Abstract
With the rapid development of industrialization, the amount of copper-containing wastewater is increasing, thereby posing a threat to the aquatic ecological environment and human health. Sludge biochar has received extensive concern in recent years due to its advantages of low cost and sustainability for the treatment of heavy-metal-containing wastewater. However, the heavy-metal-adsorption capacity of sludge biochar is limited. This study prepared a sodium pyrophosphate- (Na4P2O7-) modified municipal sludge-based biochar (SP-SBC) and evaluated its adsorption performance for Cu(II). Results showed that SP-SBC had higher yield, ash content, pH, Na and P content, and surface roughness than original sewage sludge biochar (SBC). The Cu(II)-adsorption capacity of SP-SBC was 4.55 times than that of SBC at room temperature. For Cu(II) adsorption by SP-SBC, the kinetics and isotherms conformed to the pseudo-second-order model and the Langmuir–Freundlich model, respectively. The maximum adsorption capacity of SP-SBC was 38.49 mg·g−1 at 35°C. Cu(II) adsorption by SP-SBC primarily involved ion exchange, electrostatic attraction, and precipitation. The desired adsorption performance for Cu(II) in the fixed-bed column experiment indicated that SP-SBC can be reused and had good application potential to treat copper-containing wastewater. Overall, this study provided a desirable sorbent (SP-SBC) for Cu(II) removal, as well as a new simple chemical-modification method for SBC to enhance Cu(II)-adsorption capacity.
1. Introduction
With the rapid development of industrialization, the heavy metals that enter the water environment through human activities inevitably increase [1–3]. Due to the persistence, bioaccumulation, pathogenicity, and carcinogenicity of heavy metals, the water-environment pollution they cause is attracting extensive attention [1, 4]. Copper, as an important heavy metal material, is widely used in the fields of electronics, energy, communications, and machinery production, resulting in a large amount of copper-containing wastewater. The concentration of copper in the wastewater varies from tens to thousands of milligrams per liter [5]. Once the wastewater is directly discharged into natural water bodies without proper treatment, it harms aquatic organisms and leads to a series of health problems to humans, such as nausea, diarrhea, liver, and kidney damage [1, 6]. Thus, the removal of copper from wastewater has become an important issue for heavy-metal pollution control. Several methods are presently used to deal with copper-containing wastewater, primarily including chemical precipitation, bioremediation, membrane separation, and adsorption [7]. Among these methods, adsorption has elicited extensive attention in view of its advantages of convenient operation, considerable efficiency, low cost, and strong anticontamination ability [8, 9]. For adsorption, sorbent development is an important research task [10–14].
Biochar is a porous and carbon-rich solid material obtained by pyrolyzing biomass under anoxic or anaerobic conditions [15, 16]. Due to the high porosity, large surface area, rich surface functional groups, and high pH value of biochar, it receives extensive attention in the field of heavy-metal-containing wastewater treatment [17]. In previous years, a large number of biochar materials such as discarded mushroom-stick biochar [18], corn straw biochar [19], date seed biochar [20], tobacco stem biochar [21], industrial alkali lignin biochar [22], and Ascophyllum nodosum seaweed biochar [23] have been successfully prepared to remove heavy metal from wastewater. These studies show that biochar is an environmentally friendly and cost-effective adsorption material for heavy-metal adsorption. With the deepening of research, some scholars have performed studies on biochar modification, particularly chemical modification, to improve the heavy-metal-adsorption capacity by raw biochar [24–31]. These studies have indicated that the heavy-metal-adsorption capacity of biochar can be significantly improved by an effective modifier. Such a modifier adds adsorption sites and enhances electrostatic attraction, surface complexation, or surface precipitation on biochar.
In China, the growing sewage sludge produced from sewage treatment has become a serious burden on ecology and society [32]. Accordingly, China has the urgent need for environment-friendly methods to realize the effective treatment of sewage sludge. However, the commonly used methods of incineration, sanitary landfilling, and land application [32] have some shortcomings that could lead to secondary pollution. For example, NOx, SO2, and volatile heavy metals produced from incineration can cause serious air pollution [33]. Sanitary landfilling can occupy land. Direct agricultural application may harm living organisms [34]. Many researchers have reported the related studies on converting sewage sludge to biochar for the treatment of heavy-metal-containing wastewater [35–41]. They found that preparing biochar from sewage sludge can realize the harmless and reductive treatments of sewage sludge and obtain a low-cost and sustainable sorbent for the treatment of heavy-metal-containing wastewater. However, the heavy-metal-adsorption capacity of sludge-based biochar is unsatisfactory, which limits its promotion and application to a certain extent [42, 43]. Some modifiers such as Fe3O4 [44], nanostructured CaCO3 [45], trithiocyanuric acid trisodium salt [46], α-Fe2O3 and α-FeOOH [47], K2FeO4 [48], and hydroxyapatite [42] have been successfully applied to prepare modified sewage sludge biochar, and satisfactory heavy-metal-adsorption capacity has been achieved. At present, chemical modification is a noteworthy research direction for improving the adsorption capacity of heavy metals by sewage sludge biochar [45, 49]. In the present study, we used sodium pyrophosphate (Na4P2O7), an additive widely used in the food industry, water industry, and daily chemical industry, as a modifier to prepare Na4P2O7-modified sewage sludge-based biochar (SP-SBC). The desirable Cu(II)-adsorption capacity of SP-SBC was hoped to be achieved through the precipitation between pyrophosphate or phosphate and Cu(II). Although some similar reports exist on sewage sludge biochar modified with chemical modification for Cu(II) adsorption [42, 50, 51], to our knowledge, SP-SBC and its Cu(II)-adsorption performance are reported herein for the first time. Moreover, our one-pot preparation method of SP-SBC was simpler than those of biochars reported by Chen et al. [42], Phoungthong and Suwunwong [50], and Tang et al. [51]. This study could provide an alternative path for modifying sewage sludge biochar, which is beneficial to promote the practical application of sludge biochar.
The aims of this study were as follows: (i) to prepare SP-SBC and explore its properties, (ii) to investigate the adsorption behaviors of SP-SBC for Cu(II), and (iii) to understand the application potential of SP-SBC for Cu(II)-adsorption removal. Herein, SP-SBC was prepared. The properties of SP-SBC and original sewage sludge biochar (SBC) were characterized by the yield; ash content; pH, K, Ca, Na, Mg, and P contents; SEM; and FTIR. Then, the effects of sorbent dosage, initial pH, ionic strength, contact time, and temperature on the Cu(II) adsorption by SP-SBC were determined by batch-adsorption experiments. For Cu(II) adsorption by SP-SBC, the kinetics and isotherms were explored. Lastly, a fixed-bed column experiment was performed.
2. Materials and Methods
2.1. Materials
Sodium pyrophosphate (Na4P2O7; AR, 99%), copper nitrate trihydrate (Cu(NO3)2·3H2O; AR, 99%), sodium nitrate (NaNO3; AR, 99%), silica sand, nitric acid (HNO3; AR, 65–68 wt%), and sodium hydroxide (NaOH, AR, 98%) were all supplied by Kelong Chemical Co., Ltd. (Chengdu, China) without further purification. Ultrapure water (18.25 MΩ) was used to prepare the used solutions. Sewage sludge was acquired from a local municipal wastewater-treatment plant in Chengdu city, China.
2.2. Preparation of Biochar
SP-SBC was prepared with a one-pot method (Figure 1). The specific procedures were as follows. Sewage sludge was ground and sieved to pass a 10-mesh sieve after natural drying. A part of sieved sludge was impregnated with Na4P2O7 (60 g·L−1) at a ratio of 1/20 (mass/volume) for 12 h in a constant-temperature shaker under the conditions of 25°C and 130 rpm. Afterwards, the impregnated sludge was dried at 60°C until the weight was stable. The dried impregnated sludge and the other part of the sieved sludge were loaded into different ceramic crucibles, sealed, and capped. Then, they were pyrolyzed for 2 h at 400°C in a muffle furnace. After grinding the pyrolyzed product of the original sludge and sieving (60 mesh), the acquired particles under the sieve were recorded as SBC. The pyrolyzed product of the impregnated sludge was washed several times with 55–65°C deionized water and dried at 60°C until the weight was stable. The dried product was passed through a sieve (60 mesh) after grinding. The obtained particles under the sieve were recorded as SP-SBC.

Flow chart of SP-SBC preparation.
2.3. Properties of Biochar
The analysis methods for the properties of SBC or SP-SBC were as follows. Equation (1) was used to calculate the yield (
2.4. Batch Experiments
The influences of sorbent dosage, initial solution pH, ionic strength, contact time, and temperature on Cu(II) adsorption by SP-SBC were determined with batch experiments. For all batch experiments, the Cu(II) solution with a preset concentration was prepared by diluting 1000 mg·L−1 of Cu(II) stock solution, which was in turn prepared with Cu(NO3)2·3H2O and ultrapure water (18.25 MΩ). In a typical procedure, a certain amount of SP-SBC was mixed with 50 mL of Cu(II) solution with a designed concentration, followed by adjusting the pH to a predesigned value. Then, the mixture was shaken for a certain time period at a preset temperature and at 130 rpm in a constant-temperature shaker. The specific conditions for each batch experiment are shown in Table 1. Furthermore, SP-SBC was replaced by SBC and subjected to the same experiment processes in the batch experiment of the effect of contact time to compare the difference in adsorption capacity between SP-SBC and SBC.
Specific conditions for each batch experiment.
Once a batch experiment was completed, the mixture was taken out and the suspension was passed through a 0.45 μm microfiltration membrane. In the filtrate, the Cu(II) concentration was tested with a PinAAcle900T-type flame atomic adsorption spectrophotometer (FAAS) (PerkinElmer Instrument Co., Ltd., Akron, USA). Then, Equation (3) was used to calculate the Cu(II)-adsorption capacity (
2.5. Adsorption Kinetics and Isotherms
For Cu(II) adsorption by SP-SBC, the acquired data in the batch experiment of the effect of contact time were simulated with the pseudo-first-order (PFO) model, the pseudo-second-order (PSO) model, and the intraparticle diffusion (IPD) model (Equations (4)–(6)) to analyze the kinetic characteristics [12].
2.6. Fixed-Bed Column Experiment
A fixed-bed column experiment was conducted to understand the application potential of SP-SBC for Cu(II) adsorption. The sketch map of the fixed-bed column device is shown in Figure 2. Four cycles were performed in the fixed-bed column experiment. Each cycle included two processes, i.e., adsorption process and desorption process. For the adsorption process, the Cu(II) solution (
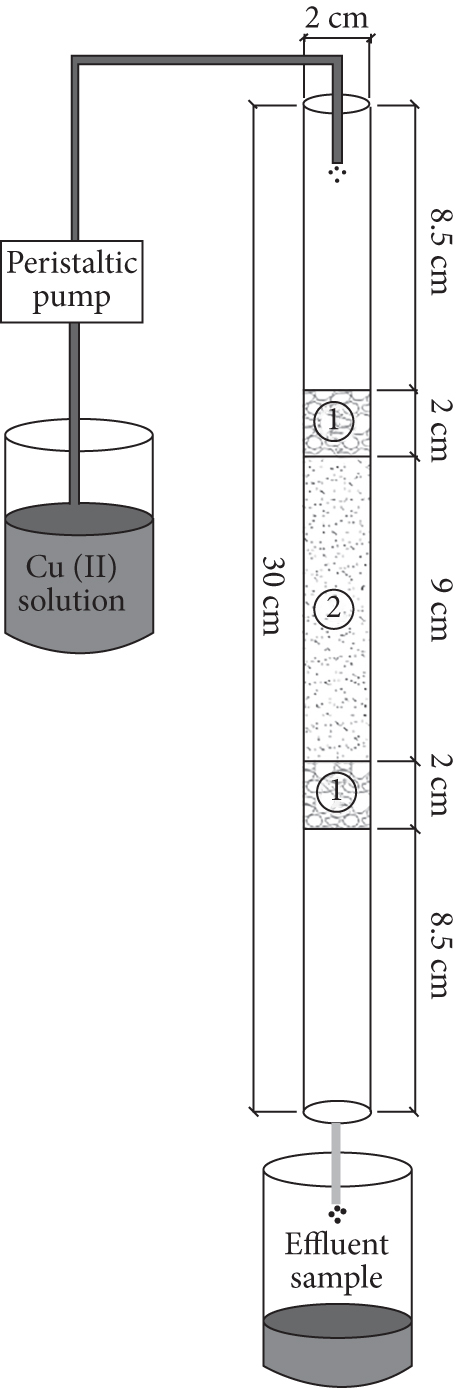
Sketch of fixed-bed column device (①: 2 cm thick quartz sand-packed layer; ②: 9 cm thick SP-SBC-packed layer).
2.7. Statistical Analysis
To ensure the reliability of experimental data, all experiments were repeated three times in this study. Origin 8.0 was used for plotting. SPSS Statistics 23.0 was used to analyze the experimental data.
3. Results and Discussion
3.1. Properties of SBC and SP-SBC
The property parameters of SBC and SP-SBC are shown in Table 2. The yields of SBC and SP-SBC were 60.57% and 68.34%, respectively, which were close to those of other sludge biochars prepared at the same pyrolysis temperature [59, 60]. Compared with those of SBC, the Na and P contents of SP-SBC significantly increased (
Some properties of SBC and SP-SBC.
Figure 3 shows the scanning electron microscopy (SEM) images of SBC and SP-SBC, which reflected the surface structural morphology. The SP-SBC surface (Figure 3(b)) was rougher than that of SBC (Figure 3(a)) and filled with many microparticles, which was caused by the introduction of sodium pyrophosphate. The results of FTIR spectra of SBC and SP-SBC (Figure 4) revealed peaks at around 790 and 1061 cm−1, which were due to the aromatic C-H out-of-plane vibrations [61] and C-O stretching vibration, respectively [15, 62, 63]. The peak at around 1433 cm−1 represented the C=C stretching vibration of aromatic hydrocarbons [64]. The peak at 1629 cm−1 was associated with the –COOH stretching vibration [65]. The observed peak at around 2924 cm−1 represented the C-H stretching vibration [66]. The intense peak at around 3405 cm−1 was due to the -OH stretching vibration [67]. Overall, the peaks of the two biochars were consistent, suggesting that the categories of functional groups did not change before and after modification. However, for SP-SBC, the intensities of these peaks weakened. These results suggested that the amounts of the functional groups on the SP-BC surface (especially for –OH and –COOH) decreased. This finding was consistent with the above result that the alkalinity of SP-SBC was higher than that of SBC.

SEM image of biochar: (a) SBC and (b) SP-SBC.


FTIR spectra of SBC, SP-SBC, and SP-SBC+Cu.
3.2. Adsorption Capacities of SBC and SP-SBC for Cu(II)
The adsorption capacities of SBC and SP-SBC for Cu(II) varied with increased contact time (Figure 5). For SBC, the Cu(II)-adsorption capacity tended to be stable after 10 h. For SP-SBC, the Cu(II)-adsorption capacity tended to reach equilibrium at 12 h. At adsorption equilibrium, SP-SBC and SBC achieved the Cu(II)-adsorption capacities of 20.01 and 4.40 mg·g−1, respectively. Results showed that SP-SBC had a higher Cu(II)-adsorption capacity (4.55 times) than SBC. First, the surface of SP-SBC was rougher than that of SBC (Figure 3), which helped in increasing the contact of Cu(II) with SP-SBC [68]. Second, for SP-SBC, the increased equivalent of sodium was greater than the sum of the decreased equivalents of potassium, calcium, and magnesium (Table 2), which enhanced the ion exchange with Cu(II) [52]. Third, the significant increase in the phosphorus and alkalinity of SP-SBC (Table 2) led to increased copper precipitation [69]. The above three reasons caused the significant improvement in the Cu(II)-adsorption capacity of SP-SBC.

Adsorption capacities of SBC and SP-SBC for Cu(II) at different contact times.
For Cu(II) adsorption by biochar, the complexation and π electron coordination of surface functional groups may be involved [6, 70, 71]. In the Section 3.1, compared with SBC, SP-SBC had fewer surface functional groups. However, the Cu(II)-adsorption capacity of SP-SBC was 4.55 times higher than that of SBC. This finding indicated that the increased Cu(II)-adsorption capacity of SP-SBC was independent of surface functional groups. Thus, the complexion and π electron coordination of surface functional groups may not be the main mechanism for the Cu(II) adsorption by SP-SBC. Additionally, the SP-SBC after Cu(II) adsorption (SP-SBC+Cu) was characterized by FTIR (Figure 4). We found inconspicuous changes in the positions and intensities of peaks between the FTIR spectra of SP-SBC and SP-SBC+Cu. These results suggested that surface functional groups had little effect on Cu(II) adsorption, which strongly supported the above discussion of surface functional group effects for Cu(II) adsorption.
Overall, modifying SBC with sodium pyrophosphate can effectively improve its Cu(II)-adsorption capacity.
3.3. Effect of SP-SBC Dosage
For the Cu(II)-adsorption capacity, the influence of SP-SBC dosage is shown in Figure 6. With increased SP-SBC dosage (from 0.5 g·L−1 to 10 g·L−1), the Cu(II)-adsorption capacity decreased (

Effect of SP-SBC dosage on the adsorption capacity of SP-SBC for Cu(II).
3.4. Effect of Initial pH
To avoid the experimental interference caused by the extra precipitation of Cu(II) at the initial pH of >6 [6, 69], the initial pH was adjusted to 3–6. Figure 7(a) shows the effect of initial pH on the Cu(II) adsorption by SP-SBC. As shown in Figure 7, with increased initial pH from 3 to 5, the Cu(II)-adsorption capacity rapidly increased from 4.52 mg·g−1 to 20.63 mg·g−1 (

(a) Effect of initial pH on the adsorption capacity of SP-SBC for Cu(II), and (b) an illustration of the effect of initial pH on Cu(II) adsorption by SP-SBC.

3.5. Effect of Ionic Strength
Ionic strength usually has a significant effect on adsorption [75]. The effect of ionic strength (Na+) on Cu(II) adsorption by SP-SBC is displayed in Figure 8. With increased ionic strength (0.005–0.5 M), the Cu(II)-adsorption capacity decreased significantly (
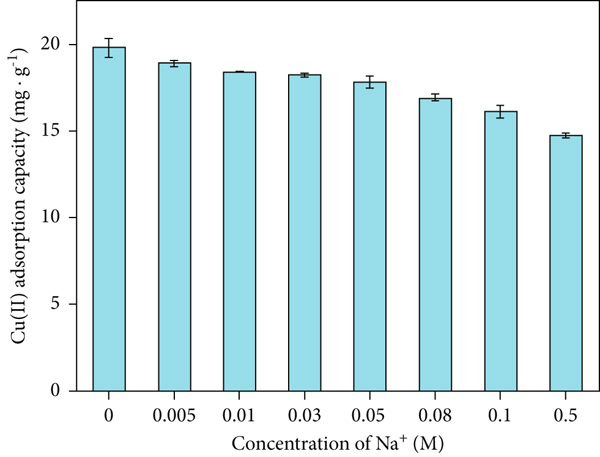
Effect of ionic strength on the adsorption capacity of SP-SBC for Cu(II).
3.6. Effect of Contact Time and Adsorption Kinetics
Figure 9 shows the effect of contact time on Cu(II) adsorption by SP-SBC. With increased contact time, the Cu(II)-adsorption capacity increased (

Effect of contact time on the adsorption capacity of SP-SBC for Cu(II) and fitting results of the PFO and PSO models (a) and the IPD model (b).

Figure 9 exhibits the fitting curves of the PFO, PSO, and IPD models for Cu(II) adsorption by SP-SBC. The fitting curve of the PSO model showed a better approximation to the experimental data than those of the PFO and IPD models. The fitting parameters, i.e.,
Fitting parameters of three kinetic models.
3.7. Effect of Temperature and Adsorption Isotherms
The Cu(II) equilibrium adsorption capacities of SP-SBC (
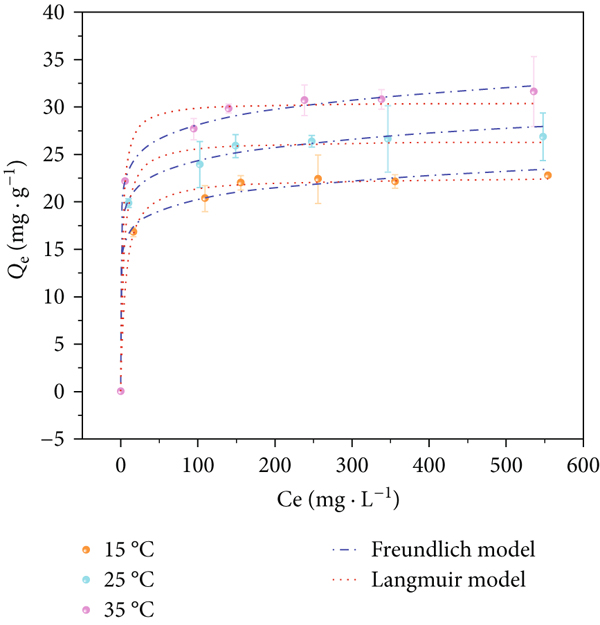
Effect of temperature on the adsorption capacity of SP-SBC for Cu(II), and fitting results of the F and L (a), L-F (b), and D-R (c) models.


To further ascertain the Cu(II)-adsorption behavior of SP-SBC, the experimental data were simulated by the L, F, L-F, and D-R models. Table 4 lists the fitting values of
Fitting parameters of four isotherm models.
Figure 11 shows the

Separation parameters (
Figure 10 shows that the experimental data were matched better by the L-F model than the other three models. Furthermore, the L-F model had the highest
Cu(II)-adsorption capacities of different biochars.
3.8. Fixed-Bed Column Adsorption
Fixed-bed column adsorption can be used to understand the application potential of SP-SBC and offer support for the large-scale treatment of real copper-containing wastewater by SP-SBC [98]. Figure 12 and Table 6 show the breakthrough curves and the corresponding calculated parameters of four adsorption–desorption cycles in the fixed-bed column experiment, respectively. The values of

Breakthrough curves of four adsorption–desorption cycles in the fixed-bed column experiment.
Adsorption parameters acquired by fixed-bed column adsorption in four adsorption–desorption cycles.
4. Conclusions
Na4P2O7-modified biochar (SP-SBC) was successfully prepared by a simple one-pot method. Compared with SBC, the Cu(II)-adsorption capacity of SP-SBC improved 4.55 times. The Cu(II)-adsorption process of SP-SBC can be better described by the pseudo-second-order and Langmuir-Freundlich models. Cu(II) adsorption by SP-SBC involved ion exchange, electrostatic attraction, and precipitation. For Cu(II) adsorption, SP-SBC gained the maximum adsorption capacity of 38.49 mg·g−1 at 35°C, which was higher than those of some reported commercial activated carbons. The fixed-bed column experiment indicated that SP-SBC can be used at least four times and had a good application potential for the treatment of copper-containing wastewater. Overall, SP-SBC can serve as an alternative sorbent to effectively remove Cu(II) from aqueous solutions. For the practical application of SP-SBC, the optimization of adsorption-operation parameters, evaluation of the desorption effect of various desorbents, and design of a feasible industrial-production scheme are required.
Footnotes
Data Availability
All data generated or analyzed during this study are included in this published article.
Conflicts of Interest
The authors have no competing interests to declare that are relevant to the content of this article.
Authors’ Contributions
Liangqian Fan, Xianda Wang, and Jiaxin Miao contributed equally to this work and should be considered as co-first authors.
Acknowledgments
This work was financially supported by the Scientific Research Innovation Team Project of Sichuan Provincial Department of Education (No. 16TD0006).
