Abstract
Owing to the COVID-19 pandemic, huge amounts of disposable face masks have been manufactured and used, and these discarded face masks have to be treated. In this study, we propose a simple approach for reusing the nonwoven polyester fabric (NWPF) from disposable face masks. In this approach, NWPF is utilized as a supporter for coating of a layer of graphene oxide/Fe3O4/chitosan (GFC) to form a GFC/NWPF adsorbent at room temperature via a simple spray coating method that does not require any solvent. The specific properties of GFC, NWPF, and the GFC/NWPF adsorbent were analysed via X-ray diffraction, transmission electron microscopy, ultraviolet–visible spectroscopy, vibrating sample magnetometry, and field-emission scanning electron microscopy. Results showed that the presence of NWPF enhanced the adsorption capacity of GFC towards organic dyes. At high concentrations of the organic dyes, the adsorption efficiency of the GFC/NWPF adsorbent to the dyes reached 100% within 24 h. The adsorption capacity (
1. Introduction
Organic dyes are widely used in many industries, such as textile, paper, rubber, plastic, leather, cosmetic, pharmaceutical, and food industries. However, the wastewater produced by these industries contains dyes and their products that contribute to water pollution, causing negative effects on humans and the environment, such as preventing the absorption of oxygen and sunlight and disrupting the respiration and growth of aquatic organisms. Furthermore, it causes adverse effects on the ability of microorganisms to decompose organic substances in wastewater [1–3]. Therefore, researchers have developed various wastewater treatment methods and have successfully applied them in removing colorants. These methods can be divided into three main categories, namely, biological, chemical, and physical methods [1]. Although physical and chemical methods, such as adsorption, photocatalysis, photocatalytic decomposition, membrane separation, ultrasonication, and wet air oxidation, are effective, they can only be applied when the concentration of the dissolved substances is sufficiently high. Moreover, some methods are expensive and still produce toxic by-products. By comparison, biological treatment methods include removing dyes via anaerobic and aerobic systems and fermenting activated sludge from filamentous fungi, yeasts, bacteria, and bacterial and fungal biomes. However, these methods also have disadvantages, such as long processing time and poor performance in removing dyes with a durable and a high-molecular polymeric structure. Moreover, the composition of these organic dyes in wastewater often harms the microorganism biomes/populations used in the sludge [4]. Among the aforementioned wastewater treatment methods, adsorption is considered one of the preeminent methods owing to its advantages, such as easy implementation, generation of nontoxic substances during the treatment process, high efficiency, and low cost [5]. Furthermore, various adsorbent materials from traditional materials can used in adsorption, such as activated carbon, clay, agricultural by-products, banana peels, straw [5], rice husk [6], and red mud [7].
Graphene oxide (GO) and its composite-based adsorbents are excellent in adsorbing organic compounds, including organic dyes, as well as heavy metal ions [8–14]. GO is notable for its simpler fabrication and easier dispersion in water than other materials [15, 16]. Accordingly, GO is easy to use with a high adsorption efficiency. Functional molecules are utilized to generate functional groups to improve the adsorption capacity of GO to heavy metal ions or organic dyes. Chitosan (CS) molecule has several functional groups, such as primary −OH, secondary –OH, and −NH2 groups. In these groups, the O and N atoms still have undivided electron pairs, making them the chemically active centres of CS. These groups are considered nucleophilic reagents and can participate in several specialized chemical responses [17] or complexed with almost all heavy metals and transition metals to help separate heavy metals from aqueous media easily [18]. Recently, Fe3O4 nanoparticles have been utilized to generate the magnetic property for adsorbents, thereby allowing them to be recovered and reused. In additions, the presence of Fe3O4 nanoparticles can improve the porosity of adsorbents [1, 2, 19, 20]. Therefore, GO/Fe3O4/CS (GFC) materials have excellent adsorption and recovery and regeneration abilities [21–23]. Hence, they are applied in the removal of organic dyes [2] and heavy metal ions [20].
Nonwoven fabric (NWPFs) is widely used in the production of medical disposable face masks and clothes [24–27]. NWPF can be combined with nanomaterials to enhance their properties, such as antibacterial, waterproof, and fireproof properties [28, 29]. The COVID-19 pandemic has necessitated the manufacturing and use of huge amounts of disposable face masks, and these discarded masks have to be treated [26]. Several recent reports have proposed using the NWPF extracted from discarded face masks and clothes as an adsorbent for the clean-up of waters polluted with petroleum and oil products [30], as a support of photocatalysts [31–33] or as a reinforcement of cement composites [27].
In this study, we propose a new approach for reusing NWPF. In this approach, NWPF is extracted from discarded disposable face masks as a support of a GFC nanocomposite adsorbent. The GFC/NWPF absorbent is fabricated by coating GFC onto NWPF via a simple and solvent-free spray coating method at room temperature (RT). Results showed that the presence of NWPF enhanced the adsorption capacity of the GFC absorbent to various organic dyes. Therefore, the GFC/NWPF absorbent will not only improve the adsorption capacity of GFC but also contribute to the efficient reuse of NWPF.
2. Experimental
2.1. Chemicals and Reagents
Graphite (99 wt.%,
GO was prepared from graphite flakes following Hummer’s method as described in previous reports [2, 20, 34, 35]. The GO product obtained was characterized via X-ray diffraction (XRD), scanning electron microscopy (SEM), and transmission electron microscopy (TEM) (Fig. SI.1). CS was prepared from shrimp shells in our laboratory in accordance with a previously published procedure [36]. The deacetylation degree (DD%) of CS was 85.0%; its average molecular weight (
2.2. Preparation of the GFC/NWPF Absorbent
The GFC/NWPF absorbent was manufactured by coating a layer of GFC ink onto NWPF via a simple spray coating method. The GFC ink was prepared by mixing 0.22 g GO, 0.85 g Fe3O4, and 0.36 g CS. Afterwards, the mixture was dissolved in 100 mL 1 wt.% acetic solution and 0.5 mL glycerol by using a homogenizer under ultrasonic conditions for 30 min to obtain a black homogeneous ink of the GFC nanocomposite. The GFC ink was spray-coated onto the NWPF at RT. Finally, the NWPF was dried at 60°C for 2 h. This process was repeated five more times to increase the thickness of the coating. Finally, the NWPF was soaked in 1 M NaOH solution overnight, washed until it reached neutral pH, and dried at 80°C for 24 h to obtain the GFC/NWPF absorbent. Subsequently, the GFC/NWPF absorbent was cut into
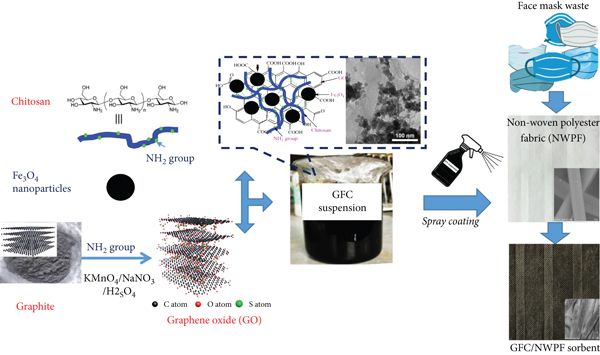
The process of fabricating a new adsorbent by coating graphene oxide/Fe3O4/chitosan (GFC) coated onto nonwoven polyester fabric (NWPF) extracted from discarded disposable face masks.
2.3. Adsorption Procedures
A piece of
Removal efficiency (
(a)Presence; (b)absence; (c)removal efficiency (
The amount of dye uptake by the GFC absorbent (
The Langmuir equation (3) and the Freundlich equation (4) isotherms were linearized into the following forms:
2.4. Characterization and Methods
The XRD patterns of each GFC sample were obtained at RT by using a D8 Advance diffractometer (Bruker ASX) with CuK
3. Results and Discussion
3.1. Characterization of GFCs
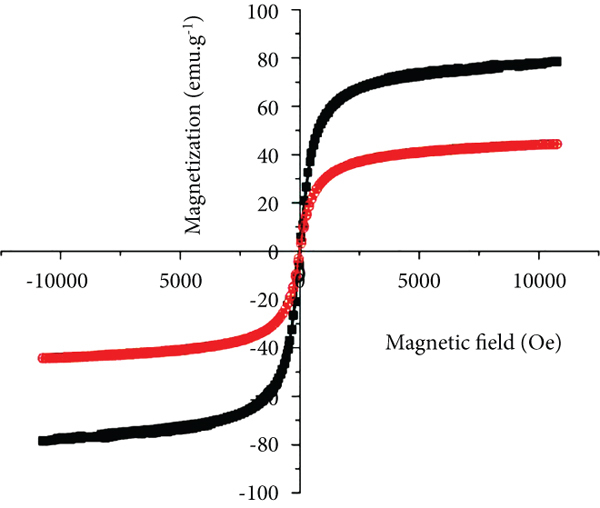
The Fe3O4 nanoparticles agglomerated quite strongly because of their large specific surface energy; thus, the nanoparticles tended to agglomerate to reduce the surface energy (Figure 1(a)). The thickness of GO sheets was thin, only over 5

(a–d) TEM images of (a) Fe3O4 nanoparticles, (b) GO, and (c, d) GO/Fe3O4/CS (GFC) nanocomposite (inset: magnetic property of GFC nanocomposite); (d) XRD patterns of (i) CS, (ii) GO, (iii) Fe3O4, and (iv) GO/Fe3O4/CS; (f) VSM of (i) Fe3O4 and (ii) GO/Fe3O4/CS.




3.2. Characterization of NWPF and the GFC/NWPF Absorbent
The GO/Fe3O4/CS materials were coated onto NWPF via a simple spray coating method. Five coats were applied to create a sufficiently thick and even coating. The mass density (
SEM images of NWPF and the GFC/NWPF absorbent are shown in Figure 2. The polyester fibres in NWPF were slippery and even, their surface was smooth, and it consisted of overlapping nonwoven fibres that were pressed by heat (Figures 2(a), 2(c), 2(e), and 2(g)). By comparison, after GFC was coated onto NWPF, the surface of the polyester fibres in the GFC/NWPF absorbent became rough and rugged (Figures 2(b), 2(d), 2(f), and 2(h)), and the coating cracked at some points (Figures 2(d), 2(f), and 2(h)). Moreover, the size of the fibres considerably increased. By contrast, the GO sheets (Figures 2(d) and 2(h)) and the Fe3O4 nanoparticles (Figure 2(h), inserted figure) became rumpled. These results indicated that GFC was successfully coated onto the surface of NWPF via the simple spray coating method adopted herein. Thus, this method can be employed in fabricating GFC/NWPF absorbents on a large scale.

SEM and FESEM images of (a, c, e, g) NWPF and (b, d, f, h) GFCs/NWPF.
3.3. Adsorption of Organic Dyes on GFC and GFC/NWPF Absorbents
3.3.1. Optimization of Adsorption Conditions
The adsorption conditions were optimized including the pH and compositions of the GFC/NWPF adsorbent to enhance its adsorption of organic dyes. Eight coated NWPF samples with different compositions were prepared (Table 1). Their ability to absorb MB (a cationic dye) and MO (an anionic dye) was evaluated at different pH 5, 7, and 9. Organic dyes can be classified into three types: cationic organic dyes with a positive charge, anionic organic dyes with a negative charge, and nonionic organic dyes with no charge. Therefore, the adsorption of the dyes onto the adsorbents can be achieved via electrostatic interactions between dye ions and groups of opposite charge as the functional groups on an adsorbent’s surface. However, this classification is only relative because an organic dye can be an anionic or a cationic dye depending on the environment (pH) (Table 2). The surface of the GFC/NWPF absorbent had abundant amine groups (−NH2) from CS and –COOH. The −OH groups of GO are suitable for nonionic dyes when they are in a neutral environment. In an acidic environment, these groups will become –NH3 ⊕, −COOH2 ⊕, and −OH2 ⊕; thus, they are suitable for absorbing MO, RS, and CR. Moreover, in an alkaline environment, these functional groups will become –NH2, −COO⊖, and –O⊖, respectively, which are suitable for absorbing MB [1, 2, 35, 44–47].
Comparison of the adsorption capacity of the adsorbents assessed herein.
Figure 3(a) shows that GO/NWPF sample (coded M3) adsorbed MB very well for all medium with removal efficiency (

Effects of pH on MB adsorption onto the GFC/NWPF absorbent: (a) pH 5, (b) pH 7, and (c) pH 9 (inserted images: corresponding color of MB solutions before and after the adsorption process).
In removing the anionic dye MO (Figure 4), the

Effects of pH on MO adsorption onto the GFC/NWPF absorbent: (a) pH 5, (b) pH 7, and (c) pH 9 (inserted images: corresponding color of MO solutions before and after the adsorption process).
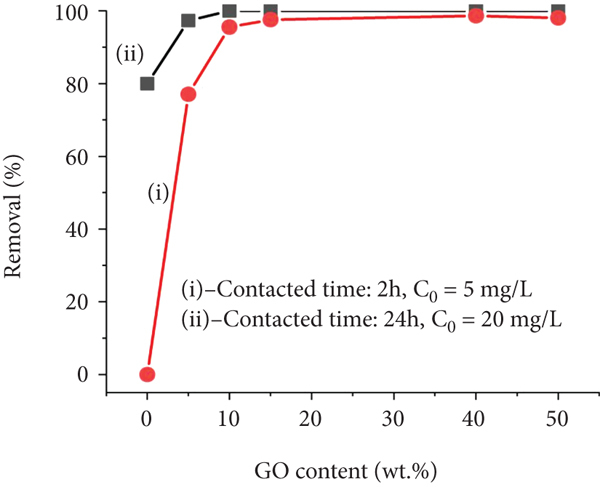
The effects of the absorbents’ components on dye adsorption capacity were evaluated. Five coated NWPF samples (S1, S2, S3, S4, and S5) were fabricated and tested for MB adsorption (Figure 5). Figure 5(a) shows the UV-Vis spectrum of the MB solution after adsorption by GFC powder for 2 h at RT and pH 9. The S1, S2, S3, S4, and S5 samples had compositions of GO, Fe3O4, and CS by mass (

(a) UV-Vis spectra of samples after adsorption of MB solutions (inserted images are the colors of the MB solutions before and after adsorption onto GO/Fe3O4/CS [percent by weight, wt.%]: S1 [50 : 40 : 10], S2 [50 : 10 : 40], S3 [0 : 50 : 50], S4 [10 : 60 : 30], and S5 [10 : 40 : 50]). (b) Influence of GO content on MB removal at various times and (i)
3.3.2. Comparison of the Adsorption of MB, MO, CR, and RS onto the GFC/NWPF Absorbent and Bulk GFC
The adsorption efficiency of bulk GFC was compared with that of the GFC/NWPF absorbent under the same conditions with the same amount of GFC converted. Both the powder and coated samples achieved very good adsorption efficiency for MB, MO, CR, and RS, and the color of the solutions was almost completely eliminated (Figure 6). In all cases, the GFC/NWPF samples seemed to remove the colorants better than the GFC:
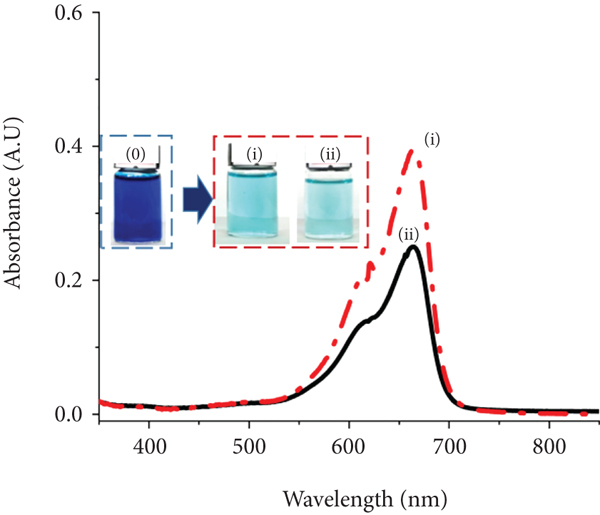
UV-Vis spectra of colorant solutions after adsorption of (a) MB, (b) MO, (c) CR, and (d) RS onto (i) GFC (red dot–dash) and (ii) GFC/NWPF (black solid) (inserted image: color of colorant solutions before and after adsorption).



3.3.3. Adsorption Isotherms of MB, MO, CR, and RS onto the GFCs/NWPF Absorbent
The adsorption isotherms of MB, MO, RS, and CR on both bulk GFC and the GFC/NWPF absorbent were built according to the Langmuir and Freundlich models by using equations (3) and (4) (Table 2 and Figs. SI.7–SI.10). On the basis of the correlation coefficient (
4. Conclusions
NWPF was extracted from discarded disposable face masks and used as a support to prepare a GFC nanocomposite-based adsorbent. GFC was successfully onto NWPF. The presence of NWPF enhanced the adsorption efficiency of GFC for the organic dyes MB, MO, CR, and RS. In all cases, the coating improved the adsorption performance of the GFC materials. The adsorption efficiency of the GFC/NWPF adsorbent for these organic dyes was higher than that of bulk GFC with the same mass. Obtained results demonstrated that the adsorption efficiency of the GFC/NWPF adsorbent was different for MB, MO, CR, and RS dyes and can be competitive to previously reported materials implying that the GFC/NWPF absorbent has a high application potential.
Footnotes
Data Availability
The data used to support the findings of this study are included within the article.
Conflicts of Interest
The authors declare that they have no conflict of interest.
Acknowledgments
This work was supported by the Vietnam Ministry of Education and Training (under project number B2020-BKA-15).
