Abstract
This work uses a new nanoadsorbent after chemically synthesis from chicken eggshell wastes for removing amoxicillin (AMX) from aqueous solution. This removal was examined as a time function, initial concentration of AMX, pH, agitation speed, and adsorbent dosage. The study achieved the optimum time for equilibration in (90) min, at
1. Introduction
A very significant natural resource is groundwater to both terrestrial and aquatic ecosystems. One third of the world’s population drinks groundwater as their primary potable source and uses it for domestic, production of food, industry [1], and irrigation of 40% of the food worldwide [2], despite its limited ecological resources consisting of a small portion of the total water distribution [3, 4]. While the standards and needs of living on our natural environment are expanding, rising populations, ongoing urbanization, and industry increase the risk of groundwater contamination [5]. In addition, numerous reports showed dangerous groundwater contamination caused by accidental spills, unsafe disposing industrial chemicals, agricultural practices, mining, etc. Contaminants are dangerous to human health. They also make natural ecosystems vulnerable, and the same is true with the long-term socioeconomic [6] including toxic metals and hydrocarbons. In addition, the organic trace pollutants, pharmaceutical polluting elements, and pesticides face the same, and if managed improperly, groundwater reservation can result in the release of these pollutants into the environment [7, 8]. Antibiotics are one of the most commonly used classes of pharmaceutical compounds, and they are perhaps the most effective class of drugs ever created for enhancing human health. They also prevent and treat animal and plant infections and raise productivity in the livestock industry [9]. Its use is expected to be on the order of 100 000 to 200 000 tons per year throughout the world [10]. Yet, the World Health Organization [11] claimed that only some nations possess national statistics of the antibiotic usage, quantities, and patterns. As a result, the quantity of antibiotics consumed is not clear. Its main sources are human and animal incompletely metabolized antibiotics in the environment. Since humans excrete a large portion of their antibiotics in the form of feces or urine, as well as through disposing leftover hospital antibiotics and wastes, antibiotics are ubiquitous in nature, and their residues have frequently been found in various environmental matrices [12, 13]. AMX was first made available for human use in the early 1970s in the United Kingdom. Since then, it has been discovered in a variety of water bodies due to its widespread use to resist many gram-positive and -negative microorganisms and its status as a first-line antibiotic in numerous nations [14]. Beside the human medicine use, amoxicillin treats and prevents animal infections for increasing growth in a variety of domestic and food animals for broiler chickens, dogs, cats, horses, pigeons, pigs, sheep, goats, preruminating calves, cattle, and fish [15]. Usually, amoxicillin environmental degradation by a number of processes, such as (1) sorption, (2) transformation in biotics, and (3) transformation in abiotics, can influence the destiny and transit of organic compounds in the environment [16]. Over the years, the developments of treatment technologies and approaches for contaminated groundwater have become available allowing flexibility in how clean-up goals can be achieved. Permeable reactive barriers (PRBs) as treatments, depending on sorption process since it removes groundwater contaminants, have been widely examined [17]. It has to be reactive. In addition, it has permeability bigger or equal to the ambient aquifer; thus, the right material choice for the barriers is important [18]. The previous research focused on using wastes, recycled, and by-product materials in PRB technology to enhance their activity in using sustainable (green) concepts, such as activated neem leaves [19], coir pith [20], recycled concrete [21], municipal composts, leaf composts, wood chips, limestone, silica sand [22], and peat and sawdust [23] in treating water contaminated with pollutants. Eggs represent as one of the most ingredient used in variety of products resulting in several daily tons of eggshell which is ranked as the 15th major food industry pollution problem by the EPA, and most of these quantities are accumulated on site or disposed annually as waste in landfills without any pretreatment and become as an organic environmental pollution source [24]. Furthermore, with increasing urgent recently to converting worthwhile waste into useful commodities for sustainable development by recycle, reuse for manufacturing high-value products, managing agricultural wastes is vital and a crucial strategy [25]. Eggshells are one such agrowaste that can be used as a useful source of material in a variety of applications, including the manufacturing of fertilizer and feed as well as other environmentally friendly catalysts in biological, environmental, and agricultural engineering applications [26]. The structural aspect of eggshell comprises of calcium carbonate as the main component with (94-95%) [27], so utilizing eggshells can provide biocompatible calcium (it is environmentally friendly and nontoxic) which lowers not only the impact of pollution but also the associated financial costs and serves as a commercial replacement for calcium carbonate, calcium hydroxide, and calcium oxide [28], and it is also utilized for fertilization, soil conditioning, or animal feed ingredients [29] and manufacturing food products including calcium additives [30]. Layered double hydroxides (LDHs) are unique in structure which is a two-dimensional structure in which the cations are in the centers of edge-sharing octahedral, with vertexes containing OH ions connecting and constituting infinite 2D sheets as [31], with high positive charge densities of layers and high contents of interlayer anionic species which result in strong electrostatic interactions that made the layers hold together in LDHs beside the hydrogen bond [32]. LDHs are active adsorbents, so most scholars have synthesized many LDHs to remove antibiotics from wastewater [33], additionally, and it is prepared relatively simply and economically through a coprecipitation method under laboratory setting in big quantities [34]. The previous studies concentrated on the using of LDHs for antibiotics removal such as in [35] adsorption of AMX that was studied were performed on Cladophora and Spirulina algae biomass immobilized in alginate beads in batch mode with algal biomass doses of 1.25 g/100 ml and 0.5 g/100 ml in respect, achieving 98% of removal, and maximal adsorption capacity was 7.89, 17.4 mg/g for AMX, while Yang et al. [36] studied synthesis as novel adsorbents Mg-Al LDH/cellulose nanocomposite beads (LDH@CB) by coprecipitation procedure utilized as a new adsorbent to remove AMX from the aqueous solution in kinetically and isothermally batch adsorption experiment and fitted with the Freundlich isotherm and pseudo-second-order model, respectively, and AMX LDH@CB qm adsorption capacity equals 138.3 mg g-1. Elhaci et al. [37] also investigated the LDH material synthesis by Mg2+/Al 3+ molar rate of 2 by coprecipitation at pH 9 to remove AMX from aqueous media by pseudo-second-order kinetics with highest adsorption monolayer capacity recorded at
2. Methodology and Materials
2.1. Reagents and Chemicals
Calcium chloride dihydrate (CaCl2.2H2O) was bought to be available and of an analytical grade from AVONCHEM, UK; ferric nitrate (Fe(NO2)3,9H2O) from Scharlau, Spain; and sodium alginate powder (food grade, China) with a
2.2. Synthesizing (Ca/Fe)-LDH-SA Beads
The main concept used in synthesizing (Ca/Fe)-LDH adsorbent as in Figure 1 involves five steps illustrated below:
From chicken eggshells, calcium ions are extracted, which gathering from household wastes, requiring mixing 20 gram of crushed and dried shells (after collecting from the source, rinsing with water several times, air-drying, crushing, drying at 105°C, passing on sieving for gaining (0.6 mm) are shown in Figure 2, then adding 100 mL of solution made of distilled water and 10 mL from 35–38% hydrochloric acid to prepare mixture stirred vigorously at room temperature for 3 h at 200 rpm. To obtain a clear calcium solution, the residual solid shell particles can be separated using filter paper from the calcium ion-rich aqueous solution Preparing aqueous solution reached with Fe+3 by dissolved specific mass of Fe (NO2)3. This dissolving process happened in distilled water (DW), then mixing this iron solution with the calcium ion-rich aqueous solution obtained from first step to obtain solution of (Ca/Fe) with molar In flask, changing the alkalinity of the mixture by adding 1 M NaOH to reach a value of around 12 ensured forming (Ca/Fe)-LDH nanoparticles with continuous agitation over 3 hours and 200 rpm, and then, the slurry that formed should be filter on filter paper, collect it and wash it with distilled water, and dry and grind to obtain the nanoparticles as a powder as shown in Figure 3. Sodium alginate is made through the same procedure of previous studies [38]. This procedure involves dissolving 2 g Na-alginate in 100 mL of DW and comprehensively agitated through magnetic stirring at room ambient over twenty-four hours.

Flowchart of preparing (Ca/Fe)-LDH-SA beads.

Eggshell (a) before and (b) after dried, crushed, and sieved.


(a) Solution of extracted calcium; (b) iron solution; (c) after coprecipitation (Ca/Fe)-LDH; (d) (Ca/Fe)-LDH nanoparticles after filtration and drying.
5.Adding the mass of (Ca/Fe) nanoparticles equal 5 g/100 mL (as optimum value obtained from [39]) that formed in the previous step for the sodium alginate introduced by 10 mL syringe into 0.1 M of CaCl2 (dissolving 1 g of CaCl2 in 100 mL DW) so that polymerization and beads are formed at 4 mm. They should cure in this solution for one hour and followed by washing it two times by DW and keeping it in 5 mM of CaCl2 (0.278 g of CaCl2 is dissolved in 500 mL DW) at 4°C for additional uses (Figure 4).

Beads of (Ca/Fe)-LDH-SA.
2.3. Batch Methodology
It is recommended that batch adsorption experiments be analysed using phenomenological models in order to obtain the appropriate values of operating conditions needed for removing the large percentage of adopted contaminant concentrations that are applicable to other systems and for reducing the number of experiments to fully describe the kinetics of adsorption, which is typically characterized on the basis of empirical models [40]. Some flasks of 250 mL were made, and simulated contaminated water (100 mL) is put in each flask with certain values of concentrations. We repeated experiments by changing variables to obtain important information about the behaviors of antibiotics sorption onto (Ca/Fe)-LDH-immobilized beads and the best parameters to remove it (primary pH, contact time, the amount of beads, and the speed of agitation or the initial concentration of the solution), for getting the isotherms and kinetics of adsorption at the end. Diverse concentration solutions of AMX were obtained from 1000 mg. L-1 stock solution through dilution methods in different intervals of time for three hours. For adsorption kinetics, different initial concentrations of AMX, i.e., from 100 mg.L-1 to 250 mg.L-1 in 100 mL volume, are connected to 0.5 g of adsorbent beads. We examined pH on AMX adsorption in range 2-12 where we adjusted pH of solutions by the NaOH and HCl solution, while the agitation speed did not exceed 250 rpm. While studying the isotherm, we added various masses of (Ca/Fe)-LDH-SA beads of 0.10-1.2 g to some flasks with 100 mL solution for everyone. We interpreted the adsorption isotherm data for various isotherm models. We used filter paper to separate the alginate beads from the water and measured AMX antibiotic concentrations by ultraviolet-visible (UV) spectrophotometer (Model Varian Cary 100 conc., England) at wavelengths of 270 nm. Also, the mean for three readings of the sorption can be gained from each point.
2.4. Data Analysis
At the equilibrium (
Here,
3. Results and Discussion
3.1. Time Influence in Batch Testing
We identified the adsorption equilibrium time through the antibiotic concentrations change with the time. Figure 5 is the change in the AMX uptake efficiencies from contaminated water onto the made adsorbent to contact time not more than three hours at operational settings of initial concentration

Contact time influence on the adsorbent bead behavior utilized to remove AMX from contaminated water.
3.2. AMX Initial Concentration Influence
According to Figure 6, the AMX antibiotic removing efficiencies onto synthesized adsorbent beads decreased intensely from 32.1% for

Effects of initial concentration on the adsorbent bead behavior used to remove AMX from contaminated water.
3.3. Impacts of pH on Removal Efficiency of AMX
The contaminated aqueous pH that contact with the synthesized adsorbent beads is important in eliminating antibiotic as this variable may influence the exterior charges of beads, the AMX ionization degree, and the dissociation of the functional groups [46]. Figure 7(a) is the plots of removing efficiencies of AMX versus various initial pH different from 3 to 12. The process of removing was low (≤14.9% at pH 3.0) because antibiotic competes H+ ions. The rise of the pH towards the neutral condition is followed by a rise in the sorption effectiveness higher than 32.1% for the used antibiotics at pH 7. The antibiotic ionization and hydration decrease at neutral conditions, enhancing removal by the hydrogen bonds and π– π stacking influence. The figure also reveals a decrease in the removing efficiency because solution changed towards the basic form, and the AMX competences are bigger than 18.2%. This efficiency reduction with higher pH could be due to generating OH- attenuating the hydrogen bonding. Figure 7(b) shows the pHpzc of Ca/Fe-LDH at 7 and for beads calculated as around 7.18, and Figure 7(c) reveals a positive charge of the

(a) Initial pH effect with removal of AMX from contaminated water, (b) pH PZC for Ca/Fe LDH, and (c) pH PZC of beads.


3.4. Influence of Agitation Speed
The agitation percentage influenced highly that of adsorption which rises when the speed of agitation rises. According to Figure 8, the increase in agitation speed is essential to enhance AMX removal from polluted water because of diffusion rate of AMX molecules that rise towards the adsorbent bead surface under operational parameters of

Effects of agitation speed on the behavior of adsorbent beads to remove AMX from contaminated water.
3.5. Adsorbent Alginate Bead Mass Impacts
Figure 9 shows the adsorbent alginate beads mass effect on removing AMX efficiencies. Thus, when the dosage changes from 0.1 to 1.2 g, the removing efficiencies increase from the least (9.8%) to more than 90% for the used antibiotic. The bead quantity rise indicates a growth in the empty sites ready to interact with contaminant molecules leading to increase the removing efficiencies of these molecules [48].
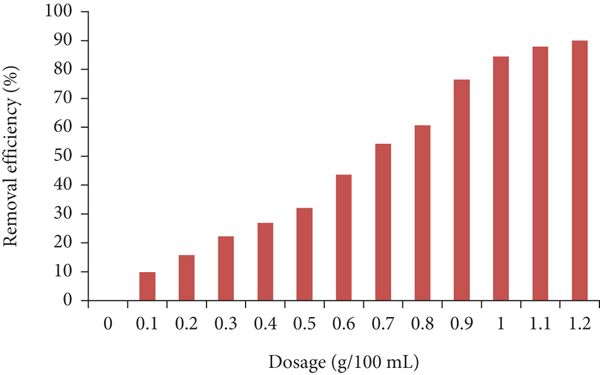
Relation between sorbent dosage and removals of AMX.
3.6. Kinetics of Antibiotic Sorption
Adsorption kinetics from batch study is one of the most important features of such processes that control the sorbent’s capacity for adsorption and rate of adsorption at the interface of solid-liquid phases due to the equilibrium state in batch mode, and the kinetics helps researchers who conducted laboratory scale research to estimate an adsorbent’s performance before applying it on a larger scale [49]. This work measures the kinetic data in the experiment by kinetic models; pseudo-first-order and pseudo-second-order models are shown in Equations (3) and (4), respectively, and intraparticle diffusion model is shown in Equation (5) [50–52]:
Here,
Kinetic models for sorption of AMX antibiotic onto the synthesized nanoadsorbent.

(a) Kinetic (b) and intraparticle diffusion models for interaction of sodium alginate beads with water-contained AMX.

3.7. Isotherms for Equilibrium Sorption Readings
Adsorption is, in general, conducted by equilibrium isotherm relationship correlating between the solute-sorbed quantity on the solid materials (
For heterogeneous sites, Freundlich model can be applied with sorption in a multilayer scheme [59].
Langmuir model suits single-layer adsorption [60]:
where
Isotherm model parameters in the AMX sorption onto adsorbent beads.

Sorption isotherms for interaction of designed sorbent—AMX water contaminated.
4. Conclusions
In this study, egg shell waste was used as crucial source of calcium ions to synthesize a nanobiosorbent from calcium and iron and immobilized into alginate to formed (Ca/Fe)-LDH-SA beads and studied the relevance of using it as a reactive material in permeable barrier utilized for removing AMX from aqueous solution. This works confirmed the AMX antibiotic efficiency in removal from solution through the sorbent. The AMX sorption was explored the time function and primary concentration of AMX, pH of AMX, and sorbent dosage. The equilibrium time for the removal of AMX on adsorbent beads was achieved over one and a half hours and maximum removed at
Footnotes
Data Availability
All the available data are incorporated in the MS.
Conflicts of Interest
No known personal relationships and competing financial interests are influencing this paper.
