Abstract
Herein, we report a simple and scalable synthesis route to prepare Ag-functionalized porous ZnO sheets and their enhanced NO2 gas sensing properties. Porous ZnO sheets functionalized with well-dispersed submicron Ag particles were prepared by using a hydrothermal method-based one-pot synthesis route from Zn and Ag precursors. NO2 gas sensing performance (response, selectivity, response time, and recovery time) was optimized at 200°C in the gas sensor fabricated with 3 at% Ag-functionalized porous ZnO sheets. We demonstrated a response (
1. Introduction
Nitrogen dioxide (NO2) is a red-brown highly oxidizing gas [1] and one of the major air pollutants [2], causes nose and respiratory problems, coughs, fatigue, eye irritation, and nausea, and affects patients with debilitating pulmonary system diseases [3–5]. It also makes severe environmental issues such as smog and acid rain [6, 7]; thus, the development of NO2 gas sensors with high reliability and performance is urgently required.
ZnO is widely used gas sensing materials due to its semiconducting nature (
In the present study, we aimed to prepare hybrid materials of Ag particles and ZnO powders in an effort to improve the NO2 gas sensing properties. Notably, by using hydrothermal method-based one-pot synthesis technique, well-dispersed submicron Ag particles (1, 3, and 5 at%) functionalized porous ZnO sheets were obtained from Zn and Ag precursors. NO2 gas sensing properties including response, selectivity, response time, and recovery time were optimized in a sensor fabricated with 3 at% Ag-functionalized porous ZnO sheets at 200°C. Possible mechanisms for enhanced sensing properties in this hybrid materials were also discussed.
2. Experimental
2.1. Synthesis of Ag-Functionalized Porous ZnO Sheets
Initially, 0.02 mol Zn(NO)3·6H2O and 0.06 mol urea were completely dissolved in a 180 mL mixture of ethanol–water (1 : 2, v–v) via mixing for 30 min. Then, 0.06, 0.12, and 0.18 g AgNO3 (1, 3, and 5 at% Ag) were added to the solution, and a transparent mixed solution (zinc nitrate hexahydrate + urea + silver nitrate in ethanol-water) was obtained. This solution was poured into a 250 mL Teflon-lined autoclave and maintained at 180°C for 24 h. After this, the white precipitate was collected and washed several times with deionized water and absolute ethanol by centrifugation. Next, the precipitate was dried under vacuum at 80°C for 6 h. Finally, the Ag-functionalized porous ZnO sheets were obtained after annealing under Ar flow at 550°C for 4 h. The corresponding schematic is shown in Figure 1.

Schematic of the synthesis method.
2.2. Characterization
Crystallinity and phases were examined by X-ray diffraction (XRD, SmartLab, Rigaku) with CuKα radiation (
2.3. Gas Sensing Test
Au interdigitated electrodes were sputter-deposited on the surface of an alumina substrate. Sputtering was performed at 80 mA for 12 min, and the final thickness of the electrodes was 300 nm. The fabricated gas sensor was placed on a ceramic heater, and its gas sensing properties were measured using a lab-made gas sensing system (Supplementary Materials (SM), Figure S1). Desired amounts of target gases were mixed with dry synthetic air as a background using mass flow controllers (MFCs). The resistance of the gas sensor in air (
3. Results and Discussions
3.1. Morphological, Chemical, and Structural Studies
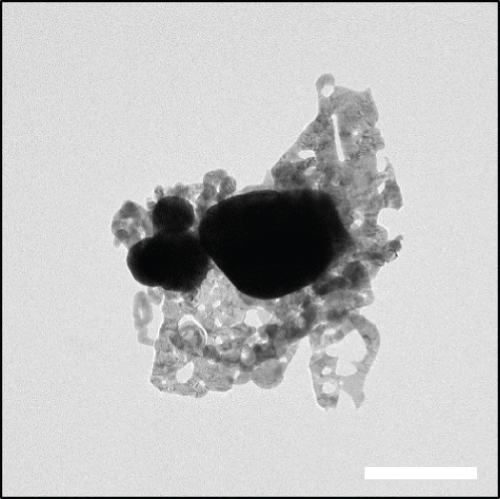
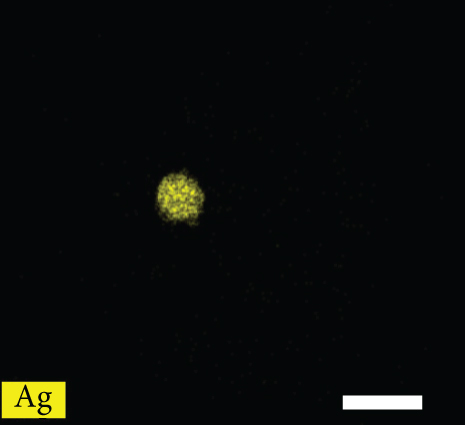
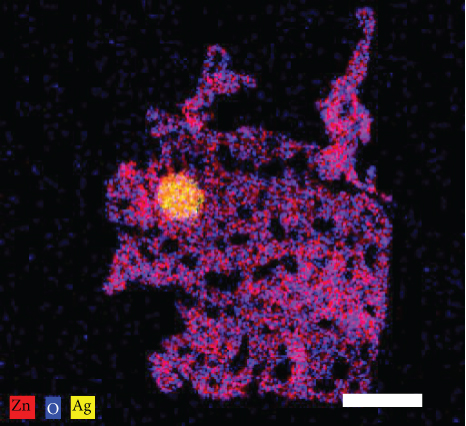
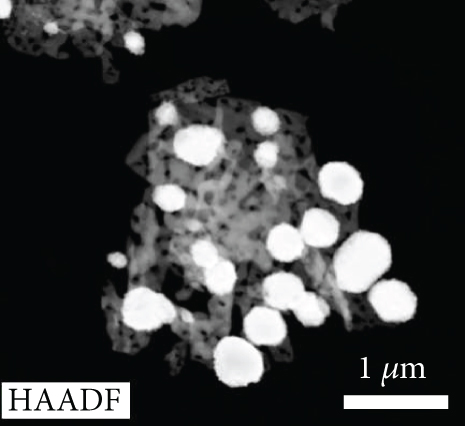
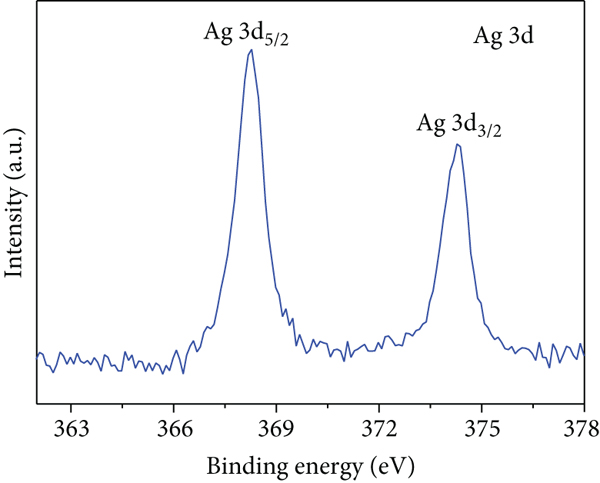
Figures 2(a)–2(f) indicate the TEM images of 1, 3, and 5 at% Ag-functionalized porous ZnO sheets, respectively. Interestingly, sheet-like ZnO with highly dense pore, which is desirable for sensing applications due to the large surface area, was formed. Dark particles represent the Ag particles functionalized on the surface of ZnO. Figures 3(a)–3(o)show the TEM elemental mapping data of 1, 3, and 5 at% Ag-functionalized porous ZnO sheets, respectively. Zn and O are evenly distributed over the sheet surface, whereas Ag is localized in certain places. Specifically in Figures 3(e), 3(j), and 3(o), it can be clearly observed that the dark points are pores, and the yellow points are Ag particles, which ensures the formation of Ag-functionalized porous ZnO sheets. The TEM–EDS point analysis results of 1, 3, and 5 at% Ag-functionalized porous ZnO sheets are given in SM Figure S2(a–d), (e–h), and (i-l), respectively. The white area in SM, Figure S2(a and b) is mostly rich in Ag, whereas the gray area in SM, Figure S2 (c and d) is rich in ZnO. Similarly, in SM, Figure S2 (e and f) and SM, Figure S2(i and j), a white area with 99.8 wt% Ag and 98.9 wt% Ag is recorded, whereas in SM, Figure S2(g and h) and SM, Figure S2(k and l), a gray area with 88.6 wt% Zn and 90.0 wt% Zn is observed. Overall, the TEM–EDS point analysis results confirmed the formation of Ag-functionalized porous ZnO sheets. SM, Figure S3(a) shows the XRD patterns of 3 at% Ag-functionalized porous ZnO sheets. Standard JCPDS files of Ag and ZnO are also shown in SM, Figure S3(b and c) for comparison. In both cases, the peaks related to both Ag and ZnO according to the JCPDS file nos. 04-0783 and 36-1451 were observed [27] without impurities. The crystallinity in XRD shows good agreement with HRTEM (Figure 4). Figure 5(a) indicates the XPS survey of 3 at% Ag-functionalized porous ZnO sheets, revealing the presence of all expected elements, namely, Zn, Ag, and O (peak related to C is due to the environment). The high-resolution Zn 2p core-level region is shown in Figure 5(b), where the two peaks at the binding energies of 1022.38 and 1045.38 eV are attributed to Zn 2p 1/2 and Zn 2p 3/2, respectively, with a spin-orbit splitting of 23.03 eV, which is in accordance with the previously reported literature [33]. This indicates that Zn is in the Zn2+ oxidation state [34, 35]. Figure 5(c) shows the high-resolution Ag 3d core-level region, where the Ag 3d 3/2 and Ag 3d 5/2 peaks can be observed at the binding energies of 374.28 and 368.28 eV, in accordance with the literature [36].

TEM images of the (a, b) 1, (c, d) 3, and (e, f) 5 at% Ag-functionalized porous ZnO sheets.





TEM elemental mapping of the (a–e) 1, (f–j) 3, and (k-o) 5 at% Ag-functionalized porous ZnO sheets.






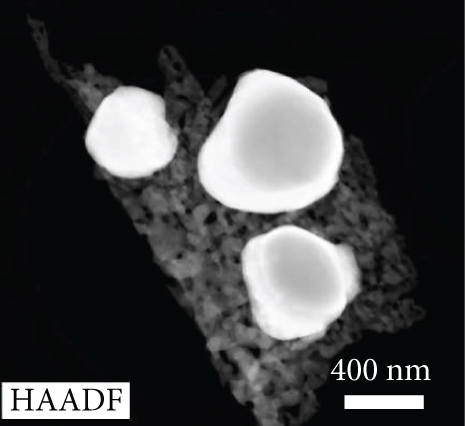

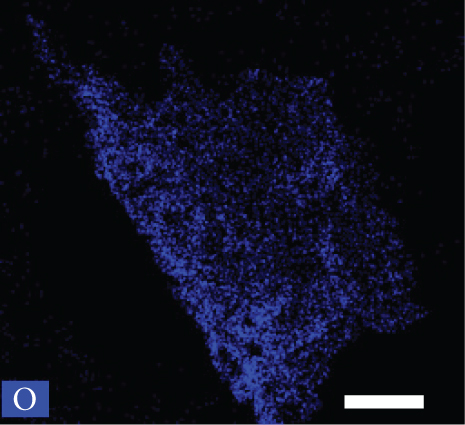
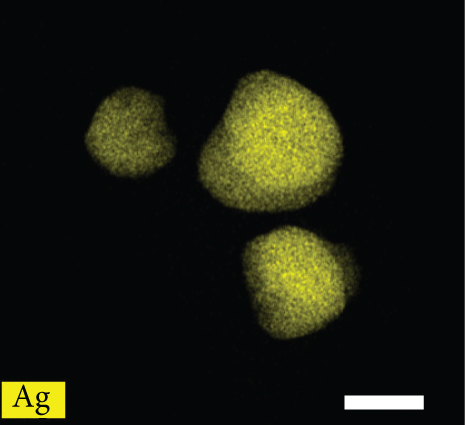


HRTEM image of the 3 at% Ag-functionalized porous ZnO sheets.
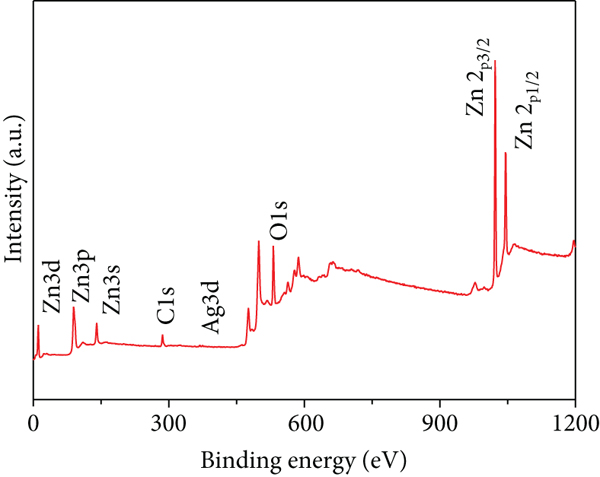
(a) XPS survey of the 3 at% Ag-functionalized porous ZnO sheets and high-resolution XPS core-levels of (b) Zn 2p and (c) Ag 3d.

SM, Figure S4 shows the PL spectrum of 3 at% Ag-functionalized porous ZnO sheets. Based on literature, the PL spectrum of ZnO indicates a band emission in the ultraviolet (UV) region and a broad defect emission in the visible range [37]. The UV emission is owing to the near-band-edge emission of ZnO, which results from the recombination of free excitons through an exciton–exciton collisions [38]. Emissions in the visible region are because of the recombination of different defects [37].
3.2. Gas Sensing Studies
We measured the resistance of the gas sensors at 100, 200, and 300°C and found the optimum operating temperature of 200°C. Figures 6(a)–6(c) show the dynamic resistance curves of (Figure 6(a)) 1, (Figure 6(b)) 3, and (Figure 6(c)) 5 at% Ag-functionalized porous ZnO sheet gas sensors at various concentrations of NO2 gas at 200°C, respectively. The sensors exhibit

Dynamic resistance curves of the (a) 1, (b) 3, and (c) 5 at% Ag-functionalized porous ZnO sheet gas sensor at 200°C to 2, 10, and 20 ppm NO2 gas.



(a) Response, (b) response time, and (c) recovery time of the 1, 3, and 5 at% Ag-functionalized porous ZnO sheet gas sensors to NO2 gas at 200°C.


Response, response time, and recovery time of 1, 3, and 5 at% Ag-functionalized porous ZnO sheets gas sensors to different concentrations of NO2 gas at 200°C.
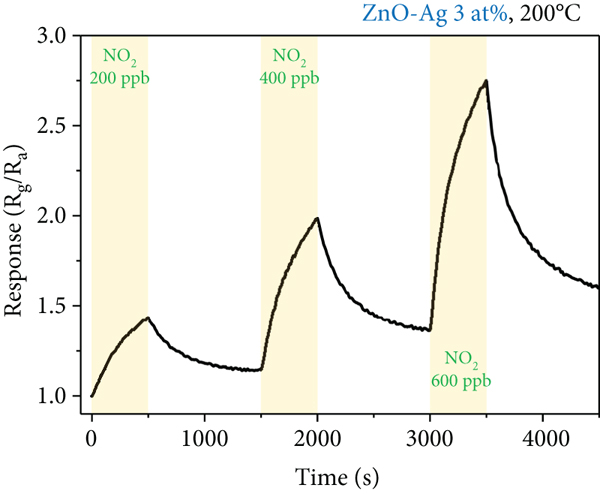
Response curves of the 3 at% Ag-functionalized porous ZnO sheets with 200, 400, and 600 ppb NO2 gas concentration at 200°C.

(a) Dynamic response curves of the 3 at% Ag-functionalized porous ZnO sheet gas sensor to 10 ppm of various gases at 200°C. (b) Selectivity pattern of (a, c). Dynamic resistance curve of the 3 at% Ag-functionalized porous ZnO sheet gas sensor in the presence of 30% RH and 60% RH. (d) Response comparison of 10 ppm NO2 gas sensor in dry and humid atmospheres. (e) Dynamic response curve of the 3 at% Ag-functionalized porous ZnO sheet gas sensor after three months. (f) Comparison between the response of the gas sensor to 10 ppm NO2 in a fresh state and after three months.
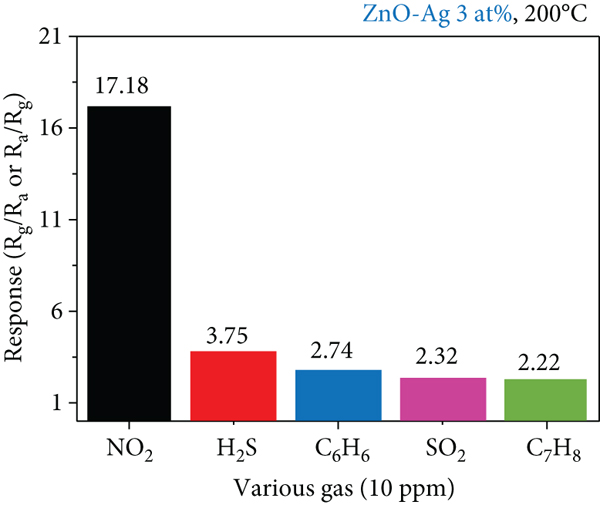




Therefore, in order to investigate the effect of humidity, it is reasonable to compare the difference in response as shown in Figure 9(d). The responses to 10 ppm NO2 gas at 0, 30, and 60% relative humidity at 200°C were 17.18, 15.21, and 14.05, respectively. Even in the humid environment, the sensor can reliably detect low concentrations of NO2 gas. Finally, regarding the performance of gas sensing, even if measured after 3 months, the response value decreases, but there seems to be no problem with the long-term use of gas sensing (Figures 9(e) and 9(f)). Then, the responses to 10 ppm NO2 gas at 200°C in a fresh state and after three months were 17.18 and 4.37, respectively.
3.3. Sensing Mechanisms
The basic sensing mechanism conductometric gas sensors are the modulation of the resistance of the sensing layer in the existence of the target gas [41, 42]. Initially, in air, when gas sensors are exposed to an oxygen environment, oxygen adsorbs electrons from the surface of ZnO because of its high electron affinity and is converted to ionic species as follows [43, 44]:
Due to the abstraction of electrons by the adsorbed oxygen species, an electron depletion layer (EDL) is developed on the ZnO surface, where a higher resistance relative to that of the core parts is expected owing to the lower concentrations of electrons. When NO2 gas is injected into the gas chamber, the surface EDL expands because NO2 has a higher electron affinity (2.28 eV) than oxygen (0.43 eV) and leads to an increase in the resistance of the gas sensor [45]. The relevant reactions are as follows [46]:
Furthermore, in the homojunctions between ZnO sheets, potential barriers are initially formed in air, and upon exposure to NO2 gas, the height of these potential barriers increases due to the further abstraction of electrons, contributing to the sensing signal.
In addition, the effects of Ag on the gas sensing characteristics of ZnO sheets were investigated. Owing to the differences between the work functions of Ag (4.72 eV) and ZnO (4.45 eV) [47], the electrons transfer from ZnO to Ag, resulting in intimate contacts between Ag and ZnO to equate the Fermi levels. Accordingly, a Schottky barrier is formed near the interfaces of ZnO and Ag in air. This increases the resistance of the gas sensor as compared to that of pristine ZnO sheets. Upon exposure to NO2 gas and further extraction of electrons from the surface of ZnO, the width and height of the Schottky barriers increase, leading to higher resistance of the gas sensor in the presence of Ag particles. This effect is an electronic effect or electronic sensitization of Ag. In other words, as the content of Ag increases, the initial resistance of the ZnO sheets may increase because more Schottky barriers are formed as seen in Figure 6. In another effect, similar to Au [48], Ag catalytically decomposes the oxygen molecules into atomic species when they come in contact with the surface of Ag particles. Then, owing to the so-called spill-over effect, the oxygen atoms move to the surface of neighboring ZnO and easily adsorb on this surface [49]. Therefore, more oxygen species adsorb on the surface of ZnO, and their more reactions with NO2 lead to a higher response of the sensor to NO2 gas. In addition, because of the process temperature of 200°C, oxygen species can react with Ag to form Ag2O, but even at this time, electrons are transferred from ZnO to Ag2O to play a role in increasing the resistance of ZnO [50]. Figure 10 schematically shows the sensing mechanism of the 3 at% Ag-functionalized sensor. Table 2 presents a comparison between the NO2 sensing capacities of ZnO-based gas sensors reported in the literature and the present optimized sensor [24, 46, 51–57]. It can be observed that the present sensor has good performance when compared with the performances of other relevant gas sensors mainly because of the catalytic effect of Ag, formation of Ag/ZnO Schottky contacts, and high surface area due to the porous nature of the synthesized sheet, where the gas molecules can diffuse into the deep parts of the sheets.

Schematic of the NO2 sensing mechanism of Ag-functionalized porous ZnO sheets.
Comparison between the NO2 gas sensing properties of ZnO-based gas sensors with the present optimized gas sensor.
4. Conclusions
We proposed the hydrothermal route to prepare hybrid materials of Ag and ZnO. Submicron Ag particles functionalized porous ZnO sheets were formed during the chemical reaction between Zn and Ag precursors in a mixed solution of ethanol and water. From the designed measurements of NO2 gas sensing properties of the sensors fabricated with Ag-functionalized porous ZnO sheets, we found that response and recovery time can be simultaneously improved by the hybridization of Ag particles due to the chemical and electrical sensitization effects even under high-humidity atmosphere. The 3 at% Ag-functionalized porous ZnO sheet sensor represents response of 17.18 and response time of 199 s under 10 ppm NO2 gas at 200°C and shows good selectivity. We believe that researches based on our synthetic approach will provide enhanced sensing properties in any oxide-based gas sensing materials and expand the applications of semiconductor metal oxide gas sensors.
Footnotes
Data Availability
All the data are available from the corresponding authors on reasonable request.
Disclosure
Conflicts of Interest
The authors declare no competing interests.
Authors’ Contributions
Min Young Kim and Jeong Yun Hwang had equal contribution as co-first authors.
Acknowledgments
This study was supported by Samsung Electronics Co., Ltd. (IO201216-08204-01). This research was also supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (2022R1A2C2005210) and the Ministry of Education (2019R1A6A1A11055660). Seung Yong Lee and Changhyun Jin were supported by the Korea Initiative for fostering University of Research and Innovation (KIURI) Program of the National Research Foundation (NRF) funded by the Korea government (MSIT) (NRF-2020M3H1A1077207).
