Abstract
The adsorption of naproxen (NAP), diclofenac (DFC), and acetaminophen (APAP) molecules from aqueous solutions using MIL-100-Fe and MIL-101-Fe metal organic frameworks (MOFs) has been analyzed and modeled. Adsorption isotherms of these pharmaceuticals were experimentally quantified at 30 and 40°C and pH 7. Textural parameters and surface chemistry of these MOFs were analyzed, and results were utilized to explain the pharmaceutical adsorption mechanism. Density Functional Theory (DFT) calculations were performed to understand the reactivity of pharmaceutical molecules, and a statistical physics model was employed to calculate the main physicochemical parameters related to the adsorption mechanism. Results showed that the adsorption of these pharmaceuticals on MOFs was multimolecular and exothermic. Both MOFs displayed the highest adsorption capacities, up to 2.19 and 1.71 mmol/g, for NAP and DFC molecules, respectively. MIL-101-Fe showed better pharmaceutical adsorption properties than MIL-100-Fe due to its highest content of Fe-O clusters and mesopore volume. Adsorption mechanism of these organic molecules could involve hydrogen bond, van der Waals forces, and electrostatic interactions with MOF surfaces. In particular, MIL-101-Fe MOF is a promising material to prepare composites with competitive adsorption capacities for facing the water pollution caused by pharmaceutical compounds.
1. Introduction
In recent years, the literature has documented an increment of the water pollution caused by emerging compounds [1]. They are unregulated pollutants by the environmental legislation that include an extensive variety of chemicals such as pharmaceuticals, personal hygiene products, and surfactants [1]. In particular, the release of pharmaceutical molecules in the environment is a relevant issue since they can affect significantly the human health causing, for example, carcinogenesis, teratogenesis, and mutagenicity even at very low concentrations (i.e., from μg/L to ng/L) [2–5]. Diclofenac (DFC), naproxen (NAP), and acetaminophen (APAP) are nonsteroidal anti-inflammatory drugs that stand out due to their worldwide prescription for human healthcare [6, 7]. NAP is employed for the treatment of osteoarthritis, rheumatoid arthritis, and migraine and to reduce inflammation and fever [7, 8]. DFC is a widely used pharmaceutical to minimize pain and inflammation caused by ankylosing spondylitis, rheumatoid arthritis, and osteoporosis [9]. This drug is commonly identified as a water pollutant in environmental samples [7]. APAP is also an anti-inflammatory, antipyretic, and analgesic drug [10]. Overall, these pharmaceutical molecules can reach wastewater via the human excreta and by their inappropriate disposal after the drug expiration. Different studies have concluded that these organic molecules can persist in the environment for a long time due to their resistance to biodegradability and stability to heat and light [11, 12].
The control and reduction of concentrations of pharmaceutical pollutants in the environment, and especially in water resources, can be performed via adsorption [13, 14] or using other treatment methods like photodegradation [15, 16]. Particularly, the removal of pharmaceutical molecules via adsorption processes can offer additional economical and technical advantages, and consequently, it is necessary to study the application of novel adsorbents to consolidate its application at industrial level. In this direction, the metal organic frameworks (MOFs) are interesting adsorbents with promising potential for wastewater treatment including the pharmaceutical depollution due to their surface area, controllable pore size (in micro and mesoporous domains), structural versatility, and composition [17–19]. There is a wide spectrum of metals and organic ligands that can be used for the preparation of MOFs, thus offering the possibility to obtain materials with different physicochemical characteristics and adsorption properties. Research on MOFs has indicated that their adsorption capacity to remove organic molecules is determined by their textural parameters (surface area and pore size) and surface functionalities. These physicochemical properties influence the
Under this perspective, this study reports the modeling and analysis of the physicochemical parameters of the adsorption of pharmaceutical molecules on MIL-100-Fe and MIL-101-Fe MOFs. These materials were synthesized and employed to adsorb DFC, NAP, and APAP as target pharmaceutical molecules from aqueous solutions. These MOFs were characterized and their adsorption properties were determined experimentally at 30–40°C and pH 7. The physicochemical parameters associated to the adsorption mechanism of these pharmaceuticals were calculated via the statistical physics-based modeling. Therefore, this study contributes with new experimental data and theoretical insights on the application of MOFs for the adsorption of pharmaceuticals molecules from aqueous solutions.
2. Methodology
2.1. Synthesis and Characterization of MOFs
MIL-100-Fe and MIL-101-Fe MOFs were prepared and employed to analyze the adsorption of different pharmaceutical molecules from aqueous solutions. MIL-100-Fe was synthetized with 1.76 g of Fe(NO3)3·9H2O and 0.57 g of H3BTC (trimesic acid). The organic linker and metal source were dissolved in 20 mL of deionized water and stirred well for 1 h. The final solution was submitted to a thermal treatment at 160°C for 12 h using a Teflon-lined stainless-steel autoclave. The solid product obtained from the reaction was separated via centrifugation, washed several times with deionized water and ethanol, and finally dried at 80°C for 24 h. The preparation of MIL-101-Fe was performed with 0.675 g of FeCl3·6H2O and 0.206 g of H2BDC (terephthalic acid), which were dissolved in 30 mL of DMF. The thermochemical conversion of this solution was done at 110°C for 20 h with the Teflon-lined stainless-steel autoclave. The final solid product was also separated via centrifugation, washed with ethanol, and dried at 80°C and 24 h. Nanosized MOF particles were used in all the pharmaceutical adsorption studies reported in this paper.
Samples of these MOFs were characterized to determine their main surface and textural properties. Surface functional groups were identified by Fourier-transform infrared (FTIR) spectroscopy with a Thermo Nicolet Is10 FTIR spectrometer (Thermo Scientific). Spectra were recorded with KBr-based sample pellets in the 4000-400 cm-1 range with a resolution of 4 cm-1. Crystallinity analysis was done via the X-ray diffraction patterns using a Malvern-Panalytical X-ray diffractometer. Samples were analyzed at room temperature with copper radiation (
2.2. Pharmaceutical Adsorption Studies
NAP (≥98%), DFC (≥98%), and APAP (≥99%) were supplied by Sigma-Aldrich and utilized to prepare the adsorbate solutions with deionized water. The main characteristics and molecular structures of these pharmaceuticals are shown in Table 1. Adsorption isotherms of these pharmaceuticals using MIL-100-Fe and MIL-101-Fe MOFs were experimentally quantified at 30–40°C and pH 7. These isotherms were determined with a MOF ratio (
Main characteristics of the naproxen, diclofenac, and acetaminophen molecules used as adsorbates in this study.
2.3. Thermodynamics and Modeling of the Adsorption of Pharmaceutical Molecules
The thermodynamics of the pharmaceutical adsorption was analyzed calculating the adsorption enthalpy (
Results of MOF characterization and the analysis of adsorbate molecular structure were utilized to define a statistical-physics-based model to calculate the physicochemical parameters of the adsorption mechanism of pharmaceutical molecules [29–32]. Therefore, this model was utilized to fit the adsorption data and to determine these parameters. It is convenient to note that the adsorption of tested pharmaceutical molecules on these MOFs could imply two surface functionalities: oxygenated functional groups (e.g., -COOH and –OH) and Fe-O clusters. But the complex molecular structure of MOFs limits the possibility of identifying, with reliability, the specific contribution of these functionalities during the pharmaceutical adsorption. Based on these facts, the statistical physic model assumed that one functional group (i.e., oxygenated functionality with or without Fe) was involved in the adsorption of NAP, DFC, and APAP where an adsorbate monolayer was also formed. This model was defined as [33]
The adsorption energies (
Calculated statistical physics parameters were used to interpret the pharmaceutical adsorption mechanism. They were obtained from the isotherm data correlation via a nonlinear regression where the next objective function (
Finally, Density Functional Theory (DFT) calculations were performed with GAUSSIAN09 software to understand the electrostatic and reactive properties of APAP, DFC, and NAP molecules. The molecule optimization was carried out with a functional hybrid B3LYP with 6-311++G(
3. Results and Discussion
3.1. MOF Characterization
The results of X-ray diffraction of the synthesized MIL-100-Fe and MIL-101-Fe MOFs are reported in Figure 1. X-ray diffractogram of MIL-100-Fe showed the characteristic diffraction peak at ~11.1° 2θ, thus denoting the formation of a pure phase [35, 36]. This result was consistent with those reported by Chen et al. [35], Forghani et al. [36], Pil-Joong et al. [37], Zhang et al. [38], Fan et al. [39], Mahmoodi et al. [40], Nehra et al. [41], Shah et al. [42], Li et al. [43], Chaturvedi et al. [44], Chen et al. [45], and Chávez et al. [46]. After the adsorption of the different pharmaceutical molecules, the MOF crystalline structure was stable but the intensity of the diffraction peaks decreased. These changes in the MOF crystallinity were attributed to incorporation of each adsorbed pharmaceutical on the adsorbent surface [47, 48]. X-ray diffraction pattern of the MIL-101-Fe showed diffraction peaks at ~5.1, 9.2, 16.7, and 19.5° 2θ, which confirmed the formation of the crystalline structure characteristic of this MOF [4, 49–53]. After the adsorption of pharmaceuticals, the X-ray diffraction patterns displayed some changes. Specifically, the disappearance of some diffraction peaks was observed and a change in the MOF crystallinity was also identified. This result could be related to the instability of MOF structure in the aqueous media generated by the incorporation of adsorbed pharmaceuticals on its surface [4, 47].

X-ray diffraction patterns of (a) MIL-100-Fe and (b) MIL-101-Fe before and after the adsorption of naproxen (NAP), diclofenac (DFC), and acetaminophen (APAP) molecules.

FTIR spectra of both MOFs are reported in Figure 2. They showed a broad absorption band at 3600–3000 cm-1 that was attributed to the stretching vibration of the O-H group [35, 36, 40, 41, 43–45, 52, 54, 55]. Iron-based MOFs showed an absorption band at ~1640 cm-1 related to the C=O stretching vibration due to carboxyl groups [24, 36, 40, 41, 44, 46, 53, 56]. The spectrum of MIL-100-Fe also displayed the absorption bands of the symmetric (~1425 cm-1) and asymmetric (~1378 cm-1) vibration of the O-C-O group [36, 41, 45], CH bending vibrations (~1112 cm-1) of the carboxylate groups in benzene rings [36, 39, 44], and CH vibrations (~760 and 708 cm-1) of these aromatic structures [46]. FTIR spectrum of MIL-101-Fe showed the absorption bands of asymmetric (~1590 cm-1) and symmetric (1390 cm-1) stretching vibration of the carboxyl groups (O-C=O) present in terephthalic acid, thus indicating the presence of the organic linker (i.e., dicarboxylate) in this sample [49, 51–53]. The absorption band of C-H bending vibration (~746 cm-1) was also identified and corresponded to the aromatic ring of dicarboxylic benzene [49, 52, 53]. Finally, the spectra of these iron-based MOFs also contained the Fe-O stretching vibration at ~550 cm-1, which also agreed with the results of other studies that have reported the synthesis of these MOFs [35, 39, 40, 43, 44, 52, 53].

FTIR spectra of (a) MIL-100-Fe and (b) MIL-101-Fe before and after the adsorption of naproxen (NAP), diclofenac (DFC), and acetaminophen (APAP) molecules.
After the NAP, DFC, and APAP adsorption, FTIR spectra of these MOFs showed a decrement in the intensity of the absorption band associated with the -OH group; see Figure 2. This result suggested the formation of hydrogen bonds between pharmaceutical molecules and MOF surface [57]. Similarly, a change in the absorption band of Fe-O stretching vibration (~550 cm-1) was also observed, which could be an indication that the metal clusters played an important role in the adsorption of these pharmaceuticals [58]. There was also a shift in the absorption band (~1590 cm-1) of the stretching vibration of carboxyl groups (O-C=O) of MIL-101-Fe. According to Tomul et al. [59] and Yaah et al. [60], this change in FTIR spectrum could be also associated with the incorporation of pharmaceutical molecules in the MOF surface. To complement the surface chemistry analysis of adsorbent samples, FTIR spectra of the single pharmaceuticals were also included and compared in Figure 2. Results showed that the specific and characteristic absorption bands of pharmaceutical molecules were also identified in the spectra of MOF samples obtained after the adsorption experiments. Therefore, these findings were an indirect evidence of the adsorption of these molecules on the external surface of these MOFs thus agreeing the results reported in other studies [59, 60].
Table 2 and Figures 3 and 4 show the results obtained from the elemental analysis, SEM images, and textural parameters of tested MOFs. In general, these adsorbents were mainly composed of carbon and oxygen. The presence of iron was confirmed in both MIL-100-Fe (4.9 wt%) and MIL-101-Fe (24.2%) thus providing additional evidence of the successful synthesis of these organometallic structures. Note that chlorine was also identified in the MIL-101-Fe sample because FeCl3·6H2O was employed as precursor in its synthesis. SEM micrographs of these MOFS indicated that the size of MIL-100-Fe crystals varied from 0.05 to 0.5 μm, while the MIL-101-Fe particles showed an average diameter of 0.5–1 μm; see Figure 3. These results were consistent with other studies [36, 43, 53, 56, 61]. It was also observed that these MOFs presented an octahedral structure; however, the octahedral form of MIL-101-Fe MOF was imperfect [50–52, 62, 63]. N2 adsorption-desorption isotherms of these MOFs are reported in Figure 4. These N2 isotherms can be categorized between types I and IV of IUPAC classification, which are typical of micro- and mesoporous materials [23, 37, 41, 43, 52, 53]. The specific surface area, total pore, micropore, and mesopore volumes of these adsorbents are given in Table 2. BET surface area of MIL-100-Fe was 768.18 m2/g with a total pore volume of 0.58 cm3/g. This surface area was similar to that reported by Nehra et al. [41] (
Elemental analysis and textural parameters of MIL-100-Fe and MIL-101-Fe MOFs.

SEM micrographs SEM (1000x) of (a) MIL-100-Fe and (b) MIL-101-Fe.


N2 adsorption-desorption isotherms (at -196°C) of (x) MIL-100-Fe and (○) MIL-101-Fe.
3.2. Adsorption of NAP, DFC, and APAP on MIL-100-Fe and MIL-101-Fe MOFs
Adsorption isotherms of NAP, DFC, and APAP obtained with MIL-100-Fe and MIL-101-Fe MOFs are reported in Figure 5. All pharmaceutical isotherms were 2L type according to the Giles classification for liquid phase adsorption [66], which indicated that the adsorption of these compounds was proportional to the adsorbate concentration until reaching the saturation of the available adsorption sites (i.e., oxygenated functionalities and Fe-O clusters of these MOFs). NAP adsorption capacities of MIL-100-Fe and MIL-101-Fe ranged from 0.186 to 1.267 and 0.205 to 2.190 mmol/g at 30°C and from 0.149 to 1.206 and 0.185 to 1.889 mmol/g at 40°C, respectively. For the case of APAP, the adsorption capacities of these MOFs were 0.012–0.151 and 0.080–0.450 mmol/g at 30°C and 0.009–0.134 and 0.073–0.382 mmol/g at 40°C, respectively. DFC adsorption capacities of MIL-100-Fe varied from 0.182 to 1.708 mmol/g at 30°C and from 0.132 to 1.616 mmol/g at 40°C, while MIL-101-Fe showed DFC adsorption capacities from 0.398 to 1.469 and 0.127 to 1.072 mmol/g at 30 and 40°C, respectively. Table 3 reports and compares the adsorption capacities of MIL-100-Fe, MIL-101-Fe, and other MOFs used in the removal of different pharmaceutical from aqueous solutions [2, 4, 8, 18, 67–69]. Several adsorption capacities reported in the literature were lower than those obtained for NAP, DFC, and APAP with MIL-100-Fe and MIL-101-Fe MOFs. Therefore, these materials could be considered as an alternative separation medium for the adsorption of these three pharmaceuticals in water depollution.
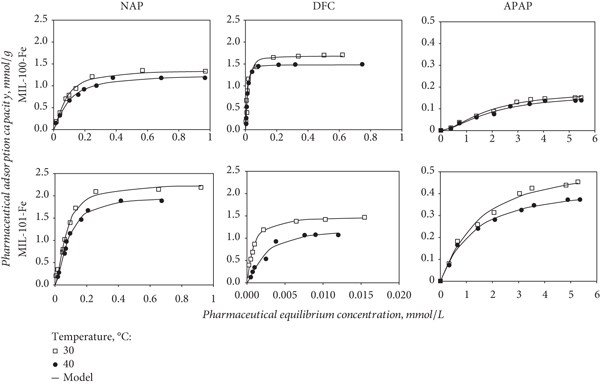
Isotherms of the adsorption of the adsorption of acetaminophen, diclofenac, and naproxen on MIL-100-Fe and MIL-101-Fe from aqueous solution at pH 7.
Adsorption capacities for different pharmaceuticals using MOFs as adsorbents.
In general, MIL-101-Fe showed a better performance than MIL-100-Fe for the adsorption of NAP and APAP, while the highest DFC adsorption capacities were obtained with MIL-100-Fe. The pharmaceutical adsorption capacities of MIL-100-Fe and MIL-101-Fe followed the next trends:
The solution temperature affected the pharmaceutical adsorption with these MOFs; see Figure 5. All the maximum adsorption capacities ranged from 0.14 to 2.19 mmol/g and decreased in 7.9–27% when the solution temperature increased from 30 to 40°C. Pharmaceutical adsorption on both MOFs was affected by the solution temperature according to the next trend:
With respect to the pharmaceutical molecular properties, the literature indicates that the molecular weight and hydrophobicity of these organic compounds could impact their adsorption on MOFs [6, 8]. Figure 6 shows the MEPs of NAP, APAP, and DFC molecules that were calculated with DFT. These electrostatic potentials are illustrated via a color mapping where the scale indicates from the most reactive to the least reactive zone of the molecule (i.e.,

Molecular electrostatic potential of (a) acetaminophen, (b) diclofenac, and (c) naproxen molecules.


Calculated physicochemical parameters for the adsorption of these pharmaceuticals on MIL-100-Fe and MIL-101-Fe MOFs are reported in Figure 7. This statistical physics model showed

Calculated physicochemical parameters of the adsorption of acetaminophen, diclofenac, and naproxen on MIL-100-Fe and MIL-101-Fe from aqueous solution at pH 7.
4. Conclusions
The adsorption of three relevant pharmaceuticals on MIL-100-Fe and MIL-101-Fe MOFs from aqueous solutions has been experimentally studied and modeled to understand their adsorption mechanism. Results showed that the pharmaceutical adsorption capacities of these MOFs were mainly related to the presence of Fe-O clusters where their mesopore structure contributed for the removal of these organic pollutants. Adsorption properties of MIL-101-Fe outperformed those obtained for MIL-100-Fe. Overall, both MOFs showed the lowest adsorption capacities for acetaminophen molecules. The adsorption of naproxen, diclofenac, and acetaminophen on these MOFs was a multimolecular and exothermic process where more than one pharmaceutical molecule can interact with one functional group from the adsorbent surface. The pharmaceutical adsorption properties of MIL-101-Fe MOF were more sensitive to increments of solution temperature and decreased up to 27% for diclofenac molecules. MOF characterization, DFT, and statistical physics calculations indicated that hydrogen bonds, van der Waals and electrostatic forces could be the main interactions involved in the pharmaceutical adsorption on tested MOFs. MIL-101-Fe is an interesting material with better pharmaceutical adsorption capacities than other MOFs reported in literature, and it can be utilized to prepare composites and other materials for water depollution. Therefore, the experimental and theoretical results reported in this study can contribute to enhance and consolidate the application of MOFs as adsorbents of emerging compounds for water treatment and purification.
Footnotes
Data Availability
Data of this paper are available on request to the corresponding author.
Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Acknowledgments
The authors acknowledge the financial support provided by CONACYT (Grant No. A1-S-10035) for the present study.
