Abstract
A new magnetic adsorbent, namely, MnFe2O4/D201, with deep-treatment ability and high selectivity adsorption for As(V) was prepared. According to isotherm adsorption and kinetics, As(V) adsorption is primarily used for chemical bonding throughout the single-layer adsorbing process. The maximum As(V) adsorption capacity of MnFe2O4/D201 can reach 35.8 mg/g at pH 3. MnFe2O4/D201 also exhibits higher selectivity adsorption against Cl-, NO3 -, SO4 2-, and PO4 3-. According to the thermodynamic results, the adsorption process was spontaneous and endothermic. The adsorption capacity is maintained at 81% of the initial after ten adsorption-desorption cycles. As(V) concentrations ranging from 1 mg/L to 10 μg/L can be treated in fixed-bed column experiments. The effectual cure volume of As(V) reaches 1332 BV (26.64 L). The removal mechanism primarily comprises electrostatic attraction and complexation.
1. Introduction
Arsenic, as a metalloid material with high toxicity, is widely available and is a significant synthetic material in the fields of industry, agriculture, semiconductor substances, and so on [1]. Because of the increasing demand for As, the issue of As-containing wastewater is also on the rise, causing severe water and soil pollution. For this reason, the World Health Organization (WHO) has set the maximum As concentration in drinking water at 10 μg/L to reduce As poisoning [2]. For a long time, approximately 150 million people in over 70 countries consumed As-contaminated water consumption, resulting in long-term damage from arsenic pollution [3]. As(V) is a class I carcinogenic element, and drinking water containing a significant amount of As poses severe health risks, including cancers and neurological diseases [4]. Therefore, it is critical and urgent to find an effective technology for reducing As(V) concentration.
Many strategies have been learnt and used in oxyanion-polluted wastewater treatment, including membrane separation, adsorption, biological treatment, chemical precipitation, and ion exchange [5]. Among various strategies, adsorption technology has attracted significant attention because of its unique treatment effect, broad application scope, important reusability, and other advantages. Therefore, many adsorbents have been proposed to treat As(V)-contaminated drinking water, including cellulose, chitosan, iron oxides, clays, zeolites, alginate, and biochar [6]. In recent years, nanomaterials have piqued interest as adsorbents because of their exceptional properties [7]. Although nanomaterials exhibit good adsorption efficiency, it is not easy to recollect the used adsorbents after pollutant adsorption [8]. The fast development of magnetized nanotechnology has provided novel possibilities for promoting the water treatment process [9]. Among these adsorbents [10], MnFe2O4 is one of the most essential chosen and worthy ecofriendly materials and has piqued considerable interest because of its easy synthesis, convenient recyclability, and biocompatibility. MnFe2O4 is extensively used in As(V) remediation because of its excellent sorption capacity and availability. Nevertheless, MnFe2O4 is inclined to self-aggregate because of its large specific surface area and minor scales, degrading the adsorption efficiencies [11].
In addition, considering the problems of aggregation and recovery difficulty of the nanomaterial MnFe2O4, supporting materials (such as activated carbon, carbon nanotubes, resins, and graphene) are employed to provide support to enable nanoadsorbent to be well recycled through a facile “filtrating-washing” process [12], thereby overcoming the disadvantages of the traditional powdered MnFe2O4 nanomaterial. Moreover, their specific surface area and dispersibility can be improved, thereby improving their reactivity [13].
Among these supporting substances, D201 resins have excellent mechanical strength, Donnan membrane impact, and stable physicochemical characteristics [14]. In particular, the D201 resin surface has positively charged functional groups conducive to heavy metal adsorption [15]. The ion exchange-surface deposition technique is one of the most widespread techniques for preparing MnFe2O4/D201 nanocomposites. The mass transfer controlling stage is the diffusion process of ionic components, because ionic components can spread out on the resin surface and the inner surface and react quickly with the functional groups on the resin surface. Thus, if modified with magnetic nanomaterials such as MnFe2O4, MnFe2O4/D201 nanocomposites can be used as excellent adsorbents for removing different types of pollutants, including anions from water. Because MnFe2O4/D201 nanocomposites contain abundant MnFe2O4 and quaternary ammonium groups, such modified materials are predicted to have a high attraction for As(V) in water.
In this work, MnFe2O4 was loaded on D201 through a coprecipitation approach for water treatment purposes, which can improve the high selectivity adsorption capacity and deep-treatment ability of materials toward pollutants such as As(V). Various characterization methods, including Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), thermogravimetric analysis (TGA), scanning electron microscopy (SEM), vibrating sample magnetometry (VSM), Brunauer-Emmett-Teller (BET) surface area, zeta, and X-ray photoelectron spectroscopy (XPS) analyses were used to characterize the composite structure and magnetic and physicochemical properties of the synthesized nanocomposite. Adsorption kinetics and adsorption isotherms were used to explore the adsorption mechanism in the presence of MnFe2O4/D201. The pH level, contact time, temperature, initial As(V) concentration, coexisting ions, and regeneration were studied through batch experiments. A fixed-bed column experiment with As(V) was conducted to assess its potential for practical applications. The underlying removal mechanisms were analyzed.
2. Materials and Methods
2.1. Materials
D201 resin was purchased from Tianjin Delite Environmental Protection Technology Co., Ltd. (Tianjin, China). Before use, D201 was treated with 1 M NaOH and 0.1 M HCl solution. Then, D201 is vacuum dried at 50°C for 24 h. KMnO4, FeSO4, NaCl, NaNO3, Na2SO4, and Na3PO4 were bought from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China).
2.2. Preparation of MnFe2O4/D201
The MnFe2O4/D201 was prepared by using a one-pot coprecipitation approach. In a standard synthesis procedure, add a certain quantity of FeSO4 and KMnO4 to 100 mL of deionized water, and stir until resolved. Afterward, 20 g of D201 was accurately added to the system. The solution was adopted by mixing at 298 K for several hours to permit the precursor to disperse inside D201. Then, 3 M NaOH solution was slowly added to the above mixed solution to obtain MnFe2O4/D201. The obtained MnFe2O4/D201 were soaked in alkaline environment for 12 h. Filter MnFe2O4/D201 out of the NaOH solution and clean them with 5 wt% NaCl solution several times until the supernatant arrives at neutral (
Adjust the mass ratios for MnFe2O4 to D201 as MnFe2O4/D201-5%, MnFe2O4/D201-10%, MnFe2O4/D201-15%, MnFe2O4/D201-20%, and MnFe2O4/D201-25%; several different adsorbents were prepared. For comparison, bare D021 without MnFe2O4 as the comparison material of the adsorption experiment. A schematic diagram for the novel preparation process can be obtained in Figure 1.
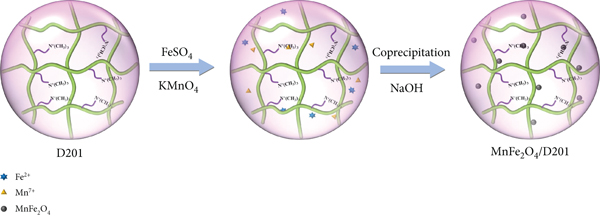
The flow chart of MnFe2O4-D201.
2.3. Batch Adsorption Experiments
The primary condition of batch adsorption experiments is to use 0.5 g/L adsorbent, keep the stirring speed at 150 rpm, and control the temperature at 25°C. The influences of pH, reaction time, initial concentration of As(V), and coexisting competing anions on the adsorption process were studied. For the pH in the As(V) solution, diluted HCl and NaOH solution (0.1 mol/L) are used to adjust the solution. The residual contents of As(V) were analyzed through an inductively coupled plasma atomic emission spectroscopy (ICP-AES) instrument [16]. The quantities of As(V) adsorbed onto MnFe2O4/D201 were estimated by comparing the changes in As(V) concentration before and after. The adsorption capacity was depicted as follows in Text S1.
Varying parameters’ impact on the adsorption process was examined, including pH, contact time, initial concentration, and temperature. The effect of pH on As(V) removal was calculated by changing the pH from 3 to 11. For kinetic, explore the adsorption performance under different adsorption times (0-360 min). For adsorption isotherm experiments, it was examined at 298 K, 318 K, and 338 K within the initial As(V) concentration altered from 30 to 100 mg/L. The effects of coexisting ions were examined in 100 mL of 30 mg/L As(V) solutions containing 50 mg MnFe2O4/D201 and three different dosages (0.01 M, 0.1 M, and 1 M) of NaCl, NaNO3, Na2SO4, and Na3PO4.
2.4. Recycle Experiment
The regeneration of MnFe2O4/D201 was studied by using adsorption-desorption experiments. NaOH solution (0.1 mol/L) was selected as eluent to release As(V) from composite adsorbents. The As(V) adsorbed was wagged with 50 mL of an aqueous NaOH solution for 3 h. Then, MnFe2O4/D201 was accumulated, washed with deionized water, and dried in a vacuum at 60°C for 12 h. After that, the As(V) removal experiment was restated with the regenerated MnFe2O4/D201. The cycle was repeated 10 times and As(V) removal efficiency was tested each time.
2.5. Fixed-Bed Column Experiments
Fixed-bed experiments were followed through 20 mL of MnFe2O4/D201 inside a 14 mm diameter and 240 mm length of glass columns. Set the experimental conditions, the empty bed contact time (EBCT) is specified as 10 min, and the solution flow rate of simulated wastewater is specified as 2 mL/min. The solution was kept to run with a peristaltic pump, and an automated fraction collector was picked out to collect the effluent samples. The feeding solutions consisted of 1 mg/L As(V), 10 mg/L SO4 2-, 100 mg/L Cl-, 10 mg/L PO4 3-, and 100 mg/L NO3 -. The adsorption temperature is at 298 K, 318 K, and 338 K.
3. Results and Discussion
3.1. Characterization of Composite Adsorbent MnFe2O4/D201
The FTIR spectra are depicted in Figure 2(a). The FTIR spectra of D021 and MnFe2O4/D201 indicate that they have near typical peaks and patterns, pointing out that the surface functional groups of D201 do not change in structure after the loading of MnFe2O4. For D201 resin, the broad peak at 3413 cm-1 is ascribed to -OH bands of H2O. The peaks at 1482 cm-1 and 1226 cm-1 are credited to -CH and -CN bands, respectively. Further, the peak at 1627 cm-1 is ascribed to C=C stretching vibration, showing the existence of D201 resin’s styrene frame. The peak at 986 cm-1 is ascribed to the feature of the -N+(CH3)3 band, implying the presence of -N(CH3)3 on D201. For MnFe2O4/D201, the raw peaks at 1160 cm-1 were the same as metal hydroxyl groups (M-OH, M represented Fe or Mn), which proved that MnFe2O4 was successfully loaded on D201’s surface [17, 18].

FTIR spectra (a), XRD patterns (b), and TGA (c) of D201 and MnFe2O4-D201.
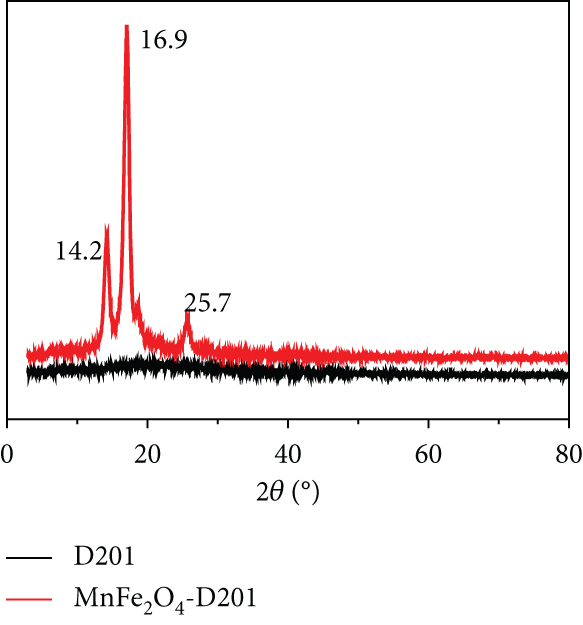

The XRD diffraction patterns of D021 and MnFe2O4/D201 before adsorption are shown in Figure 2(b). The XRD spectrum of D201 reveals no apparent typical peaks, implying the amorphous nature of D201 resin. The XRD patterns of MnFe2O4/D201 showed a clear characteristic peak of MnFe2O4 at 14.2°, 16.9°, and 25.7°, suggesting that MnFe2O4 nanocomposite had been successfully located on D201 [19, 20].
The thermal stability of D021 and MnFe2O4/D201 composite was measured by TGA in Figure 2(c). As illustrated in the curve, D021 and MnFe2O4/D201 have three steps in the thermal weight loss process, including bound water loss and composite degradation, and remain breakdown. For MnFe2O4/D201, the weight loss (18%) up to 328°C was probably due to the dehydration process. What is more, the 17% weight loss between 328 and 384°C was attributed to the carbonization of MnFe2O4/D201, and the 27% weight loss in the scope of 384-465°C was attributed to the degradation of MnFe2O4/D201 [21]. Compared with resin D201, the improvement of thermal stability of adsorbent MnFe2O4/D201 with loading of particle MnFe2O4 will help MnFe2O4/D201 to be more suitable for application in the actual environment with higher temperature.
The morphologies of D021 and MnFe2O4/D201 are shown in Figure 3. MnFe2O4/D201 exhibits a ball-shaped formation and has 0.6 mm in average diameter. Here, loaded MnFe2O4 on D201 possesses several advantages. First, the porous morphology can significantly raise the surface accessibility between MnFe2O4/D201 and As(V). Second, D201 can effectively stop MnFe2O4’s agglomeration with excellent stability. When MnFe2O4 is loaded on the resin D201, there are some particles on its surface, and the surface of MnFe2O4/D201 becomes rough [22, 23]. These findings support the successful synthesis of the adsorbent material MnFe2O4/D201. In addition, the mapping of D201 and MnFe2O4/D201 is displayed in Figure 3. For D201, N and O atomic contents are worked out, showing that the N and O are essential elements in D201. For MnFe2O4/D201, N, O, Mn, and Fe contents are calculated to be 5, 29, 14, and 52%, respectively, suggesting that there are MnFe2O4 onto D201, which are contributing to adsorb As(V). From the distribution of different elements in the mapping of MnFe2O4/D201, it can be seen that compared with the existing nanoparticle loading technology, the nanoparticle MnFe2O4 distribution in the adsorbent MnFe2O4/D201 is more uniform, which is conducive to the removal of As(V). The structure of MnFe2O4/D201 likely contributed to more adsorptive sites for As(V) removal.

SEM image and mapping of D201 and MnFe2O4-D201 before adsorption of As(V).

To confirm that the adsorbent formation existed, N2 adsorption-desorption isotherm characterization was performed. The specific surface area and pore size of MnFe2O4/D201 were also analyzed using the BET method. As depicted in Figure S1a, MnFe2O4, D201, and MnFe2O4/D201 follow a type IV isotherm and have a hysteresis loop at higher relative pressure. MnFe2O4, D201, and MnFe2O4/D201 are mesoporous materials. The BET surface areas of MnFe2O4, D201, and MnFe2O4/D201 are 9.5, 18.3, and 11.2 m2/g, respectively [24, 25]. Moreover, the diameters of D201 and MnFe2O4/D201 with a sharp distribution were approximately 2.8, 11.5, and 3.7 nm, respectively. The surface area, average pore diameter, and pore volume of MnFe2O4/D201 are less than those of D201, showing the successful functionalization through moieties as nanoparticles attached to D201 hindered the entry of nitrogen gas into the nanocomposite surface. On the other hand, D201’s higher SBET indicated that D201 could effectively prevent the self-agglomeration of MnFe2O4 nanoparticles. The possible reason is a large number of metal ions (Fe2+ and Mn7+) were loaded onto the pore channels of D201 through water at the first stage of the preparation of the adsorbent material MnFe2O4/D201. D201 with higher SBET has a large number of active sites (-N+(CH3)3 groups) uniformly dispersed in the pores. The -N+(CH3)3 groups on the resin D201 would promote the diffusion of metal ions when chemical forces occur. After in situ growth preparation process of MnFe2O4/D201, the nanoparticle MnFe2O4 was more dispersed on pore channels of the resin D201 through D201’s higher SBET. It may be expected that SBET and pore volume will advance the adsorption capacity of As(V) by providing more active adsorption sites and reducing the mass transfer resistance of As(V).
The magnetic characteristics of MnFe2O4/D201 were evaluated by using a vibrating sample magnetometer (VSM). Importantly, MnFe2O4/D201 possessed a saturation magnetization (1.6 emu/g), as demonstrated in Figure S1b. Due to the nonmagnetic D201 mediums in MnFe2O4/D201, thus MnFe2O4/D201 has a lower saturation magnetization [26]. Further, the magnetic separation and dispersion of the adsorbents were drawn by a magnet, further pointing out that MnFe2O4/D201 is well separable. At the same time, the structural properties of resin D201, including the excellent mechanical strength and spherical shape, would also bring convenience to the practical application of composite materials MnFe2O4/D201. Composite materials MnFe2O4/D201 would combine their advantages to enhance the separation and recovery of nanocomposite after adsorption.
The survey XPS spectra of D201 and MnFe2O4/D201 are demonstrated in Figure S2. The survey XPS spectrum revealed the existence of C1s, O1s, N1s, Fe2p, and Mn2p on the sample surface of MnFe2O4/D201 [27]. However, Fe2p and Mn2p spectra are not apparent in the XPS spectra of MnFe2O4/D201, probably for the reason that several MnFe2O4 nanoparticles are imbedded in D201 resin’s inner channels.
3.2. Adsorption Study of MnFe2O4/D201 for As(V) Removal
3.2.1. The Effect of MnFe2O4 Content on MnFe2O4/D201 for As(V) Removal
The effect of MnFe2O4 content on As(V) adsorption capacity of MnFe2O4/D201 is shown in Figure S3. Although the resin itself has a large specific surface area and a certain amount of effective functional groups, its adsorption capacity for As(V) still has certain limitations. Compared with many reported adsorption materials, the adsorption performance of D201 is not outstanding. With the addition of nanoparticles MnFe2O4, its adsorption performance has significantly improved. MnFe2O4/D201-15% has the most effective removal on As(V), and the adsorption capacity arrives at 35.8 mg/g. On the one hand, MnFe2O4 was developed by MnFe2O4/D201 which has the potential to adsorb As(V). On the other hand, the styrene frame of D201 can distribute MnFe2O4, and the resin surface accommodates a massive quantity of -N+(CH3)3 groups. The As(V) adsorption capacities declined in the presence of MnFe2O4/D201-20% and MnFe2O4/D201-25%, which was attributed to MnFe2O4 nanoparticle aggregation on the D201. Therefore, MnFe2O4/D201-15% was selected for further experiments.
3.2.2. The pHpzc of MnFe2O4/D201 and the Influence of pH on As(V) Removal
The pH action is a critical factor because of its double effect on the ionic form of the adsorbate and the surface charge of the adsorbent. To probe the effect of pH and achieve excellent As(V) adsorption, the zeta potential analysis of adsorbents and adsorption experiments at varying pH levels were conducted. According to the references, the nanoparticle MnFe2O4 has a low pHpzc value, so MnFe2O4 presents negative charge in a wide range of pH. MnFe2O4 has a repulsive effect with As(V), which results in poor adsorption capacity. There are a large number of -N+(CH3)3 groups on the resin D201, which can improve the electrical properties of the composite within the whole pH range and increase the material’s pHpzc value. For MnFe2O4/D201, as acidity increases, the charge of the adsorbent particles tends to be positive, as shown in Figure 4(a). Both D201 and the impregnated MnFe2O4 are positively charged and adsorb anions by electrostatic attraction [28].
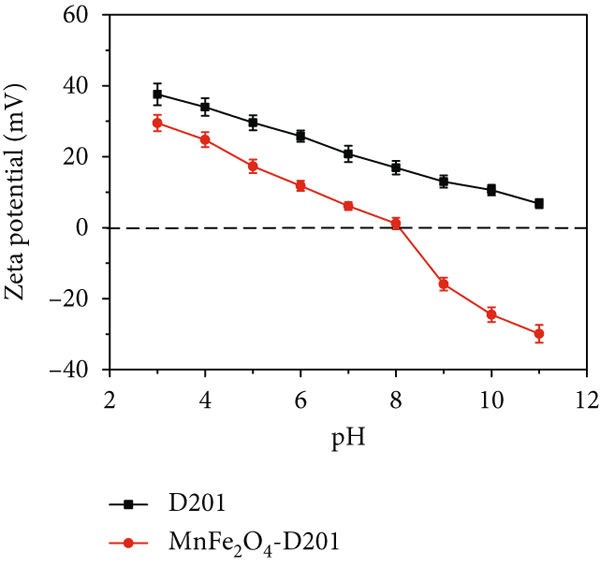
Zeta of D201 and MnFe2O4-D201 (a). Effects of pH value on the uptake of As(V) adsorption by D201 and MnFe2O4-D201 (b). Fraction of As(V) species at different pH (c) (the uniform experimental condition: 100 mL of 30 mg/L As(V) with 50 mg D201 or MnFe2O4/D201, 150 rpm, 25°C, and 360 min).


Meanwhile, for MnFe2O4/D201, the composite adsorbent’s removal efficiency gradually declines as pH increases, as shown in Figure 4(b). For As(V), H2AsO4
- is the dominate species at pH 2-7, whereas the dominant As ion formed is HAsO4
2- as pH increases, as shown in Figure 4(c). When
3.2.3. Adsorption Kinetic Analysis
Contact time was also a critical factor in the As(V) adsorption process. Kinetic experiments demonstrated that MnFe2O4/D201 could quickly reach adsorption equilibrium by supplying -N+(CH3)3 groups and MnFe2O4 (Figures 5(a) and 5(b)). As is visible, all kinetic adsorption curves could be around segmented into two consecutive adsorption steps; the As(V) adsorption capacities by the MnFe2O4/D201 were sharply raised throughout the first 60 min and the adsorption capacity would not increase after 240 min. While contact time was passed 240 min, adsorption capacity’s raises were nearly insignificant, indicating that 240 min could be considered with the adsorption equilibrium time. The maximum equilibrium adsorption capacities (

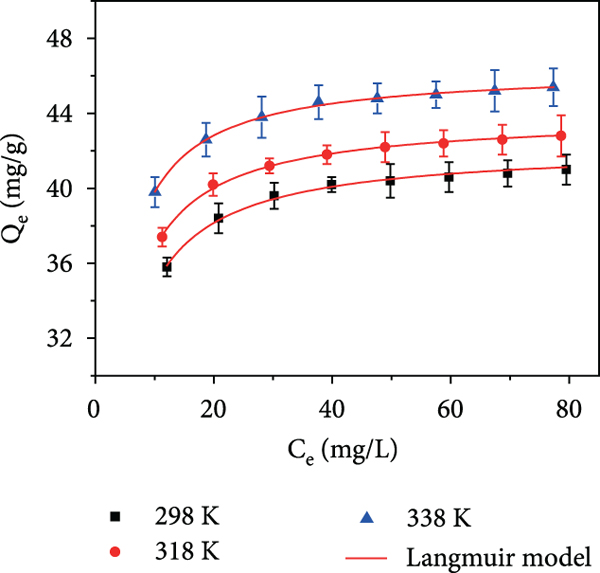
Adsorption kinetics of As(V) at 298 K (a–c). Adsorption isotherm of As(V) at 298 K, 318 K, and 338 K (d–f) (the uniform experimental condition: 100 mL of As(V) with 50 mg D201 or MnFe2O4/D201, 150 rpm; adsorption kinetics: adsorption times are 0-360 min and 30 mg/L As(V); adsorption isotherm: temperature at 298 K, 318 K, and 338 K, As(V) concentration range is 30 to 100 mg/L).

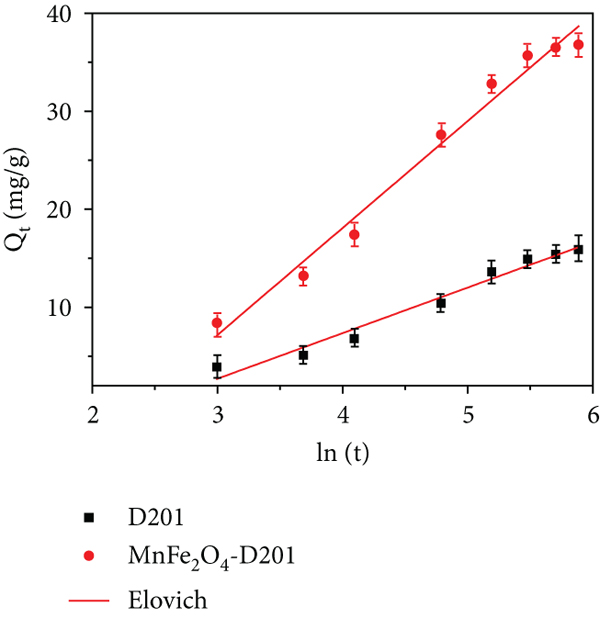


To better investigate and interpret the adsorption behavior, pseudo-first-order [32], pseudo-second-order [33], and Elovich models [34] (Text S1) were analyzed. The parameters derived from adsorption kinetic curves are listed in Table 1. The fitting data in Table 1 indicate that the pseudo-second-order model describes the sorption kinetics better than the pseudo-first-order and Elovich models. The adsorption mechanism corresponding to the pseudo-second-order kinetic is that the control factor of adsorption is chemical interaction, which occurred via electrostatic attraction or complexation between adsorbent and As(V) [35]. The
As(V) adsorption kinetic model constants derived from the pseudo-first-order, the pseudo-second-order equation, and the Elovich model.
3.2.4. Adsorption Isotherm Analysis
The effect of initial As(V) concentration on As(V) adsorption over the surface of MnFe2O4/D201 was studied as shown in Figures 5(d)–5(f). When the As(V) initial concentration was increased from 30 to 100 mg/L at 298 K, the As(V) adsorption capacity was expanded from 35.8 mg/g to 41 mg/g. That is because the adsorption process enhances in the mass transfer driving force [36]. Then, the adsorption capacity of MnFe2O4/D201 has not significantly changed as the As(V) concentration increases again, because the active sites of MnFe2O4/D201 are saturated.
To better understand the interaction mechanisms between As(V) and MnFe2O4/D201, the isotherm models of Langmuir [37], Freundlich [38], and Temkin [39] (Text S1) were evaluated (Figures 5(c) and 5(d)). The suitability of isotherm fit of experimental data was evaluated using nonlinear chi-square test (
Adsorption isotherm model constants for the adsorption of As(V).
3.2.5. Adsorption Thermodynamics
Adsorption thermodynamic characteristics were examined at 298-338 K to determine the effect of temperature on As(V) removal capacity. Three thermodynamic parameters, including Gibbs free energy, entropy, and enthalpy, are presented in Table 3 and Figure S4. The entropy and enthalpy were calculated from the incline and intercept of the adsorption data fitted adopting equation (Text S1). The Gibbs free energy as negative values for the As(V) uptake reactions by MnFe2O4/D201 indicates spontaneity of the adsorption process (Table 3). As the temperature rose, it was also noticed that the Gibbs free energy value rose, implying a greater driving force and thus a higher adsorption affinity at higher temperatures. The high positive values of enthalpy (5.34 kJ/mol) indicate that As(V) adsorption process over the MnFe2O4/D201 is endothermic and the adsorption process is involved in chemical interactions [40, 46]. The presence of a positive entropy values (0.15 kJ/(mol
Thermodynamic parameters for the adsorption.
3.2.6. Effect of Coexisting Competing Anions towards As(V) Adsorption
The ionic strength of the solution and adsorption performance would be affected by coexisting anions. Selective adsorption of As(V) with anions (Cl-, NO3
-, SO4
2-, and PO4
3-) was investigated for MnFe2O4/D201. For coexisting anions, multivalent anions (SO4
2-, PO4
3-) possess a more substantial affinity for MnFe2O4/D201 than monovalent anions (Cl-, NO3
-) in Figure 6(a). The effect of Cl- and NO3
- (0.01-0.1 mol/L) on the adsorption capacity of As(V) by using MnFe2O4/D201 is nearly unchanged. Only when the concentration of Cl- and NO3
- increases to 1 mol/L, the adsorption capacity would decrease slightly. Compared with Cl- and NO3
-, the addition of SO4
2- and PO4
3- leads to an apparent drop in adsorption capacity, but further raising the concentration of the competing anions results in an unvarying
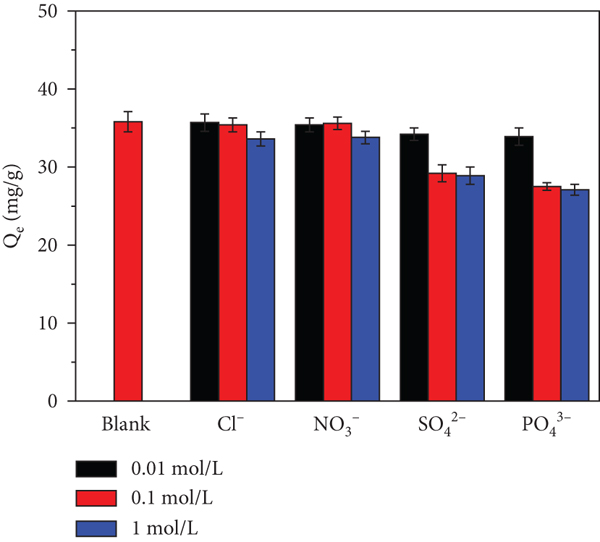
Effects of coexistent ions and ionic strength on As(V) adsorption (a). Adsorption-desorption experiments of As(V) for MnFe2O4-D201 on adsorption capacity (b) and desorption efficiency (c). Fixed-bed column experiment with simulated wastewater effluent (d) (the uniform experimental condition: 100 mL of 30 mg/L As(V) with 50 mg MnFe2O4/D201, 150 rpm, 25°C, and 360 min; (a) salt ion concentration on 0.01 M, 0.1 M, and 1 M; (b, c) 50 mL of 0.1 mol/L NaOH solution for 3 h; (d) the solution flow rate at 2 mL/min, 1 mg/L As(V), 10 mg/L SO4 2-, 100 mg/L Cl-, 10 mg/L PO4 3-, and 100 mg/L NO3 -, at 298 K, 318 K, and 338 K).


It shows that MnFe2O4 modification enhanced the selective adsorption on MnFe2O4/D201. D201 can just capture As(V) through the structure of an outer sphere complex or nonspecific electrostatic attraction. In contrast, the immobilized MnFe2O4 nanoparticles could separate As(V) following ligand exchange or a specific Lewis acid-base interaction. This study’s consequences demonstrated that MnFe2O4/D201 is efficient and selective in removing possibly hazardous As(V) from complex polluted industrial effluents and groundwaters.
3.2.7. Desorption and Reusability Study
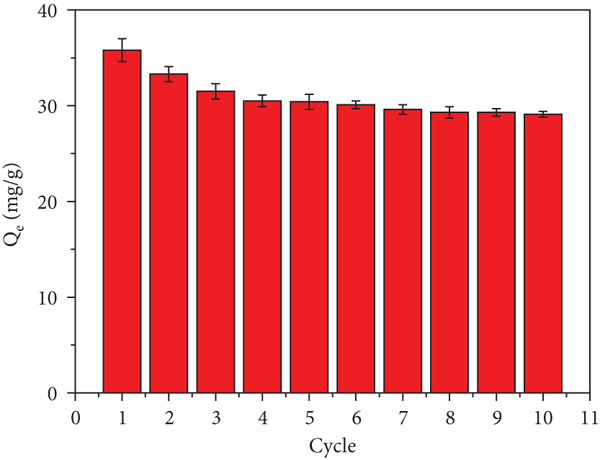
In general, the recyclability of adsorbents can enhance the economics of the remediation process. To investigate the recyclability and reusability of adsorbents, the adsorption capacity of MnFe2O4/D201 was explored for ten cycles. The adsorption capacity is 33.3 mg/g for two reusing runs, higher than 30.4 mg/g for five reusing runs, and higher than 29.1 mg/g for the ten reusing runs (Figure 6(b)). For the adsorbent MnFe2O4/D201, in addition to keeping the adsorption capacity at a high level, its other advantage over MnFe2O4 is that the material quality of MnFe2O4/D201 is basically unchanged. In the field of adsorption, most powdered adsorbents have limitations, including the quality loss of adsorbents and the difficult operation on recovery in practical application. Desorption efficiency of adsorbent MnFe2O4/D201 is shown in Figure 6(c). MnFe2O4/D201 presented a good desorption efficiency over 81.3% after ten times of adsorption-desorption cycle. The small drop in the removal efficiencies of As(V) ions over MnFe2O4/D201 nanocomposite was attributed to the damaged adsorption sites after each cycle [51]. This conclusion is consistent with SEM results of MnFe2O4/D201 before and after adsorption-desorption cycle. As shown in Figure 3, the surface of the adsorbent MnFe2O4/D201 is relatively rough before adsorption. According to the mapping results, the relative percentage contents of N, Mn, and Fe elements at this time are 5%, 14%, and 52%, respectively. After adsorption-desorption cycle, the surface of the adsorbent MnFe2O4/D201 becomes smoother, and the relative percentage content of N, Mn, and Fe elements has slightly decreased in Figure S5. The smooth surface may be caused by the shedding of loaded nanoparticles MnFe2O4 during the desorption process. The relative percentage contents of N, Mn, and Fe elements are 3%, 11%, and 42%, respectively. The active site of the adsorbent mainly contains N, Mn, and Fe elements. It is obvious that the active site of the adsorbent MnFe2O4/D201 is destroyed by the desorption agent during the desorption process, and the content of the active groups has slightly decreased. The controlled drops in sorption-desorption performances indicate the excellent stability and high recyclability of MnFe2O4/D201.
3.2.8. Fixed-Bed Adsorption
To further explore MnFe2O4/D201 applied in wastewater, fixed-bed adsorption toward As(V)-contaminated water was studied according to MnFe2O4/D201 into the separated fixed-bed columns. As described in Figure 6(d), the bed volume of MnFe2O4/D201 was about 774 BV (298 K), 1134 BV (318 K), and 1332 BV (338 K) as reported by the breakthrough curves (breakthrough level was 10 μg/L) [52], implying that MnFe2O4/D201 has good As(V) adsorption capacity in wastewater. Also, it was shown that MnFe2O4/D201 possessed the potential for As(V) species removal in water.
3.3. Adsorption Mechanism of MnFe2O4/D201
The FTIR analyses were applied to confirm chemical changes during the adsorption process. After adsorbing As(V), a raw peak corresponding with the As-O stretching vibration came out approximately 810 cm-1 in Figure 7. This suggested that As(V) was adsorbed onto MnFe2O4/D201 and composed the chemical bond. What is more, the relative intensity of -OH functional groups was decreased after adsorption of As(V), suggesting that the surface complexation between -OH functional groups and As(V) substantially contributed a lot to the adsorption process.

FTIR of MnFe2O4-D201 before and after adsorption of As(V).
The SEM and mapping of MnFe2O4/D201 after the adsorption process are exhibited in Figure S6. Compared with MnFe2O4/D201, the surface of MnFe2O4/D201 has no apparent changes. And As elements appear on the surface of MnFe2O4/D201 about 3% after adsorption, with decreased Mn and Fe element contents. This result indirectly proves that metal atoms Mn and Fe play a significant part in the adsorption of As(V).
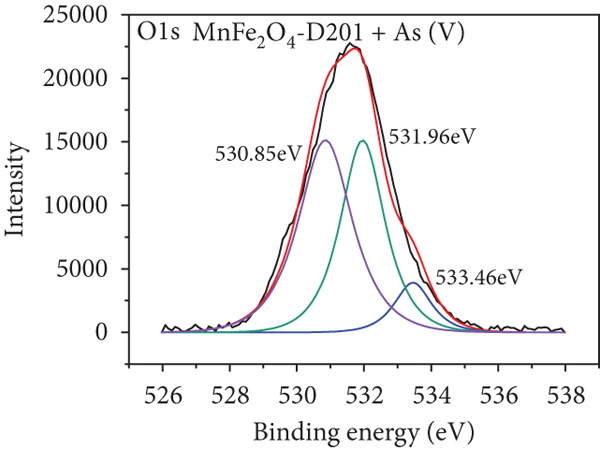
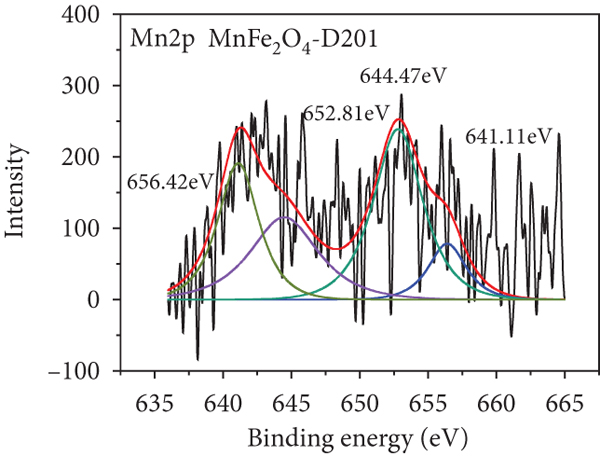
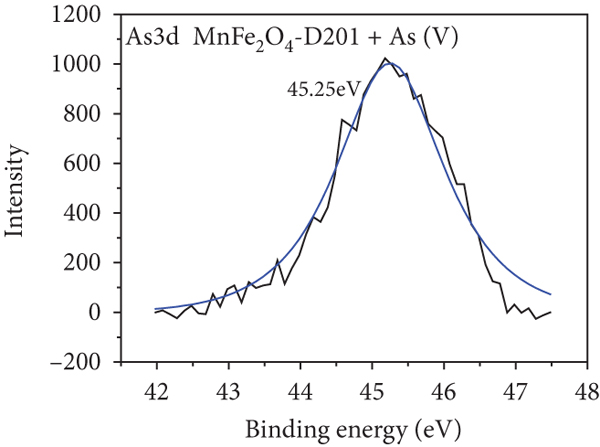
To further realize the adsorption mechanism, XPS spectra were used to probe the chemical state on the surface of the MnFe2O4/D201 composites before and after adsorption, as shown in Figure 8. The high-resolution spectra of N1s of MnFe2O4/D201 are illustrated in Figures 8(a) and 8(b). The characteristic peaks at 402.34 and 399.54 eV belong to -N+(CH3)3 and -NH, respectively. After contact with As(V), -N+(CH3)3 content is reduced from 90.47% to 76.19%, as shown in Table 4, which is ascribed to the electrostatic attraction between As(V) anions and D201 resin [53, 54]. Figures 8(c) and 8(d) illustrate the O1s XPS spectra of MnFe2O4/D201 before or after adsorbing As(V). The figures show multiple peaks centered at 533.42.2, 531.95, and 530.98 eV, assigned to adsorbed crystal water (H-O-H), the hydroxyl group bound to metal (M-OH), and the oxide-oxygen (O2-), respectively. After absorbing As(V), the peak area proportion of O2- increased significantly from 42.19% to 50.00%, whereas that of OH- declined from 50.00% to 41.18%. The results further confirmed that more -OH on the MnFe2O4/D201 surface reacted with As(V). The number of -OH decreased after the reaction, and the inner sphere complexes were formed by complexation between -OH and As(V), which was more steady than the outer sphere complexes formed by electrostatic. To further analyze the role of MnFe2O4, the high-resolution XPS spectra of Mn2p and Fe2p were observed (Figures 8(e)–8(h)). As shown in Figures 8(e) and 8(f), the Mn2p peaks exhibited major peaks before adsorption at 656.42, 652.81, 644.47, and 641.11 eV. The Fe2p peaks exhibited major peaks before adsorption at 711.13, 715.07, 724.66, and 731.55 eV. The binding energies of Mn2p and Fe2p shifted after As(V) adsorption, indicating that Mn and Fe species reacted with As(V) [55, 56]. After adsorption, a raw peak at 45.25 eV is observed in Figure 8(i), attributed to the AsV-O. The As3d’s peak indicates that the MnFe2O4/D201 surface adsorbs a significant amount of As(V) during the adsorption process.

N1s (a, b), O1s (c, d), Mn2p (e, f), Fe2p (g, h), and As3d (i) XPS spectra of MnFe2O4-D201 before and after adsorption of As(V).





Normalized peak areas of XPS spectra in Figure 8.
Concentrating on the experimental data under varying pH, the adsorption capacity of MnFe2O4/D201 decreases with the increase in pH. The physical electrostatic repulsion enhanced due to the deprotonation of sorbents accompanied by the increase of pH value. It is speculated that MnFe2O4/D201 adsorbed As(V) by -N+(CH3)3 and MnFe2O4. Afterward, they supplied electrostatic attraction to negatively charged H2AsO4 - or HAsO4 2- and bound it to the material’s surface in the form of ionic bonds.
These above consequences confirmed that the adsorption mechanism was electrostatic attraction and complexation. The results were also consistent with the isotherm and kinetic study. Besides, the schema of As(V) removal mechanisms by MnFe2O4/D201 is illustrated in Figure 9. First, D201 would increase the zeta potential of MnFe2O4/D201, which is more favorable for electrostatic attraction with As(V). Second, MnFe2O4 modified the surface structure of MnFe2O4/D201 and provided more functional groups such as -OH, contributing to complexation with As(V).

The schematic diagram of adsorption mechanism.
4. Conclusion
In this study, the successful combination of the two components (MnFe2O4 and D201) of the nanocomposite MnFe2O4/D201 was confirmed by various characterization techniques. Different parameters such as pH, contact time, and initial concentration were examined to define the optimum adsorption conditions. The adsorption of As(V) is affected by the pH value, with As(V) adsorption being the most effective at pH 3. The kinetics and adsorption isotherms of MnFe2O4/D201 for As(V) followed the Langmuir isotherm model and pseudo-second-order kinetic model, respectively. The adsorption behavior is controlled by chemical adsorption. The maximal adsorption capacity of MnFe2O4/D201 was 35.8 mg/g, and it attained equilibrium within 240 min. Thermodynamic results showed that the adsorption process was endothermic and spontaneous. The magnetic separation of MnFe2O4/D201 enhanced its reusability for As(V) removal up to ten successive cycles. Finally, the superior performance in the fixed-bed column experiments confirmed the potential use of MnFe2O4/D201 as an excellent adsorbent. The efficient As(V) removal by MnFe2O4/D201 is based on the combined electrostatic attraction and complexation. In summary, because of its facile fabrication process, excellent selectivity adsorption capacity, and deep treatment for As(V) removal, MnFe2O4/D201 has promising application capability for As(V) sequestration from polluted water.
Footnotes
Data Availability
The data used to support the findings of this study are included within the supplementary information file(s).
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Authors’ Contributions
Shaopeng Zhang contributed to the conceptualization, methodology, software, investigation, formal analysis, writing of the original draft, and funding acquisition. Jie Ding contributed to the formal analysis and writing of the original draft. Dayong Tian was responsible for the resources and funding acquisition. Feifei Liu was responsible for the investigation and formal analysis. Qiaoling Li investigated the study and wrote the original draft. Minghua Lu contributed to the resources, supervision, and funding acquisition.
Acknowledgments
This work was supported by the Innovation Practice Base for Postdoctoral of Anyang Institute of Technology and the Doctor Research Starting Foundation of Anyang Institute of Technology (BSJ2021038). Thanks are due to the Test Center of the School of Chemical and Environmental Engineering, Anyang Institute of Technology.
