the intensity of the 1042 and 1187 cm−1 bands of the C‐O stretching of sc‐PLA shells decreased, confirming the degradation of sc‐PLA shells to some extent. The results suggest that PLA could degrade to a shorter chain after UV exposure. The effect of UV irradiation on the crystalline regions of the blank sc‐PLA shells also showed similar results with sc‐PLA2U (Supporting Information S1). However, in the presence of eumelanin and ultrasonication treatment, the decrease of the crystalline regions reduced as the attached Tween 80 on the particles’ surface increased. This prevented the destruction of the sc‐PLA crystalline phase to the amorphous phase.
3.1.7. Encapsulation Efficiency of Eumelanin‐Encapsulated Nano/Microparticles
The encapsulation efficiency and eumelanin release behavior of the nano/microparticles are investigated by UV‐Vis spectroscopy. Figure 13(a) shows UV‐Vis spectra of eumelanin standard solutions at concentrations of 2.5–40 ppm. Eumelanin has high absorbance in the UV region with a gradual decrease towards longer wavelength in the visible region, typical for aromatic chromophores in its structure [3]. The wavelength of maximum absorbance is 223 nm, with a shoulder peak at ∼270 nm due to the presence of indolic and/or phenolic groups [64]. This agrees with a previous report on melanin nano/microparticles from sepia ink [65]. The strong absorption in the UV region is likely due to the energy transition caused by the delocalization of electron and a high degree of conjugation that assist in the transition of the bonding orbital π to the antibonding orbital π ∗(π⟶π ∗) and nonbonding orbital n to the antibonding orbital π ∗(n⟶π ∗) which mainly occurs in carbonyl groups existing in its indolic structure [66]. The absorption peaks were therefore used for determining the concentrations of eumelanin by constructing a standard curve correlating the absorbance and the concentrations.

UV‐Vis spectra of (a) eumelanin standard solutions with concentrations from bottom to top: 2.5, 5, 7.5, 10, 12.5, 15, 17.5, 20, 30, and 40 ppm and (b) eumelanin solutions extracted from direct and (c) indirect methods.
(b)

(c)

The eumelanin encapsulation efficiency (EE%) of the nano/microparticles is determined by two methods. The direct method was performed by extracting eumelanin inside the nano/microparticles by dissolving the shell of nano/microparticles. In contrast, the indirect method calculated EE% by measuring the concentration of unentrapped eumelanin in the supernatant after centrifugation. The corresponding spectra from both direct and indirect methods are compared in Figures 13(b) and 13(c). The spectra of extracted eumelanin from the particles exhibited an intense peak at 223 nm. A small hump at around 260–280 nm was more intense than the standard solution, indicating the presence of other components in the solutions. A strong absorption peak is also observed from the indirect method (Figure 13(c)) at around 230 nm with shoulder peaks at about 270 and 280 nm, likely corresponding to residue PLA and/or Tween 80 in the supernatant [67, 68]. The encapsulation efficiency results are summarized in Table 1. The EE% values from the indirect method vary from 84 to 97%, while those of the direct method are 32–47%. The differences likely originate from several factors. The values from the indirect method may be overestimated due to the interference from residues of other components in the system [69]. Nevertheless, the results from both techniques show similar trends. The ultrasonication‐assisted particles showed higher EE% than those without ultrasonication. This is due to the hollow structure and the larger surface area inherited by their smaller size, leading to enhanced entrapment efficiency [70]. The materials prepared from the distribution of eumelanin in the copolymer (sc‐PLA2U) also showed higher EE% than its counterparts, confirming the procedure’s efficiency.
3.1.8. In Vitro Release of Eumelanin from the Nano/Microparticles
The eumelanin release profiles of the nano/microparticles in PBS are summarized in Figure 14. Initial burst release was observed in all samples, with a release of >10% in the first 30 min, followed by a sustained release over a prolonged time. The burst release is generally associated with the unentrapped active compounds on the particle’s surface being readily released into the medium. The absence of the characteristic eumelanin bands in the ATR‐FTIR spectra, however, may be due to its low loading content and relatively‐low absorptivity of the compound’s vibrational modes. The burst release of hydrophilic eumelanin from within the particles may be associated with its diffusion through the pores of the shell structure, as evidenced in SEM images. In addition, this may be induced by the rapid water absorption or swelling of the particles caused by the osmosis mechanism [71]. A diffusion mechanism subsequently occurs after the particles’ shells are swollen upon hydration in the PBS medium, leading to a gradual release of eumelanin from the particles. The eumelanin distribution step also imposes a significant effect on the release of eumelanin. The distribution of eumelanin in the copolymer/PLLA mixture (sc‐PLA4U and sc‐PLA4) showed a more stable cumulative release than their counterparts. This might be due to the thicker sc‐PLA shell and smaller pore size, as observed from SEM images. Therefore, the swelling effect caused by the PBS medium could be controlled. In contrast, all materials showed a sustained release below 50% after 144 h, irrespective of their eumelanin distribution step and ultrasonication. These results indicate that the formed sc‐PLA shells serve as stable storage sites for eumelanin, resulting in an effective, sustained‐release behavior, which is beneficial for various biomedical and cosmetic applications.
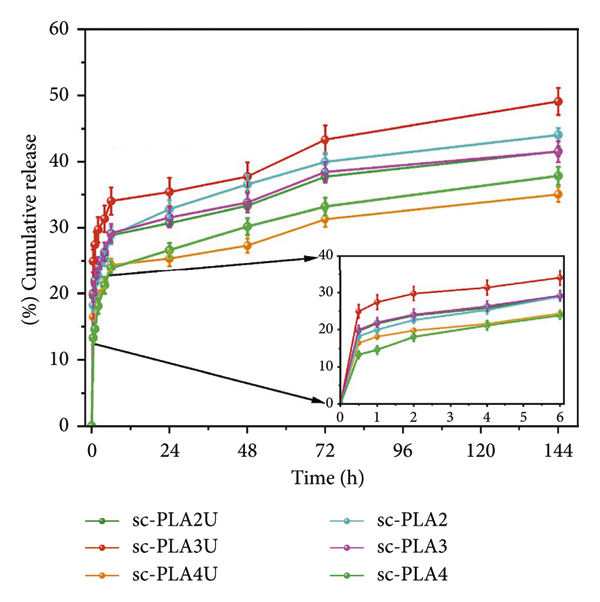
The release profiles of eumelanin from the nano/microparticles in PBS. The inset shows the behavior during the first 6 h period.
3.1.9. UV Absorbance and SPF Values of the Nano/Microparticles
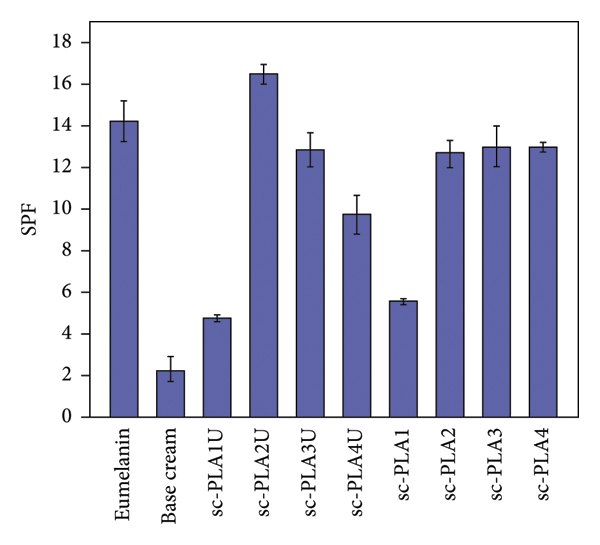
To assess the materials’ potential use as functional cosmetic products, the particles were applied to commercial sunscreen products, and their UVA‐UVB transmittance is recorded, as shown in Figure 15(a). The high UV shielding ability is confirmed, as the UV absorbance of the base cream is largely enhanced when only 1% nano/microparticles are introduced. This is due to the high UV absorption ability of eumelanin combined with the hollow structure of the particles. As shown in Figure 15(b), the base cream showed the lowest SPF value of 2.3 ± 0.6. Adding 1% nano/microparticles improved the SPF value to 9.7–16.5. The addition of the blank particles also increased the SPF value of the base cream, confirming the effect of the hollow structure [26]. The highest SPF value was exhibited by sc‐PLA2U (16.5 ± 0.5), as a result of its high eumelanin content. The inclusion of ultrasonication‐assisted particles showed slightly higher SPF values than those without ultrasonication. It was suggested that the hollow particles with smaller sizes improved the sunscreen performance due to higher UV scattering ability [26]. The results are also in accordance with lignin‐loaded nanoparticles (LNP)‐containing sunscreens, in which small LNP sunscreens (43–95 nm) possessed improved performances of UVB‐SPF and UV transmittance [28].

(a) UVA‐UVB absorbance and (b) SPF values of sunscreen products containing various eumelanin‐loaded nano/microparticles (1% content).
(b)
3.1.10. Cytotoxicity Analysis of Eumelanin and Nano/Microparticles
The cytotoxicity of eumelanin and eumelanin‐loaded nano/microparticles is evaluated by MTT assay. This colorimetric assay is based on the reduction of yellow tetrazolium to purple formazan crystals by metabolically active cells. The analysis was performed in the particle concentrations of 0–1000 μg/mL at 48 h period of treatment. As shown in Figure 16, the blank particles (sc‐PLA1U) with concentrations up to 0.1 μg/mL showed an insignificant decrease in cell viability. An increase in the concentration to 1 μg/mL slightly increased the cytotoxic activity, as reflected by the reduction of cell viability to around 50%. In contrast, eumelanin‐loaded particles showed a rapid decrease in cell viability at a concentration of 0.1 μg/mL, where the cell viability depended on eumelanin distribution. At a lower concentration (0.001–0.01 μg/mL), the particles showed insignificant cytotoxicity with cell viability of >70%. At a concentration above 0.01 μg/mL, all eumelanin‐loaded particles showed a significant decrease in cell viability. In contrast, eumelanin showed higher cell viability at a concentration of up to 100 μg/mL. This might be due to the larger particle size of eumelanin than the eumelanin‐loaded particles. It was reported that cell proliferation and viability depended on the dose and the size of nanoparticles [72]. The size dependence is associated with the initial particle size and not the hydrodynamic particle size. The authors found that smaller particles with a higher surface area showed more cytotoxic activity than larger particles. In the present work, the particle size of eumelanin was not measured. However, the eumelanin used for this cytotoxicity analysis was coarse powder. Since eumelanin has an inherent cytotoxic effect, it is expected that cytotoxicity will increase with the nano/microscopic size of the resulting particles [73]. At the highest concentration, 1000 μg/mL of eumelanin shows the most significant cytotoxic activity (10% cell viability), while eumelanin‐loaded particles show slightly higher cell viability.
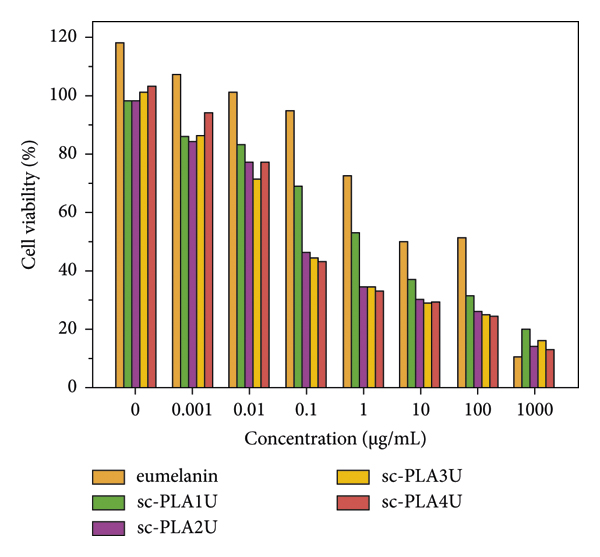
Effects of eumelanin, blank sc‐PLA particles, and eumelanin‐loaded sc‐PLA particles on human keratinocytes (HaCaT) cells viability after 48 h incubation.
The results show that the distribution of eumelanin affects the cytotoxic activity of the particles, whose effect varies with particle concentrations. As PLA and its copolymer are renowned for their nontoxicity, it is likely that the cytotoxic effect of the resulting particles might come from eumelanin. It is reported that 5,6‐dihydroxyidole (DHI) and 5,6‐dihydroxyidole carboxylic acid (DHICA), the building blocks of eumelanin, showed a wide range of cytotoxicity that is not specific to any cells [74]. DHI and its oxidization products can enter cells, where they interact with actin, other cellular proteins, and nucleic acids inside the cells in a manner that can act as a nonspecific mutagen, resulting in the rapid death of cells. However, at low concentrations, both DHI and DHICA can provide protective effects from UV radiation [74, 75].
4. Conclusions
A process for preparing eumelanin‐loaded sc‐PLA nano/microparticles employing the P(DLA‐b‐CL‐b‐DLA) triblock copolymer and PLLA as precursors was successfully developed by oil‐in‐water emulsification. The eumelanin distribution in PLA’s enantiomers and ultrasonication play an important role in the physicochemical properties, encapsulation efficiency, release behavior, and UV‐shielding performance of the resulting nano/microparticles. The shape and size of the particles can be controlled by ultrasonication treatments during emulsification. The intense energy provided by ultrasonication allows the droplet disintegration, resulting in finer particles. The hollow structure and larger surface area due to the small particle size improve the encapsulation efficiency of the resulting particles. This structure also allows for a more sustained cumulative release of eumelanin from nano/microparticles. The high energy of ultrasonication promotes the growth of sc‐PLA crystallization by increasing the nucleation rate due to faster diffusion of short polymer chains, leading to the increase in the crystallinity and thermal stability of the resulting particles. The small particle size also affects the cytotoxic activity of the resulting particles, where the cytotoxic activity increases at a relatively high concentration of particles. The effect of eumelanin distribution on the encapsulation efficiency and release profiles of the resulting particles was observed. The high content of eumelanin and the large surface area with a hollow structure improve the UV absorbance and sunscreen performance of the particles. These findings indicated that the materials have high potential in various applications, especially in cosmetic and pharmaceutical fields, as UV‐shielding products.
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
Conflicts of Interest
The authors declare that there are no conflicts of interest.
Authors’ Contributions
O.D.P. carried out all the experiments, analyzed all data, and wrote and prepared the original draft. P.O., the corresponding author, designed the experiment, got the funding, supervised the whole experiment process, and reviewed and edited the manuscript. A.P., S.B., W.S., C.K., K.T., B.N., S.B., and K.T. guided the implementation of the experiment and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Footnotes
Acknowledgments
The authors acknowledge financial support from the Thailand Science Research and Innovation Fundamental Fund and the Center of Excellence in Materials and Plasma Technology (CoE M@P Tech), Thammasat University. O.D.P. is grateful for the scholarship support from the Excellent Foreign Student (EFS) scholarship from SIIT, Thammasat University.