Abstract
In the present work, natural diatomite modified with manganese oxide
(MnO2) was prepared via direct redox reaction with
KMnO4 and HCl. The product was characterized by using X-ray
diffraction, Fourier-transform infrared spectroscopy, scanning electron
microscopy, transmission electron microscopy, energy-dispersive X-ray, and
nitrogen adsorption-desorption isotherms. It was found that the nanorod
manganese oxide was highly dispersed onto the diatomite porous matrix. The
specific surface area of the obtained manganese oxide/diatomite
(
1. Introduction
Heavy metals are commonly found in wastewater from the chemical industry, paints, ceramics, glass, mining, and battery manufacturing and severely harm humans and ecosystems [1, 2]. Among them, lead (Pb(II)) has an amphoteric structure and is a metal with a high toxic level among heavy metals. Pb(II) is easily dispersed through the soil and water ecosystem, and by entering the food chain, it causes toxic effects on human health [3]. Due to exposure to Pb(II) for a long period, the kidneys and immune systems, in particular, can be severely destroyed. Therefore, various methods have been utilized to remove heavy metal ions from aqueous solutions, such as oxidation, coagulation and precipitation, membrane filtration, adsorption, ion exchange, and biological treatment [4–9]. Among them, adsorption is the most effective because of its simplicity, low cost, and ability to treat wastewater containing low concentrations of metal ions [10–14].
Diatomite (SiO2·nH2O) is a soft, grey, low-density sedimentary rock. The main component of diatomite is the shell of diatoms consisting primarily of amorphous silica. Diatomite has unique properties, such as high porosity, high adsorption capacity, and high heat resistance [15–19]. These properties suggest that diatomite is a potential adsorbent for the pollutants present in industrial wastewater. Furthermore, natural diatomite is abundant, cheap, and environmentally friendly [18]. In Vietnam, diatomite mineral is often applied in shrimp pond treatment, sound and heat insulation, etc. Recently, the modification of diatomite to increase its applicability has also attracted scientists [20–24]. The modification via hydrothermal process exhibits advantages including nanoparticles with the desired size and shape [25] and formation of a well-crystallized powder [26]. Hence, these techniques have been widely applied to introduce the inorganic compounds with active sites to expand the application of diatomite in adsorption and catalysis [12, 27–30].
Nanoscale manganese oxide (MnO2) particles, known to be a porous material and a mild oxidant, possess a large surface area and excellent chemical stability in acidic media [31]. Manganese oxide has been widely employed as a constituent material in air batteries [32] and a catalyst for oxygen reduction [33]. In particular, manganese oxide exhibits excellent adsorption toward heavy metals in aqueous sources. However, nanosized MnO2 particles suffer from heavy agglomeration, limiting their application [34]. Moreover, using pure MnO2 as an adsorbent is not reasonable regarding economic effectiveness, and it is very difficult to separate the particles from the liquid phase [35]. To deal with these limitations and to promote the treatment efficiency and capabilities for eliminating heavy metals in an aqueous solution with natural diatomite, numerous researchers modified diatomite by coating nanosized MnO2 particles onto its surface. Al-Degs et al. [10] and Khraished et al. [11] modified diatomite with manganese oxide by treating it with manganese chloride and sodium hydroxide. The results demonstrate that its heavy metal adsorption capacity was much improved compared with that of the original diatomite. The specific surface area of diatomite and Mn/diatomite calculated with the simple and rapid methods was 33 and 80 m2 g−1. The adsorption capacity of diatomite is 24.9, 27.6, and 16.1 mg g−1 adsorbates for Pb2+, Cu2+, and Cd2+, respectively, while the corresponding values of Mn/diatomite are 99.0, 57.6, and 27.9 mg g−1. Li et al. [36] reported a two-step procedure for diatomite modification with nano-MnO2, and the material has a maximum adsorption capacity of 56.84 mg g−1 for Pb2+. Du et al. [12] also successfully synthesized MnO2 nanowires on a diatomite substrate. In addition, MnO2-modified diatomite was also used for Zn(II) adsorption [13], dye adsorption [37], catalysts [38], and electrode modifiers [39].
In this paper, a manganese-modified diatomite material (Mn/diatomite) was prepared with the hydrothermal method via the redox reaction of KMnO4 and HCl, where diatomite was a substrate. The material’s physicochemical properties and ability to remove Pb(II) ions from aqueous solutions were investigated.
2. Experimental
2.1. Materials and Chemicals
Raw diatomite was taken from Phu Yen province, Vietnam, washed several times with water, filtered, dried at 100°C, sieved, and stored in closed containers for further tests. KMnO4 (Merck, Germany), Pb(CH3COO)2·3H2O, NaOH, HCl, and KCl (Guangdong, China) are of analytical grade and are used without further purification.
2.2. Preparation of Mn/Diatomite
Diatomite was modified with manganese, according to Wu et al. [40] and Du et al. [12]. Briefly, (i) 0.5 g of diatomite, 5 mmol of KMnO4 (0.79 g), and 80 mL of distilled water were mixed to obtain mixture A; (ii) a certain volume of a 10 M HCl solution was added to mixture A under stirring to obtain mixture B; (iii) the entire mixture B was transferred into a 200 mL Teflon-lined steel autoclave with a quantity of distilled water to fill 80% of the Teflon-flask volume to obtain mixture C; (iv) the Teflon flask was placed in an oven at a specified temperature and for a certain time; and (v) the flask was cooled to ambient temperature, and the solid product was collected, filtered, washed with distilled water, and dried at 60°C for 24 h. The resulting solid is a modified diatomite material and is denoted as Mn/diatomite.
The effects of reaction conditions on the modification, including hydrothermal temperature (80, 120, and 160°C), hydrothermal time (16, 24, and 48 h), and the KMnO4/HCl molar ratio (1 : 1, 1 : 2, 1 : 4, and 1 : 8) were investigated. The details of the experimental conditions used in the diatomite modification are summarized in Table 1.
Experimental conditions were carried out during the preparation of the Mn/diatomite samples.
2.3. Preparation of Nano-MnO2
Nano-manganese oxide was prepared according to a similar procedure in Section 2.2. The 10 M HCl solution was added drop-wise (the molar ratio of KMnO4/HCl was adjusted in the ratio of 1 : 1, 1 : 2, 1 : 4, and 1 : 8). The Teflon flask was heated at 160°C for 16 h, and the yielded nano-manganese oxide was dried at 60°C.
2.4. Characterization
The morphology of the materials was observed by using scanning electron microscopy (SEM, IMS-NKL) with an acceleration voltage of 5 kV and transmission electron microscopy (TEM, EMLab-NIHE) at a voltage of 80 kV. X-ray diffraction (XRD) patterns were recorded on a VNU-D8 Advance Bruker (Germany) powder diffractometer with a Cu Kα radiation source and an angle and time scan step of 0.03° and 0.04 s, respectively. Fourier-transform infrared (FT-IR) spectra were obtained on a Jasco FT/IR-4600 (Japan), with samples being dispersed on KBr pallets and measured within the wavenumber range of 4000-400 cm-1 with a spectral resolution of 1 cm-1. Nitrogen adsorption measurements were conducted with a Micromeritics Tristar 3000 apparatus at the liquid nitrogen temperature (77 K). The textural properties of the material were examined via the specific surface area and porosity after a heat treatment at 250°C in the N2 for 5 h. The elemental composition on the material surface was analyzed with energy-dispersive X-ray (EDX) spectroscopy along with the SEM (JEOL JED-2300) at 20 kV. The Pb(II) concentration was determined with the atomic absorption spectroscopy (AAS) method on a Shimadzu AA-7000 (Singapore).
2.5. Adsorption Experiments
2.5.1. Evaluate the Pb(II) Adsorption Capacity of the Material
A quantity of adsorbent (0.1 g) was placed into an Erlenmeyer flask containing 100 mL of a Pb(II) solution with a prescribed concentration and shaken at ambient temperature for 3 h to achieve adsorption-desorption equilibrium. Then, the solution was filtered to remove the adsorbent, and the Pb(II) ion concentration in the solution was determined.
The adsorption capacity at equilibrium (
2.5.2. Pb(II) Adsorption Kinetics
Mn/diatomite (0.1 g) was placed into a double-necked flask containing 100 mL of the Pb(II) solution with a prescribed concentration under magnetic stirring at a prescribed temperature, under natural pH. At a specified interval, a certain volume of solution was withdrawn and centrifuged to remove the adsorbent, and the Pb(II) concentration in the supernatant was determined with the AAS technique.
The adsorption capacity at time
The pseudo-first-order (Eq. (3)) and pseudo-second-order (Eq. (4)) kinetic models are commonly used to test
experimental data [13, 41–47].
2.5.3. Effect of Solution pH
Mn/diatomite (0.02 g) was placed into an Erlenmeyer flask containing 50 mL of the Pb(II) solution with a specified concentration and pH (the pH value of the solution was adjusted with a 0.1 M HCl or 0.1 M NaOH solution) and shaken at ambient temperature for 3 h to achieve adsorption-desorption equilibrium. Then, the adsorbent was removed, and the Pb(II) concentration was determined.
2.5.4. Equilibrium Studies
In this section, the adsorption was performed at 303 K, and the
initial concentration of Pb(II) varied between 30.8 and
188.3 mg L-1 (
The Freundlich model assumes heterogeneous energy distribution of the active
sites on the adsorbent surface with the interactions within the adsorbate.
The Freundlich isotherm can be expressed in the following equation [13, 37, 41, 42, 45–47].
The Langmuir isotherm is a theoretical model for monolayer adsorption to a
surface containing a finite number of adsorption sites with uniform
adsorption energies without the displacement of the adsorbate in the plane
of the adsorbent surface. The Langmuir isotherm is expressed in the
following equation [10, 11, 37, 41–43, 45–47].
The Sips isotherm has a form similar to the Freundlich isotherm, but it has a
finite limit when the concentration is sufficiently high. The Sips equation
can be expressed in the following equation [48].
Isothermal and kinetic parameters were determined from nonlinear regression
with the Solver tool in Microsoft Excel [49–51] with the
root-mean-square error (RMSE) function described in the following equation
[49, 50].
2.5.5. Thermodynamic Studies
In this section, the initial concentration of Pb(II) was kept constant
(
The free energy change
The adsorption rate constant (
2.5.6. Desorption Experiments
After each experiment, the adsorbent was recycled. The desorption was
performed by mixing the used adsorbent with 0.1 M HCl solution (a
ratio of 0.5 g/50 mL). The mixture was then shaken with a
shaker at ambient temperature (
3. Results and Discussion
3.1. Modification of Diatomite with Manganese Oxide and Pb(II) Adsorption Capacity Assessment
3.1.1. Effect of Hydrothermal Temperature
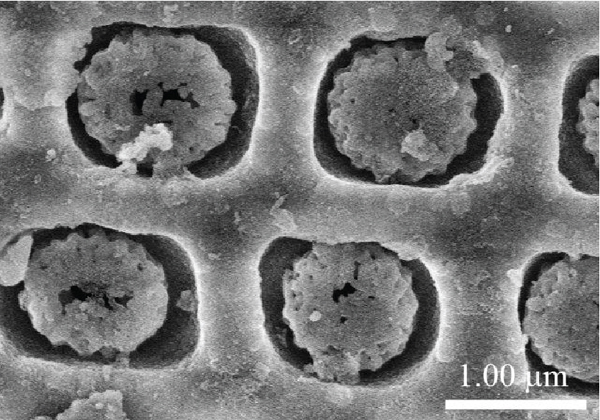
The morphology of natural diatomite and the Mn/diatomite samples prepared at different hydrothermal temperatures is presented in Figure 1. Natural diatomite exists as cylindrical tubes with a 5-7 μm diameter. Its surface is relatively smooth and has pores with a diameter of about 0.5 μm (Figure 1(a)). The morphology of manganese oxide varies from a flower-like structure to a needle-like one with an increase in the hydrothermal temperature. The Mn/diatomite sample prepared at 80°C exhibits flower-like particles attached to the diatomite surface and pores (Figure 1(b)). The morphology of the flower-like particle with some hundred nanometers in diameter consists of thin plates embroiled together. At the hydrothermal temperatures of 120 and 160°C, needle-like manganese oxide appears and is dispersed on the surface and in the pores of natural diatomite (Figures 1(c) and 1(d)).

SEM images of diatomite (a) and the Mn/diatomite samples prepared at different hydrothermal temperatures: (b) 80°C; (c) 120°C; (d) 160°C, and the insets present TEM images (experimental conditions: hydrothermal time of 16 h and molar ratio of KMnO4/HCl 1 : 8).



Natural diatomite has a moderate adsorption capacity
(15.6 mg g
3.1.2. Effect of Molar Ratio of Precursors
The molar ratio between KMnO4 and HCl was chosen at 1 : 1, 1 : 2, and 1 : 4. At the ratio of 1 : 1 (Figures 2(a) and 2(b)), the sample has a large number of circular or rod-shaped clusters attached to the surface of diatomite. At the ratio of 1 : 2 (Figures 2(c) and 2(d)), the circular clusters become larger, and spheres with a flower-like membrane structure appear (like the Mn/diatomite sample prepared at the molar ratio of KMnO4/HCl 1 : 8; Figure 1(b)). Finally, at the ratio of 1 : 4 (Figures 2(e) and 2(f)), besides the spheres with the flower-like membrane structure, film-like structures are formed uniformly on the surface of the diatomite, thus increasing the Pb(II) adsorption efficiency. In this case, the structure of Mn-modified diatomite decides the Pb(II) adsorption efficiency, and the ratio of 1 : 4 provides the highest efficiency (43.1 mg g-1, Table S1). Therefore, this ratio was chosen for subsequent experiments.
SEM images of Mn/diatomite samples prepared at different KMnO4/HCl molar ratios: (a, b) 1 : 1; (c, d) 1 : 2; (e, f) 1 : 4 (experimental conditions: hydrothermal time and temperature are 16 h and 80°C, respectively).





3.1.3. Effect of Hydrothermal Time
In this section, we investigate the hydrothermal time under the conditions found above (Figure 3). The figure shows the SEM images of the 24 and 48 h samples with flower-like spheres and a regular layer of membranous structures. This topography is similar to that of the 16 h samples (Figures 2(e) and 2(f)). Concerning the content of manganese, it seems that a longer hydrothermal time favours the amount of this element in the sample and thus, together with the membranous structures, enhances Pb(II) adsorption (Table S1). From these results, we choose 24 h (54 mg g-1 Pb(II) adsorption efficiency) as the appropriate time for further studies. The 24 h sample was denoted as Mn/diatomite and used for characterization.

SEM images of Mn/diatomite samples prepared at different hydrothermal times: (a, b) 24 h; (c, d) 48 h (experimental conditions: hydrothermal temperature of 80°C and molar ratio of KMnO4/HCl 1 : 4).



The crystalline phase of natural diatomite and the Mn/diatomite was analyzed based on the X-ray data. For natural diatomite, the XRD pattern displays characteristic diffraction peaks of amorphous silica (broad diffraction centered at 20-25°) [52–56]. As for Mn/diatomite samples prepared at different hydrothermal temperatures, no characteristic diffraction peaks of manganese oxide are observed (Figure 4(a)), which is probably because manganese oxide particles are very small and evenly distributed on the surface of diatomite or because diatomite hinders the X-ray diffraction of manganese oxide. This result is consistent with that reported by Li et al. [36].
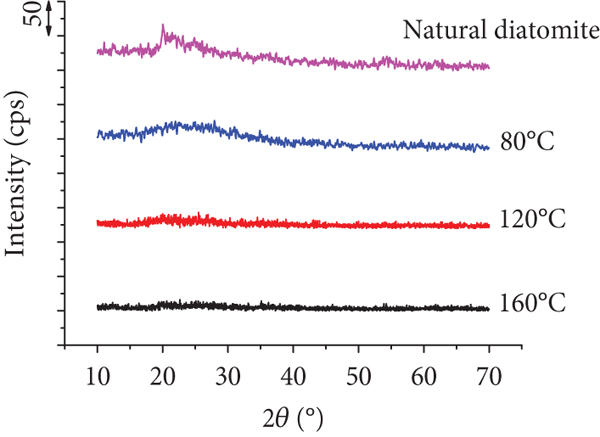
(a) XRD patterns of natural diatomite and the Mn/diatomite samples prepared at different hydrothermal temperatures (experimental conditions: hydrothermal time of 16 h and molar ratio of KMnO4/HCl 1 : 8); (b) XRD pattern of the Mn/diatomite sample (experimental conditions: hydrothermal time and temperature of 24 h and 80°C, respectively, and molar ratio of KMnO4/HCl 1 : 4); (c) nitrogen adsorption-desorption isotherms; (d) pore-size distributions of diatomite and Mn/diatomite (experimental conditions: hydrothermal time and temperature of 24 h and 80°C, respectively, and molar ratio of KMnO4/HCl 1 : 4).


The XRD pattern of Mn/diatomite (Figure 4(b)) shows characteristic diffraction peaks of amorphous silica
at 20–25° [52
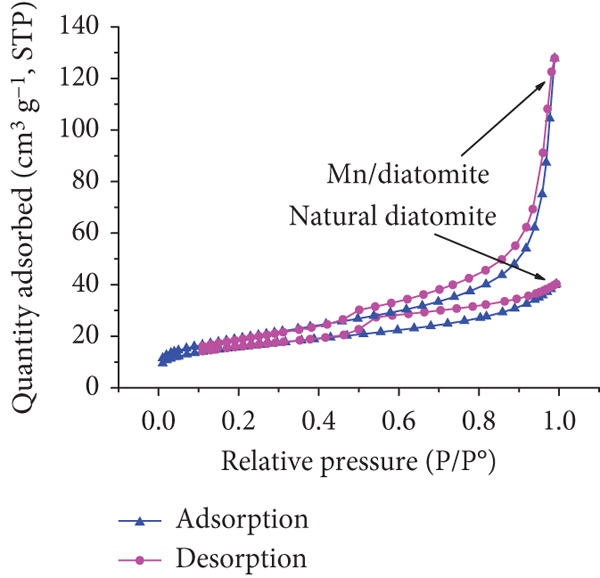
The textural properties of natural diatomite and Mn/diatomite indicate the
type II isotherm and a H3-type hysteresis loop (Figure 4(c)), corresponding to the existence of
macropores of nonuniform size and/or shape [57]. Besides, the Mn/diatomite sample with significant
condensation at high relative pressures (
The pore-size distribution curve (Figure 4(d)) indicates that natural diatomite has pores of about
4.3 nm in diameter, while the Mn/diatomite sample has smaller pores
(3.9 nm). This decrease is caused by the manganese oxide film coated
on the surface. However, the specific surface area of Mn/diatomite
(
3.2. Characteristic Properties and Pb(II) Adsorption Capacity of Nano-MnO2
Nano-MnO2 was prepared from KMnO4 and HCl with four molar ratios of 1 : 1, 1 : 2, 1 : 4, and 1 : 8. All the samples have a bar shape with sides of about 60 to 100 nm (Figure 5), very similar to what was prepared by Wu et al. [40]. Some bars have a hollow tip like a tube. Except for the 1 : 1 KMnO4/HCl sample, which has the bars stacked together, the other three samples have more or less separate bars with an insignificant number of stacks.

SEM images of MnO2 samples synthesized at different molar ratios of KMnO4/HCl: (a) 1 : 1; (b) 1 : 2; (c) 1 : 4; (d) 1 : 8 (experimental conditions: hydrothermal time and temperature are 16 h and 160°C, respectively).



The MnO2 samples have the crystalline structure of an α-MnO2 crystal with characteristic XRD peaks at 28.8, 37.6, 42.1, 49.8, 56.0, and 60.2° (Figure 6), corresponding to the (310), (211), (301), (411), (600), and (521) planes (JCPDS No. 00-024-0735) [40, 58, 59]. The sharp peaks with high intensity of the 1 : 2 to 1 : 8 KMnO4/HCl samples indicate that the formed α-MnO2 has high crystallinity and order. The sample at the KMnO4/HCl 1 : 1 ratio has sharp peaks with low intensity, showing a low-ordered material. This observation is completely consistent with the results observed with SEM in Figure 5(a).
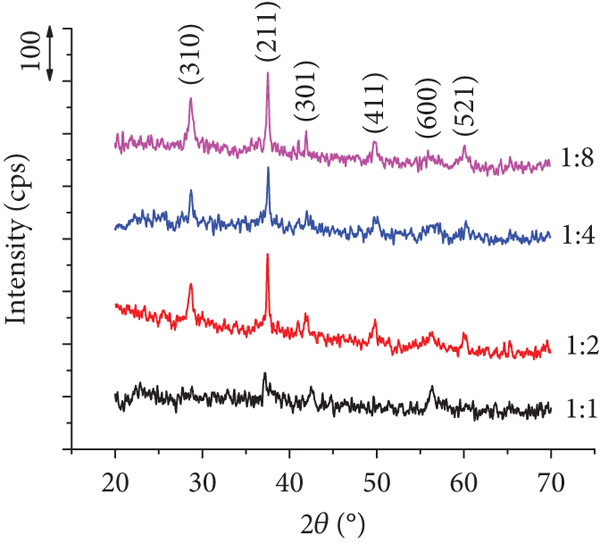
XRD pattern of MnO2 samples synthesized at different KMnO4/HCl molar ratios (experimental conditions: hydrothermal time and temperature are 16 h and 160°C, respectively).
The nano-MnO2 samples were subjected to Pb(II) adsorption in the
aqueous solution. Their adsorption capacities are similar with low values
(16.2
The FT-IR spectra of natural diatomite, Mn/diatomite, and Mn/diatomite after Pb(II) adsorption are relatively similar (Figure 7(a)), with a broad band at 1100 cm-1 and two narrow bands at 797 and 470 cm-1, corresponding to the asymmetric and symmetric stretching vibrations and bending vibration of the Si-O-Si bonds, respectively [13, 19, 55]. The peaks at 3697 and 3621 cm-1 are typical for surface hydroxyl groups in diatomite, with the former corresponding to the isolated hydroxyl group (Si-OH) on the surface of diatomite [55, 60, 61], and the latter due to O-H stretching vibration of the aluminol groups (≡AlOH) [61]. These bands of Mn/diatomite have a lower intensity, indicating the chemical interaction between the surface silanol groups and the oxides [13]. The broad bands at 3441 and 1637 cm-1 are thought to be due to the vibrations of the O-H bonds of adsorbed H2O molecules, including free adsorbed water and hydrogen-bonded water with surface hydroxyl groups. The absorption band at 533 cm-1 is thought to be the stretching vibration of the Fe-O bond [55] or the Me-O bond (with Me being a metal). The bands observed at 431-435 and 419-426 cm-1 in Mn/diatomite and nano-MnO2 (Figure 7(b)) are probably another vibration characteristic for Mn-O binding (not present in the diatomite sample). For the Mn/diatomite sample after Pb(II) adsorption, the intensity of the absorption bands is very low, probably because of the adsorbed Pb(II) layer. In particular, no vibrations are observed at wavenumbers lower than 470 cm-1. This is probably because the Pb(II) ions are bound to the manganese oxide adsorption sites.

FT-IR spectra of natural diatomite, Mn/diatomite, Mn/diatomite after
Pb(II) adsorption, and MnO2: (a) wavenumber region
4000
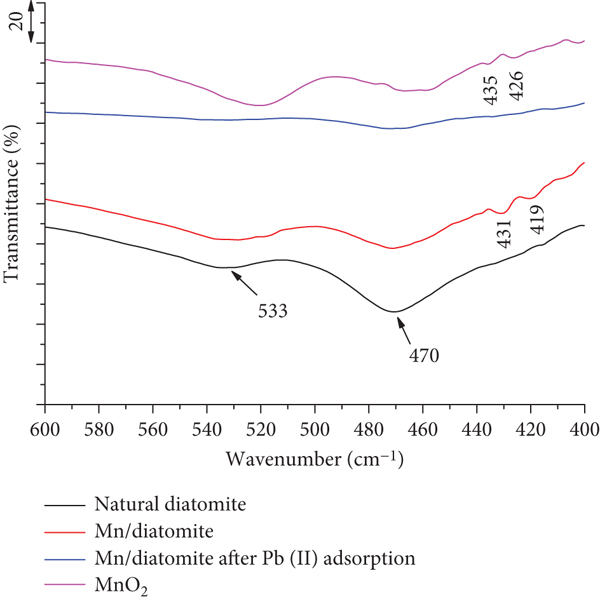
3.3. Adsorption of Pb(II) on Mn/Diatomite
3.3.1. Adsorption Kinetics of Pb(II) on Mn/Diatomite
Chemical kinetics is indispensable in adsorption studies to determine the adsorption rate of the adsorbate at the solid interface. Kinetic models allow the estimation of adsorption rates and lead to suitable rate expressions and suggestion of possible reaction mechanisms.
We can see that the adsorption efficiency of Pb(II) increases with an increase in contact time and initial concentration (Figure 8). This increase probably depends on the concentration difference of Pb(II) ions on the adsorbent surface and in the solution (driving force). When the initial concentration is small, the driving force is also small, leading to a low adsorption efficiency. The adsorption efficiency increases with the driving force, but this increase becomes smaller when the initial concentration is higher. Figure 8 also shows that the Pb(II) adsorption occurs very rapidly during the first 10 min of contact; then, it decreases slowly in the next 60 min and becomes stable after 240 min. Thus, the time required for the adsorption of Pb(II) on Mn/diatomite to reach the adsorption-desorption equilibrium is 60 min.

Adsorption kinetics of Pb(II) onto Mn/diatomite with the different
initial concentrations (
The adsorption kinetics were described by using the pseudo-first/second-order
kinetic models, and the results are presented in Tables 2 and 3. It was found that the equilibrium adsorption capacity values,
Kinetic parameters of Pb(II) adsorption on Mn/diatomite at different initial concentrations of Pb(II) solutions.
Pb(II) adsorption kinetic parameters on Mn/diatomite at different adsorption temperatures.
3.3.2. Effect of Solution pH
Figure 9(a) displays the pH effect on
the Pb adsorption efficiency. Overall, it is clear that the adsorption
efficiency increases consistently with increasing pH. The adsorption
efficiency soars from pH 2 to pH 5 and then increases steadily
up to pH 6. The Pb(II) species can be in the forms of
Pb2+, Pb(OH)+,
Pb3(OH)4
2+, Pb(OH)2, and
Pb(OH)3
-, depending on the solution pH [62]. The cation form Pb2+
mainly exists at
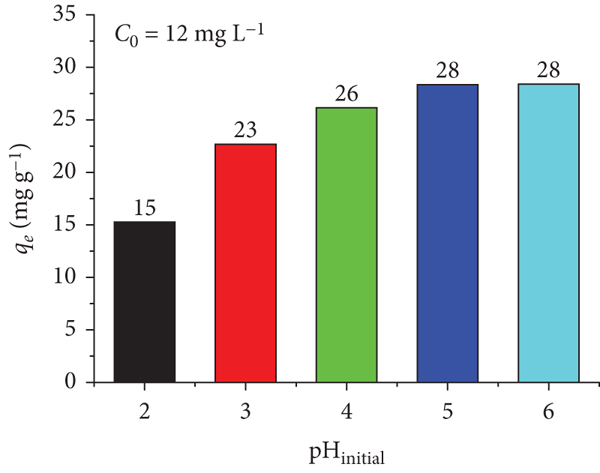
(a) Pb(II) adsorption capacity on Mn/diatomite at different solution pHs; (b) point of zero charge of Mn/diatomite in 0.01 M KCl solution and in 0.05 mM Pb2+ solution determined by the pH drift method [64].

At low pH, Pb(II) reacts with hydroxyl groups (silanol, Mn-OH) on the surface
of Mn/diatomite to form complex adsorption:
At high pH, hydroxyl anions of lead react with hydroxyl groups (silanol,
Mn-OH) on the surface of Mn/diatomite to form complex adsorption:
3.3.3. Adsorption Isotherms
The study of adsorption isotherms is essential in describing the characteristic relationship between the concentration of the adsorbate and the adsorption capacity of the adsorbent, especially when designing an ideal adsorption system in industry [42, 45].
The results of the determination of isotherm parameters of Pb(II) adsorption
on Mn/diatomite, natural diatomite, and nano-MnO2 are presented
in Table 4 (and Figure S6). Based on the RMSE value, it can be assumed that the
Freundlich and Sips models are more suitable to describe the adsorption of
Mn/diatomite and natural diatomite than the Langmuir model. On the contrary,
the Sips and Langmuir models are more suitable than the Freundlich model to
describe the adsorption on nano-MnO2. To evaluate the
compatibility of models with experimental data, numerous authors have used
statistical methods, such as the paired-sample
Parameters of the nonlinear models for the adsorption of Pb(II) in an aqueous solution on Mn/diatomite, natural diatomite, and nano-MnO2.

(a) Relationship between the experimental and the calculated adsorbed
amount for the adsorption of Pb(II) on Mn/diatomite; (b) effect of
temperature and contact time on the adsorption capacity of Pb(II) in
aqueous solution of Mn/diatomite (

The maximum capacity of monolayer adsorption determined with the nonlinear Langmuir model is 81.42 mg g−1. Our value is slightly smaller than that published by Khraished et al. [11] (99.00 mg g−1) but higher than Li et al.’s value (56.843 mg g−1) [36] and other authors’ values (Table 5). The maximum adsorption capacity of natural diatomite and nano-MnO2 is 16.31 and 19.42 mg g−1, respectively. These values are significantly smaller than the maximum adsorption capacity of Mn/diatomite. Because a layer of manganese oxide coats the diatomite, this material has an increased adsorption capacity, indicating that Mn/diatomite is not merely a physical mixture of natural diatomite and nano-MnO2 but is a composite with a high adsorption capacity.
Pb(II) adsorption capacity of different adsorbents.
3.3.4. Adsorption Thermodynamics
We can see that the adsorption capacity depends on the temperature in a similar manner to that of the initial concentration (Figure 10(b)). At lower temperatures (303–323 K), the maximal capacity increases more rapidly than at higher temperatures (323–338 K), and it remains practically the same at 333 and 338 K. This behaviour is also revealed from the slope of the lines during the first 10 min of adsorption. The higher the temperature, the steeper the line is. For all the temperatures, the adsorption-desorption equilibrium is reached after around 60 min.
The plot of

(a) Linear plot of

Thermodynamic parameters of Pb(II) adsorption on Mn/diatomite.
The negative value of
Since the Pb(II) adsorption on Mn/diatomite follows the pseudo-second-order
kinetic model, the adsorption rate constant
3.3.5. Recycling Studies
The regeneration of the spent Mn/diatomite adsorbent was carried out in three
adsorption-desorption cycles. The readsorption experiments were performed on
50 mL of Pb(II) solution with pH and initial concentration of 5.45
and 80.1 mg L-1, respectively, and 0.05 g of
the adsorbent. The mixture was shaken with a shaker at 200 rpm for
3 h at ambient temperature (
Mn/diatomite after each recycle was subjected to FT-IR measurement, and it was found that the characteristic vibration bands of the diatomite modified by manganese oxide remain (Figure S8a). However, the peaks at the wavenumbers less than 470 cm-1 are observed only in the first recycled sample (Figure S8b). This indicates that the chemical bonding between Pb(II) and the manganese oxide adsorption sites reduces desorption. This is also the cause of the reduced adsorption capacity of the recycled samples.
4. Conclusions
The manganese-modified diatomite material was successfully prepared with the
hydrothermal method from KMnO4, HCl, and diatomite. The resulting
Mn/diatomite possesses a high manganese content (20.47% by weight) and a
large specific surface area (
Footnotes
Data Availability
The data used to support the findings of this study are available from the corresponding authors upon request.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Acknowledgments
The authors would like to thank the Center for Scientific Research and Practice, Institute of Applied Technology, Thu Dau Mot University (Binh Duong province, Vietnam), for the support with the experimental equipment and AAS measurements.
