Abstract
Water pollution being a potential risk to mankind is treated in several ways which includes chemical treatments. Among them, adsorption took a prominent position for the removal of many hazardous dyes from waste water. Here in this study, an environment-friendly, inexpensive, and broadly available leaves of Brassica oleracea were utilized for adsorption of two carcinogenic dyes, i.e., Congo red and brilliant green. The adsorbent Brassica oleracea leaves were collected, dried, and characterized by FTIR and SEM and then utilized in batch manner for dye removal. Isothermal modeling was carried out on data obtained after experiment which show the best fitting of Langmuir with
1. Introduction
Water pollution is one of the major risks for our planet earth. It is established that about 70-80% of water-related issues in emerging countries are associated with industries, which produce tones of colored effluents in their discharge going directly into water bodies [1]. Almost 40000 pigments and dying agents are already found in industrial wastes [2–4]. About 10000 tons per annum of textile dyes are used in industries and along with printing industry, it makes 10-15% of total dying agents released in water bodies [3, 5, 6]. All these effluents coming out of industries cause severe damage to the ecosystem especially in these water bodies. Whenever this polluted water is consumed by humans for daily shore, it causes life-threatening damage towards human health as it is responsible for high mutation index and results into different kinds of cancers [4, 5].
Previous literature revealed many hazardous effects of water containing dyes like brilliant green and Congo red which includes cancers of different types, skin diseases, gastrointestinal poisoning, and also issues related to respiratory system in humans [6, 7]. Minor concentration of such dyes in water cannot be overlooked because even 0.005 ppm of such dyes in water can interfere in light path which results into inefficient photosynthesis by the aquatic plants and rest of the ecosystem suffers as a result. Hence, the dye removal is a must before throwing all the effluents directly into water bodies [8].
Various in-practice traditional methods that include photochemical, biological, and chemical treatments [9–11] are in practice for the removal of such effluents, such as ozonation [12], coagulation [13], fungal decolorization [14–16], adsorption, flocculation, and electrochemical technique [17, 18]. All these techniques are efficient in one way or the other but are complicated and problematic towards various environmental aspects being expensive and produce several hazardous by-products [19, 20]. So, adsorption techniques emerged as the most suitable method among all these in response to its availability (of adsorbent), efficiency, and cost effectiveness. A number of different adsorbents have been used both as raw as well as chemically modified for the removal of dyes, i.e., brilliant green and Congo red (Figure 1).
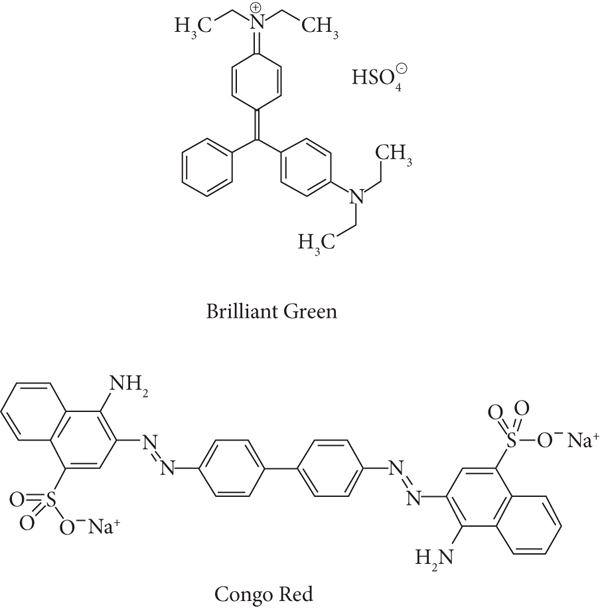
Structural formulas of brilliant green (BG) and Congo red (CR).
Brassica oleracea commonly known as cauliflower is an annual plant which belongs to family Brassicaceae. It grows in sunny and moist climate in temperature range of 21-29°C [21]. In recent years, 25.5 million tons of Brassica oleracea was produced globally. 92% of it is composed of water and carbohydrates along with some essential vitamins. Apart from all this, it contains number of nonnutrient phytochemicals and dietary fibers [22]. From decades, brassica is a part of major cuisines of the world but mostly its stalk and leaves are not used in cooking that goes directly into waste. It is also reported as anticancerous in nature by different researchers [23–25]. Current study Brassica oleracea is picked for adsorption of selected dyes, and its adsorption efficiency is studies under appropriate operation conditions.
2. Experimentation
2.1. Chemicals
All the chemicals as well as apparatus used were of analytical grades which include brilliant green dye (
2.2. Preparation Characterization of Biosorbent
The biosorbent Brassica oleracea is nontoxic and is available easily almost all parts of Punjab, Pakistan. The sample used in this particular study was acquired from the rural areas of Layyah district, Punjab. After collecting the leaves of Brassica oleracea (BO) ,it was thoroughly washed with tap water to remove all the dust trapped. Later on, it was trimmed and washed again with distilled water; then, the material was sundried for 8-10 days. This again was placed in oven at 80°C for 3 days to get rid of all the remaining moisture present in it. This dried sorbent material was then ground and sieved to 60-70 mesh size, and a fine powder was obtain [26]. To eliminate any remaining moisture, it was oven dried once again and then poured into air tight plastic jars to use it further. It is summarized in Figure 2.

Schematic representation of the steps involving preparation of adsorbent. The biosorbent BO was characterized using various useful techniques, i.e., Boehm titration (to determine acidic and basic moieties) pH, and other physioanalytical data Table 1 [27]. FTIR and SEM were also carried to get further understanding of the sorbent behavior.
2.3. Synthetic Waste Water Preparation
1000 ppm solutions of both brilliant green (BG) and Congo red (CR) dyes were prepared by adding 1 g per 1000 mL of distilled water in a measuring flask. All other solutions of dyes for further use were prepared by diluting this stock solution of 1000 ppm. For optimization of the parameters to determine the adsorption capacity of BO against dyes, 25 mL of each dye was taken in 100 mL flasks, and operational conditions were finalized.
2.4. Optimization of Operational Conditions
Various operational conditions were optimized to get better results in a batch adsorption technique. These operational parameters include the following: adsorbent dose (range 0.2-2 g with variation 0.2 g), pH (range 1-10), temperature (range 20-70°C), contact time (range 5-60 min), and agitation rate (range 40-240 rpm difference 40 rpm). To check the reliability of the adsorption results, isothermal and kinetic modeling was done for both the dyes. Following formulas were utilized to determine the % adsorption as well as amount of dye adsorbed for each dye.
Here,
3. Results and Discussions
3.1. Characterization of Adsorbent
FTIR spectrum of Brassica oleracea leaves presented here shows a number of major functional groups present that it is responsible for the binding of dye onto the adsorbent (Figure 3). Here, the band at 3855 cm-1 and 3738 cm-1 indicates the presence of OH group along with carboxylic moieties at 3297 cm-1 while the bands at 2917 cm-1 and 2849 cm-1 show the presence of C–H functional moieties. The bands present at 1653 cm-1 and 1510 cm-1 indicate N-H stretching. Graph also shows a band at 1420 cm-1 which shows carboxylic bending frequency. Figure 3 presents the FTIR spectra of adsorbent BO before and after dye loading, BO, BO-CR, and BO-BG, respectively. Here, the shift in the region of 3855 and 3738 cm-1 to 3881 cm-1 (for CR) and 3898 cm-1 (for BG) depicts the involvement of OH bond because of hydrogen bonding between the molecules of adsorbent and dyes. Also, a clear change in wavenumber at carboxylic region from 3297 to 3281 cm-1 (Figure 3(b)) and 3288 cm-1 (Figure 3(c)) advocates the involvement of OH from carboxylic moiety. N-H stretching region shifts to 1639 and 1535 cm-1 in BO-CR and to 1636 and 1540 cm-1 in BO-BG spectrum, respectively. A hypsochromic shift in COOH bending peaks was also observed for CR 1374 cm-1 and for BG 1399 cm-1. These shifts in major functional moieties accompanied by oxygen containing groups advocate a clear interaction between adsorbent and dye molecules, as the involvement of such functional groups promotes adsorption of dyes on adsorbents [28].

FTIR spectra of (a) Brassica oleracea (BO), (b) Congo red loaded Brassica oleracea (BO-CR), and (c) brilliant green loaded Brassica oleracea (BO-BG).
The data given in Table 1: the presence of small amount of moisture, volatile matter, and ash content indicates little particle density that favors adsorption over this particular biosorbent [29]. Porosity, i.e., number of pores per unit area, depicts good adsorption capacity. It is described that in case of some cationic dye adsorption occurs spontaneously due to presence of electrostatic interaction along with hydrogen bonding between the dye (cationic) and the adsorbent while for anionic dye adsorption hydrogen bonding play vital role [30].
Physioanalytical data of Brassica oleracea (BO).
SEM image of the adsorbent BO shown in Figure 4 represents the presence of rough and porous surface which always play effectively towards the adsorption of dye on to the adsorbent surface. More porous adsorbent surface have large physiosorption tendency to adsorb more dye molecules [31].

SEM image of BO.
3.2. Operational Condition Optimization
pHpzc (point of zero charge) was measured for Brassica oleracea (BO) which turned out to be 8 that provides a strong basic medium for the adsorption of dyes. pHpzc actually helps in determining the sensitivity towards linear range of pH. It also reveals the surface-active centers and adsorption capacity of that adsorbent surface [30]. Literature has widely reported about the pHpzc of agriwaste by a number of researchers. Cationic dyes get more adsorption if

Adsorption parameter for Congo red (CR) ( % age adsorption y- axis on the right hand side) and brilliant green (BG) ( % age adsorption y-axis on the left hand side) adsorption on Brassica oleresea (BO): (a) pHpzc, (b) contact time, (c) adsorbent dose, (d) pH influence, (e) agitation rate, and (f) temperature change.




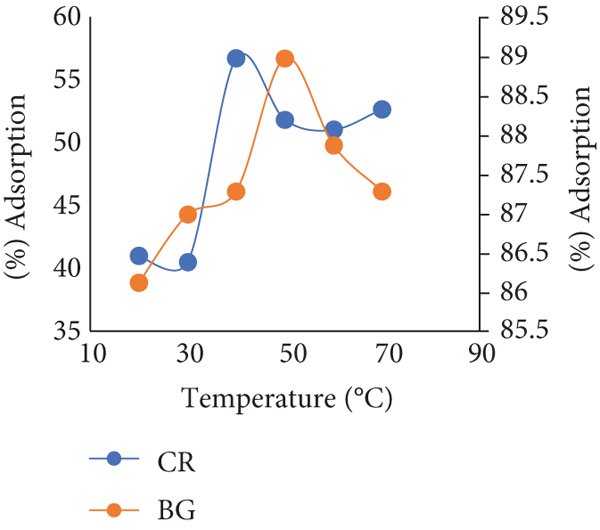
Optimum time of contact of BO for both the cases, i.e., CR and BG shown in Figure 5(b) which depicts optimum time for CR and BG is 30 and 40 minutes, respectively. Both the trends present a gradual increase in % age adsorption until their optimums were reached, and a downward trend started over there. Optimum adsorbent dose of BO for CR and BG was turned out to be 1 and 1.4 g, correspondingly. Figure 5(c) shows an increasing trend initially, and then the values of % age adsorptions decreased for both of the dyes. Values obtained from the procedure for optimum pH of Brassica oleracea present 1 and 7 pH for CR and BG, respectively (Figure 5(d)). Literature shows that Congo red (CR) is more likely to be adsorbed at acidic pH because of its anionic nature and interacts with adsorbent surface with electrostatic interaction [34]. This electrostatic interaction between the two decreases as the pH of the system gets more basic. It occurs because of the increase in negatively charged particles in the solution and attraction converts into electrostatic repulsion [35, 36]. On the other hand, BG acts as cationic dye and as literature states that cationic dyes show spontaneous adsorption because of growing number of positively charged particles. Hence, there occurs strong hydrogen bonding along with electrostatic attraction [37, 38].
Agitation speed of BO for the removal of CR and BG was found to be 120 rpm and 100 rpm, respectively. Shaking speed of solution-containing adsorbent is related to the interaction of molecules between dye, and speeds higher than mentioned above were not suitable for this particular case as there occurs a clear decline in % age adsorption of dyes (Figure 5(e)).
Temperature optimization was also done for BO for both CR and BG dyes that turned out to be 40°C and 50°C. The higher temperature of the solution higher will be the diffusion rate of dye at the adsorbent surface that results into increased adsorption of the dye but as temperature increased over the optimum, a clear decline in the % age adsorption was observed. This decline is related to very high movements of molecules that reverses the adsorption process [10] indicated in Figure 5(f).
3.3. Adsorption Isothermal Studies
The relationship established between the adsorbate and adsorbent is determined by isothermal modeling. It also assessed the authenticity and mechanism of the experiment performed [39]. Following isothermal models were applied to get all this information, i.e., Langmuir, Freundlich, Temkin, and the Dubinin–Radushkevich isotherms.
3.3.1. Langmuir Isotherm
It conveys about the relation between equilibrium concentration and the amount of adsorbate per unit weight of sorbent. Langmuir isotherm established on the idea that adsorption occurs as monolayer on homogenous surface of adsorbent [40]. Langmuir isothermal model (linear) given in Eq. (2) is as follows:
where
where

Adsorption isotherms. A comparative plot of CR and BG for adsorption on Brassica oleracea BO. (a) Langmuir isotherm, (b) Freundlich isotherm, (c) Temkin isotherm, and (d) Dubini-Radushkevich isotherm.
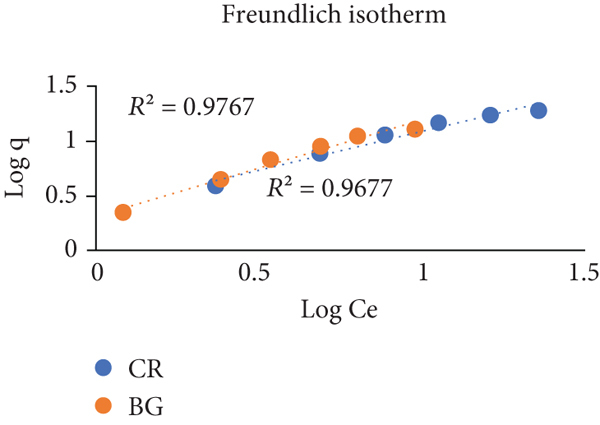

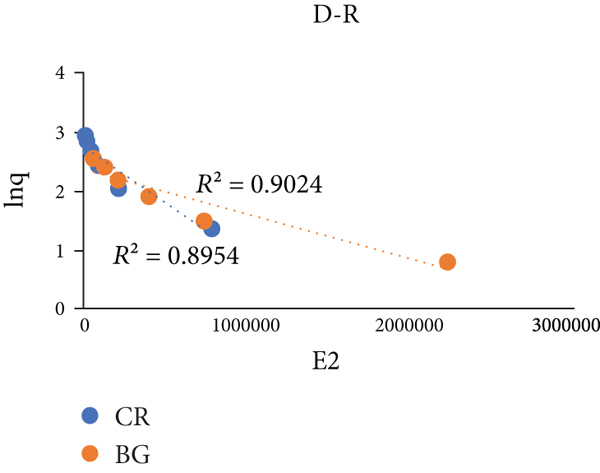
A summary of adsorption isothermal data of Congo red (CR) and brilliant green (BG) dyes adsorbed on Brassica oleracea (BO).
Previously announced comparative adsorption capacity (
3.3.2. Freundlich Isotherm
It is employed to determine multilayer, on-ideal heterogeneous adsorption pattern. This isotherm supports the idea of reversible adsorption on a rough adsorbent surface [43]. Equation Eq. (4) shows the linear for of Freundlich isothermal model
where
3.3.3. Temkin Isotherm
It validates the relative decrease of sorption energy. It says heat of adsorption (
where
3.3.4. Dubinin–Radushkevich Isothermal Model
It is considered to be relatively broader than Langmuir and Freundlich isotherms. It contributes in determining adsorption energy and distinctive absorbency mechanism [50]. The equation given illustrates the biosorption of gases on adsorbents (especially microporous). This model supports the multilayer adsorption supported by Vander Waal’s forces [51]. Linear form
All the adsorption isotherms applied, Langmuir, Freundlich, Temkin, and Dubinin-Radushkevish, and regression factor
3.4. Kinetic Studies
Biosorption rate of reaction was studied by applying the standard kinetic models which include Elovich, Lagergren pseudofirst order model, and Ho’s pseudosecond order model. To get the best fittin,g among them, percent relative deviation was measured by eq. (9):
where
3.4.1. Elovich Model
It favors multiple coating of dye as it is based on the idea of exponential increase in adsorption, powered by the rise of sorption locations on adsorbent [70]. This model was first applied in favor of chemisorption process of gases [65]. Linear equation of the Elovich model is shown in eq. (10) [71]:
where

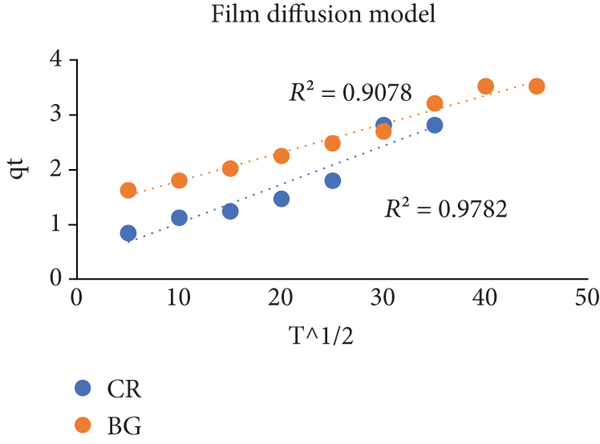
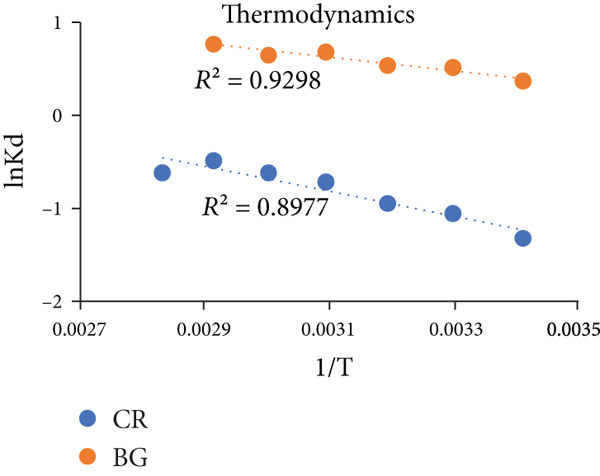
Comparative kinetic and thermodynamic studies plots for CR and BG dye removal by BO. (a) Elovich plot, (b) pseudofirst order, (c) pseudosecond order, (d) intraparticle diffusion model, (e) film diffusion model, and (f) thermodynamic parameters of CR and BG dyes.



3.4.2. Pseudofirst Order Model
It is also known as the Lagergren model which is recognized on the postulate that adsorption is comparable to the number of attaching sites for the dye on the adsorbent surfaces [72].
where
3.4.3. Pseudosecond Order Model
Ho’s model is based on the statement that reaction rate of adsorption process is related to concentration of dye and square of attaching sites for dye on adsorbent surface [73, 74]. Following equation presents linear form of this model:
where
Here, the lesser difference between
A brief summary of kinetic parameters applied on adsorption of dyes on BO.
3.5. Adsorption Mechanism
To determine the mechanism followed by adsorption process, two different models were applied, i.e., intraparticle diffusion model and film diffusion model [76, 77].
Intraparticle diffusion or Weber Morris model is presented as
The plot (
Boyd’s model is built on partially attained equilibrium related to variable time as given below [78],
where
3.6. Thermodynamic Studies
The variation in temperature of the adsorption system visibly affects the kinetic energy of the dyes, CR and BG. This factor speeds up rate of diffusion as the interaction of dye molecules occurs with porous and spongy surface. Certain operational parameters of thermodynamics, i.e., Gibbs free energy
Thermodynamic parameters of dyes removal by
where
The value of
Here, the value of
4. Conclusion
Brassica oleracea is an abundantly available agriwaste material that is effectively utilized to eradicate two potentially damaging dyes: Congo red (anionic) and brilliant green (cationic) from waste water by applying batch sorption. Surface characterization and morphological studies by FTIR and SEM analysis provided that active uptake or removal of both the dye CR and BG is carried out by electrostatic interface by carboxylic as well as hydroxyl moieties present in adsorbent. Also, the interspaces present on sorbent surface expedited the uptake of dye molecules in the form of ions traveling towards its surface. These ions (dye) followed intraparticle as well as film diffusion mechanism to get attached on the surface of adsorbent (BO). Adsorption isotherms carried out also exhibit best fitting of the Langmuir isotherm model on the basis of

Summarizing whole work in one frame.
Footnotes
Abbreviations
Data Availability
All data related to this work is presented in Results along with references.
Conflicts of Interest
The authors declare that they have no conflicts of interest regarding the publications of this paper.
Acknowledgments
The authors are thankful to the home institute for providing facilities for this work.
