Abstract
A hybrid composite based on metal-organic framework (MOF) was chemically fabricated by embedding the magnetic Fe3O4 nanoparticles within amino-functionalized porous La-MOF (MOF/NH2) to produce a highly efficient and reusable composite of MOF/NH2/Fe3O4. Different proper techniques were used for the characterization of surface morphology and chemical arrangement of the prepared MOF/NH2/Fe3O4 composite. The characterization results using various techniques including Fourier transform infrared spectroscopy (FT-IR), X-ray diffraction (XRD), scanning electron microscope (SEM), Brunauer, Emmett, and Teller analysis (BET), and vibrating sample magnetometer (VSM) approved the successful fabrication of MOF with amino arms on its surface besides the well magnetization using magnetic nanoparticles. The MOF/NH2/Fe3O4 composite showed enhanced adsorption capacity (618 mg/g) toward methyl orange (MO) anionic dye which is higher than many commercial reported adsorbents due to the presence of many types of adsorption sites (NH2 groups and lanthanum sites), large surface area of MOF, and the synergetic effect of magnetic nanoparticles. Moreover, the MOF/NH2/Fe3O4 composite showed selective adsorption of MO dye from dye mixtures owing to the electrostatic attraction. Also, the MOF/NH2/Fe3O4 composite retained over 90% of its efficiency for the dye removal even after six successive cycles. So, the present study provided a practical strategy for the design of functional MOF hybrid composites. Furthermore, due to the adaptability of its architectural form, it is a potential adsorbent material for industrial wastewater treatment uses.
1. Introduction
Water pollution due to the existence of organic dyes and toxic heavy metals is a serious problem that faces the world [1]. This problem resulted from the release of industrial wastes directly to the running water and sewage [2]. The real danger arises from the toxic effect of these dyes and metals on the human health and living organisms [3]. Among all pollutants, organic dyes represent the most dangerous class on the environment [4]. These dyes appear in water due to the discharge of industrial wastewater including paper industries, leather industries, and textile industries. Dyes’ danger arises from its stable structure and the difficulty to biodegrade their aromatic complex structure [5]. This makes the organic dyes to be carcinogenic, mutagenic, and very toxic [6]. Subsequently, their removal from wastewater before discharge is mandatory to protect human health and other microorganisms. Great efforts have been done by scientists to achieve proper methods for dye removal from wastewater. Different methods have been accepted for the removal of organic dyes from water including ion exchange [7], membranes [8, 9], adsorption [10–19], precipitation [20], reverse osmosis [21], coagulation-flocculation [22], ozonation [23, 24], electrochemical oxidation [25], and biological treatment [26–28]. Among all methods, adsorption is the widely used method for the removal of dyes due to many factors including low cost of processing, high efficiency, fast and easy separation, no secondary pollution, and high recyclability [29, 30]. The commonly used adsorbents such as natural fibers, zeolites, and activated carbon have limited applications for the removal of organic dyes due to the poor selectivity and low adsorption capacity. So, the fabrication of new adsorbents for dye removal has become an urgent necessity. In this context, new fabricated materials such as graphene [31], carbon nanotubes [32, 33], quantum dots [34], nanocelluose [35], and metal-organic frameworks (MOFs) [36–38] have been investigated as adsorbents for water treatment. Among all adsorbents, MOFs have attracted the attention recently for the water treatment applications. MOFs are classes of fabricated inorganic–organic hybrid porous crystalline materials with magnificent properties such as high thermal stability, large surface area, large amounts of unsaturated metal sites, and tunable pore structure allowing their wide use in different fields of applications such as water treatment [39], gas separation [40], gas storage [41], sensors [42], luminescent materials [43], and drug delivery and storage [44]. In the recent decade, engineered MOFs are widely investigated for the removal of water pollutants. Because metal ion is frequently used as an active site for a variety of applications, choosing the right metal ion for the framework is always critical. Lanthanide-based metals are excellent choice for the fabrication of MOFs due to their high valence state (normally +3) giving high complexation mode and flexibility giving architectural diversity into the MOFs [45]. All these advantages of MOFs make them appropriate applicant for adsorption of different pollutants from water.
However, MOFs as adsorbents have limitations due to the difficulty of separation from adsorption environment as their separation requires filtration and high-speed centrifugation. This makes the magnetization of the MOFs essential for their application as adsorbents for water treatment to ease their separation using an external magnet. Magnetic nanoparticles are favored because they have various advantages, including a wide surface area, low toxicity, low cost, environmental friendliness, and reusability, in addition to being collected by an external magnet and not requiring centrifugation. The dispersibility and magnetism loss are the essential disadvantage of magnetic nanoparticles that result from its tendency to oxidation. Magnetic nanoparticles are usually coated and treated with different materials to promote stability and prevent aggregation. The hybrid magnetic adsorbents have advantages over magnetic nanoparticles, like providing active sites for effective capturing of pollutants and enhance the selectivity and adsorption ability. Subsequently, the association of MOF blocks with magnetic nanoparticles will enhance the capacity of the hybrid during treatment process with solving the issues of adsorbent separation from the medium after adsorption process. This magnetic separation provides the ability for using the adsorbent several times for water treatment. This makes the magnetic MOFs as adsorbent cost-effective from the economical view. In the present study, all these features are taken into account during the fabrication of the adsorbent to enhance its large-scale (industrial) application.
Herein, a nanosized, highly efficient, and recyclable adsorbent based on La-MOFs was synthesized. The La-MOFs were amino-functionalized to produce La-MOFs-NH2 to allow the surface modification of the La-MOFs using the magnetic Fe3O4 nanoparticles and fabricate a composite, namely, La-MOFs/NH2/Fe3O4. The synthesized nanomaterials were characterized using proper techniques including scanning electron microscopy (SEM), Fourier transform infrared spectroscopy (FT-IR), X-ray diffraction (XRD), thermogravimetric analysis (TGA), and Brunauer−Emmett−Teller (BET). The synthesized composite was examined for the removal of methyl orange (MO) as a model dye. Moreover, the adsorption kinetics and adsorption isotherm for MO uptake on the surface of MOFs/NH2/Fe3O4 composite were studied. Also, the reusability of the MOFs/NH2/Fe3O4 composite as adsorbent was determined for MO removal for up to five cycles. Finally, the efficiency of MOFs/NH2/Fe3O4 composite as adsorbent for dye removal was evaluated. The originality and novelty of the present study are based on the following criteria: (1) the magnetization of La-based MOF to enhance its adsorption capacity and reusability toward the adsorption of organic dyes; (2) to the best of our knowledge, there is no information about the capacity of the synthesized composite toward MO dye removal; (3) information on the removal of MO dye will be added to the current literature as a result of this study.
2. Experimental
2.1. Chemicals
Methyl orange (MO) was purchased from Aladdin Co. N,N-Dimethylformamide (DMF), hydrochloric acid, and sodium acetate were purchased from El-Gomhouria Co., Egypt. Ethylene glycol, ferric chloride hexahydrate, lanthanum nitrate hexahydrate, and 2-aminoterephthalic acid were purchased from Sigma-Aldrich. All chemicals were used as received without any further purification. Deionized water was used for the preparation of all solutions.
2.2. Synthesis of Magnetic Nanoparticles
The magnetic Fe3O4 nanoparticles were synthesized using solvothermal method as described in the literature [46]. Typically, a colloidal solution was prepared at room temperature under strong stirring by dissolving 8.2 g of sodium acetate and 2.7 g of ferric chloride hexahydrate in 60.0 mL of ethylene glycol. The stirring process was continued for half-hour then poured into stainless steel autoclave and heated for 10.0 hours at 200°C. After that, the mixture was kept to cool down in room temperature and the formed magnetic Fe3O4 nanoparticles were collected using a magnet. The collected magnetic nanoparticles were washed several times using deionized H2O and methanol. Finally, the Fe3O4 nanoparticles are dried in the oven for 10.0 hours at 65°C.
2.3. Synthesis of Amino-Functionalized La-MOFs
The synthesis of amino-functionalized La-MOFs (MOFs/NH2) was synthesized as described in the literature [47]. Typically at room temperature, 30 mL of DMF solvent was used to dissolve 0.363 g of 2-aminoterephthalic acid and 0.886 g of lanthanum nitrate hexahydrate with continuous stirring for 15 minutes. Then, the solution was poured into stainless steel autoclave and heated for 6.0 hours at 150°C. After that, the solution was kept to cool down at room temperature and the obtained MOFs/NH2 was washed several times with deionized H2O and methanol. Finally, the MOFs/NH2 was dried for 1 day at 65°C in vacuum.
2.4. Synthesis of MOFs/NH2/Fe3O4 Composite
Firstly, 30 mL of DMF solvent was used to dissolve 0.363 g of 2-aminoterephthalic acid and 0.886 g of lanthanum nitrate hexahydrate. Then, 10 mL of DMF solution was used to dissolve 0.115 g of magnetic Fe3O4 nanoparticles under ultra-sonication to prevent the nanoparticles’ aggregation. After that, the magnetic Fe3O4 nanoparticles solution was added to the firstly prepared solution with stirring for half-hours. The mixture was then poured into a stainless steel autoclave and heated for 6.0 hours at 150°C. The obtained solution was kept to cool down at room temperature and the formed MOFs/NH2/Fe3O4 composite was collected using an external magnet and washed several times using methanol and deionized H2O. Finally, the formed composite was dried for 1 day at 65°C. Scheme 1 illustrates the synthesis method of the MOFs/NH2/Fe3O4 composite.

The schematic diagram for the synthesis of MOFs/NH2/Fe3O4 composite.
2.5. Batch Adsorption Study
The adsorption performance of the synthesized MOFs/NH2/Fe3O4 composite was evaluated using MO dye as a model at different experimental conditions. All experiments were performed at 25°C. The adsorption conditions were optimized by studying the effect of different parameters including pH, contact time, initial dye concentration, and adsorbent dose. In each experiment, a certain amount of adsorbent is shaken with 80 mL of dye solution of a certain concentration at 200 rpm. After that, the adsorbent was collected using an external magnet and the solution was examined for the presence of MO dye using UV–Visible spectrophotometer at
The removal efficiency (%) was calculated according to Eq.(2).
Moreover, the reusability of MOFs/NH2/Fe3O4 composite as adsorbent was studied for up to five successive cycles. During the adsorption-desorption, the adsorbent was used to adsorb the dye followed by desorption of the dye from the surface of the adsorbent using methanol. Then, the adsorbent was used for another cycle. After each adsorption experiment, the adsorbent was collected using an external magnet. Finally, depending on the removal efficiencies, the reusability of the adsorbent was evaluated.
3. Results and Discussion
3.1. The Characterization of Materials
The characterization of synthesized materials was achieved using familiar techniques. For the determination of the present functional groups, FT-IR spectra for the synthesized MOF/NH2/Fe3O4, MOF/NH2, and magnetic Fe3O4 nanoparticles were performed in the range of 4000 to 400 cm−1 as shown in Figure 1(a). According to Figure 1(a), the FT-IR spectrum of magnetic nanoparticles showed the appearance of a broad strong band of Fe-O at 584 cm-1 [48]. This characteristic band of magnetic nanoparticles was also observed in the spectrum of MOF/NH2/Fe3O4 which indicated the successful embedding of magnetic nanoparticles within the synthesized composite. Moreover, the MOF/NH2/Fe3O4 and MOF/NH2 spectra showed the appearance of absorption bands at 3475 cm-1, 3354 cm-1, and 1546 cm-1 which are corresponding to the bending and stretching vibration of the NH2 group [49].

XRD (a) and FT-IR (b) of the synthesized Fe3O4 nanoparticles, MOF/NH2, and MOF/NH2/Fe3O4.

For the determination of the phase structure of synthesized materials, XRD patterns of MOF/NH2/Fe3O4, MOF/NH2, and magnetic Fe3O4 nanoparticles were performed in the range of
For the characterization of size and morphology, the SEM analysis was performed for the synthesized Fe3O4 nanoparticles, MOF/NH2, and MOF/NH2/Fe3O4 composite as shown in Figure 2. Figure 2(a) illustrates the SEM image of Fe3O4 nanoparticles that clearly appears to have round biscuit-like morphology with nanoscaled size. Moreover, Figure 2(b) shows the SEM image of amino-functionalized MOFs (MOF/NH2) that appear to have wool ball-like morphology. Figure 2(c) shows the SEM image of MOF/NH2/Fe3O4 microstructured composite that clearly shows the excellent coating of magnetic nanoparticles on the surface of MOF/NH2 to correctly fabricate the composite. Subsequently, the SEM results are in agreement with XRD and FT-IR results confirming the successful loading of magnetic Fe3O4 nanoparticles on the surface of MOF/NH2.

SEM images of Fe3O4 nanoparticles (a), MOF/NH2 (b), and MOF/NH2/Fe3O4 composite (c).


To study the thermal stability of the synthesized materials, TGA curves are shown in Figure 3(a). According to Figure 3(a), Fe3O4 nanoparticles showed the highest thermal stability, while MOF/NH2 showed a similar trend with lower thermal stability than MOF/NH2/Fe3O4 which was attributed to the addition of magnetic Fe3O4 nanoparticles. MOF/NH2/Fe3O4 composite showed gradual weight loss within two stages. The first stage occurred in the temperature range of 30°C to 400°C with a weight loss of 19% and attributed to the evaporation of adsorbed solvent molecules. The second stage occurred in the temperature range of 400°C to 700°C with a weight loss of 21% and attributed to the decay of organic linkers and decomposition of MOF. The thermal stability results according to Figure 3(a) revealed the good stability of the synthesized materials. Moreover, the differences in TGA results indicated the successful construction of the MOF/NH2/Fe3O4 composite.
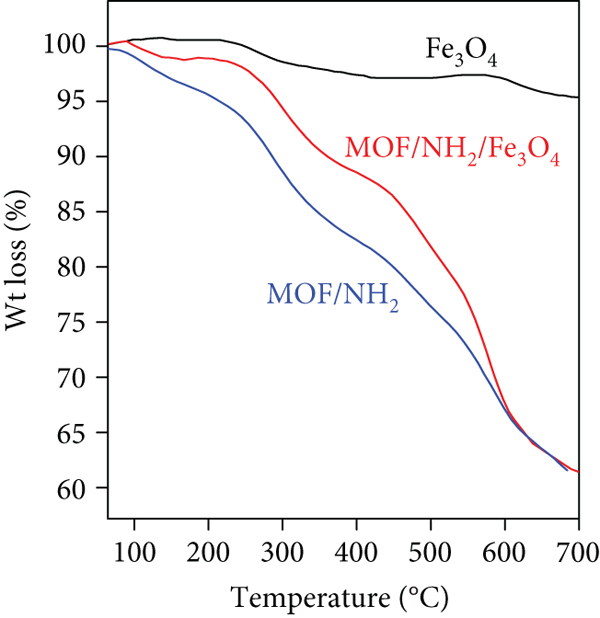
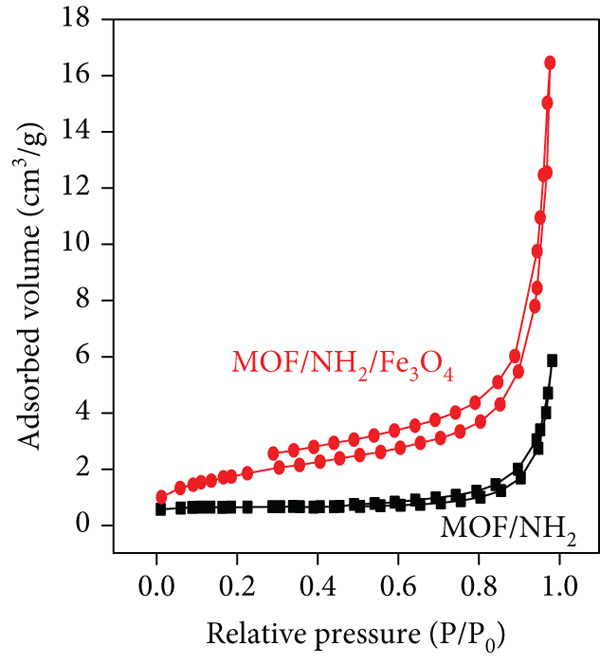
TGA curve (a), magnetization curve (b), and N2 adsorption-desorption isotherms (c) of the synthesized Fe3O4 nanoparticles, MOF/NH2, and MOF/NH2/Fe3O4.

One of the advantages of magnetic adsorbents is the magnetic separation that allows the ease and simple separation from the adsorption medium. Subsequently, the magnetic behavior of the synthesized Fe3O4 nanoparticles and MOF/NH2/Fe3O4 composite was studied using VSM as shown in Figure 3(b). According to Figure 3(b), the MOF/NH2/Fe3O4 composite showed a lower saturation magnetization (16 emu g-1) than Fe3O4 nanoparticles (89.34 emu g-1). This is attributed to the addition of nonmagnetic MOF/NH2 to the magnetic Fe3O4 nanoparticles that caused the drop in the saturation magnetization. So, the paramagnetic behavior of the synthesized MOF/NH2/Fe3O4 composite allows their simple separation using an external magnetic field and enables their reusability several times for water treatment that in turn reduces the overall cost of the treatment.
Furthermore, N2 adsorption-desorption isotherm results of the synthesized materials were used for the characterization of geometrical properties as shown in Figure 3(c). These properties include pore volume, average pore diameter, and specific surface area (SBET). According to Figure 3(c), the MOF/NH2/Fe3O4 composite (36.2 m2 g-1) showed a higher SBET than MOF/NH2 (32.11 m2 g-1) due to the addition of magnetic Fe3O4 nanoparticles. Additionally, the MOF/NH2/Fe3O4 composite and MOF/NH2 showed average pore diameters of 16.35 nm and 16.21 nm (BJH method), respectively, with mesoporous structure. This average pore diameter is appropriate enough for entrapping the dye molecules within their building. So, the addition of magnetic Fe3O4 nanoparticles to MOF/NH2 provides more spaces and active sites to dye uptake and enhances the adsorption capacity.
3.2. Factors Affecting the Adsorption Capacity
For the optimization of adsorption conditions, the effect of different parameters (initial MO concentration, contact time, and pH value) on the dye adsorption on the surface of Fe3O4 nanoparticles and MOF/NH2/Fe3O4 composite was studied as shown in Figure 4. Figure 4(a) shows the plot of initial MO concentration against the adsorption capacity of Fe3O4 nanoparticles and MOF/NH2/Fe3O4 composite. According to Figure 4(a), the increased initial dye concentration from 0 to 400 ppm showed an increase in the adsorption capacity due to the availability of large number of adsorption sites on the surface of the Fe3O4 nanoparticles and MOF/NH2/Fe3O4 composite. After 400 ppm, the adsorption capacities of Fe3O4 nanoparticles and MOF/NH2/Fe3O4 composite were almost constant due to the increased number of dye molecules with regard to the adsorption sites (saturation process). So, 400 ppm is considered the optimum initial concentration at which the adsorption process reached its equilibrium. Also, there is a significant difference in the adsorption capacity between Fe3O4 nanoparticles (124 mg/g) and MOF/NH2/Fe3O4 composite (618 mg/g) indicating the enhanced adsorption capacity resulted from the magnetization of MOF crystals. This enhanced capacity is attributed to the highly porous construction of MOF/NH2/Fe3O4 besides the large surface area of MOF that speeds the mass transfer for quick dye uptake [51]. Another important factor affecting the adsorption process is the contact time. Subsequently, the adsorption capacity of Fe3O4 nanoparticles and MOF/NH2/Fe3O4 composite for MO adsorption was studied in the range of 0 to 250 min as shown in Figure 4(b). According to Figure 4(b), the increased contact time caused an increase in the adsorption capacities of Fe3O4 nanoparticles and MOF/NH2/Fe3O4 composite until it reaches the equilibrium at 180 min and 60 min, respectively. After that time, the adsorption capacity almost remains constant. This behavior is attributed to the availability of large numbers of active sites for the dye uptake in the first stage, while the second stage in which the equilibrium was reached represents the saturation of all active sites with MO dye. Also, there is a significant difference between optimum contact time for Fe3O4 nanoparticles and MOF/NH2/Fe3O4 composite reflecting the role of MOF in driving the adsorption process.

The effect of initial dye concentration (a), effect of contact time (b), effect of pH (c), and Zeta potentials (d) for the adsorption of MO on the surface of Fe3O4 nanoparticles and MOF/NH2/Fe3O4.



Moreover, pH as a significant factor greatly affecting the adsorption process was studied in the range of 4 to 10 as shown in Figure 4(c) for the adsorption of MO on the surface of Fe3O4 nanoparticles and MOF/NH2/Fe3O4 composite. According to Figure 4(c), the adsorption capacity for Fe3O4 nanoparticles showed a decrease with the increased pH values from 4 to 10. This behavior could be attributed to the –ve charge on the nanoparticle surface that increased with pH increase that causes an electrostatic repulsion between nanoparticles and anionic dye. But, pH value has a great effect on the adsorption capacity of MOF/NH2/Fe3O4 composite. The adsorption capacity of MOF/NH2/Fe3O4 composite toward MO dye was increased by increasing pH value from 4 to 6 due to the protonation of functional groups (+ve) on the surface of composite that induces the electrostatic attraction between adsorbent and anionic MO dye (as shown in Zeta potential results in Figure 4(d)), while at pH>6, the adsorption capacity of the MOF/NH2/Fe3O4 composite was decreased with the any increase in the pH value. This behavior is attributed to the repulsion between the anionic dye and the negatively charged adsorbent at high pH values. Additionally, the decreased adsorption capacity of MOF/NH2/Fe3O4 composite at high pH values may be attributed also to the repulsion between the anionic MO dye and excess OH- groups in the solution. So, pH has complicated effect on the adsorption of pollutants on the surface of MOF/NH2/Fe3O4 composite.
3.3. Adsorption Isotherm and Kinetics
To understand the adsorption of MO dye on the surface of MOF/NH2/Fe3O4 composite, it is important to study the isotherm and kinetic models [52, 53]. Herein, the Langmuir and Freundlich isotherm models were used to fit the adsorption data of MO dye on the surface of MOF/NH2/Fe3O4 composite and Fe3O4 nanoparticles. The Langmuir and Freundlich isotherm models can be represented according to Eq.(3) and Eq.(4), respectively [54, 55].
The isotherm and kinetics parameters for the adsorption of MO dye on the surface of Fe3O4 nanoparticles and MOF/NH2/Fe3O4 composite.
The Langmuir model suggests that the adsorption active sites on the surface of adsorbent are energetically identical and therefore capturing the adsorbate molecules as a monolayer over the homogenous surface of adsorbent [56, 57], while the Freundlich model suggests that the adsorption active sites on the surface of adsorbent are not the same energetically and therefore capturing the adsorbate molecules as multilayers over the heterogeneous surface of adsorbent [58, 59]. The fitting of experimental data for the adsorption of MO dye on the surface of MOF/NH2/Fe3O4 composite and Fe3O4 nanoparticles with the Langmuir and Freundlich models are presented in Figures 5(a) and 5(b). Regression coefficient (

The fitting of adsorption data to the Langmuir model (a), the Freundlich model (b), pseudo 1st order (c), and pseudo 2nd order for the adsorption of MO dye on the surface of MOF/NH2/Fe3O4 composite and Fe3O4 nanoparticles.



For more understanding of the adsorption mechanism and speed, the adsorption data were analyzed using kinetic models. So, the experimental adsorption data for the removal of MO dye on the surface of MOF/NH2/Fe3O4 composite and Fe3O4 nanoparticles were fitted using the well-known kinetic models, pseudo 1st order model and pseudo 2nd order model as shown in Figures 5(c) and 5(d). The pseudo 1st order and pseudo 2nd order models can be expressed according to Eq.(5) and Eq.(6), respectively [60, 61].
The pseudo 1st order model suggested that the adsorption of adsorbate ions was achieved on the adsorbent surface via physisorption mechanism and subsequently depending on the number of the vacant active sites [62]. But, the pseudo 2nd order model suggested that the adsorption of adsorbate ions was achieved on the adsorbent surface via chemisorption mechanism through exchange or sharing the electrons [63]. Like the isotherm study, the regression coefficient (
3.4. Thermodynamic Parameters of Adsorption
For a better understanding of the behavior of MO dye adsorption on the surface of MOF/NH2/Fe3O4 composite, adsorption thermodynamic parameters were calculated. Moreover, the thermodynamic parameters help the determination of whether the adsorption process is spontaneous. The thermodynamic parameters include entropy change (
The thermodynamic parameters for the adsorption of MO on the surface of MOF/NH2/Fe3O4 composite.
According to Table 2, the MO adsorption on MOF/NH2/Fe3O4 surface is a spontaneous process as the
3.5. Comparative Study
For the assessment of adsorbent performance, the maximum adsorption capacity of MOF/NH2/Fe3O4 composite was compared with many commercial adsorbents of MO dye as tabulated in Table 3. According to Table 3, those previously reported adsorbents showed clearly lower adsorption capacities than MOF/NH2/Fe3O4 composite. By comparing,
Comparison the adsorption capacity of MOF/NH2/Fe3O4 composite with previously reported adsorbents for the removal of MO dye.
3.6. Selective Adsorption
The selective adsorption of MO by MOF/NH2/Fe3O4 composite was performed using dye mixtures as shown in Figure 6. The mixtures of RB/MO and MB/MO were used to perform the selective adsorption study with dye initial concentrations of 10 mg/L for RB and MB dyes and 100 mg/L for MO dye, mixture volume of 50 mL, and an adsorbent dose of 20 mg. The solution was mixed for 30 minutes for adsorption followed by the collection of the adsorbent using an external magnetic field and the solution was examined for the presence of dyes. Then, the selective adsorption efficiency was calculated according to Eq.(9).

Uv-vis absorption spectra of MO/MB (a) and MO/RB (b) before and after selective adsorption on the surface of MOF/NH2/Fe3O4 composite.
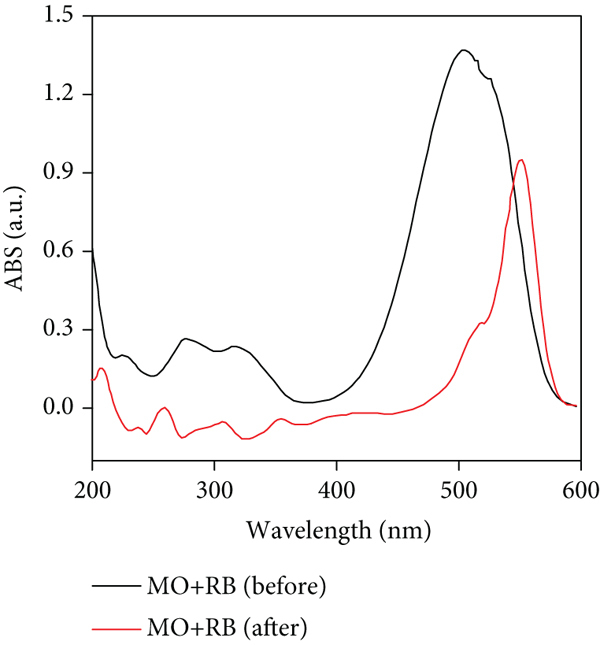
According to Figure 6, MO dye was adsorbed selectively in the presence of RB and MB dyes on the surface of the MOF/NH2/Fe3O4 composite.
Uv-vis spectra of dye mixtures after adsorption showed the reduction of the MO dye band indicating selective adsorption. After adsorption, the selective adsorption efficiency of MO dye was 93% and 90% in the presence of MB and RB dyes, respectively. These results indicate the efficiency of the synthesized composite for the removal of MO from an aqueous solution. The results can be interpreted based on electrostatic attractions where the positively charged composite can adsorb the anionic dyes stronger than the cationic dyes.
3.7. Desorption and Reusability Study
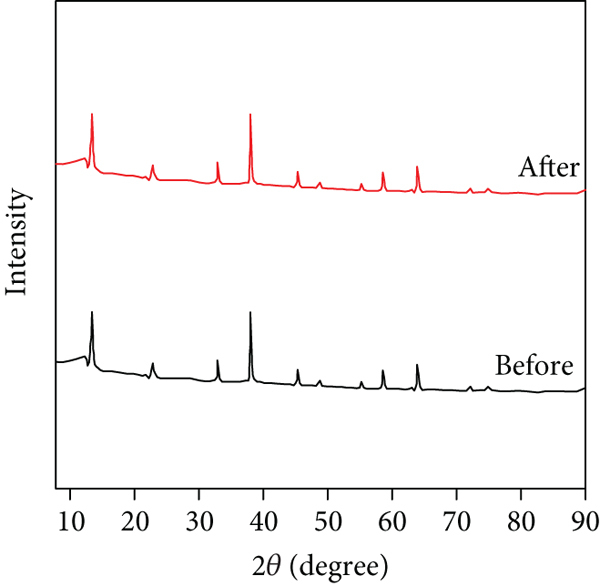
A reusable adsorbent is cost-effective and in agreement with green chemistry [77–79]. In this context, the reusability of MOF/NH2/Fe3O4 composite was examined using methyl alcohol as an eluent for up to six successive cycles. The adsorption–desorption cycles were investigated six times as shown in Figure 7(a). During each cycle, the adsorbent was mixed with dye solution for adsorption. After that, the adsorbent was collected using an external magnet for desorption. The dye was desorbed from the surface of the adsorbent using methyl alcohol as eluent and the adsorbent was dried for usage in the next cycle. After each cycle, the solution was examined for the presence of dye, and the removal efficiency was calculated. According to Figure 7(a), the 1st cycle has the highest efficiency due to the presence of a large number of fresh adsorption sites. The next cycles showed a little drop in the removal efficiency due to the damage to nonrenewable adsorption sites. However, the last cycle showed that MOF/NH2/Fe3O4 composite retained more than 90% of its efficiency. As can be shown in Figure 7(b), the composite’s XRD after the sixth cycle of MO dye adsorption-desorption is identical to that obtained before dye adsorption, emphasizing the composite’s stability during the reusability study. According to the reusability results, MOF/NH2/Fe3O4 composite is considered a good regenerable adsorbent. The MOF/NH2/Fe3O4 composite had advantages such as reusability at several stages without any change in the magnetic property, easy magnetic separation from aqueous solutions by the magnet, and fast pollutant adsorption.

Reusability of MOF/NH2/Fe3O4 composite for the removal of MO dye up to six cycles (a) and XRD pattern of MOF/NH2/Fe3O4 composite before and after MO dye adsorption (b).
4. Conclusion
A reusable, highly efficient, and low-cost dye adsorbent, amino-functionalized MOF embedded with magnetic nanoparticles (MOF/NH2/Fe3O4) was well fabricated and the proper techniques were used for characterization. The synthetic approach involved the functionalization of porous MOF with amino groups to act as arms for dye capturing followed by the embedding of magnetic nanoparticles within MOF pores to increase its adsorption efficiency besides the ease of collection with an external magnet. The lanthanum sites and the large surface area of the synthesized adsorbent could be responsible for the effective adsorption of MO dye from an aqueous solution with
Footnotes
Data Availability
The research data used to support the findings of this study are included within the article.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Acknowledgments
The authors extend their appreciation to the Deanship of Scientific Research at King Khalid University for funding this work through the research group program under grant number RGP.2/57/43. Also, this research was funded by Princess Nourah bint Abdulrahman University Researchers Supporting Project number PNURSP2022R42, Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
