Abstract
Aims. The cardiobenefits of empagliflozin are multidimensional, and some mechanisms are still unclear. The aim of the present study was to evaluate the effect of treatment with empagliflozin on biometric parameters and gene expression in the local cardiac RAS, oxidative stress, and endoplasmic reticulum pathways in a mouse model. Main Methods. Forty male C57BL/6 mice were fed with control (C) or high-fat (HF) diets for 10 weeks. After that, the groups were redistributed according to the treatment with empagliflozin—CE or HFE. The empagliflozin was administered via food for 5 weeks (10 mg/kg/day). We performed biochemical analyses, blood pressure monitoring, oral glucose tolerance test, left ventricle (LV) stereology, RT-qPCR for genes related to classical and counterregulatory local RAS, oxidative stress, and endoplasmic reticulum stress. Key Findings. In comparison to HF, HFE decreased body mass and improved glucose intolerance and insulin resistance. The cardiac parameters were enhanced after treatment as expressed by decrease in plasma cholesterol, plasma uric acid, and systolic blood pressure. In addition, LV analysis showed that empagliflozin reduces cardiomyocyte area and LV thickness. The local RAS had less activity of the classical pathway and positive effects on the counterregulatory pathway. Empagliflozin treatment also decreased oxidative stress and endoplasmic reticulum stress-related genes. Significance. Our results suggests that empagliflozin modulates the local RAS pathway towards alleviation of oxidative stress and ER stress in the LV, which may be a route to its effects on improved cardiac remodeling.
1. Introduction
Cardiovascular diseases (CVDs) are the leading cause of death globally, and obesity is the third cause of death from noncommunicable disease. The latter is an established risk factor for diabetes mellitus type 2 (DM2) and leads to development of CVDs, affecting the cardiac structure and function [1].
The current treatments for DM2 are based on strict glycemic control, without substantial effect on CVD reduction, often leaving a residual vascular risk [2]. In 2008, the Food and Drug Administration issued guidelines to ensure that DM2 drugs had cardiovascular safety as a prerequisite. In this context, type 2 sodium and glucose cotransporter inhibitors (SGLT-2i) were developed as a new class of antihyperglycemic agents that inhibit, concomitantly, the reabsorption of glucose and sodium in the renal proximal convoluted tubule [3]. The ensuing glycosuria and natriuresis translate into an approximate 0.5 to 1.2% reduction in glycated hemoglobin, a 4-5/2 mmHg reduction in blood pressure (BP), and a 2 to 3 kg reduction in body mass [4].
The cardiobenefits of SGLT2i are multidimensional, and empagliflozin was the first drug in the SGLT2i class to show a significant reduction in CVD risk [5], presumably related to the weight loss [6]. Empagliflozin has also been reported to decrease cardiac fibrosis, arteriolar wall, and cardiovascular oxidative stress levels in obese mice, in addition to reduced left ventricular (LV) weight [7] and diameter of cardiomyocytes [8].
Another target of empagliflozin is the renin-angiotensin system (RAS). About 14% of patients with DM2 and resistant hypertension have a partial or complete autonomous aldosterone hypersecretion of RAS [9]. In the EMPA-REG-OUTCOME trial, such activation cannot be easily demonstrated since the vast majority of patients received treatments that block RAS activity. Therefore, the reduction in CV mortality could be a consequence of pharmacological interactions (SGLT2i+RASi) [10].
RAS has also been described as capable of enhancing the production of intracellular reactive oxygen species (ROS) by stimulating Nox2 and Nox4 and inhibiting SOD and catalase [11]. This stress increases cardiac hypertrophy as well as blood pressure (BP), both factors increasing the stimulus for endoplasmic reticulum (ER) stress [12]. However, it is known that there is an increase in both classical and nonclassical RAS pathway activities after SGLT2i use [13]. Besides, the local RAS activity, in cardiac tissue, activates the nonclassical pathway via Ang(1-7), leading to vasodilation, anti-inflammatory effects, and positive inotropic effects [14].
Although it was previously described that SGLT2i improves cardiac function and reduces cardiac fibrosis, there is a lack of studies evaluating the role of SGLT2i on cardiac RAS. The hypothesis that the cardioprotective effect of SGLT-2i is mediated by interaction with RAS is intriguing, and the present study was aimed at investigating whether empagliflozin treatment affects local RAS so that it would contribute to the reduction of cardiac changes caused by high-fat diet and the consequent changes in body mass, blood glucose, and BP, in addition to a reduction of oxidative stress and ER stress.
2. Material and Methods
2.1. Animals and Diets
The study was approved by the local ethics committee CEUA/031/2017, and the protocol followed the recommendations of the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health (NIH Publication number 85-23, revised in 1996). We used three-month-old male mice (C57BL/6) maintained in ventilated cages under controlled conditions (NexGen System, Allentown Inc., PA, USA,
Forty male mice were randomly divided into two groups (
Diet compositions. Protein, mineral, and vitamin mixes of all diets are in accordance with AIN-93M. The experimental high-fat diet had added 40% of lard +10% soybean oil.

Experimental design. C: control; CE: control with empagliflozin; HF: high fat; HFE: high fat with empagliflozin.
2.2. Biometric Analysis
Body mass was measured once a week on a precision balance (model B320H, Shimadzu, Brazil). Food and water intake were measured daily and recorded as the difference between the supplied food/water and the amount left in the cage after 24 h.
2.3. Oral Tolerance Test (OGTT)
Oral glucose tolerance test (OGTT) was performed before and after the treatment. After a 6 h fast, 1 g/kg glucose was administered by orogastric gavage (25% in sterile saline, 0.9% NaCl). Blood glucose measurement from the tail tip using a glucometer (Accu-Chek Go, Roche Diagnostic, Mannheim, Germany) was taken before glucose administration (time 0) and thereafter at 15, 30, 60, and 120 min.
2.4. Plasma Analyses
The fasting insulin concentration was measured by ELISA (enzyme-linked immunosorbent assay) using a commercial kit (Cat. # EZRMI13K, Millipore, Missouri, USA) and with TP Reader Thermoplate equipment (Tek Instruments, Inc., Highland Park, USA). Plasma uric acid and total cholesterol were quantified by an enzymatic colorimetric method using 25 μL of plasma in an automated spectrophotometer and commercial kits (Bioclin System II, Quibasa Ltda., Belo Horizonte, MG, Brazil).
2.5. HOMA-IR (Homeostatic Model Assessment of Insulin Resistance)
The insulin resistance which was analyzed by HOMA-IR was used:
2.6. Urinary Analyses
The urine was collected for 24 h in metabolic cages for individualized animals and stored at -20°C until the time of analysis. An aliquot of 25 μL was diluted in water at 1 : 10, and the uric acid was determined with an automated spectrophotometer and commercial kits (Bioclin System II, Quibasa Ltda., Belo Horizonte, MG, Brazil).
2.7. Systolic Blood Pressure (SBP)
SBP was measured by the noninvasive caudal plethysmography method using a calibrated tail-cuff system (Insight, Ribeirao Preto, SP, Brazil). Mice were kept warm using a warming pad and were acclimatized and trained at least two weeks (excluded from the analysis) before the actual measurements to be used for data analysis were taken. A mean of 3 measurements per day/animal was used to express the final systolic BP.
2.8. Euthanasia
Mice were fasted for 6 h and anesthetized intraperitoneally with ketamine (240 mg/kg) and xylazine (30 mg/kg), after which the blood was collected via a cardiac puncture and centrifuged at room temperature (712 g for 15 min). The plasma obtained was then stored individually at -80°C for the under-described analysis.
2.9. Tissue Extraction and Analyses
Scherle’s method [16] was applied to weigh the heart and the LV, dissected through the identification of the valvar plane. The samples were frozen and stored at -80°C for future molecular analyses or fixed in formaldehyde. Half of the fixed samples were used for collagen analyses, while the other half was embedded in Paraplast Plus (Sigma-Aldrich, St. Louis, USA), sectioned at 5 μm thickness, and stained with hematoxylin and eosin for microscopy. The observations and digital photomicrographs were obtained with a Nikon microscope (model 80i) and DS-Ri1 digital camera (Nikon Instruments, Inc., New York, USA).
2.10. Stereology and Morphometry
Stereological analysis was performed to estimate the mean area of cardiomyocytes (A[car]), and LV thickness was assessed by morphometric analysis.
A 36-point test system produced with STEPanizer (http://www.stepanizer.com) was used for data acquisition. Thus, the volume density (Vv) of the cardiomyocytes was estimated by the ratio between the partial points that touched them (Pp) and the total points of the test area (Pt), following the formula
2.11. Concentration of Total Collagen
The LV obtained was finely minced, washed three times for 30 minutes in distilled water, immersed in acetone for 24 hours, and submitted to two changes of 24 hours each in 40 mL of chloroform : methanol (2 : 1,
2.12. Quantitative Real-Time PCR (RT-qPCR)
RT-qPCR was performed to detect mRNA expression of genes related to oxidative stress, ER stress, and RAS. Total RNA of LV was extracted using TRIzol reagent (Invitrogen, CA, USA). mRNA concentration was determined by spectroscopy using NanoVue (GE Life Sciences). Then, 1 μg of RNA was treated with DNAse (Invitrogen). The cDNA was synthesized using oligo (dT) primers and reverse-transcriptase Superscript III (Invitrogen, CA, EUA). The Biorad CFX96 cycler and the SYBR Green mix (Invitrogen, CA, USA) were used. The endogenous β-actin was used to standardize the expression of the selected genes. PCR reactions were performed following a polymerase denaturation and activation program (4 min at 95° C), with 44 cycles, each consisting of 95°C for 10 s and 60°C for 15 s, followed by a fusion curve (60-95°C, with a heating rate of 0.1°C/s). Negative controls consisted of wells in which the cDNA was substituted for deionized water. The signals were quantified using the ΔΔCt method to estimate the difference between the number of target gene cycles and the endogenous control. Sense and antisense primer sequences used for amplification are described in Table 2.
Primers from Plin2, renin angiotensin system, endoplasmic reticulum stress, and oxidative stress.
Abbreviations: Plin2: perilipin 2; Ace: angiotensin-converting enzyme; Ace2: angiotensin-converting enzyme 2; AT1r: angiotensin II type 1 receptor; At2r: angiotensin II type 2 receptor; Masr: Mas-receptor; Atf4: activating transcription factor 4; Chop: Caat-enhancer-binding protein homologous protein; Gadd45: growth arrest and DNA damage-inducible gene 45; Nox4: NADPH oxidase 4; Sod1: superoxide dismutase [Cu-Zn]; Sod2: superoxide dismutase [Mn]; Timp1: metallopeptidase inhibitor type 1.
2.13. Data Analyses
To analyze both groups in the pretreatment phase, the Student
3. Results
3.1. Body Mass Gain and Fasting Glucose
During pretreatment, the BM was higher in the HF than in the C group (+14%,
In fasting glucose, a difference was observed between C and HF groups, with an increase of 78% (
Biometric results, plasma and urinary analyses, and pretreatment and posttreatment.
Data are presented with
3.2. Glucose Tolerance and Insulin Resistance
Regarding glucose tolerance, a higher area under the curve (AUC) was observed in the HF than the C group before treatment (+25%,
The plasma insulin increased in the HF when compared with the C group (108%;
The HOMA-IR index showed that the HF group had significantly higher insulin resistance than the C group (+165%,
Two-way ANOVA revealed that diet accounted for 77.06% OGTT and for 51.65% in HOMA-IR index (
3.3. Urinary Volume and Glucose Urinary
Empagliflozin increased urinary volume in both treated groups compared to their untreated counterparts (+88%,
There was no difference in glucose urinary between the C and HF groups (
3.4. Plasma Cholesterol
Total cholesterol was 13% higher in the HF when compared with the C group (
Two-way ANOVA showed that the plasma cholesterol was influenced simultaneously by diet and empagliflozin (54.9%
3.5. Uric Acid Analyses
Uric acid plasma of the HF presented significantly higher concentration than the C group (+24%,
In urinary, uric acid decreased -42% in the HF compared to the C group (
Two-way ANOVA showed that diet, empagliflozin, and the interaction between them influenced plasma uric acid, but it is noteworthy that the interaction accounted for 46.04% making it the most influential factor (
3.6. Systolic Blood Pressure (SBP)
Before treatment, significant difference was observed in SBP between the HF and C groups (+25%,

Systolic blood pressure (mmHg) pre- and during treatment. Data presented with
3.7. Left Ventricle Results
3.7.1. Hydroxyproline Measurement
The content of total collagen in the myocardium, as determined by a hydroxyproline assay, did not differ significantly between C and HF groups (
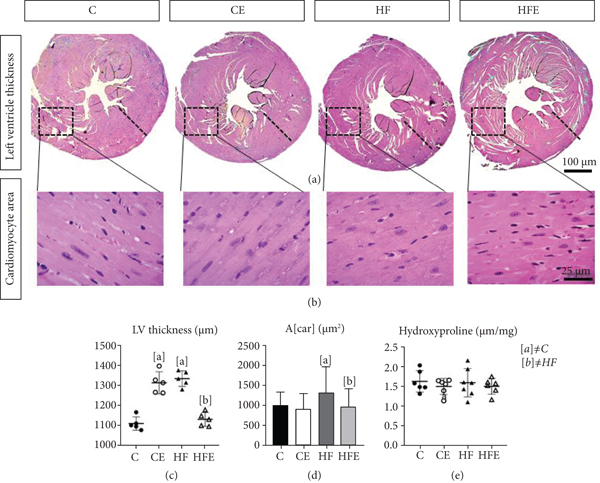
(a) Left ventricular thickness, cross-section caudal view at the valvar plane showing the differences in the wall thickness. (b) Photomicrographs of the cardiomyocytes, paraffin sections stained by hematoxylin and eosin (×600). (c) Left ventricular thickness (μm). (d) Cross-sectional area of cardiomyocyte (A[car] (μm2)). (e) Hydroxyproline concentration in dry tissue of left ventricle. All data are presented as
3.7.2. Left Ventricle Remodeling
Concerning the LV mass, the HF presented a higher LV/tibia ratio than the C group (+17%;
The cardiomyocyte cross-sectional area (A[car]), in the HF, showed hypertrophied cardiomyocytes, with an increase of 30% (
Influenced by these results, the LV thickness increased by 20% in the HF compared to the C group (
Two-way ANOVA showed that diet was the most influential factor in the LV/tibia ratio (50.17%,
3.8. Gene Expression
3.8.1. Local RAS System
There was an improvement in local LV RAS with decreased expression of these pathway-related genes after empagliflozin treatment, while its counterregulatory pathway was increased (Figure 4).
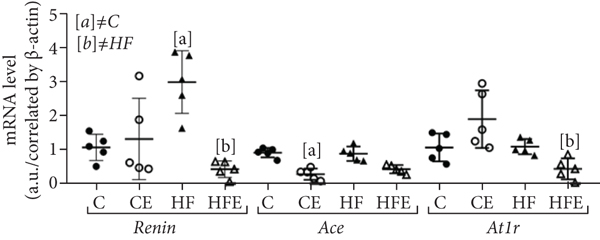
Effect of empagliflozin on mRNA levels of genes involving in renin angiotensin system (RAS). (a) Classic pathway: renin, angiotensin-converting enzyme (Ace), angiotensin II type 1 receptor (AT1r). (b) Contraregulatory pathway: angiotensin-converting enzyme 2 (Ace2), Mas-receptor (Masr), angiotensin II type 2 receptor (At2r). (c) Ace/Ace2 ratio. Data are presented as


Renin is the rate-limiting enzyme of the renin-angiotensin system, and its gene expression was higher in the HF compared to the C group (+179%,
AT1r is the major receptor for Ang II-mediated cardiovascular functions in mice, and its expression did not differ between the C and HF groups (
Concerning the effects of the counterregulatory axis, AT2r has been recognized as an integrative part of the protective arm of the RAS, and the current results showed 48% reduction in the gene expression of AT2r in the HF group (
In the two-way ANOVA, treatment significantly influenced the AT2r, ACE1, and MASr gene levels (
3.9. Oxidative Stress System
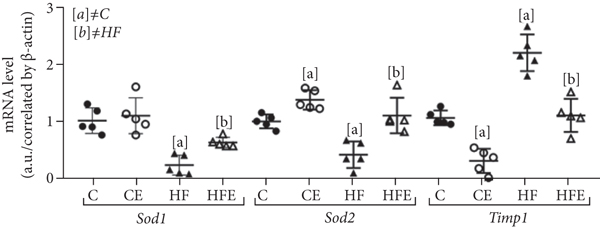
Oxidative stress is associated with metabolic disorders, such as hypertension, and is conducive to LV hypertrophy and heart failure. However, there are antioxidant enzymes that protect biological systems. Therefore, some antioxidant enzymes were analyzed, and the gene expression of catalase decreased in the CE in comparison with the C (-64%,
A role of Nox4 in cardiac and vascular protection has been reported, and it presented higher expression in the HF compared to the C group (+95%,
Timp1 had higher expression in the HF compared to the C group (+106%,
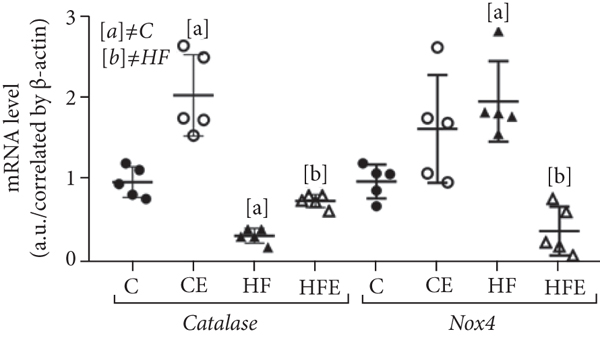
Effect of empagliflozin on mRNA levels of genes involving in oxidative stress. Catalase, NADPH oxidase 4 (Nox4), superoxide dismutase [Cu-Zn] (Sod1), superoxide dismutase [Mn] (Sod2), metallopeptidase inhibitor type 1 (Timp1). Data are presented as
Two-way ANOVA highlighted that the primary effect of Nox4 expression was from an interaction between diet and treatment (58, 35%,
3.10. ER Stress System
Experimental evidence suggests that ER stress deregulation is implicated in heart diseases [17]. Atf4 is a master regulator for ER stress, and we found that its gene expression was increased in the HF compared to the C group (+60%,
Chop is a widely investigated biomarker involved in ER stress-associated apoptotic signaling in CVD, whose gene expression was enhanced in HF in comparison with the C group (+37%,
Gadd45 is a stress signal gene that is expressed in response to physiological or environmental factors. Its expression was augmented in the HF compared to the C group (+73%,

Effect of empagliflozin on mRNA levels of genes involving in endoplasmic reticulum (ER) stress. Atf4: activating transcription factor 4; Chop: Caat-enhancer-binding protein homologous protein; Gadd45: growth arrest and DNA damage-inducible gene 45. Data presented with
4. Discussion
This study investigated an experimental mouse model with a high-fat diet that triggered detrimental alterations in SBP, LV thickness, and important metabolic pathways, like local RAS in the LV, ERE, and oxidative stress. On the other hand, the SGLT2 inhibitor, empagliflozin, showed positive results by mitigating most of these alterations.
Empagliflozin treatment reduced BM by 7%, and this can be explained because empagliflozin leads to a continuous excretion of about 60-100 g glucose in the urine, corresponding to a calorie loss which contributes to weight loss [18]. Both glucose and insulin of the treated animals improved, characterizing a decrease in insulin resistance. Insulin resistance could be one factor for damage to cardiac tissue by inducing de novo lipogenesis and increased lipotoxicity in target tissues such as the heart [19]. This study showed an influence of empagliflozin on the decrease in total cholesterol; these effects probably stemmed from reduced LDL clearance and reduced intestinal cholesterol absorption [7, 20].
It is given that hyperuricemia is a contributing factor in the development of hypertension, a risk factor for CVD [21]. Our results demonstrated that empagliflozin decreased plasma uric acid but increased its urine excretion. Reductions in uric acid have also been reported in the EMPA-REG OUTCOME trial, and this effect by empagliflozin is possibly due to the increasing renal urate elimination. Almost all of the filtered uric acid is reabsorbed via the urate transporter and the facilitative glucose transporter 9 in the basolateral membrane of the proximal tubule and the consequent increase in uricosuria [22].
More than 50% of hypertensive patients have additional cardiovascular risk factor as diabetes, lipid disorders, overweight and obesity, and hyperuricemia and metabolic syndrome, as well as unhealthy habits. For all the diabetes patients the treatment should lowered BP, decreased RAS activation, prevention cholesterol rising and include glucose and lipid lowering per current guidelines [23]. In this context, our data showed that empagliflozin has positive effects in reducing SBP in the HFE group, which is in agreement with some evidences suggesting that the SGLT2i causes a remarkable reduction in BP [6, 24]. In spite of this, the precise mechanism of the BP reduction remains incompletely elucidated, with some involved mechanisms being related to natriuresis, reduced plasma volume, nonfluid weight loss, direct vascular effects [6], and RAS alternative axis [25].
Of note, hypertension is responsible for increased LV thickness as a compensatory mechanism [26]. In accordance with the current data, the HF group presented increased LV thickness and LV hypertrophy, which could be related to an increase in the LV mass and SBP, while empagliflozin treatment decreased LV thickness. Indeed, SGLT-2i is associated with improvements in regression of LV hypertrophy and cardiac remodeling and can also mitigate cardiac fibrosis remodeling [27].
Although our results showed that HF-fed mice had cardiac hypertrophy and higher SBP, the histological analysis and a specific biochemical assay showed that collagen content in the myocardium was not significantly affected in these animals, which could be due to the shorter duration of the high-fat diet in the current study. Indeed, when mice are fed a high-fat diet for longer periods, such as 24 weeks, interstitial fibrosis in the myocardium has been reported [28].
Empagliflozin reduces oxidative stress and monocyte/macrophage infiltration in the heart of metabolic syndrome rats [8]. The increase in oxidative stress promotes cardiomyocyte hypertrophy and increased expression of collagen type I and TIMP1 [11]. The latter is a glycoprotein that inhibits collagen degradation, thereby increasing its interstitial concentration. In this study, an increase in TIMP1 expression was observed in HF-fed animals, while it was reduced in treated groups. Besides, based on present data, there was an increase in antioxidant enzymes through the catalase, Sod1, and Sod2 gene expression after treatment. Few studies are investigating these enzymes in the heart of empagliflozin-treated animals, and they showed that the expressions of Sod2 [7, 29] and catalase [29] are increased while those of Nox1, Nox2 [30], and Nox4 [7] are decreased after treatment. However, this study demonstrated that empagliflozin can also downregulate the level of NOX4 in the LV of the HF-fed animals. NOX4 is the major NAD(P)H oxidase isoform in cardiomyocytes, which is associated with cardiomyopathy in the diabetes model [7]. In this way, these results indicate that empagliflozin can attenuate oxidative stress by elevating the expression of the antioxidant enzymes and reducing oxidation products in the LV of the high-fat-fed mice.
The RAS is a central component of the pathophysiological responses of cardiovascular system and has two activation routes with opposite actions, the classical pathway [14, 31] and a nonclassical axis, in which Ace2 produces angiotensin 1-7, activates the Masr, and leads to systemic arteriolar vasodilation, diuresis, reduced oxidative stress, and antiproliferative activity by increasing nitric oxide. Moreover, through the Masr, angiotensin reduces signaling pathways considered responsible for fibrogenesis and chronic inflammation [14].
Despite these facts, the role of SGLT2i on local RAS modulation in the LV is still unclear. The intrarenal RAS suppression has been demonstrated in response to SGLT2 inhibition in experimental models of T2D [32] and may contribute to the reduction in cardiovascular complications [33]. Our data suggest that empagliflozin leads to beneficial LV remodeling through decreased gene expression of the RAS classical pathway (renin, Ace1, and Atr1), in contrast to their high expression in the HF-fed mice, complying with the increased BSP and the LV hypertrophy found.
On the other hand, in the nonclassical pathway, considered as the counterregulatory axis, empagliflozin increased Atr2 and Masr gene expression in LV. Ace2 did not show any decrease in its expression, so we performed the analysis of the relationship between Ace/Ace2, which was above 1, indicating a preference for the classic pathway [34]. This ratio ensured that empagliflozin tends to act on the nonclassical pathway, leading to cardiovascular protection. It has also been reported that there is an increase in Ang II levels after the use of empagliflozin, but this increase is not harmful due to the action of this hormone via Atr2 and the concomitant increase in angiotensin 1-7 [10]. However, some studies show a decrease in RAS through a decrease in angiotensinogen and Ang II and in renin [35] excretion after dapagliflozin treatment, but the present study is the first one to ascertain the local expression of this pathway in the heart after empagliflozin use.
Regarding ER, it is responsible for the degradation of damaged proteins and organelles and plays a critical role in obesity-related cardiac injury [36] and cardiac hypertrophy [37]. Increasing Ang II due to hypertension is known to enhance the expression of ER stress-related genes such as Chop, Atf4, Bip, and Gadd45 [12]. In our study, the markers of ER stress in the LV increased in the HF group, implying ER stress in this group, which was reversed by empagliflozin. This effect has already been described after treatment with empagliflozin [38] and other SGLT2i [39].
The limitation of this study was the lack of the analysis of cardiac function, but previous studies have shown an improvement in cardiac function using SGLT2 inhibitors, and dapagliflozin attenuates the activation of the inflammasome and deterioration of left ventricular ejection fraction in BTBR mice [40]. In ob/ob-/- mice, empagliflozin improves coronary microvascular function and contractile performance indicated by coronary flow velocity reserve (CFVR) and fractional area change (FAC) [41]. In addition, treatment with empagliflozin has been shown to improve the left ventricular function in a model of diabetic cardiomyopathy [42] and also to reduce oxidative stress, inflammatory activity, and cardiac dysfunction in obese C57BL/6 mice [43].
In conclusion, our data showed that treatment with empagliflozin for 5 weeks decreases body mass, plasma uric acid, and systolic blood pressure in a diet-induced obesity model. By acting on the left ventricle, empagliflozin may enhance the local RAS pathway by preventing the oxidative stress and endoplasmic reticulum stress. Empagliflozin thus ameliorates the cardiac remodeling by reducing the cardiomyocyte area and left ventricle thickness, as evidenced in high-fat diet mice.
Footnotes
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
Conflicts of Interest
The authors have declared that no competing interests exist.
Acknowledgments
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Brazil (CAPES), Finance Code 001. This study was supported by FAPERJ (Rio de Janeiro State Foundation for Scientific Research), grant number: E-26/202.819/2019 for S.B.S. The authors thank Aline Penna de Carvalho for the technical assistance.
