Abstract
In order to assess the interactions between process factors, the experiments involving the liquid-phase adsorption of cephalexin (CEX) onto silicon-coated iron nanoparticles (Fe3O4@SIO2) were designed using the Box-Behnken Design-Response surface methodology (BBD-RSM). Optimal circumstances were used to investigate the synergistic influence on the process’s efficiency. In addition, the data was used to test and fit an artificial neural network (ANN) model. Molecular-level DFT calculations on the CEX molecule were carried out. The PW6B95D3/Def2-TZVP level of theory was used to build DFT-based descriptors for the CEX molecule. At 25°C, pH 5.83, 37.67 min, a dosage of 0.8 g Fe3O4@SIO2 and 118.01 mg/L CEX, the removal efficiency achieved a maximum of 99.01 percent. For example, we found that OH --- O, NH --- O, CH --- O hydrogen bonds, NH --- π, OH --- π, CH --- π interactions as well as dipole-dipole interactions between CEX and the nanoparticles could all be used to connect the CEX and the nanoparticles. There is a strong correlation between the output and target values acquired by BBD-RSM and ANN fits. Fe3O4@SIO2 proved to be an excellent tool for eliminating CEX.
1. Introduction
Antibiotic residues on the environment have been increasingly controversial in recent years. As a result, antibiotic depletion control has been explored extensively [1]. The development of antibiotic resistance is a big worry, even though medications are unlikely to have an immediate negative effect on aquatic microbes [2]. Antibiotic Cephalexin (CEX), which belongs to the penicillin group, is used in medicine and veterinary medicine to treat gastrointestinal and bacterial infections of the intestines and urinary tract [3].
One of the most widely used antibiotics is the beta-lactam family, which includes the molecule CEX [4, 5]. More serious side effects include limited biological decomposition capacity, high toxicity, carcinogenic, mutagenic, mutagenic effects, DNA and lymphocytes being damaged, allergic responses being increased, and the development of medication resistance [6]. Because of their complicated structure and limited biodegradability, the degradation of these substances is seen as a significant environmental concern [7].
Cephalexin (CEX) is an antibiotic that comes from the cephalosporin family and is widely regarded as the second-most consumed group of antibiotics globally [8]. According to research evidence, it is known to be used as a treatment method normally to treat respiratory and urinary infections [9]. However, the discharge of contaminated water from pharmaceutical facilities, hospitals, homes, and agro-industrial facilities containing pharmaceuticals such as CEX into the environment can have adverse impacts on human health, increase antibiotic resistance, and inhibit the growth of algae and beneficial microorganisms in the environment [10]. Furthermore, the removal of cephalexin from effluents has not been fully studied. Adel et al. [11] worked on the removal of cephalexin antibiotic and heavy metals from pharmaceutical effluents using Bacillus subtilis strain. The removal of cephalexin from effluents by activated carbon prepared from alligator weed was studied by Miao et al. [12]. Furthermore, Miao et al. [12] studied the removal of cephalexin and erythromycin antibiotics and their resistance genes, by microalgae-bacteria consortium from wastewater treatment plant secondary effluents. In their findings, cephalexin and erythromycin had a removal efficiency of 96.54% and 92.38%, respectively. According to Miao et al. [12], cephalexin has been observed to be flowing out of hospital wastewaters, effluents, and wastewater treatment plants at mg L–1 levels. In the light of the above, it becomes imperative to develop and deploy new methods to effectively remove these contaminants from aqueous solutions using sustainable technologies under favorable economic conditions. Consequently, standard physicochemical and biological treatments can only breakdown a limited number of these compounds [13]. Reverse osmosis, ozonation, and other sophisticated treatment procedures can remove antibiotics from wastewater; however, these technologies may not be cost-effective, energy-efficient, operational, or ecologically justifiable [14, 15]. As a result, scientists are always searching for new approaches that are more effective, less expensive, and less harmful to the environment. Antibiotics, for example, are well-suited for elimination by adsorption [16]. Adsorption is a well-known approach for removing antibiotics that is both effective and ecologically safe, based on past research.
Imaging, sensing, targeted medicine administration, gene transfer systems, and artificial implants are all examples of how nanotechnology is being applied in medical research today [17, 18]. With these uses, nanotechnology can be beneficial to human health care, and it can be hoped that it could lead to breakthrough advancements in fields such as medicine, communication, genomics, and robotics [19]. Fe3O4@SIO2 magnetite nanoparticles have been the primary focus of CEX adsorption. It is difficult to extract, process, and investigate uncoated magnetite nanoparticles since they agglomerate easily and have limited usefulness. As a result of this, we have recently reported on the electrochemical production of magnetite nanoparticles without the need of any organic solvent or surfactant [20].
The number of experiments to be conducted has been the subject of several theories and methodologies over the last few decades. Using statistical and mathematical methods, the response surface methodology (RSM) examines the influence of several independent variables [21]. There are various benefits to using the Box Behnken design approach (a sort of RSM), including a statistical model that describes the process as exactly as possible while decreasing the number of test stages, time, cost, and material consumption [22]. Due to its ability to apply learning methods to discover input–output relationships for complex, nonlinear dynamic systems, artificial neural networks (ANNs) are increasingly being used as prediction tools in a variety of industries, including engineering [23].
As a result of this, the researchers conducted a study on the removal of CEX from an aqueous medium using silica-modified iron nanoparticles (Fe3O4@SIO2). CEX antibiotics were also shown to have a wide range of efficacious factors ranging from contact time to temperature to dose. We used response surface methodology (RSM) in our research to better understand how different processes interact with one another and how to improve adsorption efficiency. Tested/fitted for the data were the artificial neural network (ANN) models CEX molecules and Fe3O4@SIO2 were also studied using density functional theory in a computer investigation of the interaction/mechanism (DFT). In order to get a better knowledge of the CEX adsorption, the DFT was also employed to determine the chemical potential (
2. Materials and Methods
2.1. Materials
Cephalexin-CEX (C16H17N3O4S, 347.39 g/mol, 99.6%) were supplied by Sigma-Aldrich USA. The following diagram depicts the molecular structure of CEX (Figure 1). All solutions and buffers were prepared using distilled water. Citric acid and citrate acid were utilized to make a citrate buffer with a certain pH. The Thermo Scientific GENESYS 180 UV-visible spectrophotometer was used to obtain the concentration of CEX at 263 nm wavelength.

The chemical structure of CEX.
2.2. Synthesis of Fe3O4 Nanoparticles
The nanoparticles were produced using the chemical precipitation process. A 100-milliliter flask of dehydrated water with nitrogen gas, 4.5 g of FeCl3.6H2O and 2.78 g of FeCl2.4H2O is stirred constantly. This was achieved by adding ammonia solution (25 percent) and letting it sit until the solution became black. 30 minutes of stirring and heating to 80°C resulted in a precipitate that had developed. It was rinsed three times with ionized water, then twice with ethanol, after the precipitate produced during the heating process. The magnetic nanoparticles (Fe3O4) were finally isolated from the magnetic solution by using a magnet.
2.3. Coated Iron Nanoparticles with Silica (Fe3O4@SIO2)
Using the sol-gel process, silica was used to coat the nanoparticles. A solution comprising 150 mL ethanol, 2 mL water and 4 mM tetraethyl orthosilicate (TEOS) was added to the previous step’s nanoparticles after they had been washed and dried. After 30 minutes of stirring, it was cleaned five times with ethanol before being dried. Drying and powder production of coated nanoparticles were carried out in a laboratory setting at room temperature.
2.4. Characterization of the Fe3O4 and Fe3O4@SIO2
The Fe3O4@SIO2 and Fe3O4 morphological structures were examined using a LEO instrument, model 1455VP scanning electron microscopy (SEM). Experimenters employed Fourier-transform infrared spectroscopy to recognize the chemical groups contained in the Fe3O4 and Fe3O4@SIO2 samples.
2.5. Batch Treatment Study
As part of each experiment, 10 milliliters of the CEX antibiotic solution were in contact with Fe3O4@SIO2 for a predetermined time duration at a predetermined temperature and pH. After reactions, 0.5 cc of samples were taken and filtered through a 0.22-micron filter. UV-Vis
2.6. Experimental Design and Statistical Analysis
An RSM of Design expert version 8.0.7.1 software, USA, was utilized to create 54-run experiments that included 8 axial and 16 factorial point locations as well as 6 replicates at the center points (Table 1) which included the dependent factors including pH, time, dose, and CEX concentration. In this investigation, the independent variables were coded in accordance with Equation (2):
Predicted and experimental values for CEX by Fe3O4@SIO2.
The experimental levels tried for CEX onto Fe3O4@SIO2 particles are indicated in Table 2. The empirical second-order polynomial regression model (Equation (3)) was used to fit the experimental data. The experiments were run following the method mentioned in Section 2.5 following the variables’ conditions in Table 1.
Experimental levels tried for CEX onto Fe3O4@SIO2 particles.
2.7. Computational Methodology
To get further understanding on CEX adsorption, a computer analysis of the CEX molecule was conducted. The computations were carried out using the density functional theory (DFT) functional PW6B95D3 in conjunction with the Def2-TZVP basis set. The optimizations were carried out using the tight option selected for accuracy. We utilize the ultrafine grid for integrals because it is suggested for molecules with a large number of tetrahedral centers, for optimizing molecules with a large number of soft modes, and for calculating extremely low-frequency modes of systems. The energies of the orbitals were calculated, especially those of the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO). The HOMO and LUMO energies were utilized to determine the chemical hardness (
In addition, the ionization potential (IP) and the electronic affinity (EA) were calculated using the following formula [29, 30]:
3. Result and Discussion
3.1. Computational Analysis
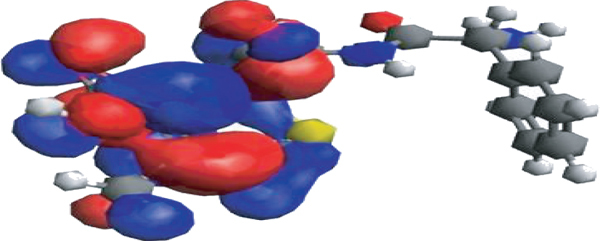
As can be seen in Figure 2(a), the CEX molecule’s structure has been optimized to the PW6B95D3/Def2-TZVP level of theoretical theory. Additionally, we have shown in Figure 2 the structural distribution of the Homo and Lumo, as well as the optimization of the structure (Figures 2(b) and 2(c), respectively). Table 3 lists the CEX molecule’s computed DFT-based characteristics.

(a) Structure of CEX molecule as optimized at the PW6B95D3/Def2-TZVP level of theory. (b) HOMO; (c) LUMO of the CEX molecule.

DFT-based descriptors of the CEX molecule as calculated at the PW6B95D3/Def2-TZVP level of theory. The energy unit used in the table is eV.
There appears to be a consistent distribution of the HOMO across the structure. CEX will be attracted to the nanoparticles through the various -NH, -OH, and -CH groups of the nanoparticles molecule’s molecule, as seen in this diagram. The LUMO is also found on the other side of the molecule, but not in any of the benzenoic rings. The CEX molecule acts as a proton donor for the nanoparticles, making it possible to interact with them. Hydrogen bonds, NH —- O, OH —- O, CH —- O, NH --- π, OH --- π, CH --- π interactions as well as dipole-dipole interactions are all demonstrated in the HOMO and LUMO experiments as possible mechanisms for mediating interactions between the CEX molecule and nanoparticles. Because of this, it is been discovered that nanoparticle molecules may strongly adsorb the CEX molecule via the various interactions indicated above in studies of its HOMO and LUMO. And the molecule’s moderate chemical hardness value (2.79) eV confirms that it is highly reactive.
Computational studies were conducted in this work to give an explanation for the interactions between the CEX molecule and iron nanoparticles covered with a layer of iron oxide. A thorough description of these interactions would include all the molecules involved and compute the interaction free energy at the relevant experimental temperature. Because the experiment is being replicated, it is necessary to make computations in solution. Molecular dynamics simulations or a combination of quantum mechanics and molecular mechanics molecular dynamics simulations are unable to handle the complexity of these computations. Our calculations have been restricted to the analysis of the CEX molecule by offering certain DFT-based descriptors that might explain the probable interactions, due to tractability and affordability.
3.2. Characteristics of Nanoparticles
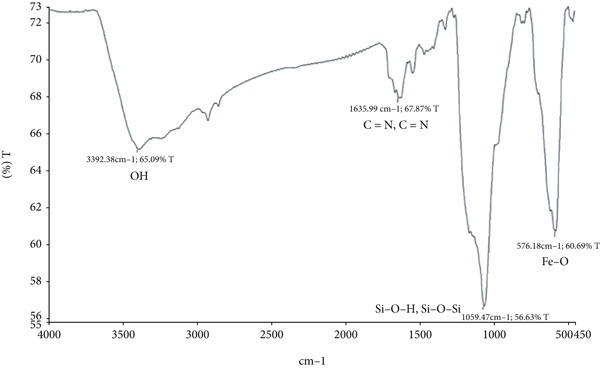
Particle morphology, shape, and size may be determined using scanning electron microscopy (SEM) techniques [31, 32]. Images of Fe3O4 and Fe3O4@SIO2 particles are revealed in Figure 3. Compared to the Fe3O4, the Fe3O4@SIO2 particles seem rounder and more uniform in shape in SEM images. Cheng et al. [33] also obtained round Fe3O4@SIO2 particles from their study. Figure 4 shows the FTIR data for the Fe3O4 and Fe3O4@SIO2 particles. Si-O-H and Si-O-Si have been detected in both spectra after silicon has been applied to Fe3O4@SIO2 particles, confirming that silicon has been applied to the Fe3O4 particles. Both particles shared functional groups including OH, Fe-O, and C=N. Similar result was observed by Du et al. [34] when FTIR analysis was done on both particles.

SEM micrograph of Fe3O4 (a) and Fe3O4@SIO2 (b).


FTIR spectra of (a) Fe3O4 and (b) Fe3O4@SIO.
3.3. Model Fitting, Statistical Analysis, and Optimization
According to Table 4, the adsorption experiments were executed. Design expert 8.0.7.1 software, USA, was used to evaluate and interpret the produced data. As shown in Table 4, the quadratic model outperforms all other models (interactive, linear, and cubic) except the cubic model in terms of
Fit summary of the analysis.
Model summary statistics and sequential model sum of squares for CEX by nanoparticles.

The plot of the predicted values versus the observed values of CEX adsorption on Fe3O4@SIO2.
Predicted and experimental values for CEX by Fe3O4@SIO2.
Response surface quadratic model ANOVA findings are shown in Table 7. Analyzing ANOVA results helps to determine if the quadratic model produced is statistically appropriate for describing processes like CEX in this study’s study range. An
ANOVA for the response surface quadratic model of CEX adsorption on Fe3O4@SIO2.
The model conveying the response (CEX removal efficiency) and process factors is shown as Equation (6):
In this regard: (
To accurately forecast the output response, the above model equation’s unimportant terms can be deleted (Grenni et al. 2018). CEX concentration, however, has a synergetic effect on the adsorption of CEX. Also, the interaction between pH and time has a synergetic effect on the process as well as the interaction between pH and CEX concentration. Solution pH, reaction duration, dose, temperature, and the interaction between pH and dosage have an antagonistic effect on CEX oxidation looking at the signs in Equation (6). The higher
Figure 6 depicts the normal percent probability vs. internally studentized residuals (Figure 6), which show that there was little divergence from the norm. There is no need to alter the data because the graphical points follow a straight line [39]. Independent process factors are examined for their combined influence on a response using the RSM statistical approach [40]. Response surface plots of any two process factors versus any two additional process variables were used to analyze the interaction of the process variables and their associated impacts on CEX adsorption.
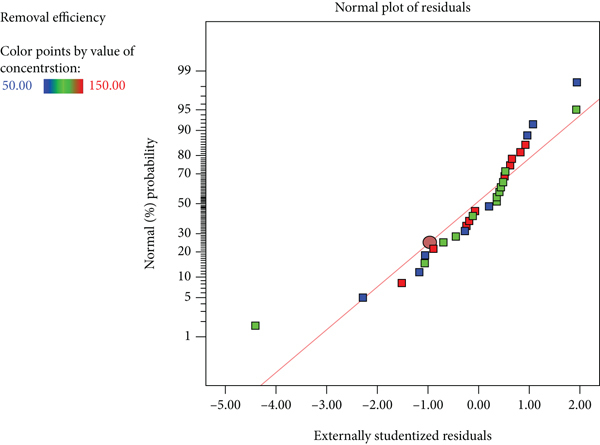
Normal probability plots of the residuals for CEX adsorption on Fe3O4@SIO2.
The 3D surface plots of the relation between the process variables and their associated output responses are shown in Figures 7(a)–7(f). The adsorbing material’s functional groups and the pH of the solution play an important role in the adsorption process [32, 41]. The enzyme ionizes the amino acids in the molecule and alters their nature when the pH is acidic, resulting in the placement of the hydrogen ion on the nitrogen atom of the protein molecule [36, 38]. There are hydroxide ions attached to reactive groups at alkaline pHs, which have positive charges and prevent electrons from being transferred from the substrate to active sites of lacasses [42]. In somewhat acidic circumstances, the pH range of 4-6 is optimal for most fungal laccase oxidation [36, 38]. Increasing or reducing the pH of this value has the greatest effect on the elimination of CEX in both free and dose methods. There was also less oxidation. In contrast to the efficiency decline caused by nanoparticles, this decrease happens in the adsorption process and has a lesser slope. Figures 7(a) and 7(b) demonstrates that the reaction time and elimination of CEX is maximum and occurs in 60 minutes at pH 3.

3D surface plots of the interactive effect of CEX on Fe3O4@SIO2.





Increasing the contact period from 15 to 60 minutes significantly improves the effectiveness of antibiotic CEX removal via adsorption, since nanoparticles have a direct relationship with removal efficacy. The maximum removal efficiency is achieved in 60 minutes for both procedures. Figures 7(a)–7(c) show the effect of Fe3O4@SIO2 dose on the efficacy of eradicating CEX antibiotics in the range of 0.5 and 1 g during nanoparticle adsorption. CEX removal was increased with elevated Fe3O4@SIO2 dose; similar observation was made when zeolite coated with magnetic Fe3O4 nanoparticles was used for CEX removal [43]. By raising the CEX concentration from 50 mg/L to 150 mg/L, the clearance rate was significantly decreased (Figures 7(a), 7(d), and 7(f)). Increasing the concentration led to more saturation of the Fe3O4@SIO2; similar trend was observed in past study where activated carbon derived from pomegranate peel was used [44]; the same authors observed an optimum pH of 5. In addition, the percentage of deletions rose as the interaction duration grew from 15 minutes to 60 minutes (Figures 7(c), 7(e), and 7(f)). At 25°C, pH 5.83, 37.67 min, 0.8 g Fe3O4@SIO2, and 118.01 mg/L CEX, the removal efficiency achieved a maximum of 99.01 percent, according to numerical optimization data (Figure 8). Fe3O4@SIO2 was shown to remove CEX with an efficiency of

Desirability effect for the CEX-Fe3O4@SIO2 process.
3.4. Artificial Neural Network (ANN) Modeling
Using a tanh-sigmoid activated hidden neuron and a linearly activated output neuron in a two-layered feed-forward network, the parameter was estimated. Levenberg-Marquardt backpropagation was used to train the network. Shown below is an example of how a neural network may look (Figure 9). An input into a neuron with a value between +1 and -infinity may be squashed to produce an output of -1 to 1 using the tanh-sigmoid activation function. Tanh-sigmoid activation function is a mathematical representation of this function.

The proposed neural network architecture.
The linear transfer function takes the input to the neuron (in this case output from the tanh-sigmoid hidden layer) and turns it into a continuous number. The linear activation function calculates the neuron’s output by simply returning the value passed to it [46, 47]. Mathematically, the linear activation function is represented as.
The cost (error) function is the function used to minimize the error between the output (result by the network) and the target [48]. Because parameter estimation is a multiple regression analysis problem that output continuous values. mean squared error cost function was used to train the network. The mean squared error cost function is given below:
The procedures with which the network was trained can be summarized in the following steps:
Import or read the training data (time and pressure data of the well test) from an excel data sheet to MATLAB as variables Choose the training algorithm in this case the Levenberg-Marquardt backpropagation Choose the number of neurons in this chase twenty (8). Preprocess the input data to prevent outliers Divide the input data randomly into three parts for training, validating, and testing of the algorithm (in this case the division was made in the ratio of 80 : 10 : 10) to prevent overfitting Select the cost or error (in this case the mean square error) function to be minimized Train the network to meet the goal (until the cost function is minimized and the variables converge).
Figure 10 shows how well the data were fitted to provide the coefficient of determination for the training, validation, and testing sets after 13 iterations. The total index of fitness is 0.9777, with a mean squared error of 31.44702. A suitable stopping point (Figure 11) was found at epoch 7 when the best validation performance of 13.203 was achieved. The

Regression plot for CEX onto Fe3O4@SIO2.

Performance plot for CEX onto Fe3O4@SIO2.
4. Conclusion
Box-Behnken design-response surface (BBD-RSM) approach was used to examine the interactions between process factors on the liquid-phase adsorption of cephalexin (CEX) onto silicon-coated iron nanoparticles (Fe3O4@SIO2). PW6B95D3/Def2-TZVP level of theory was used to calculate CEX’s density functional theory- (DFT-) based descriptors. The elimination efficiency was found to be at its peak at 99.01 percent in the study. The elimination efficiency was found to be 99.01 percent at 25°C, pH 5.83, 37.67 min, dosage 0.8 g Fe3O4@SIO2, and 118.01 mg/L CEX, with the highest concentration tested. A variety of interactions between the CEX molecule and the Fe3O4@SIO2 might be established through OH --- O, NH --- O, CH --- O hydrogen bonds, NH --- π, OH --- π, and CH --- π interactions as well as dipole-dipole interactions. There is a strong correlation between the
Footnotes
Data Availability
No data were used to support this study.
Conflicts of Interest
There are no conflicts of interest declared by the authors.
Acknowledgments
We are thankful to Dr. Swagata Ghosh, Assistant Professor of English, Kumaraguru College of Arts and Science, Coimbatore, India, for editing this manuscript.
