Abstract
Teff straw, a by-product of Teff, mainly available in Ethiopia, has not been studied much for biosorbent production. The present study has investigated the effects of modification and optimization of process parameters (viz., concentration of modifying agent (H3PO4 and KOH), modifying temperature, and modifying time) on the Cr (VI) removal efficiency of using chemically activated Teff straw biosorbent by RSM followed by BBD. The maximum Cr (VI) removal was obtained using an H3PO4-modified Teff straw biosorbent of 92.5% with 2 M concentration of the modifying agent, 110°C, and 4 h. Similarly, maximum Cr (VI) removal using KOH-modified Teff straw biosorbent of 95.2% was obtained with 1.5 M activating agent concentration, 105°C activation temperature, and 3.5 h activation time. In addition, the effects of adsorption parameters (viz., biosorbent dosage, temperature, initial concentration of Cr (VI), and contact time) were investigated. The maximum removal efficiency was attained at 2 g of biosorbent dosage, 4 h contact, 75 mg/L of initial Cr (VI) concentration, and 25°C sorption temperature. In addition, isotherm, kinetic, and thermodynamic studies for Cr (VI) biosorption were studied. The experimental adsorption data were well fitted with the Langmuir isotherm and pseudo-second-order kinetic model with higher correlation coefficient in both untreated and chemically modified Teff straw biosorbent. The investigated thermodynamic parameters (
1. Introduction
The frequent utilization of fossil-based products becomes a significant cause of economic breakdown and environmental degradation. Therefore, alternative, cost-effective, abundant, environmental-friendly, and biorenewable feedstocks must be investigated [1, 2].
Lignocellulosic biomass is a typically nonedible material mainly deduced from woody biomass, nonwoody biomass, and organic wastes. In the last few decades, interests have been grown almost in all developed, developing, and underdeveloped countries for the utilization of lignocellulosic biomass for various economic sectors, namely, renewable energy, dispersants, biocomposites, fertilizer, textile, pharmaceuticals, adsorbents, additives, phenolic compounds, and food industry [3, 4]. However, due to rapid industrial development, discharges of pollutants such as heavy metals to the environment through wastewater have remarkably been increased. Thus, environmental pollution with heavy metals is a serious issue worldwide, posing threats to humans, animals, and plants and the overall ecosystem’s stability [5, 6].
In the industrial wastewater treatment sector, heavy metals such as chromium, copper, cadmium, lead, zinc, and nickel are considered more toxic and receive more researchers’ attention. Chromium (Cr) is a harmful heavy metal and exists in various oxidative forms. Cr (VI) is considered more harmful and toxic due to its high carcinogenic and resistant properties than Cr (III) [7, 8]. Various Cr contamination sources include electroplating, leather tanning, textile industries, metal finishing, nuclear power plants, and chromate preparation. Effluents from tannery industries are among the problematic environmental issues faced by the Ethiopian manufacturing sector. Tannery effluents have been reported to contain a high amount of heavy metals such as chromium and organic pollutants [1, 2, 8].
In developing countries, 70% of industrial effluents are discharged into the environment without any treatment action. Industries that contribute as a source for such effluents are the processing of radioactive materials, the manufacture of electrical equipment, electroplating, leather tanning, metal finishing, dyes and pigments, mining operations, fossil fuel combustion, metallurgical operations, mineral processing, fly ash from incinerators, refining ores, pesticides, and preservative [8, 9]. The increasing levels of toxic and hazardous heavy metals that interfere with the biosphere cause immediate economic crisis and hazardous health effects.
To date, researchers have focused on investigating cheap and readily available biomass as a source of biosorbent instead of using very expensive, nonrenewable, non-eco-friendly, nonbiodegradable, and continually depleted fossil sources [7, 10]. Synthetic adsorbents like zeolites, silica gel, alumina, concrete, and polymers also have high production costs and less adsorption capacity than biosorbents obtained from biomass sources [11, 12]. Various researchers have given special attention to biosorbents from agricultural and industrial residue due to high porosity and their availability in large quantities at low cost [1, 2, 13].
Biosorbents prepared from lignocellulosic biomass could be promising materials to remove various toxic, hazardous, and nonbiodegradable organic and inorganic pollutants such as heavy metals, recovery of high-value proteins, specific organic compounds (phenolic compounds, polycyclic aromatic hydrocarbons, organic pesticides, and herbicides agrochemicals), dissolved compounds, and suspended solids of water from the effluents of various industries [10, 14, 15]. Biosorption has been more feasible, economical, simple, and advantageous over the well-known conventional techniques enclosed phytoremediation, ultrafiltration, ion exchange, reverse osmosis, electrodialysis, chemical precipitation, and electrochemical oxidation [12, 16]. Various authors have addressed the biosorption process which provided various advantages: low operating cost, readily available, high adsorption capacity, minimum chemical and/or biological sludge, no additional nutrient requirements, regeneration of biosorbent and the possibility of metal recovery, and short operation time and improved selectivity for specific metals of interest. However, the commercial treatment methods are less effective and more expensive in capital and operational costs [17–20].
In Ethiopia, various potential agricultural/industrial waste sources are unlimited [18, 21]. Among the many biomass wastes, Teff straw is one of the abundantly available, feasible, and locally sourced materials for synthesizing low-cost biosorbent. Various researchers have shown that adsorbents obtained from various agricultural wastes, such as Teff straw, have been widely used to remove different toxic heavy metals from wastewater. Characterization and utilization Teff straw for chromium removal from wastewater, viz., kinetics, isotherm, and thermodynamic modeling, have been reported by Wassie and Srivastava [22]. Wassie and Srivastava [23] have also studied the chemical modification of Teff straw using NaOH, H3PO4, and ZnCl2 for adsorptive removal of chromium from aqueous samples. Tadesse et al. [18] have reported Teff straw as a potential low-cost material for removing Cr (VI) from aqueous samples. Although these researchers have studied Teff straw as a potential low-cost material for biosorption of Cr (VI) from aqueous samples, they have not reported the response surface methodology parametric optimization (viz., concentration of acid/base, modification temperature, and time) of chemical modification of Teff straw using various acids and bases to enhance the adsorption capacity.
The present study has addressed (1) the synthesis of modified biosorbent using chemical modifying agents (H3PO4 and KOH) and characterization of the modified biosorbent from Teff straw biomass and (2) the effects of modification and optimization of process parameters (viz., concentration of the activating agent, activation temperature, and activation time) on the removal efficiency of Cr (VI) using the modified biosorbent from Teff straw. In addition, the kinetics and thermodynamics of Cr (VI) adsorption using modified biosorbent from Teff straw were also analyzed.
2. Materials and Methods
2.1. Materials
All chemicals used throughout this study were attaining the analytical reagent grade. The reagents were required to prepare stock solutions for chemical modification, adsorbate preparation, and dilution purposes. The Teff straw modification process required phosphoric acid, H3PO4 (purity 85.5%), and potassium hydroxide, KOH (98.08% pure). Sodium hydroxide, NaOH (98.08% pure), was employed for the determination of hemicellulose content of Teff straw, whereas sulfuric acid, H2SO4 (purity 98%), was used to determine the lignin content. Cr (VI) stock solution was prepared from potassium dichromate (K2Cr2O7). All required solutions were prepared with analytical reagents using distilled water. 1,5-Diphenyl carbazide (DPC) was used to provide complexation with Cr (VI) and form pink color to determine the equilibrium concentration of residual Cr (VI) in the range of visible region in UV-visible spectrophotometer characterization. Methanol was required to dissolve the ligand DPC.
2.2. Equipment and Analytics
A laboratory test sieve (BS 410-1, Endecotts Ltd., England) was used to separate the required working size of the sample (pan to 0.50 mm). An electrical blender (OE-999, 220-240 V, 50/60 MHz, 350 W Seven 7 STAR, Germany) was employed to reduce the straw up to the desired average size. Digital balance (Mettler Toledo, 1118330367, 2000, Switzerland) was used to measure weights. Different size conical and Erlenmeyer flasks and beakers (Borosilicate Glass: 2000, 1000, 500, 250, 100, and 50 mL) were used in the preparation and dilution of the solutions.
A water bath (Weston-S-Mare Avon, Nickel Electro Ltd. 1008E, 50/60 Hz, 1000 W, England) was used to heat the solutions to determine the hemicellulose and lignin content of Teff straw. Jenway 3510 pH Meter (Barloworld Scientific Ltd., Dunmow, Essex, CM6 3LB, UK) was employed to fix the appropriate pH of the solution during the preparation of biosorbent and batch adsorption experiments. UV-visible spectrophotometer (Cary 50, Varian Australia Pty Ltd., Australia) was required to determine equilibrium concentrations of the adsorbate Cr (VI) after adsorption. Fourier transform infrared (FTIR) spectroscopy (IR Affinity-IS, Shimadzu, Japan) was used to determine the availability of organic and inorganic chemicals and/or functional groups that favor high adsorption capacity. Scanning electron microscope (SEM) (JEOL JCM-6000Plus, Germany) was provided to determine topography, morphological properties, essential information, crystallography, and orientation of grains, for acid- and base-treated straw. X-ray diffraction analysis (40 kV, 30 mA) was employed to analyze the crystalline structure of the prepared biosorbent on the adsorption capacity.
2.3. Collection and Pretreatment of Teff Straw
The natural Teff straw was collected from farmlands around Bishoftu, Ethiopia. The straw was thoroughly washed with tap water several times to eliminate the surface adhered particles and other soluble impurities. The washed samples were kept initially drying in sunlight for 12 h. After removing excess water content, the straw was transferred into an electric oven to eliminate extra moisture for 6 h at 105°C. In order to improve the modification process, the dried Teff straw was grounded and sieved to reduce cellulose crystallinity and attain homogenous particle-sized material in the range of pan to 0.50 mm using an electrical blender. The grounded straw was again washed thoroughly with hot distilled water to remove the extra impurities and soluble components like reducing sugars. Then, it was oven dried for 8 h, at 105°C. Finally, the untreated Teff straw (UTS) was placed in a well-dried and airtight desiccator for further use.
2.4. Biosorbent Preparation
The procedure followed by Doboy et al. [24] was adapted to prepare biosorbent from Teff straw biomass. The grounded and well-dried Teff straw was used to prepare a potential biosorbent by chemically modifying the original structure of the straw using H3PO4 and KOH as activating agents. Biosorbent preparations were carried out by applying batch reactor mode using separate identical beakers of 500 mL. During the preparation of biosorbent, the effects of process variables (viz, concentration of activating agents (H3PO4 and KOH), activation temperature, and activation time) were investigated in the case of one variable at a time (OVAT) by fixing other variables constant. The grounded dried material, i.e., UTS, was chemically modified on the heating mantel (450°C max.) with the help of a magnetic stirrer in the stated temperature scales. On the other hand, the concentration of activating agents and activation temperature vary (0.5, 1, 1.5, 2, 2.5, and 3 M and 40, 60, 80, 100, 120, and 160°C), respectively, for both activating agents. The activation temperature was adjusted by using a thermometer (250°C max.).
Moreover, the activation time was set at 1, 2, 3, 4, 5, and 6 h corresponding with activating agent concentration and activation temperature. Finally, 15 g of UTS sample was dehydrated with 100 mL of H3PO4 and KOH solutions of the activating agents. The reactor (beaker) was covered by aluminum foil throughout the modification process to prevent escaping of condensable materials. After the modification, the material was cooled at ambient temperature and washed with plenty of distilled water until the filtrate attains neutrality. Meanwhile, the modified and filtered Teff straw was transferred into an electric oven until attaining constant weight at 110°C. The resulting Teff straw biosorbent material is named as follows: acid-treated biosorbent (ATB) and base-treated biosorbent (BTB), modified with H3PO4 and KOH, respectively, as given in Figure 1.
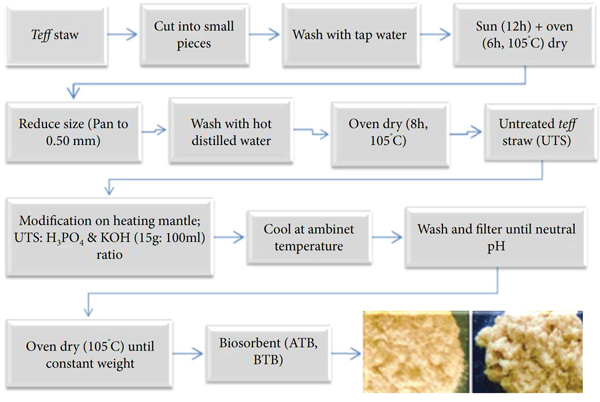
Schematic representation of Teff straw biosorbent preparation.
2.5. Compositional Analysis of the Teff Straw
Agricultural wastes mostly contain three major compositional components, cellulose, lignin, and hemicellulose, along with trace amounts of pectin, protein, extractives, and ash [25, 26]. Determining structural components of lignocellulosic material is crucial to analyze the overall efficiency (i.e., the compositional analysis of biomass and its effects in the metal adsorption process) of the process designed to convert biomass to biosorbent. Mainly, the biosorption of metal ions depends on the available binding active sites of the biosorbent. However, the available binding active sites of the biosorbent are also dependent on the composition of the biomass, i.e., its cellulose, hemicellulose, and lignin. Thus, the modifying/activating agents such as acids and bases to enhance the available active sites of the original biomass altered these compositions of biomass [18, 25]. Various methods existed to examine the compositional constituents of given agricultural biomass, such as TGA, X-ray diffraction, and chemical analysis. However, the chemical gravimetric analysis is most promising than others due to its simplicity, economically viable, and readily available procedure to identify the constituents of the straw [24, 27, 28]. Specifically, under TGA, compositional analysis of biomass might have some errors. However, hemicellulose decomposed up to 270°C cellulose also decomposed 240–350°C, and lignin might be hydrolyzed 280–500°C [22, 24]. Therefore, the great challenge is that cellulose might be interfering in the structural value of others and vice versa at the specified temperature ranges. Thus, in this study, gravimetric analysis was employed to identify the amount of three lignocellulose components (cellulose, hemicellulose, and lignin) in straw by the method proposed by Abbas et al. and Maisyarah et al. ([27]; Maisyarah et al., 2019). Experiments under a compositional analysis of Teff straw were replicated three times and reported the average value of each result.
2.5.1. Determination of Extractives
The Soxhlet extraction unit setup was applied to separate extractive free biomass from extractive-laden material following the method represented by Adeeyo et al. [28]. 400 mL of acetone solvent was used to determine the extractive content with transferred 3.0 g dried straw (A) into cellulose thimble. The extraction process was carried out at 70°C for 4 h on the heating plate. After 4 h extraction, the extracted material was dried in an electric oven at 110°C until a constant weight was obtained (B). Therefore, the amount of extractive (%
2.5.2. Determination of Hemicellulose Content
The hemicellulose content of Teff straw was determined using chemical gravimetric analysis [28] with some modification. 250 mL Erlenmeyer flask was required to mix 1 g of dried extractive-free UTS biomass (B) with 150 mL of 0.5 M sodium hydroxide (NaOH) and boiled at 80°C for 3.5 h in hot plate. The sample was washed with distilled water until neutral pH ≈7.0 and filtered through Whatman 42 filter paper. The sample was dried in an oven at 110°C until a constant weight (C). Finally, the hemicellulose content (%
2.5.3. Determination of Lignin Content
The lignin content of Teff straw was also determined using chemical gravimetric analysis used by Adeeyo et al. [28] with some modification. First, 3 g of extractive-free sample (B) was transferred into 30 mL of 98% sulphuric acid in a 250 mL conical flask. At ambient temperature, the mixture was leftover 24 h. The black liquor was formed and then boiled at 120°C for 1 h on the hot plate. The soluble lignin derivates, lignosulfonates, formed by treating biomass at elevated temperatures (≈150°C) with solutions containing sulfate ions (1.5 to 3%). After filtering the mixture, the solid residue was washed using distilled water until the sulfate ion was undetectable. The washed water of sulfate ion was analyzed and/or detected via titration process with 10% of barium chloride solution obtaining precipitate in a colloidal form. The sample was dried in an oven at 110°C until a constant weight was obtained (D). The final insoluble lignin content (% weight) was determined.
2.5.4. Determination of Cellulose Weight
Considering an assumption that cellulose, hemicelluloses, lignin, and extractives are only components of the untreated Teff straw, the cellulose content was determined using the difference. Assuming 100% to the total amount of Teff straw sample applied in the experiment and then calculating the difference between the initial sample weight with other three components (viz., extractives, hemicellulose, and lignin) weight during the experimental process, the weight of cellulose (E) would be as represented in
2.6. Batch Biosorption Studies
An adsorption equilibrium data is critical to provide optimum parameters and understand the mechanism of adsorption. This study used batch adsorption experiments to investigate parametric effects such as contact time, biosorbent dosage, initial Cr (VI) concentration, and temperature. The 1000 mg/L Cr (VI) synthetic stock solution was prepared by adding 0.1 g of dried K2Cr2O7 in 100 mL distilled water in a volumetric flask. To examine effect of parameters, contact time (1-6 h), initial Cr (VI) concentration (50-200 mg/L), biosorbent dosage (0.5, 1, 1.5, 2, 2.5, and 3 g), and temperature (25-50°C) were applied. The Cr (VI) solutions were taken in a 250 mL conical flask with the pH adjustment at 2. The solutions were stirred at a constant speed in an automatic shaker to achieve a high interfacial contact area for a better mass transfer process. Vacuum filtration was applied to separate the supernatant from the chromium-containing biosorbent. Finally, the remaining or residual concentrations of Cr (VI) were determined by a Varian UV-visible spectrophotometer (
2.7. Adsorption Isotherm Studies
Langmuir and Freundlich’s models were the most widespread and preferred expression of adsorption equilibrium applied in vast adsorbate concentrations. Shokoohi et al. [29] adopted the assumptions in the Langmuir model: monolayer coverage, all surface sites are alike and only can accommodate one adsorbed atom, and the ability of a molecule to be adsorbed on a given site independent of its neighboring sites occupancy. Based on these assumptions and a kinetic principle (rate of adsorption and desorption from the surface is equal), the Langmuir equation can be written in the general form of nonlinear equation (6a). The values of
The
2.8. Adsorption Kinetic Studies
Adsorption kinetic studies were conducted in batch reactions using 100 mL of Cr (VI) solution with 100 mg/L initial concentration of Cr (VI) in 250 mL Erlenmeyer flasks at constant pH (=2). 1 g of treated Teff straw biosorbent was added to the solution and agitated in the shaker at 30, 40, 50, and 60°C and analyzed at a time interval of 1 to 6 h. The adsorption kinetics was analyzed by pseudo-first-order and pseudo-second-order models. According to the procedure described by Overah [30], the linear functions of pseudo-first- and pseudo-second-order kinetic models are given in equations (9a) and (9b), respectively.
2.9. Thermodynamic Studies
Thermodynamic studies under the biosorption process showed us the spontaneity of the process, i.e., whether the process is spontaneous or not. The main parameters analyzed under thermodynamic studies were Gibb’s free energy (
3. Results and Discussion
3.1. Effect of Modification Parameters on the Adsorption Capacity of Biosorbent
As mentioned in Introduction, the modification variables (viz., agent concentration, activation temperature, and activation time) significantly affect the adsorption capacity of Teff straw biosorbent. Modification of the Teff straw plays a significant role in increasing porosity and specific surface area of the biosorbent. In this study, the chemical activation method was employed using base (KOH) and acid (H3PO4) activating agents that significantly affect the extent of activation. Strong base activation resulted in highly microporous adsorbents with high surface areas and created more active reaction sites than other activating agents [23, 31–33]. In this study, maximum adsorption capacity attained with acid- and base-treated Teff straw were 9.2 and 9.5 mg/g, respectively. KOH modification of Teff straw has increased the concentration of oxygen molecules of hydroxide, which strongly reacts with metal ions, whereas in H3PO4 treated, bulk phosphate groups cannot pass fast in the aqueous solutions resulting in adsorption capacity of Cr (VI) onto Teff straw surface to decrease. Generally, the results show that Teff straw was a potential biosorbent prepared at 1.75 M, 100°C, and 3.5 h for both activating agents (KOH and H3PO4). All three process variables (viz., activating agent concentration, activation temperature, and activation time) were investigated with the corresponding value of percentage yield and removal efficiency of Cr (VI). The percentage yield was determined from equation (11), and the removal efficiency,
3.1.1. Effect of Activation Temperature
The activation temperature ranges from 40 to 160°C were performed for both activating agents (H3PO4 and KOH) by fixing the concentration of chemical activating agents and activation time at 1 M and 3 h, respectively. The percent yield (yield%) and removal efficiency of the activated biosorbent changed significantly with activating agents. The activation with H3PO4 and KOH resulted in the highest percentage yield of 91.67 and 88.60%, respectively. This result indicated that KOH solubilizes the lignin structure of biomass, while H3PO4 also decomposed only the cellulose and hemicellulose structure of the straw. The activation temperature increased causing a decrease in the yield values due to more volatile matter from the straw. Therefore, the final biosorbent dry weight becomes lesser than the precursor dry weight of the base-treated Teff straw. Figure 2(a) shows that the yield (%) decreased whereas removal percentage,

Results of yield (%) and


3.1.2. Effect of the Activation/Modification Time
During the activation process, the activation agent concentration and temperature were kept constant at 80°C and 1 M, respectively. The effect of activation time on the yield (%) and
3.1.3. Effect of Activating Agent Concentration
The activating agents’ concentrations were set at 0.5, 1.5, 2, 2.5, and 3 M to investigate the effect of different concentrations on the yield (%) and
3.2. Characterization of Teff Straw and Modified Biosorbent
Both untreated and modified Teff straw were subjected to the physicochemical characterization that determines the adsorption capacity of the biosorbent, i.e., the compositional analysis of Teff straw, Fourier transform infrared (FTIR) spectroscopy, scanning electron microscope (SEM), X-ray diffractometer (XRD), and UV-visible spectrophotometer analysis.
3.2.1. Compositional Analysis of Teff Straw
Cellulose, hemicellulose, and lignin content of Teff straw were determined using chemical gravimetric analysis as
3.2.2. Fourier Transform Infrared (FTIR) Spectroscopy Analysis
The FTIR spectra of raw and chemically modified natural Teff straw biosorbent were used to determine the functional groups’ availability and vibrational frequency changes capable of binding metal ions. Different biomass has various types and amounts of binding groups that actively participate in adsorption. Figure 3 shows that Teff straw has some functional groups such as hydroxyl (-OH), amino (NH3 +), carboxyl (-CH2COOH), C=O), and amide (-CONH2) that are involved in the removal of Cr (VI) metal ions. The peak in the range of 1700-1450 cm-1 due to carboxylic and carbonyl groups from aldehydes, ketones, and aromatic rings from the lignin part is not detected after KOH treatment of Teff straw due to lignin removal. In the peak range of 1700-1450 cm-1, stretching vibration caused by -C=O vanished for the KOH treatment Teff straw as compared with the untreated Teff straw, which inferred that the addition reaction might be happened in the -C=O group. Moreover, the band at 1251.00 cm-1 triggered by -C-O- group divided into two small peaks, which showed that some substitution reaction took place on the side chain of -C-O- group. Thus, the results of FTIR showed that the improved adsorption efficiency of Teff straw due to KOH modification resulted from the increase of ether bond [23, 32, 33]. From 3700-3150 cm-1, peak range of -OH group stretching vibration, in both untreated and chemically modified Teff straw, is observed due to the presence of water. Therefore, it is predictable that peak strength rises in some functional groups owing to the increment in surface area and loss of specific functional group as a result of removal of specific lignocellulosic portion during chemical activation.

FTIR spectral of the raw and treated Teff straw.
3.2.3. Scanning Electron Microscope (SEM) Analysis
SEM analysis has been employed to observe the surface texture and morphology changes of both untreated and chemically modified biosorbents. The SEM images of untreated and chemically modified Teff straw biosorbents are given in Figure 4. The untreated Teff straw biosorbent, UTS, shows a relatively smooth surface (Figure 4(a)). However, after H3PO4 and KOH modification, the presence of pores and cracks made the surface of Teff straw more uneven and irregular (Figures 4(b) and 4(c)), which may be more helpful for adsorption. Specifically, for KOH-modified biosorbent, BTB shows significant surface modification (Figure 4(c)) that developed honeycomb, rough surfaces, nonuniform pores, and cavities. These were due to lignin and hemicellulose structure removal during the reaction between KOH and ester bonds. Nevertheless, H3PO4-modified Teff straw showed somehow lesser porous surface structures (Figure 4(b)) than KOH-modified Teff straw due to less reactivity of the phosphate groups.

The SEM micrographs of (a) UTS, (b) ATB, and (c) BTB.


3.2.4. X-Ray Diffractometer (XRD) Analysis
Figures 5(a)–5(c) depict XRD patterns of the untreated (UTS) and chemically modified Teff straw with acid (ATB) and base (BTB), respectively. In order to observe the crystalline structure of Teff straw biomass, X-ray diffractometer equipped with copper (Cu) radiant source with energy of 40 kV, an electric current of 30 mA, scanning speed of 3°/min, and scanning range of 10 to 80 degrees was carried out.
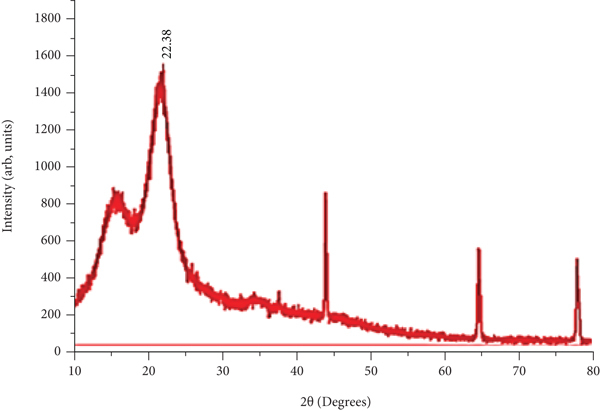
XRD patterns of untreated and treated Teff straw: (a) UTS, (b) ATB, and (c) BTB.


As shown in Figure 5(c), peak height formed due to the diffraction of crystalline regions of cellulose around 2θ of 22° increased in KOH-treated Teff straw compared to the untreated and acid-treated Teff straw. From XRD data, the area of all crystalline peaks (10876.42, 12081.26, and 10959.6) and area of all peaks (18048.3, 18838.56, and 16159.36) of untreated and modified Teff straw, respectively, could be found using Origin-pro and Excel software. The crystallinity index (equation (12a)) of UTS was found to be 60.3%. The crystallinity index increased from 60.3% to 64.1% and 67.8% with ATB- and BTB-modified Teff straw. The increase in the crystallinity of KOH-modified Teff straw is due to removing hemicellulose and lignin content. In XRD data, the broadening (
The XRD analysis results showed that both crystallinity index and crystallite size increased with acid- and base-treated Teff straw biosorbent due to removing amorphous lignin and hemicellulose with cellulose domination [23, 32, 33].
3.3. Optimization of Process Variables for Teff Straw Modification
Box-Behnken design (BBD) under response surface methodology (RSM) was conducted to study the effects of independent process parameters (viz., activating agent concentration, modification time, and temperature) involved in the preparation of Teff straw-based biosorbent and their interaction that affects the characteristic of biosorbent and biosorption efficiency to remove Cr (VI) metal ions. The response was optimized using Design Expert 7.0 statistical software tools through a two-level three-factor design via RSM to develop correlations between the biosorbent preparation variables and the response value that is Cr (VI)
Independent variables’ range and level: (a) the BBD of the experimental matrix with experimental and predicted values for ATB and BTB (b).
1: acid-modified biosorbent (ATB); 2: base-modified biosorbent (BTB); Exp: experimental; Pred: predicted.
3.3.1. Optimization of Process Variables for Biosorption of Cr (VI) Using ATB
The positive coefficient values in equations (13a) and (13b) indicated the positive interaction and impact of factors on the biosorption process, whereas the detrimental and interfering effect of the parameters on overall adsorption capacity is from negative coefficient value points. In order to identify the relevant model terms and fit the generated experimental data, the highest order polynomial empirical equation for Cr (VI) removal efficiencies (
ANOVA for the surface response model of Cr (VI) removal efficiency: (a) ATB and (b) BTB.
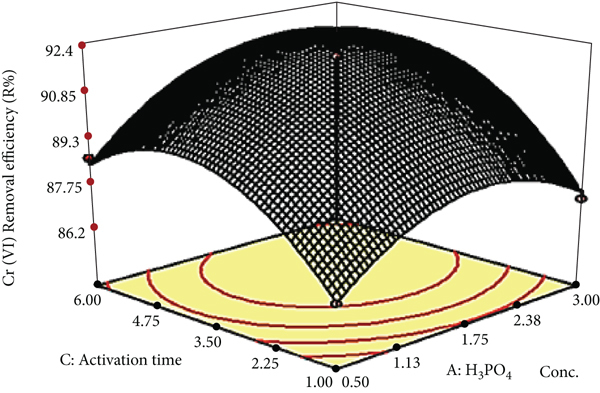
Figure 6 shows 3D response surface plots for the interaction effects of the independent variables on the Cr (VI) removal efficiency of the acid-treated biosorbent. Figure 6(a) depicts the combined effect of activating agent concentration versus activation temperature on the removal efficiency of Cr (VI) by fixing the activation time at the center point (3.5 h). The increase inactivating agent concentration and activation temperature improved the Cr (VI) removal efficiency slightly at the beginning. This effect was due to the formation of uniform porous structure and evolution of condensable and noncondensable volatile substances. In contrast, higher temperature and concentration of activating agent causes the widening of micropores to mesopores and macropores, thereby decreasing the performance of adsorbents [38]. The optimum values for maximum Cr (VI) removal efficiency of 92.5% under this model was as follows: 2 M, 110°C, and 4 h of concentration of the activating agent, activation temperature, and activation time, respectively, by using acid-treated Teff straw biosorbent.

Surface plots of acid-treated Teff straw biosorbent Cr (VI) removal efficiency. (a) H3PO4 concentration vs. activation temperature, (b) H3PO4 concentration vs. activation time, and (c) activation temperature vs. activation time.

3.3.2. Optimization of Process Variables for Biosorption of Cr (VI) Using BTB
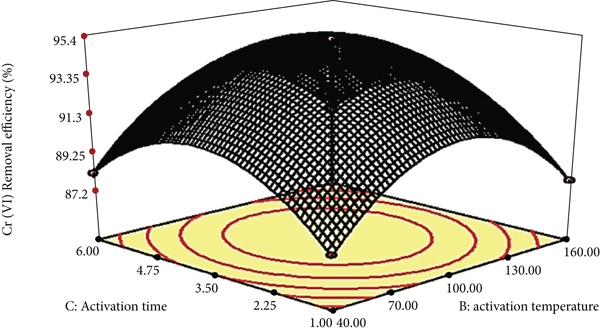
Figure 7 shows 3D response surface plots for the interaction effects of the independent variables on the Cr (VI) removal efficiency of the acid-treated biosorbent. Table 2 (b) shows the ANOVA results for the response surface quadratic model. Large

Surface plots of base-treated Teff straw biosorbent for Cr (VI) removal efficiency: (a) KOH concentration vs. activation temperature, (b) KOH concentration vs. activation time, and (c) activation temperature vs. activation time.

Significant model terms are A, B, C, AB, AC, BC, A2, B2, and C2. Model terms with values greater than 0.1000 are not significant. Model reduction may improve the model if there are many nominal terms (not counting those required to support hierarchy). The “Pred R-Squared” of 0.9985 is in reasonable agreement with the “Adj R-Squared” of 0.9997. “Adeq-Precision” measures the signal-to-noise ratio. A ratio greater than 4 is desirable. The ratio of 212.735 indicates an adequate signal. In order to fit the generated experimental data and to identify the relevant model terms, the most widely used highest order polynomial empirical equation for Cr (VI) removal efficiencies (
3.4. The Adsorption Capacity of the Modified Biosorbent
The adsorbent was prepared from Teff straw using chemical activating agents: H3PO4 which acts as the most common acid activating agent for synthesis of biosorbent due to high carbon yield, ease of acid recovery, and low operation time and KOH which acts as a primary activating agent that creates a high specific surface area in the preparation of biosorbent [31, 35]. The adsorption capacity of Teff straw-based biosorbent was investigated by analyzing adsorption parameters of adsorbent dosage, initial Cr (VI) concentration, contact time, and temperature effect on the prepared biosorbent.
3.4.1. Effect of Adsorbent Dosage on Adsorption Capacity
The effect of biosorbent dose on the removal efficiency of Cr (VI) metal ion was examined by varying the quantity of biosorbents from 0.5 to 3 g by keeping other parameters constant at a contact time of 4 h, pH of 2, initial Cr (VI) concentration of 100 mg/L, and agitation speed of 150 rpm. Percentage Cr (VI) removal increased with increasing biosorbent dosage (Figure 8(a)) due to increased adsorptive surface area, and the availability of more active sites on the adsorbent surface until the equilibrium point was reached. However, beyond equilibrium, all active sites of the biosorbent were fully occupied by Cr (VI) metal ions, i.e., totally no active binding sites to accumulate extra metal ions, which is no significant increase in percentage removal of Cr (VI). Figure 8(a) shows that the maximum percentage Cr (VI) metal ion removal was attained around 1 g. Optimum Teff straw biosorbent dosage was 47%, 83.1%, and 89.3% for UTS, ATB, and BTB, respectively. The Cr (VI) removal percentage increased from 35% to 47%, 63 to 83.1%, and 72 to 89.3% as the biosorbent quantity increased from 0.5 to 3 g at room temperature (25°C).

Effect of parameters on adsorption capacity of Cr (VI) metal ion: (a) biosorbent dosage, (b) initial Cr (VI) metal ion concentration, (c) temperature, and (d) contact time.
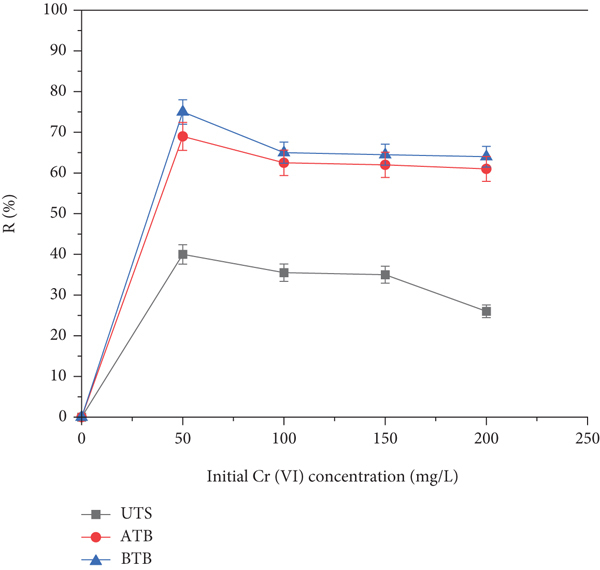

3.4.2. Effect of Initial Cr (VI) Concentration on Adsorption Capacity
Mainly, the biosorption of metal ions depends on the available binding active sites of the biosorbent. Figure 8(b) shows the influence of initial Cr (VI) concentration on the metal ion biosorption. The removal efficiency decreased by keeping other parameters constant and varying, the Cr (VI) concentration 50 to 200 mg/L.
3.4.3. Effect of Temperature on the Adsorption of Cr (VI) Metal Ions
The temperature has a significant effect on the adsorption of heavy metals onto the prepared biosorbent. This effect is because the pollutants might be soluble at given temperatures. Thus, the experiments were conducted magnetically at different temperatures (25, 30, 40, and 50°C) in the present study. Figure 8(c) shows that as the temperature increased, the percentage removal of Cr (VI) ions decreased from 92% at 25°C to 85% at 50°C. The main reason for the adverse effect of temperature on percentage adsorption has been that a higher temperature destroys the binding sites on the biosorbent. Thus, increasing temperature favors adverse effects for the adsorption of aqueous chromium ions [35, 39].
3.5. Adsorption Isotherm Studies
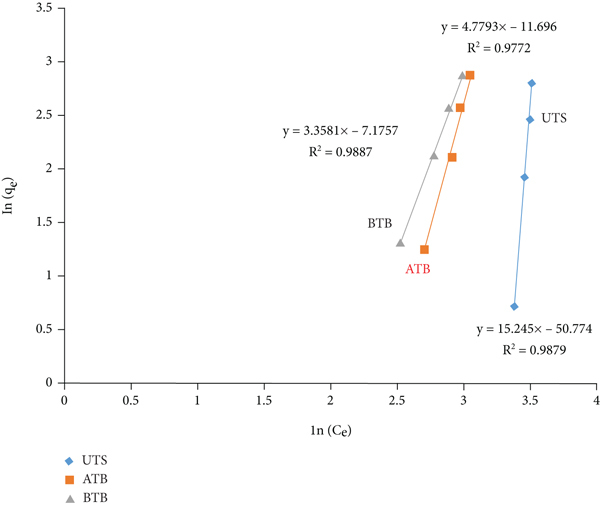
To analyze the isotherms data, the Langmuir and Freundlich equilibrium models were the most common and familiar models. The Langmuir isotherm assumes that the adsorbent forms monolayer coverage, and adsorption takes place at the specific surface of the adsorbent with no lateral interaction between the sorbed molecules, whereas the Freundlich isotherm model adsorption takes place on heterogeneous surface (multilayer adsorption). The most appropriate correlation of the equilibrium data was examined by taking various initial concentration values,
Isotherm and kinetic constants for Cr (VI) adsorption onto unmodified and modified Teff straw biosorbent.
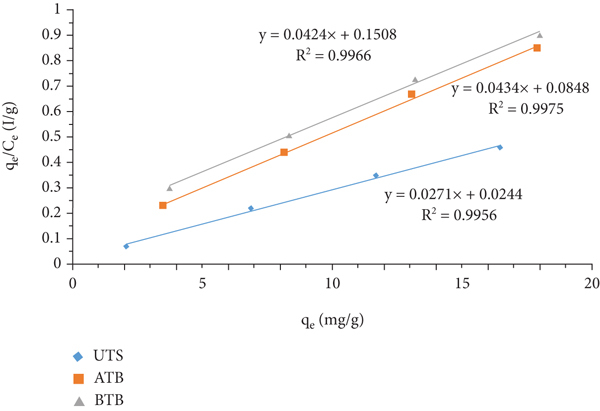
Linear fitting for adsorption of Cr (VI) metal ion: (a) Langmuir isotherm and (b) Freundlich isotherm.
3.6. Adsorption Kinetic Studies
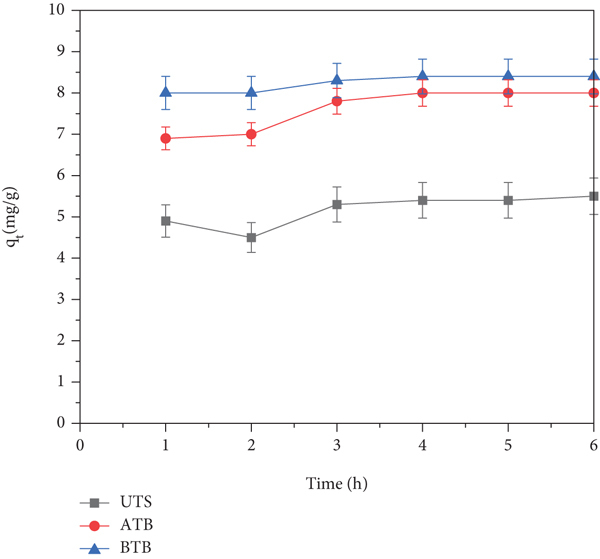
The prediction of kinetic variables has given important data about designing and modeling of adsorption process as well as used to select the optimized conditions of a batch process. Adsorption experiments were conducted to investigate the behavior of Teff straw biosorbent and to determine the rate-controlling mechanism of the biosorption of Cr (VI) ions onto Teff straw biomass. In the present study, pseudo-first-order and pseudo-second-order kinetic models were applied to scrutinize the nature and pathway of Cr (VI) adsorption on Teff straw-based biosorbent. First contact time effect on adsorption was investigated by varying soaking time from 1 to 6 h in 100 mg/L of Cr (VI) of 100 mL solution transferred into 1 g chemically activated biosorbent in 250 mL conical flask.
As shown in Figure 8(d), the amount of adsorbed

Plots of Cr (VI) adsorption onto Teff straw biosorbent: (a) pseudo-first order and (b) pseudo-second order and (c) Van’t Hoff’s plot.


As shown in Table 3, the pseudo-second-order model was fitted with the experimental data compared to the pseudo-first-order model, as confirmed by higher correlation coefficient values,
3.7. Adsorption Thermodynamics
Adsorption thermodynamics were determined using thermodynamic parameters (viz., change in free energy,
Table 4 indicates the calculated values of thermodynamic parameters investigated in this study as follows: change in enthalpy,
Thermodynamic parameters for adsorption of Cr (VI) onto Teff straw biosorbent.
3.8. Comparison of the Obtained Adsorption Parameters with the Literature Values
Various researchers have shown that adsorbents obtained from various agricultural wastes have been widely used to remove different toxic heavy metals from wastewater. For example, Tadesse et al. [18] have reported Teff straw as a potential low-cost material for removing Cr (VI) from aqueous samples. The maximum Cr (VI) adsorption capacity of 3.51 mg/g was obtained at initial pH of 2.0 and an adsorbent dose of 0.6 g. The results of their adsorption study showed that the Langmuir model was found to fit better to the Freundlich model and pseudo-second order was found to fit as compared to pseudo-first-order kinetics. Gonçalves et al. [42] have studied the adsorption of Pb+2 ions from canola meal using H2SO4 modification. Their study revealed that the removal efficiency of 80% Pb+2 ions was obtained at 140 min contact time. The adsorption model study also showed that the Langmuir model was found to fit better to the Freundlich model and pseudo-second order was found to fit as compared to pseudo-first-order kinetics. Similar results with this study were obtained in the present study with acid- and base-treated Teff straw biomass. Table 5 summarizes the results of various literature for adsorption of different metal ions using various biomass sources. The results of the present investigation are well competent compared to the reported values with some alleviate. The adsorption capacity varies and depends on the individual adsorbent’s characteristics, the adsorbate’s initial concentration, and the extent of surface modification. It is to be noted that Teff straw could be considered as one of the potential low-cost, local availability, and environmentally friend adsorbent materials to be used reliably for the efficient removal of toxic metals, such as Cr (VI), from contaminated wastewater.
Summary of literature values for adsorption of metal ions using various biosorbent sources.
4. Conclusion
The chemically modified Teff straw-based biosorbent exhibited effective Cr (VI) metal ion removal efficiency from aqueous solution due to the availability of active binding sites on the surface of the modified biomass.
The effect of concentration of activating agents (H3PO4 and KOH), activation temperature, and activation time on Cr (VI) removal efficiency of chemically modified Teff straw biosorbent was better described by quadratic polynomial model adequately. The good agreement between experimental and predicted values was revealed from the analysis of variance outputs.
The characterization showed that the Teff straw biosorbent has good properties compared with other biomass source biosorbents. The optimum values for maximum Cr (VI) removal efficiency 92.5% of 2 M, 110°C, and 4 h of activating agent (H3PO4) concentration, activation temperature, and activation time, respectively, were obtained by using acid-treated Teff straw biosorbent (ATB). Similarly, the optimum values for maximum Cr (VI) removal efficiency of 95.2% under this study was as follows: 1.5 M, 105°C, and 3.5 h of activating agent (KOH) concentration, activation temperature, and activation time, respectively, by using base-treated Teff straw biosorbent (BTB).
The isotherms exhibited the Langmuir behavior at all temperatures, which indicates that adsorption took place via monolayer surface binding. The adsorption kinetic data agreed with the pseudo-second-order kinetic model for untreated and chemically modified Teff straw biosorbent. Furthermore, based on thermodynamic parameters, the adsorption of Cr (VI) onto untreated and chemically modified Teff straw biosorbent was a spontaneous and endothermic process.
Footnotes
Abbreviations
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Acknowledgments
The first author acknowledges the Defence University College of Engineering, Ethiopia, for providing experimentation support to do the research.
