Abstract
Water hyacinth (Eichhornia crassipes) is a wild floating plant that can be found widely in pond or river areas. The plant grows fiercely and causes many harmful issues to the ecosystem around its covered area. This work provides a utilization method that converts wild water hyacinth to reliable magnetic biochar which can be used as a very effective adsorbent for the removal of lead ion Pb(II) in industrial wastewater. The mentioned magnetic biochar can be prepared via a modified pyrolysis process at 550°C with the support of cobalt sulfates as magnetite precursors and limited oxygen from the sweeping gas (the gas mixture ratio is 4 : 1 nitrogen/oxygen). The produced samples were hydrophobic biochar with high oxygen-containing functional groups that are suitable for the removal of inorganic contaminants. The impregnation of cobalt (II, III) oxides provided high magnetic separation performance and additional adsorption sites on the produced magnetic biochar. As indicated by the obtained result, the WHB-Co2M sample possesses a highly porous structure (0.126 cc/g), higher thermal stability (thermal durability reaches 900°C), relatively stable magnetic properties (14.74 emu/g), and a larger surface area (192 m2/g). These beneficial properties led to its suitability to serve as an adsorbent in removing lead ions in the contaminated effluent, recording 95% of removal efficiency and adsorption capacity of 67.815 mg/g. As indicated in the result, all prepared magnetic biochar samples were fitted to two-parameter (Langmuir models) and three-parameter (Sips model) isotherm models. Therefore, the adsorption process in this work could be carried out on both homogeneous and heterogeneous adsorbent surfaces. The adsorption kinetics of the removal process also was described by the pseudo-first-order, pseudo-second-order, and Elovich models to reveal the adsorption and desorption rate of the as-prepared magnetic biochar. This work indicates a successful waste refinery route of converting lignocellulosic biomass such as water hyacinth into value-added material for use as promising heavy metal adsorbents.
1. Introduction
Nowadays, the intense industrialization and modernization in many countries around the world have led to several environmental problems, especially the heavy metals pollution in industrial wastewater. Contamination of heavy metals could be related to wastes from many industries, such as the plating industry, mining, tanneries, and the circuit board industry [1]. Lead, tin, and nickel are the most popular metals used in the manufacturing of circuit boards as resistant plates [2]. In particular, lead is the most commonly used material, despite its high toxicity and widespread presence in the environment [3]. Lead is also the main factor causing anemia, kidney malfunction, brain damage, and even death in some significant poisoning conditions [4]. These facts motivate the development of effective solutions to remove lead ions Pb(II) from contaminated sources. In that manner, this work proposed a method to eliminate Pb(II) in the printed circuit boards manufacturing wastewater, using adsorbents derived from water hyacinth (Eichhornia crassipes). Water hyacinth is an aquatic plant typically found in lakes and other basins, causing numerous conservation problems. The growth of the plant is very aggressive, and its ability to spread over the surface of body water was reported to lead to the degradation of water quality by altering the physical, chemical, and biological processes in the covered areas [5]. The floating plants form dense and thick brush over the water surface, providing a breeding ground for mosquitoes, restricting access to water, damaging the natural wetlands, and also eliminating other aquatic plants [6].
Generally, the water hyacinth is utilized as livestock feed; however, the effectiveness of this utilization plan has been reduced over time due to the popularization of formulated feeds. Therefore, turning overgrowth aquatic plants into a useable product, such as biochar, can not only provide an alternative disposal route for unwanted biomass but also produce quality value-added materials with a wide variety of applications [7, 8]. Biochar is a stable carbon-rich material that provides the ability to deal with issues of water contamination; however, biochar in powder form is very difficult to be separated from the aqueous solution in practical applications [9]. Accordingly, a solution to introduce magnetic content into the carbon phase would be a suitable strategy to deal with the separation problem of the biochar [10]. The addition of magnetic contents to the biochar leads to the creation of magnetic biochar which not only possessed excellent magnetic separation performance but also provides additional adsorption sites, and a higher surface area in comparison with the original sample [11]. The creation of traditional biochar can be achieved via a pyrolysis process which is a popular method to synthesize carbonized materials such as biomass from agriculture waste and unwanted biomass sources [12, 13]. However, the magnetic biochar can only be fabricated with the support from magnetic precursor materials as described previously in the work of Cai et al. [14] with amino-functionalized peanut hull biochar, Niu et al. [15] with Mn-Zn ferrite biochar from battery industrial waste, or Alchouron et al. [16] with Guada bamboo and iron oxide magnetic precursors.
In this work, water hyacinth representing abundant lignocellulosic biomass but also a problematic aquatic plant was collected from rivers and pond areas in the southwest of Ho Chi Minh City, Vietnam. The plant was then impregnated with cobalt sulfates under varying input concentrations and subsequently served as feedstock for pyrolysis conversion into magnetic biochar. The main objective of this work is to determine the performance of the magnetic biochars as adsorbents for the removal of Pb(II) from contaminated effluent. The adsorption isotherm and kinetics of the magnetic biochar are measured and used for the correlation of the Pb(II) uptake. Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), scanning electron microscopy (SEM), surface area analysis, pore size distribution, energy dispersive spectroscopy (EDS), and magnetic hysteresis measurement were adopted to characterize the magnetic behavior of the prepared biochar.
2. Materials and Methods
2.1. Materials and Chemicals
The water hyacinth was collected from rivers and pond areas of Lai Vung district, Dong Thap province, Vietnam. Nitrogen and oxygen gas were purchased from the local manufacturing company in Vietnam (Sovigaz Vietnam Company). The chemicals of sodium hydroxide (NaOH) were supplied from Sinopharm Chemical Reagent Co., Ltd., China. Cobalt(II) sulfate heptahydrate (CoSO4·7H2O), and lead(II) nitrates (Pb(NO3)2) were purchased from Sigma Aldrich. All chemical solutions were prepared using deionized (DI) water (18.2 MΩ), and the same DI water also was used to rinse and clean samples during the work (DI water was purchased from Vinachem chemical company). Plastic containers were purchased from Bach Khoa Chemical Company. Chemicals were used at analytical grade and without any further purification.
2.2. Production of Water Hyacinth Magnetic Biochar
The conversion of water hyacinth biomass into biochar is depicted in Figure 1. Briefly, the biomass was cleaned with tap water and cut into small chunks with the size of 1-2 cm long and then dried under the sun for 96 hours. The dried plant was soaked in sodium hydroxide NaOH 0.1 M at 50°C and then dried to the constant mass (Table S1 concludes the physicochemical properties of the dried water hyacinth used in this work as an input material). The final pretreatment stage includes the second drying process which happened at 105°C in an oven for 24 hours. The dried samples were then impregnated with CoSO4 solution at a varying concentration of 1, 1.5, and 2 M via magnetic stirring for 30 minutes at 45°C (mixing speed 150 rpm).

The preparation process of the magnetic biochar is derived from water hyacinth biomass.
The solution of NaOH 0.5 M was added drop-wise to maintain the pH value at 10 which leads to the creation of a blue gel of Co(OH)2 (mixing speed 200 rpm). In order to accelerate the impregnation reaction, the mixture containing the dried water hyacinth and the Co(OH)2 solution was put under a temperature of 70°C and maintained stirring for 24 h (200 rpm). The reaction of the cobalt sulfate at the alkaline condition can be described as below:
Following the impregnation process, products should include water hyacinths impregnated with various concentrations of CoSO4 which are water hyacinths impregnated with CoSO4 1 M (WH-Co1M), 1.5 M (WH-Co1.5M), and 2 M (WH-Co2M), respectively. The system we used in this work is very similar to the pyrolysis system in our previous work [17]. The carbonization of impregnated water hyacinth was carried out at 550°C (heating rate value 30°C/min) for 70 minutes. The Co(OH)2 obtained in this process is in the form of blue precipitation. The layers of hydroxide and cobalt possessed residual positive charges and alternated quickly with other anions such as nitrate, carbonate, or chloride [18, 19]. Following the impregnation process, products should include water hyacinths impregnated with various concentrations of CoSO4 which are water hyacinths impregnated with CoSO4 1 M (WH-Co1M), 1.5 M (WH-Co1.5 M), and 2 M (WH-Co2M), respectively. Figure S1 in the supplementary files provides more information about the pyrolysis chamber.
The mixture of nitrogen/oxygen with the ratio of 4 : 1 serves as sweeping gas, and the flow rate has been carefully controlled at 50 mL/min throughout the process. The use of nitrogen/oxygen mixture as the sweeping gas was crucial, which supports the cause of the creation of the Co3O4, reduces the tar generation, exhibits the creation of char, and saves the investment cost for running the pyrolysis process [20]. The low flow of a sweep gas maintains the limited amount of oxygen residue in the chamber. The creation of Co3O4 from Co(OH)2 can only be operated in the presence of air and at a temperature of 300°C or above [21, 22]. The reaction below describes the synthesis of Co3O4:
The overall reaction would be the combination of reactions (2) and (3):
2.3. The Batch Experiment of Lead Ion Removal
The wastewater sample originated from an industrial circuit board manufacturer in Cu Chi Province, Ho Chi Minh City, Vietnam. All samples were collected using 10-L plastic lab containers and taken to the laboratory within two hours. The collected samples were then stored in the refrigerator for 12 hours at 10°C for metal preservation. After storage, the wastewater can be used as an influent for the process of removal of lead ions using magnetic biochar. Table S2 in the supplementary file provides characteristics of the influent used in this work, and the mentioned parameters are compared to the regulatory standard for industrial wastewater discharged.
All the experiments of lead ion removal were operated using a batch adsorption technique with the magnetic biochar used as adsorbent (dosages value varied from 0.5 to 3 g/L). The prepared biochar then was mixed with 50 mL of influents in 250-mL digestion vessels with the adjustment of pH values at 7 using NaOH and HNO3 0.01 M. The vessels were then shaken in a mechanical shaker at 200 rpm at room temperature. At the end of the experiment, the vessels were collected and filtered to determine the concentration of Pb(II) remains in the liquid phase. The determination of Pb(II) concentration was carried out using inductively coupled plasma atomic emission spectroscopy (ICP, Aivo 200 by Perkin Elmer). All the adsorption experiments were performed in duplicate, and the average value possessed relative errors smaller than 5%. Kinetic experiments were studied by mixing 10 mg of sorbent with 50 mL of influent with a concentration of 200 mg/L at 35°C at different time intervals (0, 100, 200, and 300 min). Sorption isotherm was studied using similar experiments with 10 mg of sorbent mixed with 50 mL of influent at 25, 35, and 45°C and with different concentrations of Pb(II) (from 0 to 200 mg/L).
The adsorbed Pb(II) (
Adsorption kinetics of the lead ion removal process was performed by adopting the selected magnetic biochar samples as adsorbents for the treatment under selected conditions, and various working times varied from 0 to 300 min. The pseudo-first-order, pseudo-second-order, and Elovich models were applied to indicate the adsorption mechanism. The equations of the pseudo-first-order, pseudo-second-order, and Elovich models are described as follows:
Adsorption isotherm of the lead ion removal process can be determined using the two-parameter models such as Freundlich and Langmuir models and three-parameter model such as Sips model. Langmuir’s model was employed to assess and calculate the adsorption behavior of the WHB-Co samples. In accordance with Wang et al. [23], the adsorbates binding to the surface of the adsorbents is mainly related to the physical forces and assumes that all adsorption sites possess an equal affinity for the sorbate. The Langmuir model can be described as:
Freundlich isotherm model can be adopted to evaluate the adsorption on diverse surfaces and indicates the stronger binding sites that are occupied first during the adsorption process. The model can be applied to multilayer sorption to describe the relationship between the adsorbent and the sorbate [24]. The Freundlich model can be described as:
On the other hand, the Sips model provides the prediction of an adsorption process in a heterogeneous system with monolayer adsorption associated with the Langmuir model and prevents the rise of adsorbate concentration associated with the isotherm described by the Freundlich model. The Sips adsorption isotherms can be determined following the equation:
All the isotherm models were also recorded and verified with the sum of squares errors and correlation coefficient value. Nonlinear equations of Langmuir, Freundlich, and Sips models were used to fit the experimental data by adopting the least square method.
2.4. Material Characterization and Analysis Procedure
Different characterization methods were adopted to study the affection of initial magnetic precursor concentration on the creation of the magnetic biochars. All the measurements were carried out in duplicate and repeatable to strengthen the quality of the work. Average values and standard deviations are presented along with figures throughout the work. Elemental analysis of the water hyacinth biomass and the as-prepared magnetic biochar was carried out using the elemental analyzer Fison’ EA 1108 CHNS, according to the ASTM 5291-96. The proximate analysis includes the measurement of moisture, volatile matter, carbon content, and ash contents and was carried out following the standard methods. Both proximate and elemental analyses possessed deviation values less than 5%.
The microstructure and morphology of each sample were examined by SEM Hitachi-S4800 type 2 with Thermo NORAN NSS EDS: cold field emission electron gun. The resolution of the machine was 1 nm at 1 kV and 1.4 nm at 15 kV, with magnifications ranging from 20 to 800,000 times. In addition, the STEM detector showed the best performance at 200 kV acceleration voltage. The as-prepared magnetic biochar sample was coated with gold for high conductivity and a high-quality image. Functional groups and their interaction with the as-prepared magnetic biochars were determined using FTIR Aplphe II by Bruker in the scanning range of 4000 to 600 cm-1. The phases and crystalline structure analysis were examined by XRD Malvern Panalytical, with X’pert pro-MRD, using copper (Cu) K (alpha) radiation.
The surface area of each sample was determined by the Brunner-Emmet-Teller (BET) method, and the sample porosity was analyzed by porosimetry of the adsorption/desorption isotherms using nitrogen and argon at 77 K. All samples were degassed at 300°C for 2 h. Both of the measurements are carried out by porosimetry and accelerated surface area system ASAP 2420 equipment by Micromeritics.
Thermogravimetric analysis was performed using TG/DTA analyzer from Perkin Elmer Diamond. The heating rate was set to 10°C/min in the temperature range of 30 to 900°C with nitrogen flow (100 mL/min) to determine the thermal properties of the as-prepared magnetic biochar and therefore indicate the role of magnetic precursor added to the biochar via the preparation steps.
Inductively coupled plasma (ICP, Aivo 200 by Perkin Elmer) equipment was used to determine the concentration of heavy metals in the effluent. The applied plasma gas was at 15 L/min, the auxiliary gas was at 1.4 L/min, carrier gas was at 1.5 L/min, and the pump speed was at 15 rpm.
The magnetic measurement was carried out using a vibrating sample magnetometer (VSM) (BHV-55, Riken, Japan). The magnetic field was set in the range of 0 to 3.0 T, while the scope of the magnetic moments was at 10-2-300 emu (sensitivity:
3. Results and Discussions
3.1. Characterization of the Magnetic Biochars
3.1.1. The Creation of the Cobalt Magnetic Biochar
The physicochemical properties and the mass yield of the as-prepared magnetic biochar are summarized in Table 1. As observed, the magnetic biochar mass yield is increased (from 42.08% to 42.71% at max yield) as the concentration of magnetic precursors was increased along with the samples (1 M, 1.5 M, and 2 M of cobalt sulfate were added to the dried biomass). Following the proximate analysis, the moisture content in all samples was reduced after the pyrolysis process (from 6.34% in the WHB-Co1M to 6.15% in the WHB-Co2M). The changes in the moisture content of the WHB-Co1M, WHB-Co1.5M, and WHB-Co2M samples are small and depend on the extent of the hydrophobicity phase in the materials.
Physicochemical properties and mass yield of the magnetic biochar. The obtained data values are the mean ± standard deviation of triplicate experiments.
The fixed carbon content of magnetic biochar decreased slightly from 20.56% in WHB-Co1M to 20.49% in WHB-Co2M. It is clear that the fixed carbon content increases when the concentration of the cobalt precursor was increased. On the other hand, the ash content in the magnetic biochar samples is composed of inorganic elements that remained after the pyrolysis process and slightly increases from 21.57% in the WHB Co1M to 21.71% in the WHB-Co2M. The difference in volatile compounds content is also indicated and reflected in only small changes (51.53% in the WHB-Co1M, 51.57% in the WHB-Co1.5M, and 51.64% in the WHB-Co2M). Following the elemental analysis, the carbon content decreases from 43.88% in the WHB-Co1M to 42.74% in the WHB-Co2M. The amount of carbon was expected to be lost due to contact with the oxygen in the sweeping gas during the heating process, which may result in partial calcination. In this work, the obtained carbon content and hydrogen content via the elemental analysis are lower than expected.
The mentioned fact can be explained by the presence of a limited volume of oxygen during the pyrolysis process. Although the contact with oxygen during the pyrolysis is controlled (sweep gas ratio is 4 : 1, the flow rate of 50 mL/min) and the volume of oxygen joined in available reactions is small (10 mL/min), that was still sufficient for the small reduction in fixed carbon content in magnetic biochar samples. Similar results were found in other works of greenhouse crop [24], corn stalk [25], and rice husk [26] which indicate the affection of pyrolysis temperature at 550°C to the change in both elemental analysis results and proximate analysis results. According to the results concluded in Table 1, the introduction of cobalt precursors at different concentrations did not affect the physicochemical properties of the final products afterward, while the pyrolysis temperature and the oxygen content from the sweeping gas-only may cause the smallest change in both the content of ash and hydrocarbon elements.
3.1.2. Chemical Structure and Phase Analysis
Fourier transform infrared spectroscopy (FTIR) was used to determine the chemical structure of the magnetic biochars in this work. During the pyrolysis process, temperature, heating time, and the ratio of cobalt precursors on biomass affected the properties of the produced samples. In most cases, the temperature is the most important factor and directly shows its influence on the magnetic biochar substance [27]. However, the results obtained in this work indicate how the initial concentration of magnetic precursors affects the chemical structure of the as-prepared magnetic biochars. In Figure 2(a), we observe sharp peaks at around 577, 604, and 597 cm-1 together with peaks at 615, 627, and 631 cm-1 for WHB-Co1M, WHB-Co1.5 M, and WHB-Co2M, respectively. Those mentioned spectrums are associated with the (Co3+)-O (in an octahedral hole) and the (Co2+)-O (in a tetrahedral hole) vibration [28, 29].

The FTIR spectrum (a) and the XRD patterns (b) for the different samples of cobalt magnetic biochars.

The phenomena also confirm the existence of the magnetic cobalt that has been successfully impregnated on the biochar following the preparation steps in this work. These peaks of about 1099 to 1106 cm-1 are attributed to the stretching vibrations of C-O and carbonate anions that occurred during the pyrolysis process. Those peaks at round 1383 to 1387 cm-1 indicate the C-H deformation vibration of hemicellulose (at 1383 cm-1), lignin (at 1387 cm-1), and cellulose (at 1385 cm-1) content in all three samples, and similar results can be found within the work of Zhang et al. [30] and Khandalou et al. [31]. Broad peaks at around 3361, 3362, and 3365 as well as those at 1623, 1628, and 1629 cm-1 would belong to the stretching of the O-H bond which is assigned to the content of water in the sample of WHB-Co1M, WHB-Co1.5M, and WHB-Co2M, respectively. The FTIR analysis successfully indicates the changes in the chemical structure of each magnetic biochar sample under the influences of the initial concentration of magnetic precursors added during the preparation steps, previously. Accordingly, the difference in cobalt precursors does not dramatically affect the nature of the final product, but most likely confirms a successful preparation process.
For a more in-depth investigation of the phase properties and crystallinity of the prepared magnetic biochars, X-ray diffraction (XRD) analyses were conducted. Figure 2(b) concludes the XRD patterns of the three different samples with a series of diffraction peaks at the 2θ degrees of 32.69, 37.21, 43.77, and 63.58 which can be assigned to the plane of (220), (311), (400), and (440). Accordingly, the XRD patterns we obtained in this work are well uniformed in comparison to each other and similar to values reported for Co3O4 spinel cubic in the JCPDS card number 42-1467 [32].
As we can observe in Figure 2(b), the modification of cobalt precursor concentration indicates small changes to the creation of the final samples, including that the weak intensity of those peaks at around 32.69 (220) at 2θ degrees is getting smaller and weaker from WHB-Co1M to WHB-Co2M. In the case of the WHB-Co1M sample, the (220) peak was small and broad which indicates poor magnitude properties and an amorphous phase. For the case of WHB-Co1.5M and WHB-Co2M samples, peaks at around 32.69 (220) were also very low and can be explained by the influence of pyrolysis temperature on the size distribution of nanocrystal materials in the sample. The orientation of Co3O4 strongly depends on the pyrolysis temperature which means the cobalt contents increase in nanocrystalline size for the cost of the amorphous phase at the pyrolysis temperature of 550°C [33].
The diffraction peaks at 37.21 2θ (311) which are the main peaks of Co3O4 show small sharp peaks with similar intensity for the three samples. Therefore, there are only very thin crystalline single phases of the cubic crystal structure that are formed. The difference in peak intensity at this orientation is not much different. The peak found in the WHB-Co2M sample XRD pattern appears to be better and clearer in comparison with the other samples due to its highest peak intensity. The same phenomena were found at the peaks of 43.77 and 63.58 of 2θ degrees, and no other peaks for impurities were detected in these patterns. All the carbon content included in the XRD analysis was at the amorphous phase due to their broad peak around 25 of 2θ degrees. Similar confirmations can also be found in the work of Yang et al. [34], Tian et al. [35], and Kemmou et al. [36]. The results obtained via XRD analysis can determine the successful preparation of magnetic biochar with the support of cobalt precursors. However, the change in the concentration of the used precursors reflects only small differences between the peak intensity of three magnetic biochar samples.
3.1.3. Morphological Analysis
The morphology of these as-prepared magnetic biochar samples including dried biomass sample, alkaline-treated sample, biochar sample, WHB-Co1M, WHB-Co1.5M, and WHB-Co2M is shown in Figure 3. As we can observe, Figures 3(a) and 3(b) show the morphological image of dried biomass and alkaline-treated samples, respectively. The surface of the biomass was changed and got rougher after the alkaline treatment process, and the use of the alkaline solution is meant to remove hemicellulose, lignin, and some other organic compounds that may contact with the dried biomass from previous preparation steps [37, 38]. The morphology of the alkaline treated sample in Figure 3(b) indicates the successful solubilization of hemicellulose and lignin with a notable amount as well as enhancing the porosity and surface area of cellulosic content in the sample [39, 40].

SEM images of (a) dried biomass sample; (b) alkaline-treated biomass sample; (c) water hyacinth biochar: WHB; (d) biochar WHB-Co1M sample; (e) biochar WHB-Co1.5M sample; and (f) biochar WHB-Co2M sample (with higher calibration image attached).





Figure 3(c) indicates a clear image of the produced biochar in similar sizes and shapes. However, the morphology of the cobalt impregnated biochar samples reveals a very different story and confirms that the concentration of magnetic precursors is associated with the loading of the metal content during pyrolysis. In Figure 3(d), the WHB-Co1M provides the morphology of a surface with Co3O4 anchored. It is clear that the Co3O4 particles are scattered on a rough surface of the biochar and were not uniformed, and the creation of a dense surface was not achieved. Figure 3(e) describes a more transparent surface of sample WHB-Co1.5M with more particles of Co3O4 can be recognized. The quantity of cobalt content increases clearly. There were some pore-shaped Co3O4 particles from the size of 100 nm that can be found uniformly anchored to the sample and covered across the surface of the biochar.
In the case of the WHB-Co2M sample, Figure 3(f) shows the morphology of a dense and well-covered surface of biochar with the size, and the form of the created cobalt(II, III) oxides is not so different in comparison with those in the WHB-Co1M sample in Figure 3(e). The small images on the left corner indicate a clearer state of the sample; uniformed pore-shaped particles of Co3O4 cover the biochar surface and almost create a dense layer with similar morphology.
The increase of magnetic precursor concentration extends the loading of the metal on the biochar, whereas the creation of Co3O4 was at around 300°C and the completed magnetic biochar was at 550°C. Consequently, WHB-Co2M seems to have better properties with larger surface properties, higher porosity, and thermal stability compared to the other two. The discussion about porosity, surface, and other properties of magnetic biochars can be confirmed later in this work. Similar results were obtained in the work of Liu et al. [41], Wang et al. [42] from a bloom-forming, and Sewu et al. [43]; the effects of the initial concentration of the magnetic precursors on the metal loading on the biochar are confirmed following the results presented in this work. The small EDS graph at the corner of Figures 4(d), 4(e), and 4(f) confirms the successful creation of the cobalt impregnated biochar in all three samples. The confirmation of the successfully impregnation process of cobalt content is also supported via the EDS analysis in Figure S2 in the supplementary file.

The magnetic hysteresis loop (a) and the thermogravimetric-TGA analysis (b) of the magnetic biochar samples.

3.1.4. The Magnetic Hysteresis and Thermal Properties of the Impregnated Magnetic Biochar
The difference in cobalt content for each magnetic biochar sample would directly affect its properties and performance. In this way, the efficiency of magnetic biochars can be determined through the study of their surface, thermogravimetric and magnetic properties. Figure 4 shows the magnetic hysteresis loop and the thermal gravimetric analysis results of WHB-Co1M, WHB-Co1.5M, and WHB-Co2M samples, respectively.
As indicated in Figure 4(a), the WHB-Co2M sample possesses the highest saturation magnetization value (14.74 emu/g), followed by the WHB-Co1.5M (12.51 emu/g) and the WHB-Co1M (11.77 emu/g). All three samples react positively with the applied magnetic field varied between 10 and -10 kOe and can be easily separated from the aqueous phase by a magnet.
The input of different cobalt precursor concentrations from the previous preparation steps strongly affects the magnetic properties of the magnetic biochars. The cobalt loading during pyrolysis was crucial in this case, where the higher cobalt content in the magnetic biochar provides better magnetization saturation. The cobalt content can be determined using the digestion method, and the results are given in Table 2.
Surface area analysis and cobalt content in the prepared samples.
With the largest surface area (192 m2/g) and pore volume (0.126 cc/g), the WHB-Co2M sample also includes the highest cobalt content (38.94 mg/g) which indicates the great saturation magnetization value over the other two samples. This may be explained following the thermogravimetric analysis illustrated in Figure 4(b). Light hydrocarbon components and moisture were removed at temperatures ranging from 100°C to 150°C. At 300°C with only 15-20% weight loss, Co(OH)2 was converted to Co3O4 with the support of oxygen from the pumped ambient air (45 mL/min) which also means the creation of the magnetism cobalt occurred before the completed creation of the biochar at 550°C with more than 70% mass weight remains for all samples. The large surface area and pore-rich structure of the biochar provide a notable adsorption capacity of metal which resulted in the higher cobalt content included in the sample treated with the higher input magnetic precursor concentration (WHB-Co2M > WHB-Co1.5 M > WHB-Co1M) [44]. In comparison with the work of Ouyang et al. [45] and Han et al. [46], the results obtained in this work provide the homogeneous confirmation of the relationship between the metal loading on the biochar and the metal concentration in the magnetic precursor solution. In that manner, the WHB-Co2M sample is derived from the preparation process including the higher concentration of the magnetic precursor solution, and provided not only a larger surface area, but also higher thermogravimetry property, and higher magnetization saturation than the other samples. Not only that, the fact that the addition of metal sites to the biochar has the potential to extend the surface area, the pore volume, and the sorption of the biochar has been confirmed by Chen et al. [47].
3.2. The Removal of Lead Pb(II) from Industrial Wastewater
3.2.1. Kinetics Study and the Effects of Adsorption Times on the Removal Efficiency
The original concentration of 95 mg/L of Pb(II) of the influent (as described in Table S1) would be completely removed in about 7-10 minutes so that in these batch experiments, longer adsorption times were used (0 to 300 mins). Figure 5(a) determines the effects of adsorption times on the removal efficiency of the process. The adsorption of lead ions Pb(II) on the prepared magnetic biochars was fast and reached equilibrium in 100 mins.

Plots of (a) the removal efficiency versus the adsorption time and the adsorption kinetics for Pb(II) on WHB-Co1M (b), WHB-Co1.5M (c), and WHB-Co2M (d) sample. The adsorption kinetics data were calculated following the pseudo-first-order, pseudo-second-order, and Elovich models.
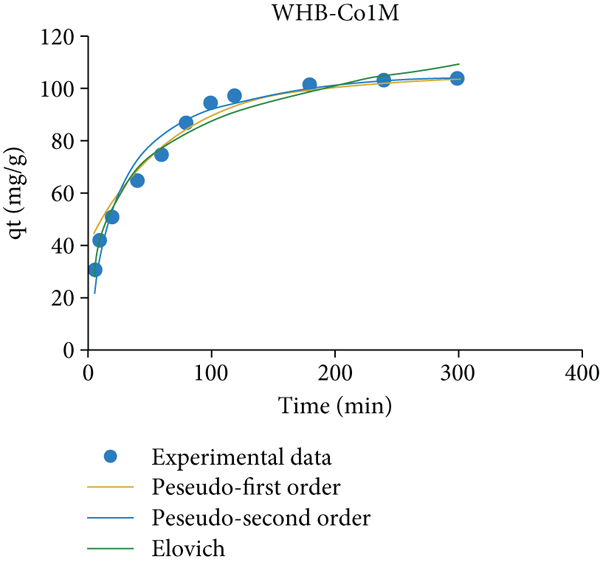


All three magnetic biochar samples, WHB-Co1M, WHB-Co1.5M, and WHB-Co2M, indicate a fast rate adsorption process with more than 80% of the Pb(II) and were removed in the first 75 mins; the removal rate then slowed down apparently until the equilibrium was achieved after 100 mins and stable to the end of the process. The adsorption in the early stage of this work can be attributed to reactions such as electrostatic and ion exchange which were reported to reach equilibrium quickly after a few minutes [48, 49]. The WHB-Co2M sample provided the highest removal efficiency (up to 85%) after 100 mins, at room temperature (35°C).
It is reasonable to argue that the higher surface area (192 m2/g) and high porosity (0.126 cc/g pore volume) of the WHB-Co2M sample provide the ability to adsorb more Pb(II) ions. In addition, results in Table 3 also showed that the faster mass transfer during the adsorption is governed by an excellent pore-filling mechanism [ 50, 51].
The kinetic parameters of magnetic biochar sample adsorption.
Figures 5(b) and 6(c) and 6(d) determine the Pb(II) adsorption kinetic of the WHB-Co1M, WHB-Co1.5M, and WHB-Co2M magnetic biochar sample that were used as adsorbents in this work, respectively. The mononuclear and binuclear adsorption were described using the pseudo-first-order and pseudo-second-order models in a solid-solution system. The Elovich model was also applied to evaluate the contribution of desorption throughout the process. The calculated kinetic parameters from the three models are listed in Table 3.

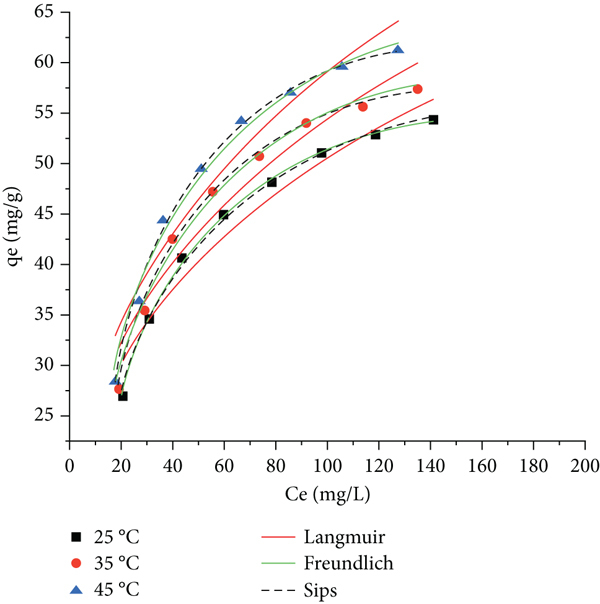
Effects of adsorbent dosage on the removal efficiency (a), the adsorption isotherm for Pb(II) on WHB-Co1M (b), WHB-Co1.5M (c), and WHB-Co2M (d) sample.


In the case of the WHB-Co1M sample (described in Figure 5(b)), the correlation coefficient value (
3.2.2. The Adsorption Isotherm and the Influence of Adsorbent Dosage
The dosage of adsorbent plays a crucial role that greatly influences the adsorption process, due to its affection for the available adsorption sites. The determination of adsorption equilibrium for all magnetic biochar samples in this work includes 24-h adsorption time, and the relationship between adsorbent dosage and the removal efficiency of this work are described in Figure 6(a), while the sorption isotherm of each magnetic biochar sample which was used as adsorbent would be studied using the two-parameter (Langmuir and Freundlich) and three-parameter (Sips) isotherm models.
As observed in Figure 6(a), the plot indicates two different areas, whereas the first area shows that the removal efficiency of all three magnetic biochar samples was significantly increased (from 27% to 63% with WHB-Co1M, 32% to 67% with WHB-Co1.5M, and 41% to 72% with WHB-Co2M sample) as their dosage increase from 0.5 g/L to 1.5 g/L for all three samples. The second area indicates the removal efficiency of the work with a smaller change, with the adsorbent dosage increase from 2 to 3 g/L for all three samples and the efficiency increase with a slower rate (from 73% to 80% with WHB-Co1M, from 80% to 83% with WHB-Co1.5M, and 82% to 85% with WHB-Co2M sample). Completed removal was not yet reached due to the lack of adsorption sites from all three applied magnetic biochar samples, and all active sites would be fully occupied by the Pb(II) during the process [54]. The result obtained in this part suggests that the higher Pb(II) removal percentage would be achieved with a higher adsorbent dosage, thanks to its additional active sites.
The isotherm study includes a 24-h process with the adsorbent dosage of 2 g/L, and the concentration of Pb(II) in the influent varied from 0 to 200 mg/L. Experiments were carried out under three different temperatures to determine the effect of working temperature throughout the adsorption process. According to the acquired correlation coefficient value (
Langmuir, Freundlich, and Sips isotherm constants for the adsorption of Pb(II) on the magnetic biochar sample.
According to Table 4, the Sips exponent value (
The Langmuir Pb(II) adsorption capacity of water hyacinth magnetic biochars from this work can also be used to compare the performance of various adsorbents. The maximum uptake capacity for WHB-Co1M, WHB-Co1.5M, and WHB-Co2M was 43.55, 65.57, and 75.53 mg/g at 45°C, respectively. The adsorption capacity for Pb(II) with other carbon-based adsorbents has been reported as 99.85 mg/g with modified sewage sludge biochars [57], 45.70 mg/g with magnetic energy cane biochar [58], or 66.23 mg/g with the sugarcane bagasse treated with H3PO4 and pyrolyzed at 400°C [59]. Table S3 in the supplementary file concludes with more comparison references [60–62] for the adsorption performance of Pb(II) with different applied adsorbents.
The adsorption mechanism of Pb(II) using magnetic biochars derived from water hyacinth can be described by both chemical and physical adsorption. As evidenced from the SEM micrograph and BET measurement, the high surface area and highly pore structure of the WHB-Co2M provide more active adsorption sites that benefit the adsorption on its surface. As the pH value in this work is controlled 7, the results obtained from kinetic experiments indicate that the fast early adsorption stage (initial stage) can be attributed to the electrostatic attraction and ion exchange, and the removal capacity of Pb(II) depends on chemical adsorption rather than physical adsorption. It is reasonable to claim that the adsorption that happened in this work could be related to many other mechanisms such as Van der Waal forces, hydrophobic force, and π-π interaction. Furthermore, the isotherm experiment provides insight into the monolayer and multilayer adsorption on the heterogeneous surface.
4. Conclusion
In this work, the preparation and characterization of cobalt impregnated water hyacinth magnetic biochars via a modified pyrolysis process were successfully carried out. The role of the impregnated cobalt (II, III) oxides on biochars is confirmed following the described methods, which mainly focus on the difference in cobalt sulfate concentration throughout the preparation for the final products. The WHB-Co2M sample was found to be the most effective adsorbent for the removal of lead ions in industrial wastewater, thanks to its high porosity (0.126 cc/g pore volume), higher surface area (192 m2/g), and higher saturation magnetization value (14.74 emu/g). The adsorption kinetics and isotherms on all magnetic biochar samples are better fitted to the pseudo-second order and the Sips model rather than Langmuir and Freundlich model which demonstrate a strong adsorption process with high removal efficiency (up to 95%) in a notably fast process where the equilibrium is reached after about 100 minutes. However, the results of adsorption performance obtained from the comparison with commercialized adsorbents indicate that the magnetic biochar in this work still needs and still be able to improve. The advantages of this work are concluded as a simple method that can be able to convert waste to economic materials, as well as contribute greatly to the decrease and control of wild and natural species such as water hyacinth.
Footnotes
Data Availability
All the data were concluded in the submission. A supplementary file is available for more details descriptions and explanations throughout the work.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Acknowledgments
This work has been supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (NRF-2019R1C1C1007907 and NRF-2017M3A9E2065287). The author would also express gratitude toward Van Lang University (VLU) for its excellent support for research funding (VLU Research Funding 2022) and the experimental facility. All experiments were carried out in the School of Engineering and Technology (VLSET) laboratory, Van Lang University (VLU). Sogang University (South Korea) supported analytical processes on each sample.
