Abstract
Nanofluids made of TiO2 and multiwalled carbon nanotubes are the focus of our study. Using TiO2 and water as the basic fluids, nanofluids were created in 100 : 0, 75 : 25, and 50 : 50 proportions. Carbon nanotubes (CNTs) were disseminated into these base fluids of three types at 0.125, 0.25, and 0.5 percentages. Over the course of two months, the change of zeta potential is monitored to assess the stability of the dispersion. XRD analysis and SEM and TEM analysis are carried out for TiO2 nanofluids. Nanoparticles were analyzed using EDAX analysis to identify their composition. SEM examination was used to investigate the morphology for TiO2 nanoparticles. Particles of TiO2 produced in this manner seemed to have an average diameter of 27 nm. Nanofluids have a poor stability, which is the most important aspect of employing them, although most studies did not focus on this. When it comes to nanofluids, this study is unique in that it provides an in-depth look of surface modification approaches that have been employed by researchers to address these issues, as well as an evaluation of their stability over two months. Solubility is improved by acid treatment of CNTs, which results in functional groups on the surface of CNTs.
1. Introduction
1.1. Nanotechnology
Nanotechnology is an innovative idea presented in the 21st century, which has the potential to change a material’s characteristics in many ways. A nanosized component in coolants has emerged as a potential alternative additive. The development of nanofluids has been a recent advance in nanotechnology. Nanofluids are colloids of nanometric materials in liquids like water/ethylene glycol/oil. A particle is demarcated as a small entity that, in terms of its transport and properties, acts as a whole unit.
On the other hand, particles of ultrafine size range from 1 to 100 nanometers. Nanoparticles might or might not show size-linked properties that substantially differ with those present in small particles or whole materials. Research on nanoparticles is currently a field of extreme scientific importance due to a broad range of possible uses in mechanical, biochemical, and electronics [1–3].
1.2. Nanofluids and Their Uses
Nanofluids have been proved to be useful in a wide range of applications throughout the last several decades. Industrial equipment’s heat transfer efficiency may be improved if these materials were used. Research on nanofluids has focused on improving thermal conductivity and heat transfer rather than viscosity since its introduction relatively recently [4, 5]. In a nanofluid, one or more different kinds of nanoparticles are dissolved in a base fluid.
Compared to micro fluids, the benefits of nanofluids in heat transfer applications is owing to their more excellent thermal conductivity and lesser viscosity. Researchers have been studying nanomaterial technologies to be applied for improved heat transfer since the 1990s. They concentrated on the research work relating to highly efficient cooling technology heat transfer. Nanofluids have tremendous potential for application in electricity, chemical engineering, automotive, building, microelectronics, knowledge, and other fields and became a research hotspot in the fields of materials as well as physics and chemistry. As of date, several metallic oxide nanoparticles like CuO, Al2O3, TiO2, and Fe3O4 have been used as mixtures to prepare nanofluids and assess the transport properties [6].
1.3. Carbon Nanotubes and Their Properties
A carbon nanotube is composed of carbon, arranged in a tubular pattern with a diameter in nanometer scale. CNT is unusual due to the strong bonding between the atoms, and the tubes may have drastic aspect ratios. The carbon nanotubes’ tensile strength is about 100 times greater than that of the same-diameter material. That strength accounts for two things. The first is the strength that the interlocking covalent carbon-to-carbon bonds provide. There are abundant applications of carbon nanotubes which take full benefit of CNTs’ distinctive aspect ratio and properties like mechanical strength and electrical and thermal conductivity. Among these options for particles, carbonaceous materials, mainly carbon nanotubes (CNTs), absorb the most spectrum energy from the sun possible [7].
CNTs will likely play a central part in the creation of solar thermal plants based on nanofluids. It has been difficult to use CNTs because of their poor dispersion in base fluids. CNTs are difficult to mix with the base liquid because of their thick interparticle contact caused by Van der Waals interactions and their water-repelling nature.
Several physical and chemical approaches to attain stable dispersion of CNTs have been developed till date [8], mainly for dispersion stability at temperatures significantly inferior to those expected in traditional solar thermal plants. Although physical procedures, including ultrasonication and surfactant usage, can efficiently mix CNTs at low temperatures, it has been found that the stability of the resulting dispersion decreases significantly as temperatures touch (or surpass) 100°C [9]. Furthermore, chemical surface modification has been commonly used in the literature to create extreme stability. The production of stable MWCNT nanofluids for usage in high-temperature devices necessitates the optimum blend of the base fluid and CNTs and modification technique for the anticipated working temperature range to be defined. Carbon nanotubes are characterized using FESEM and FTIR spectroscopy which was explained by Feizy et al. [1] who explained the same process using graphene oxide.
It is possible to use CNTs in a variety of ways. The initial usage of CNTs as electrically conductive fillers in polymers was in the form of a powder. They are also employed in fiber composites nowadays. The blades of wind turbines and the hulls of maritime security vessels are two examples. Lithium batteries integrated carbon nanofibers that are wires produced from CNTs, before the end of 2005. Even sports equipment including tennis rackets, baseball bats, and bicycle frames may benefit from the use of carbon nanotubes. Figure 1 shows the structure of carbon nanotube.

Carbon nanotube.
Table 1 shows the properties of carbon nanotubes.
Carbon nanotube properties.
1.4. Titanium Dioxide and Its Properties
As you can see, titanium dioxide is made up of two elements: barium and sulfuric acid. In this case, four oxygen atoms are joined to a sulfur atom. As the mineral barite, titanium dioxide (TiO2) is a barium sulfate salt. Insoluble in water and alcohol but soluble in strong acids, it is a white solid crystal. It does not have a smell. Titanium dioxide is a divalent metal that is found in alkaline environments. It is nontoxic and may be used in medical settings without fear of side effects. It is commonly utilised in the production of oil and natural gas to acquire high-density drilling fluids by keeping the boreholes free of rock.
Table 2 shows the properties of TiO2.
Properties of TiO2.
2. Literature Review
Aviles et al. [2] investigated mild acid oxidative treatments for modification of MWCNTs. They studied the effect of acids like HNO3, H2SO4, and H2O2 at moderately low concentrations, the small-time duration of treatment, and low power of sonication to achieve efficient functionalization of MWCNTs with minimal damage to CNTs.
Chen et al. [3] proposed wet-mechanochemical modification to functionalize MWCNTs. The FTIR results validate that the nonreactive surfaces of CNTs could be successfully modified with numerous hydroxyl groups. Surface-modified MWCNTs exhibited greater values of zeta potential demonstrating their outstanding dispersibility in polar solvents.
Vaisman et al. [8] considered the impact of surfactants in the preparation of a stable suspension of carbon nanotubes. Deagglomeration and even dispersion are challenges for producing high property enhancement as carbon nanotubes incline to form microscale aggregates. This would lead to inferior mechanical and thermal performance.
Xuan et al. [10] selected salt and oleic acid as the surfactant to improve the stability of transformer oil-copper nanofluid and water-Cu nanofluid, correspondingly. However, the use of surfactant could produce abundant amounts of foam which would reduce the effectiveness of nanofluids at higher temperatures. In order to overcome the issue, oxidation of carbon nanotubes using acid or bases is suggested. CNTs due to their large surface areas react with chemicals which could form COOH- and OH- functional groups on the surface of CNTs.
Yang et al. [11] presented a simplistic method for surface treatment of multiwalled carbon nanotubes (MWCNTs) with an acidic solution. During the oxidation with acids, the surface of MWCNTs was added with hydroxyl, carbonyl, and carboxyl groups. MWCNTs oxidized in the nitric and sulfuric acids were found to be having more functional groups compared to MWCNTs oxidized with hydrogen peroxide and sulfuric acids.
Zhang et al. [12] functionalized carbon nanotubes using potassium permanganate aided by a phase transfer catalyst. XPS characterization confirmed that functional groups are formed on the surface of CNTs. This technique is identified as an economical method for large-scale functionalization of CNTs.
3. Purification of Carbon Nanotubes
M/s Ultrra Nanotech sold multiwalled carbon nanotubes made utilising the CVD technique. The CNTs have a diameter of 30–50 nm and a length of 3–15 nm. Analytical-grade chemicals are used for all other purchases. The immaculate CNTs have a purity of 95 percent and are strongly intertwined. As received, pristine carbon nanotubes contain lots of impurities like metal particles and amorphous carbon in the soot. With impurities, the carbon nanotubes tend to get entangled and form agglomerates.
Numerous recent studies have proven that dispersion stability is indeed a critical property for optimising the thermophysical characteristics of nanofluids, most notably thermal conductivity [4–7]. The ultimate properties of nanofluids were deduced from their dispersion stability. Multiwalled carbon nanotubes in their purest form are hydrophobic and insoluble in polar solvents, such as water, monoethylene glycol, and ethanol. Clusters of nanotubes form and settle inside the liquid medium because to their hydrophobicity, resulting in property loss. The researchers’ standard method is to distribute carbon nanotubes in base fluids using a surfactant. Surfactants increased fluids’ foaming propensity, resulting in reduced heat transfer rates [9, 13–15].
The CNTs are purified and oxidized in a three-step procedure. CNTs were initially calcined for 1 hour at a temperature of roughly 575°C to eliminate any amorphous carbon as from powder. In the second phase, hydrochloric acid is utilised to eliminate any impurities that may have collected during the CVD process of producing CNTs. Following thereafter, the residue was continuously rinsed with DI water until it reached a pH of zero. The third stage included oxidizing the modified CNTs in a 3 : 1 volume ratio combination of H2SO4 and HNO3 4 molar each for three hours. We then used distilled water to wash and dry the residue overnight at 60°C in an oven after the reflux process.
3.1. Stability of Nanofluids
The clustering of nanoparticles leads to settling and blockage with a penalty of reduction in nanofluid thermal conductivity. Therefore, the factors affecting the dispersion stability of nanoparticles need to be examined and analyzed. This section will include the methods of stability assessment, ways to improve nanofluid stability, and nanofluid stability mechanism.
3.2. Zeta Potential Analysis for Measuring Stability
Zeta potential indicates the difference in potential amid the dispersal medium (nanomaterials) and the motionless film of the fluid close to the suspended object. The significance of zeta potential is that its outcome might be connected to stability. While low zeta potential solutions appear to coalesce or oscillate, a value of 25 mV (+ or − value) is taken as the point dividing less-charged surfaces concerning highly charged surfaces. Zeta potential colloids between 44 and 65 mV were assumed to have excellent stability, and those with more than 65 mV have exceptional stability. Besides, surfactant mixing in base fluids will increase the stability of the nanofluid suspensions. Zetasizer is used to assess zeta potential and also to analyze the absorption spectrum of the surfactants on the nanoparticles.
3.3. Carbon Nanotube Purification and Oxidation
Many studies have shown that improving the thermophysical properties of nanofluids, notably their thermal conductivity, necessitates ensuring that dispersion stability is maintained. For this reason, they cannot be dispersed in water, monoethylene glycol, alcohols, or any of the other more hydrophobic polar solvents. Agglomeration of nanotubes would occur owing to hydrophobicity, which would result in the loss of characteristics. CNTs are often dispersed in base fluids by using a surfactant. The foaming propensity of fluids was increased by the addition of surfactants, resulting in lower heat transfer rates.
3.4. Synthesis of TiO2 Nanoparticles
To synthesize TiO2 nanoparticles, titanium tetra isopropoxide was purchased (TTIP Sigma-Aldrich, AR grade,

Preparation flowchart of TiO2 nanoparticles.

TiO2 nanopowder.
3.4.1. Characterization
An investigation into the properties of nanoparticles was conducted. The structural characteristics of TiO2 nanoparticles were investigated at temperatures ranging from 10° to 80°C using an X-ray diffractometer equipped with 40 kV/15 mA radiation. To measure titanium dioxide nanoparticle absorption, we used a UV-visible spectrophotometer, the Shimadzu 2700 UV-visible spectrophotometer. Synthesized TiO2 nanoparticles were subjected to high-resolution scanning electron microscopy (SEM Carl ZEISS) and energy-dispersive X-ray spectroscopy before being subjected to chemical analysis using PerkinElmer FT-IR spectroscopy 400-4000 cm–1 (EDXS). The surface shape and texture of a nanoparticle were examined using a TEM (Titan).
3.4.2. Base Fluid and Nanofluid Production
TiO2 nanofluids of three kinds (50 : 50, 75 : 25, and 100 : 0) are used in this experiment. An Ultra probe sonicator was used to disseminate the oxidized carbon nanotubes into the coolant at 0.125, 0.25, and 0.5 Wt percent.
3.5. HRTEM Images of CNTs
3.5.1. Microstructural Analysis of CNTs
Figure 4 shows the HRTEM image, and Figure 5 shows the SEM image of CNTs.

HRTEM image of CNTs.

Microstructural analysis of CNTs.
3.5.2. XRD Analysis
Constructive interference between monochromatic X-rays and crystals is the basis of X-ray diffractometers. CRTs are used to create the radiation, which is then filtered to produce monochromatic radiation, collimated, and focused on the specimen. To check the nature of the materials using XRD patterns, you have to look at the nature of Bragg’s peaks appearing in the XRD pattern. If you get a very broad humped peak, then the material will be amorphous with short range ordering. If you get sharp peaks in the XRD pattern, then the material is crystalline.
3.5.3. SEM and TEM Analysis of TiO2 Nanoparticles
An electron beam scans a sample to provide a magnified picture for study in scanning electron microscopy (SEM). For solid inorganic materials, it is known as SEM analysis and SEM microscopy and is utilised for microanalysis and failure analysis. To create indications on the interaction of electrons, the scanning electron microscope utilises kinetic energy as its working principle. Figures 6(a)–6(d) show the SEM, TEM, and EDX spectral images of TiO2 nanoparticles.

(a, b) SEM images of TiO2 NPs. (c) TEM micrograph of TiO2 NPs. (d) EDX spectra of the TiO2 NPs.



In order to see crystalline elements and photons, we require secondary electrons, which include backscattered electrons and diffracted backscattered electrons. SEM examination was used to investigate the morphology for TiO2 nanoparticles. Particles of TiO2 produced in this manner seemed to have an average diameter of 27 nm, as seen in Figure 6(a). Particles of powder were discovered to be a little clumped. The TiO2 nanoparticles were analyzed using EDAX analysis to identify their composition. Only Ti and oxygen (O) were shown to have peak values in Figure 6(b), with no additional peak values for any other metals. TiO2 particles are thus verified to exist. Figure 6(c) shows the TEM micrograph of TiO2 NPs, and Figure 6(d) shows EDX spectra of the TiO2 NPs.
4. Results and Discussions
4.1. XRD Analysis
The XRD pattern indicated the TiO2 nanoparticle phase, purity, structure, and average crystallite size values displayed in Figure 7. The X-ray diffraction peaks are observed at 25.38°, 37.97°, 48.14°, 54.40°, 55.20°, 62.71°, and 75.30° and correspond to Miller indices (hkl) value of (101), (004), (200), (105), (211), (118), and (215), respectively. The obtained results were confirmed using JCPDS Card No. 78-2486 (Joint Committee on Powder Diffraction Standards) [16, 17]. Debye-Scherrer’s equation was used to get the average crystalline particles size (Equation (1)).

XRD crystalline nature of TiO2 nanoparticles synthesized.
Debye-Scherrer’s equation [18] is given by
To calculate the crystalline size, the (101) plane was chosen. The as-prepared TiO2 NPs produced have an average crystalline size of 27 nm.
4.2. UV-DRS Analysis
The bandgap of synthesized TiO2 nanoparticles was investigated using a UV-visible spectrophotometer. The prepared TiO2 nanoparticles display significant absorption in the UV range because of the broad forbidden bandgap transition and reduced absorption in the visible range due to surface imperfections in TiO2 nanoparticles.
It is possible to quantify the amount of light that a material absorbs using UV-Vis (or spectrophotometry). Light passing through a sample is measured in relation to the light passing through a reference sample or blank. The absorbency of a material may be measured using ultraviolet spectroscopy. Measurements are made by mapping the interaction between light and matter. Excitation or deexcitation occurs when matter absorbs light, resulting in the formation of a spectrum.
The absorption of intrinsic bandgap in TiO2 nanoparticles results in a prominent absorption peak at 356 nm [19] as shown in Figure 8(a). Using Tauc’s plot method, the optical bandgap of TiO2 NPs was determined to be 3.1 eV (Figure 8(b)). The measured absorption edge of TiO2 nanoparticles at 356 nm is markedly blue-shifted compared to tetragonal anatase TiO2 nanoparticles’ bulk bandgap wavelength (345 nm) [20]. The presence of a significant 356 nm spike in the wavelength range 200–600 nm supports the production of TiO2 nanoparticles. Equation (2) can be used to calculate the energy gap of TiO2 manufactured using the green method.

(a) Spectrum of UV-DRS absorption of synthesized TiO2 NPs. (b) Tauc’s plot of synthesized NPs.

The UV-visible absorption spectra of TiO2 demonstrate that it operates as a photocatalyst as well as responds significantly due to the production of reactive species in the exposed to UV light. For each molecular orbital, the particle’s potential energy state is multiplied by the number of molecular orbitals in its vicinity. Thus, absorbance will occur at greater energies, resulting in shorter wavelengths, which will be absorbed.
4.3. FT-IR Analysis
TiO2 NP spectra in the 400–4000 cm1 range produced via a chemical method are shown in Figure 9. O–Ti–O bonding occurs in a highly crystalline phase at a peak of 698 cm1 in this curve [21–23]. Surface-adsorbed water and hydroxyl groups are represented by bands centred at 1624 cm1 and 3422 cm1, respectively. For TiO2 NPs, C–H stretching band peaks at 2918 MHz and 2849 MHz indicate that all organic components have been eliminated from the samples following calcination. The 1381 cm-1 band indicates the elongation of the aromatic ring’s C-C bond as well as the bending of the phenolic molecule’s C-O link. The band at 1120 cm-1 is caused by the asymmetric stretching of the C-O bond in polymeric material [24–26].
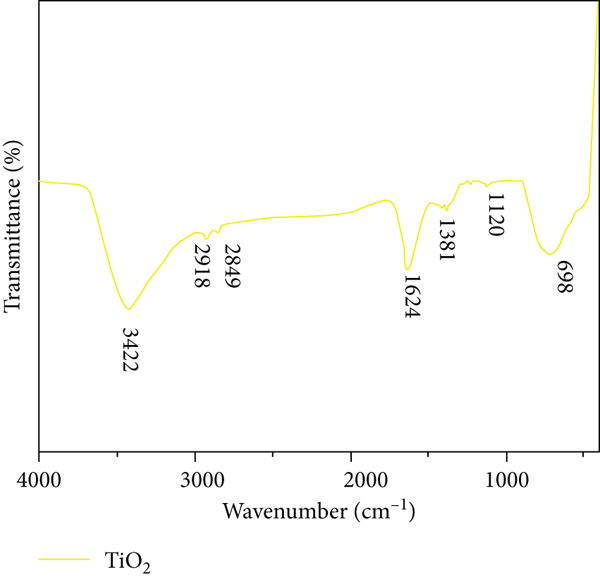
Synthesized TiO2 nanoparticle FT-IR spectrum.
4.4. Stability of Nanofluids
The stability of colloidal fluids is determined by their zeta potential. The zeta potential describes charged particle repulsion. The nanofluids are stable above and below the threshold value of 25. The zeta potential for TiO2-water solutions containing oxidized CNTs is evaluated for 60 days [27, 28].
Figures 10–12 depict the evaluation of the stability among all nanofluids during a two-month period. Considering a 100% TiO2 water combination, the zeta potential of nanofluids, comprising pristine and oxidized CNTs, is shown in Figure 10. Considering a TiO2-water 75 : 25 combination, the zeta potential of nanofluids comprising pristine and oxidized CNTs is shown in Figure 11 over the course of 60 days. Using a 50 : 50 mixture of TiO2 and water, the graph in Figure 12 demonstrates the zeta potential variations of nanofluids, including pure and oxidized CNTs, over the course of 60 days.
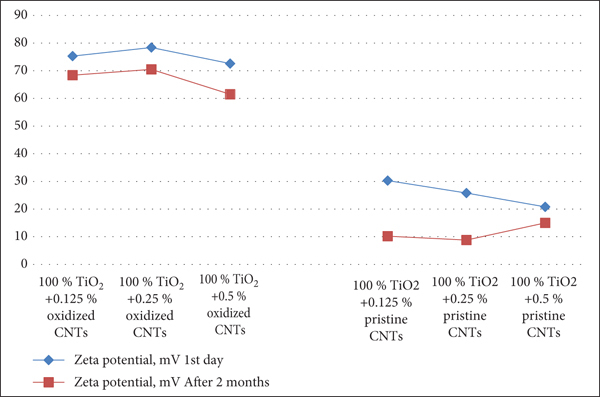
Zeta potential changes of nanofluids including pristine and oxidized CNTs during 60 days for a 100 percent TiO2-water combination.

Zeta potential changes of nanofluids including pristine and oxidized CNTs during 60 days for a TiO2-water (75 : 25) combination.

Zeta potential changes of nanofluids including pristine and oxidized CNTs during 60 days for a 50 : 50 TiO2-water combination.
Figures 10–12 demonstrate that CNTs with changed surfaces are far more stable with coolants than CNTs with unmodified surfaces. Day by day, the zeta potential for coolants containing clean CNTs fluctuates dramatically. The zeta potential difference of oxidized CNT dispersion for two months is small, showing strong suspension stability. For example, CNTs may establish hydrogen bonds with monoethylene glycol and water molecules, making them stable in nanofluids because of the carbonyl and carboxyl groups on them. There is evidence that surface-modified CNTs are more stable in coolants than pure CNTs. Day by day, the zeta potential of coolants containing clean CNTs fluctuates dramatically.
5. Conclusion
This work demonstrated in detail how to make TiO2 nanoparticles in an ecologically benign and cost-effective way, using just a few simple steps. The XRD analysis of the generated NPs verified their tetragonal form.
It was determined that the nanoparticles were 27 nm in diameter by using the Debye-Scherrer equation Scanning electron microscopy and transmission electron microscopy were used to confirm the nanoparticles’ size and structure UV-Vis tests confirmed the creation of TiO2 nanoparticles, which had a high peak at 298 nm, according to the results. The bandgap of the nanoparticles was calculated using the Tauc plot and was found to be around 3.48 eV. As exposed to ultraviolet light, the nanoparticles created deteriorated by 94 percent when compared to the MB dye The environmentally friendly synthesis of TiO2 nanoparticles is a critical performer in the wastewater cleanup and environmental remediation activities that take place TiO2 nanofluids distributed with pristine CNTs have extremely low zeta potential on the first day and after two months, and as a result, one might assume that the zeta potential is relatively low Dispersion of CNTs in nanofluids, which resulted in good stability from day one to day 60, was clearly obvious. According to this research, surface-modified CNTs are far more stable in coolants over pure CNTs Over the period of 60 days, the zeta potential for coolants containing pristine CNTs varies substantially. Over the course of two months, the zeta potential of dispersed oxidized CNTs does not change, showing that the suspensions are stable
Footnotes
Data Availability
The data used to support the findings of this study are included in the article.
Conflicts of Interest
The authors declare that they have no conflicts of interest regarding the publication of this paper.
