Abstract
Effective and sustainable separation processes for critical metals, especially for the physicochemically similar elements nickel and cobalt in battery recycling, are of great interest in the future. Selective adsorption represents a highly potential process for this purpose. In this publication, a silica adsorbent functionalized with an amino-polycarboxylate derivate (HSU331) was investigated regarding the selective adsorption of Ni(II) in the presence of Co(II) in acidic solution (pH range at equilibrium 1.8–2.3) at elevated temperature. Comparable maximum equilibrium loadings (
1. Introduction
Rising global warming requires an energy transition away from fossil fuel-based power generation to a sustainable generation from wind and solar energy. For the success of energy transition, electricity storage systems for wind power and photovoltaic systems or electric vehicles are mandatory. As a result, state-of-the-art lithium-ion batteries (LIB), whose mixed oxide cathodes consist of lithium (Li) and certain transition metals such as cobalt (Co) and nickel (Ni), will be increasingly demanded in the future. In 2015, 49% of the globally produced refined Co was already used in the rechargeable battery market [1] and the European Union (EU) classified this metal already as critical raw material [2]. According to estimates, the application of Ni in batteries for electric vehicles will grow by 39% annually until 2025. At similar growth rates, this corresponds to a Ni requirement in 2030 of more than 50% of the current global Ni production [3].
From 2030 onwards, the EU thus demands material recovery rates from batteries, i.a. for Ni and Co of 95% each [4]. Klimenko et al. postulate that an improvement of current global Co recycling rates from 30 to 50% is imperative to address an otherwise inevitable Co shortage by the middle of the century [5]. Consequently, accelerated development of effective and sustainable recycling approaches for end-of-life Li batteries becomes an essential technological task.
Today, LIB recycling e.g. based on the Batrec, and Duesenfeld process, respectively, which combine mechanical and thermal treatments with hydrometallurgical methods, mainly acidic leaching in the presence of reductants [6, 7]. After leaching, Ni and Co normally exist as Ni(II) and Co(II) in an acidic aqueous phase, from which they are subsequently recovered by solvent extraction, and precipitation, respectively. Depending on the leaching agent employed, the recovery rates for Co(II) and Ni(II) range from approximately 80 to 99% [6, 8, 9]. Thus, considerable amounts of up to 20% of these valuable metals get lost via wastewater. In view of the required economic and ecological sustainability of industrial processes, further treatment of such sewage effluents are expedient. Another typical industrial process is electroplating [10–12], where Ni(II) and Co(II) are removed from acidic wastewater during plating or at the end of the process [10–13].
Common separation technologies for bivalent metal ions from aqueous solution are based on adsorption methods, membrane techniques, or electrochemical methods [14]. Especially, Ni(II) and Co(II) separation processes from aqueous solution include complexation by chelating agents [10, 15] combined with electrodialysis [16, 17], adsorption [11, 13, 18, 19], and precipitation [20, 21]. Nevertheless, a selective separation of Ni(II) from Co(II) is challenging because of their similar physicochemical properties [7, 20].
Particularly, adsorption represents an advantageous elimination method for heavy metal ions in aqueous phase because of low operational costs, efficient separation at low concentrations, and an adjustable selectivity towards a specific target component [14, 18, 22, 23]. Adsorption selectivity can be achieved by surface modification of an adsorbent matrix, like porous silica gels, with target-directed ligands [24, 25]. These matrix materials offer adjustable porosities according to the selected process conditions, defined structures, and immense possibilities of specific surface functionalization [25, 26].
Generally, amino-polycarboxylic acids in a non-immobilized form show high complex stabilities with Ni(II) and Co(II) [27] and are consequently suitable for functionalization of adsorbents for heavy metal recovery. For instance, Repo et al. [28] showed high adsorption capacities for Ni(II) and Co(II) using silica gels functionalized with amino-polycarboxylic acids. A selectivity towards Ni(II) was suggested when utilizing chitosan [29, 30] as matrix matter. Several other matrices, e.g. acrylonitrile-divinylbenzene copolymer (AN-DVB) [31] or silica polyamine composites [32], were functionalized with amino-polycarboxylic acid derivatives which showed high adsorption capacities for Ni(II) and Co(II).
In this article, we demonstrate the outstanding performance of a mesoporous silica gel functionalized with an amino-polycarboxylate derivate for the selective adsorption of Ni(II) from Co(II) from an acidic model laboratory solution (
Equilibrium batch experiments were performed in order to investigate its adsorption behavior in single- and dual-component systems and the influence of different experimental parameters (temperature, pH, and various molar ratios) on the selective Ni(II) adsorption. Furthermore, a desorption method at 20°C is presented.
This study provides substantial information for the development of a selective adsorption-based separation process as a basis for a sustainable Ni(II) and Co(II) recovery in lithium-ion battery recycling and for a process-integrated Ni(II) and Co(II) recovery step in electroplating.
2. Materials and Methods
2.1. Materials
For the selective adsorption experiments, a silica-based adsorbent, designated as HSU331, with the following characteristics was applied: specific surface area of 510 m2·g-1, pore diameter of 58 Å, pore volume of 0.74 mL·g-1. It consisted of an irregular silica matrix (particle size: 40–63 μm, pore diameter: 60 Å), functionalized with an amino-polycarboxylate ligand. Ultrapure water of type 1 was utilized in all experiments and generated with the water purification system B30 Integrity (AQUAlab, Höhr-Grenzhausen, Germany). Cobalt(II) nitrate hexahydrate (Co(NO3)2 · (H2O)6,
2.2. Elemental Analysis of the Functionalized Silica Gel
The amount of functionalized ligands onto the silica surface (surface coverage
2.3. Metal Ion Quantification
Ni(II) and Co(II) concentrations in the experimental samples were determined by Inductively Coupled Plasma Mass Spectrometry (ICP-MS) with an Agilent 7800 system (Agilent Technologies, Waldbronn, Germany) according to Kriese et al. [36].
2.4. Adsorption Experiments
Discontinuous adsorption experiments with 50 mg HSU331 in differently concentrated aqueous metal samples (
Initial concentrations of metal ions in the prepared single- and dual-component systems (molar ratio Ni(II) : Co(II) of 1 : 1 and 1 : 3) for the different adsorption experiments.
Initially, the pH value of each sample solution was adjusted to 3.5 with 65 wt% nitric acid. For equilibrium achievement, the centrifuge tubes rotated in an overhead rotator (Sunlab, Mannheim, Germany) with 40 rpm for 24 h at 20°C. Subsequently, an analogous series of experiments at
Based on the measured equilibrium concentrations compared to the initial ones, specific dynamic and thermodynamic equilibrium parameters (equilibrium loading, stability constant, enthalpy, entropy, and free enthalpy of formation) were calculated to clarify the underlying adsorption mechanism and to verify the selectivity of HSU331 (expressed by the selectivity coefficient and reaction engineering selectivity).
According to the measured experimental concentrations in μmol · L-1, each corresponding molar equilibrium loading
Usually, the adsorption of bivalent metal ions on functionalized surfaces proceeds according to the following equilibrium reaction:
The enthalpy (
By plotting
Subsequently, the free enthalpy of formation (ΔG0) according to the Gibbs-Helmholtz Equation (Equation (6)) substantiates the endergonic or exergonic character of the adsorption [37]:
Two characteristic parameters, (1) the selectivity coefficient (
2.5. Desorption Experiments
Prior to the desorption investigations, corresponding adsorption experiments (
After equilibration (24 h) and rejection of the acidic supernatants, two washing steps (
The desorption efficiency (η
DE) was calculated appropriate to the following equation:
3. Results and Discussion
3.1. Surface Coverage
Elemental analysis of the adsorbent HSU331 determined a nitrogen mass fraction of 2.2 wt% which corresponds to a surface coverage of HSU331 of 524 μmol(Ligand) · g(Adsorbent)-1.
3.2. Single-Component Adsorption
Investigations of the adsorption performance of HSU331 at

Adsorption isotherms for the single-component systems Ni(II), and Co(II) at
In comparison to our results (see Table 2, column 5), Repo et al. [28] reported Ni(II) and Co(II) maximum loadings (mg(M(II)) · g(Adsorbent)-1) in the same order of magnitude. However, contrary to our results, the difference between the maximum loadings for Ni(II) and Co(II) was ten times smaller than that in our study, indicating a lower selectivity potential at competitive adsorption conditions.
Characteristic Langmuir isotherm parameters (
Taking into account the industrial process conditions (e.g. elevated temperatures) for the Ni(II) and Co(II) separation, the adsorption experiments were also performed at
3.2.1. Adsorption Mechanism
By virtue of (1) the observed discoloration of HSU331 after contact with the metal ion solutions and (2) the significant pH shift in the sample solutions by approximately two units into the strongly acidic range at equilibrium, it can be assumed that the adsorption occurred as an adsorptive ligand complexation.
The complexation of Ni(II) and Co(II) resulted in a blue, and pink coloration of HSU331, respectively. Generally, blue octahedral or tetrahedral high-spin Ni(II) complexes and pink octahedral high-spin Co(II) complexes are known from literature [41, 42] supporting our assumption of adsorptive ligand complexation. Additionally, similar pink colored Co(II)/DTPA octahedral complexes, immobilized onto a melamine-formaldehyde resin were reported in [43].
Due to the chelating character of the non-immobilized amino-polycarboxylic acids, e.g. EDTA and DTPA, complex formation of free hexa- or octadentate ligands with Ni(II) or Co(II) ions usually generates octahedral complexes with high stability constants between
Correlation between proton release per adsorbed metal ion during adsorption and stability constant
In particular, the pH of the equilibrated system is a decisive factor influencing
During the adsorption experiments, this pH shift was evident. When only adsorbent HSU331 is dispersed into an aqueous phase with an initial
3.2.2. Influence of Temperature on Adsorption
The fact that at elevated temperature (
Thermodynamic state variables for the adsorption of Ni(II) and Co(II) onto HSU331 in a single-component system (metal ion : ligand ratio of 1 : 1).
Overall, the more negative free enthalpies (
The positive entropies for Ni(II) and Co(II) adsorption onto HSU331, presumably caused by the release of metal ion-coordinated H2O molecules throughout the complexation [38], signified a generation of chelate complexes onto the HSU331 surface, in which a multidentate ligand occupies at least two coordination sites of a central metal ion [27, 42]. As summarized in Table 3, the release of more than two protons per adsorbed metal ion implies an occupation of the same number of metal coordination sites by the ligands’ dentates and a simultaneous release of at least two water molecules per adsorbed metal ion causing the observed entropy increase in the system (Table 4, column 3).
A significantly higher
3.3. Competitive Adsorption
The resulting isotherms for the competitive adsorption onto HSU331 at

Adsorption isotherms of the equimolar dual-component system Ni(II) : Co(II) at
At higher initial concentrations, the adsorption loading of HSU331 with Ni(II) increases steadily until saturation, while the adsorption loading of Co(II) decreased by around 80% with rising initial concentrations compared to that in the single-component system. This can be attributed to the onset of competitive adsorption from a
Table 5 demonstrates the relations between pHeq, released protons per adsorbed metal ion, and the stability constant
Correlation between proton release per adsorbed metal ion during adsorption, stability constant
From an initial concentration of greater than or equal to 2,800 μmol(Ni(II)/Co(II)) · L-1 (metal ion : ligand ratio ≥ 1), the stability constants of the Ni(II)/HSU331 complexes always exceed those of the Co(II)/HSU331 complexes (Table 5, column 6), that is Ni(II)/HSU331 complexes are preferentially formed in the presence of ligand deficiency. Additionally, the equilibrium reached at pH 2.0 is more favorable for Ni(II) than for Co(II) complexation as demonstrated for the single-component system (Table 3, column 4). Overall, the resulting
In general, the temperature rise of 30°C has no impact on the adsorption performance of HSU331 towards both metal ions (refer to Figure 2). However, the remarkable decrease of the maximum Ni(II) loading at
Additionally, an isotherm for the molar ratio of Ni(II) : Co(II) of 1 : 3 was generated in order to investigate the selectivity of HSU331 towards Ni(II) under non-equimolar conditions (Figure 3). Interestingly, even if Ni(II) represents the minor system component, still relative high maximum adsorption loadings of HSU331 with Ni(II) (0.52 μmol(Ni(II)) · μmol(Ligand)-1) at both temperatures are achievable (Figure 3), which can be explained with the about 1.5 times higher

Adsorption isotherms of HSU331 for the dual-component systems with a molar ratio of Ni(II) : Co(II) 1 : 3 at
In the experiments, where the total metal ion amounts were lower than the amount of ligands (Figure 3, first three data points), Co(II) was still adsorbed with a
The decrease of the Co(II) loadings (Figure 3, data points with a
Calculated selectivity coefficients (
Since the calculated selectivity coefficients showed no trend, the reaction engineering selectivity related to the formed Ni(II)/HSU331 complex (
Selectivity of reaction engineering (
In the equimolar system,
According to the calculated
3.4. Desorption Studies
In order to find suitable desorption conditions for Ni(II) and Co(II), nitric acid with varying concentrations of 0.1 to 2 M (pH 1 to -0.3) was investigated as desorption solution (DS). Desorption efficiencies for both metal ions depending on the acidity of the DS are presented in Figure 4. The application of a DS with a
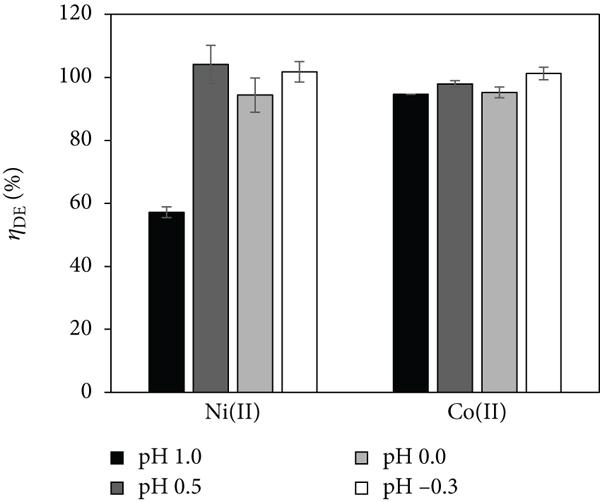
Desorption Efficiency (
Similar findings were reported by Repo et al. [28]. They demonstrated that Ni(II) was still adsorbed at
4. Conclusion
Our experimental results evidence that the investigated adsorbent HSU331 selectively separates Ni(II) from Co(II) at 20°C and 50°C in acidic aqueous solution (
Determined positive entropies of formation indicated that the generation of metal ion chelate complexes during adsorption is very likely. The thereby accompanied decrease of free enthalpies at
Furthermore, based on our results from the desorption experiments, a two-step desorption process can be developed in which the selectivity of the adsorbent HSU331 is maintained. In terms of the recovery of both critical metals, the achieved results demonstrate the outstanding performance of HSU331 for a development of selective and thereby sustainable industrial processes for nickel and cobalt separation.
Footnotes
Data Availability
The experimental data used to support the findings of this study are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Acknowledgments
The present work was partly financed by the Ministry for Environment, Agriculture, Conservation and Consumer Protection of the State of North Rhine-Westphalia (MULNV). This publication has been funded by the Open-Access-Publication-Fund of the Helmut-Schmidt-University/University of the Bundeswehr Hamburg.
