Abstract
Predicting the adsorption performance to remove organic pollutants from wastewater is an essential environmental-related topic, requiring knowledge of various statistical tools and artificial intelligence techniques. Hence, this study is the first to develop a quadratic regression model and artificial neural network (ANN) for predicting biochemical oxygen demand (BOD) removal under different adsorption conditions. Nanozero-valent iron encapsulated into cellulose acetate (CA/nZVI) was synthesized, characterized by XRD, SEM, and EDS, and used as an efficient adsorbent for BOD reduction. Results indicated that the medium pH and adsorption time should be adjusted around 7 and 30 min, respectively, to maintain the highest BOD removal efficiency of 96.4% at initial BOD
1. Introduction
Recently, adsorption has been employed in several types of research as an efficient and reliable process for wastewater treatment [1–3]. The adsorption systems neither consume a lot of electricity nor generate large amounts of sludge [4, 5]. Moreover, the adsorbent material could be appropriately synthesized to provide effective adsorption sites to capture the pollutants from wastewater [6, 7]. However, the adsorption process is highly influenced by several operational factors such as time, pH, and mixing speed [8]. The correlation between these environmental factors and pollutant removal efficiency could be described by nonlinear and complex modeling methods [9]. Hence, more studies are required to investigate the applicability of various statistical tools and artificial intelligence techniques for predicting adsorption performance.
Artificial neural network (ANN) models have been used in recently published studies as a proper method to describe the adsorption performance in relation to operational conditions [10–12]. The architecture of ANN is composed of multiple processing elements (or units) arranged in layers. These units, known as neurons, are highly interconnected and work in parallel to solve complex problems and get relevant relationships among the input attributes. For instance, Mahmoud et al. [13] found that an ANN model could simulate and predict phosphate removal in adsorption experimentation, showing a predictive accuracy of
Several researchers have also used regression models and a combination of statistical tools to predict the treatment performance under different operational factors. For example, Fawzy et al. [15] used a quadratic regression model to predict the Ni(II) removal efficiency via adsorption onto plant biomass. The quadratic equation described the correlation between Ni(II) and several inputs (e.g., pH, biomass dosage, and adsorption time) with high predictive accuracy of
Given the aforementioned aspects, the application of the computational approaches to describe the adsorption process is an essential point of research. However, further investigations are required to verify the implementation of ANN and quadratic models to predict organic matter removal. This objective would offer a feasible and sustainable approach to domestic wastewater treatment.
Hence, this research focused on predicting and optimizing the BOD removal performance in adsorption experimentation by a computational-based approach (quadratic regression and ANN models). In particular, the study objectives are fourfold: (1) characterization of adsorbent material synthesized by the entrapment of nanozero-valent iron into cellulose acetate polymer (CA-nZVI), i.e., this material has been widely used in the adsorption system due to its proper mechanical strength, thermal stability, and accessibility; (2) use of the CA-nZVI adsorbent to reduce organic matter from wastewater, expressed by BOD, i.e., BOD is considered the standard criterion for assessing the organic pollution of domestic wastewater; (3) describe the influence of various adsorption factors, i.e., pH, adsorbent dosage, time, mixing rate, and initial BOD concentration (
2. Materials and Methods
2.1. Preparation of Adsorbent Material
For preparing an iron solution, around 0.0037 M of ferric chloride hexahydrate (FeCl3·6H2O; 98.5% pure, Arabic lab.) was dissolved in 60 mL of a mixture of 4 (ethanol; C2H6O, 95% pure, World Co.) : 1 (deionized water). In parallel, 0.7564 g of reducing sodium borohydride (NaBH4; 99% pure, Win lab.) was dissolved in 200 mL of deionized water to prepare the NaBH4 solution. Further, the reducing NaBH4 solution was placed in a burette and added drop by drop into the prepared iron solution. Black iron nanoparticles precipitate as a result of the direct reaction (Equation ((1)). The iron nanoparticles, known as nZVI, were then washed with distilled water and dried at 75°C for 5 h:
Further, the prepared nZVI was capsulated into cellulose acetate (CA; 99%, Oxford) polymer, using the phase inversion approach [17]. Briefly, at room temperature, 4 g of CA was dissolved in 25 mL dimethylformamide (DMF; 99.99%, Fisher Chemical) solution and then mixed at 300 rpm until complete dissolution (within approximately 60 min). About 0.4 g of nZVI was mixed into the dissolved CA solution for 10 min before being cast into a gelation bath. The preparation of the gelation bath included 2 L of nonsolvent distilled water, 2 wt% DMF, and 0.2 wt% sodium lauryl sulphate (SLS). The prepared CA/nZVI beads (around 3–4 mm in diameter) were collected and washed with distilled water and then used for the adsorption experimentation.
2.2. Preparation of BOD Containing Solution (Adsorbate)
Raw wastewater samples were collected from a sewage treatment plant located in New Cairo, Egypt. The samples were analyzed for BOD and subjected to different dilution regimes with ultrapure water. Working stock solutions with BOD concentrations of about 100, 200, 300, 400, and 500 mg/L were prepared and used for the individual experiments.
2.3. Batch Studies on Adsorption
Batch experiments were conducted to determine the effects of adsorption factors on the BOD removal efficiency. For this objective, a one-factor-at-a-time approach was used to prepare the batch assays statistically (Table 1). The factors (pH, CA/nZVI dosage, time, stirring rate, and
Operating conditions of batch adsorption experiments for BOD removal.
2.4. Analytical Analysis
The concentrations of BOD in the aqueous solutions were determined using the procedures of
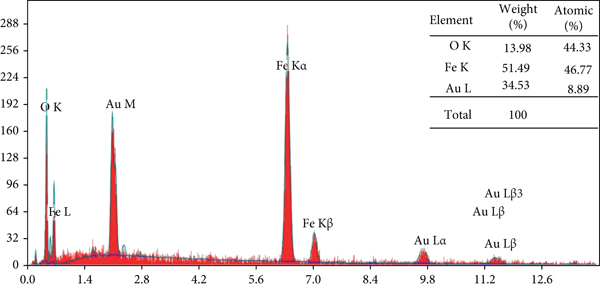
The nanoparticles’ surface morphology was measured by a scanning electron microscope (Philips SEM, Quanta 250 field emission gun (FEG), USA). The elemental composition of nZVI was analyzed using energy-dispersive spectroscopy (EDS) in conjunction with SEM at a high magnification of 16kx. For determining the pH at the point of zero charge (pHPZC), the solution pH was incrementally adjusted from 2 to 12 using either 1 N H2SO4 or 1 N NaOH (pHi) in a 100 mL Erlenmeyer flask. About 0.1 g of nZVI was added to the flasks and kept at 23°C for 24 h, and then, the final pH readings were recorded (pHf).
2.5. Computational-Based Studies
2.5.1. Artificial Intelligence Neural Networks
Figure 1 shows the ANN architecture used to predict BOD removal from a given dataset of five input variables (pH, adsorbent dose, time, stirring rate, and

Flowchart of feed-forward back-propagation ANN model for predicting BOD removal efficiency using five adsorption factors, i.e., solution pH, CA/nZVI dosage, time, stirring speed, and initial BOD concentration.
During the ANN learning phase with a back-propagation technique, the weights and biases were adjusted using several epochs (trials). The mean squared error (MSE) between the ANN output and measured BOD removal reached its minimum value at the best network performance. This feed-forward ANN model was used for its simplicity (no cycles or loops) to describe complex input-output relationships and to cope with the weighting adjustment issues [23]. In this study, the number of neurons (
2.5.2. Regression Analysis
A quadratic regression model (Equation (5)) was developed to predict BOD removal and estimate the optimum adsorption condition. Moreover, the results of the polynomial model were used to visualize the correlation between BOD removal and the adsorption factors. The model parameters were estimated based on the least square method [24] to fit the BOD removal data. The goodness-of-fit criteria (
3. Results and Discussion
3.1. Characterization of nZVI
Figure 2(a) shows the XRD pattern in the

Characterization of the prepared CA/nZVI adsorbent (a) XRD, (b) SEM, (c) EDS, and (d) pHPZC.


3.2. Effect of Operating Conditions on BOD Removal
The BOD removal efficiency varied considerably in response to the change in the adsorption factors (Figure 3). For instance, adapting the solution pH to 7–8 would provide a suitable condition for BOD reduction (Figure 3(a)). This pH range complied with the pHPZC, supporting the involvement of strong attractive and binding forces to remove organic impurities. Moreover, the optimum pH condition would facilitate the generation of hydroxyl radicals (⋅OH) to degrade and oxidize a series of organic compounds [29]. However, decreasing the pH level below pHPZC was associated with unsatisfactory BOD removal, probably due to the dissolution and/or separation of Fe from nZVI [34]. The BOD removal efficiency was also maximized (above 90%) at pH of 7 in an adsorption process using mixed adsorbent carbon [35]. Their study also demonstrated that the alkaline condition (

Influences of adsorption factors on BOD removal efficiency: (a) solution pH, (b) adsorbent dosage, (c) contact time, (d) stirring rate, and (e) initial adsorbate concentration.
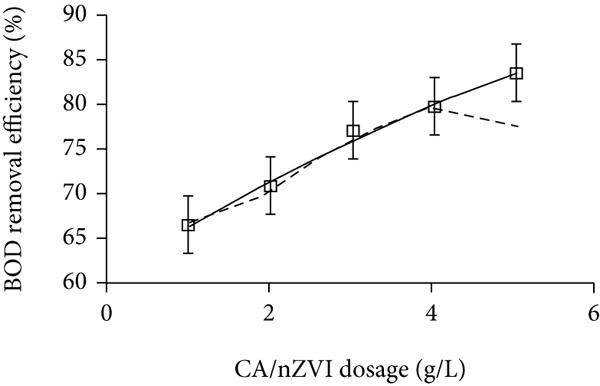

The BOD removal efficiency was also enhanced by increasing the CA/nZVI dosage for the 1–5 g/L range (Figure 3(b)). For instance, the BOD removal efficiency improved from 66.3% to 83.2% when the CA/nZVI dosage increased from 1.0 g/L to 5.0 g/L, respectively. Increasing the adsorbent dosage provided more vacant sites to capture large amounts of organic ions. Similar behavior was observed for organic matter removal via an adsorption system with wood fly ash (adsorbent), showing an increase in BOD removal from 4 to 24% with elevating the dosage from 20 to 160 g/L, respectively [36]. Their study demonstrated that raising the adsorbent dosage was accompanied by greater surface area and carbon content, finally promoting higher sorption of organic pollutants [36].
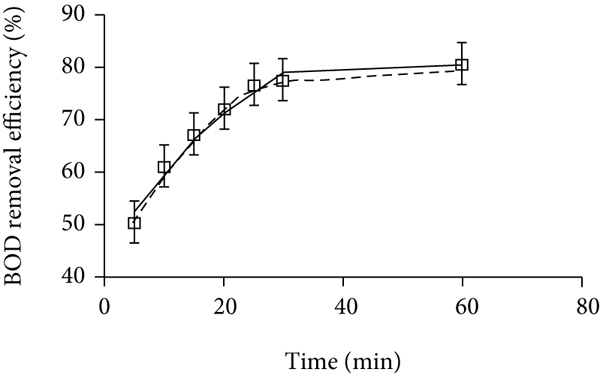
The adsorption time within the range of 5–60 min also influenced the BOD removal efficiency (Figure 3(c)). A high BOD reduction (61.5%) occurred rapidly within the first 10 min, assigning to the availability of a large number of vacant sites at the initial stage. This BOD removal reached 76.8% after 25 min and then slightly increased to 81.2% after 60 min (Figure 3(c)). It could be observed that the adsorption process started to reach the equilibrium state after 25 min due to nZVI saturation. This time was shorter than 60 min used to remove BOD with an efficiency of 91.3% via adsorption onto green synthesized nanomaterials [37].
The BOD removal efficiency also varied according to the rate of mixing nZVI particles in the aqueous solutions (Figure 3(d)). The mixing speed of about 200–300 rpm was suitable to improve BOD removal due to facilitating the transfer and diffusion of organic ions through the nZVI pores. However, increasing the mixing speed over 300 rpm would not be recommended in the adsorption process, probably due to further desorption of the captured contaminants under fast agitation. Moreover, the operational cost of the adsorption system would be expensive due to the surplus electricity input to reach 500 rpm.
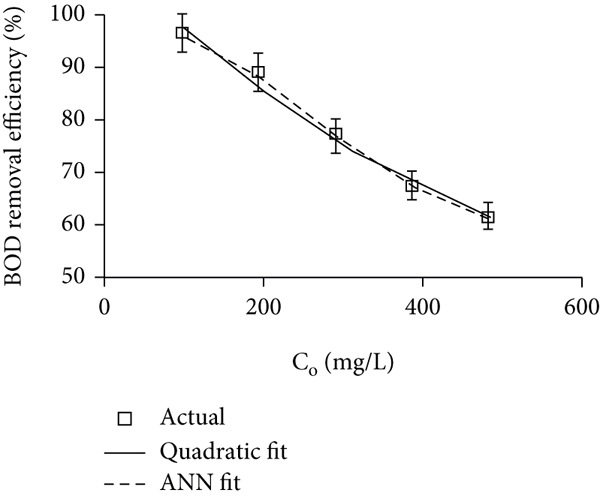
The results in Figure 3(e) depict that increasing
3.3. BOD Removal by Different Adsorbents Reported in the Literature
Table 2 includes the removal efficiencies of BOD using various adsorbent materials reported in the literature compared to CA/nZVI applied in this study. For example, Mahmoud et al. [37] used soft black tea to prepare nZVI, which removed 91.3% of BOD at pH 8, stirring rate 200 rpm, and adsorbent dosage 3.2 g/L within 60 min. To avoid the unmanaged disposal of wood residues, Laohaprapanon et al. [36] used wood fly ash to remove BOD via batch sorption experiments. Their study achieved BOD removal of 24% using an ash dosage of 160 g/L within 20 min. Due to its high carbon content and quite accessibility, date palm waste was used to prepare activated carbon, which is further employed for organic matter adsorption [39]. Their study showed that 1 g/L of this activated carbon could eliminate 92.8% of BOD at
Removal efficiencies of BOD using various adsorbent materials reported in literature.
3.4. Isotherm and Kinetic Studies
Three isotherm models were used to describe the adsorption equilibrium between organic pollutants and CA/nZVI (Figure 4(a)). These models were Langmuir [40] (Equation (6)), Freundlich [41] (Equation (7)), and Tempkin and Pyzhev [42] (Equation (8)):
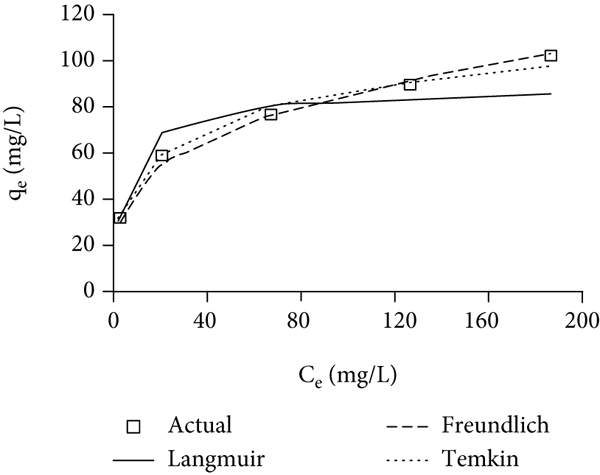
Fitting of adsorption data to (a) isotherm models and (b) kinetic models.

Table 3 lists the values of isotherm parameters and the corresponding fitting accuracies (
Results of isotherm and kinetic studies for BOD removal by CA/nZVI adsorbent.
The pseudo-first-order (PFO) (Equation (9)) and pseudo-second-order (PSO) (Equation (10))) kinetic models [45] were used to quantitatively describe the kinetic adsorption of organic impurities onto CA/nZVI:
The plot of
3.5. Artificial Neural Network (ANN) for Adsorption Computation
3.5.1. ANN Optimization
Table 4 lists the predictive performances of several ANN structures derived by altering the number of hidden layer neurons and the back-propagation learning algorithm. The input layer (with 5 neurons) received data from the five adsorption factors. Only one hidden layer was used in these ANNs to avoid an overcomplex network’s architecture and obey the optimality criterion (e.g., save computational cost, minimize MSE, and fasten the learning speed). The output layer included a single node, and hence, the ANN configuration could be expressed as
Determining the best ANN predictive performance by adapting the number of hidden layer neurons and the back-propagation learning algorithm.
3.5.2. ANN Training, Validation, and Test
During network optimization, the predictive accuracies for the training, validation, and testing processes were recorded (Figure 5(a)). In these figures, the theoretical and best regression fittings are given by the dashed and solid lines, respectively. These processes showed

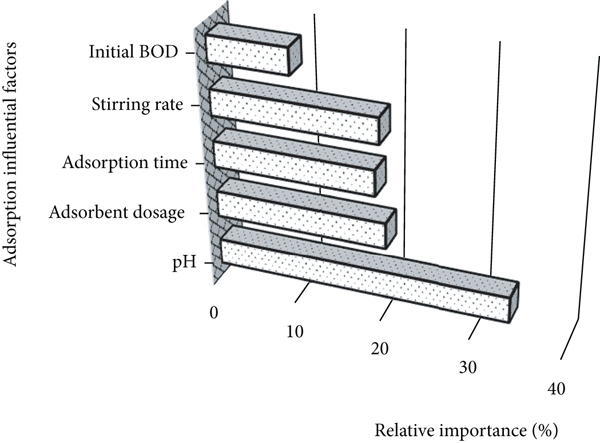
Performance of ANN model for predicting BOD removal efficiency: (a) regression plot, (b) validation checks, (c) best validation, and (d) relative importance. The number of data points in the training, cross-validating, and testing sets had 70%, 15%, and 15% proportions, respectively.


3.5.3. ANN Applicability for Adsorption Studies
In this study, a three-layer feed-forward back-propagation ANN with a “
Figure 5(d) shows each experimental factor’s relative importance, where the solution pH experienced the most influence on the BOD removal efficiency. Accordingly, the medium pH should be adjusted to around 7.5 for maintaining the highest adsorption performance. Controlling and adjusting the medium pH would be essentially considered to design and scale up the adsorption system. The relative importance of adsorbent dosage, time, and mixing speed was almost comparable at around 18%. Lower relative importance for
3.6. Quadratic Regression Model for Adsorption Computation
Table 5 lists the statistical results generalized from the
3.7. Model Verification
The accuracy of the developed computational models to predict BOD removal under new conditions was estimated. In particular, additional 25 experimental runs were performed by varying the adsorption factors, followed by the analysis of BOD concentrations. In parallel, these inputs were incorporated into the quadratic and ANN models to predict the corresponding BOD removal efficiencies. The average of the absolute differences between the experimental results and model outputs was used to estimate the mean absolute error (MAE). The results in Table 6 demonstrate that the MAE values for the ANN and quadratic regression models were 0.73% and 1.91%, respectively. Apparently, both models showed a promising ability to predict the BOD removal efficiencies remarkably close to experimental values. However, the ANN model was more reliable and robust than the quadratic regression method in providing the predictions closer to the measured data. Each of the modeling techniques has advantages, regarding the prediction, optimization, and recognition applications in wastewater treatment processes. However, ANN is able to overcome some shortages that could arise during regression analysis implementation. In particular, the input factors do not require a statistical experimental design to train the ANN model (compared to the regression analysis that only provides first- or second-order polynomial models). ANN as a soft computing technique and a black-box model depends on the analysis of available data to simulate any form of nonlinearity. In parallel, the regression models utilize a small number of experiments to generate manifold information, provide graphical illustrations for input-output relationships, and establish significance analysis. Hence, the authorities are encouraged to develop and scale up these modeling approaches in real-scale wastewater adsorption systems.
Verification of quadratic regression and ANN models for predicting BOD removal using additional experimental runs.
4. Conclusions
This study focused on the application of computational-based techniques to predict BOD removal in an adsorption process. The adsorbent material was characterized by XRD, SEM, and EDS, showing a successful preparation of Fe nanoparticles in the zero-valent state. The highest BOD removal efficiency (96.4%) was observed at
Footnotes
Nomenclature
Data Availability
The article includes all data generated or analyzed during the investigation.
Conflicts of Interest
No known competing financial interests or personal relationships could have appeared to influence the study.
Authors’ Contributions
All authors contributed to conceptualization, methodology, formal analysis, and writing—review and editing.
Acknowledgments
The authors would like to thank Badr University in Cairo (BUC), Egyptian Russian University (ERU), and Housing and Building National Research Center (HBRC) for supporting this research. The last author acknowledges Nasr Academy for Sustainable Environment (NASE). This work was supported by The Academy of Scientific Research and Technology (ASRT)/the Bibliotheca Alexandrina (BA) Research Grants (grant number 1469) awarded to the first author of this paper (Associate Professor Mohamed K. Mostafa, BUC).
