Abstract
The ZIF-8 crystals were successfully postsynthetically modified using methylamine (MA), ethylenediamine (ED), and N, N
1. Introduction
Nowadays, the rising level of carbon dioxide in the atmosphere has become one of the biggest problems worldwide. As the main component of greenhouse gases, CO2 contributes the most to global warming and climate change [1]. To date, the global CO2 concentration has increased from 270 to 400 ppm with the industrial development and is anticipated to approach 950 ppm by 2100, which will bring about sincere environmental problems such as extreme weather, glacial melting, and ocean acidification [2, 3]. Therefore, reducing carbon dioxide emission has been proposed to be a scientific challenge of the highest order and thus has been explored by many researchers throughout the world utilizing different technologies [4–7]. Among these technologies, chemical absorption with aqueous organic amines like diethanolamine (DEA) has been widely used for CO2 capture from industrial waste gases for some time, but they are still subject to volatilization or degradation of organic amines, instrument corrosion, and high energy consumption for solvent regeneration [8–10]. Therefore, adsorption technology using various kinds of solid porous adsorbents has been proposed for CO2 capture and separation.
Different types of adsorbents have been investigated and developed for CO2 adsorption, for example, carbon-based materials, zeolites, metal oxides, and mesoporous silica [7, 11–18]. Nevertheless, these conventional porous materials usually display limited performance toward CO2 adsorption capacity as well as selectivity. Metal-Organic frameworks (MOFs) composed of metal ions and organic linkers, also known as porous coordination polymers, have developed rapidly and attracted significant attentions in the past two decades [19–26]. Owing to their diverse framework structure, high crystallinity, large surface area, and unlimited adjustability of pore structures and surface functionalities, MOFs exhibit great application prospects in the field of CO2 capture [14, 15, 21, 23, 27–37]. Nevertheless, it still remains a challenge to design MOFs with quite high CO2/N2 selectivity as well as CO2 adsorption capacity since the large surface area of MOFs usually leads to both increased CO2 and N2 adsorption capacities, resulting in limited CO2/N2 adsorption selectivity.
Many researches have tried to improve both the CO2 adsorption capacity and selectivity of MOFs, for instance, ligand functionalization [38, 39], incorporation of open metal sites [29, 40, 41], construction of specific pores [17], and pre-/postmodification for amine grafting/impregnation [36, 42–48]. Zhu et al. [42] reported that the polyethyleneimine-modified UiO-66-NH2 (Zr) exhibited a considerable enhancement toward CO2/N2 adsorption selectivity (48 vs. 25) and moderate desorption energy (68 kJ/mol CO2). Additionally, Justin et al. [49] described that the tris (2-aminoethyl) amine- (TAEA-) appended-Zn4O (NH2-BDC)1.2(BrAcNH-BDC)1.8 displayed a dramatically increased CO2/N2 (15/85) selectivity (143) at 313 K and a zero-coverage
In the current work, in order to improve its CO2 adsorption capacity and CO2/N2 selectivity, the ZIF-8 will be postsynthetically modified by using methylamine (MA), ethylenediamine (ED), and N, N
2. Materials and Methods
2.1. Materials
Zn (NO3)2·6H2O (Alfa Aesar, 99.99% purity), was purchased from Alfa Aesar Chemical; 2-methylimidazole (98% purity), N, N
2.2. Synthesis and Modification
Preparation and amine modification of ZIF-8 were carried out according to the reported procedures with a few modifications [50, 51]. The concentration gradients of methylamine (MA), ethylenediamine (ED), and N, N
2.3. Characterization
The PXRD patterns were obtained by an X-ray diffractometer (D8 advance, Bruker) using Cu Kα line focused radiation (wavelength
2.4. CO2 and N2 Adsorption Isotherms at Ambient Temperature
The specific adsorbed amounts of pure CO2 and N2 were measured using a static volumetric method on the Quantachrome Autosorb IQ2 instrument at three temperatures (273 K, 288 K, and 298 K, respectively). The initial activation of amine@ZIF-8 materials was performed at 523 K for 8 h under vacuum. Helium (ultrahigh purity) was the purge gas; CO2 and N2 were both of 99.999% purities.
2.5. Reusability Assessment
The regeneration performance was studied to evaluate the recyclability of the amine@ZIF-8. In this study, both volumetric and gravimetric adsorption-desorption cycles were carried out. For the volumetric experiments, multiple CO2 adsorption-desorption cycles on amine@ZIF-8 were operated by the Quantachrome Autosorb IQ2 instrument at 298 K. During each cycle, the maximum adsorption pressure was set at 1 bar; while the minimum desorption pressure was 1 mbar. The whole desorption process was under vacuum at 473 K for 8 h. For the gravimetric method, the six adsorption-desorption cycles were determined on a TA550 system under different regeneration temperatures (125°C and 150°C, respectively) [52, 53]. Before the recycle experiment, 5 mg of modified ZIF-8 sample was put in a platinum pan and heated to 150°C with a speed of 10°C/min and under high-purity N2 (99.999%, 60 mL/min) to desorb any preadsorbed gases on the surfaces of the sample. Then, the sample was retained under N2 atmosphere until a constant weight was reached at 25°C. After that, the gas was switched to a CO2/N2 mixture stream (60 mL/min of total flow, 90% CO2, balance N2) and kept for 30 min.
3. Results and Discussion
3.1. Characterization of Amine@ZIF-8 Materials
The postsynthetic modification route is demonstrated in Scheme 1. The amount of amine grafted on the ZIF-8 material is the key factor that may greatly affect its porous structure. Figure 1 shows the PXRD patterns of amine@ZIF-8 materials. Clearly, the modified ZIF-8 materials all show diffraction peaks at 7.29°, 10.32°, 12.65°, 16.50°, and 18.01°. These peaks are the same as the characteristics of the diffraction spectrum of the original ZIF-8. The peak positions are also consistent as compared to the simulated ZIF-8, implying that amine@ZIF-8 materials still maintain a complete crystal structure without structural collapse [30].
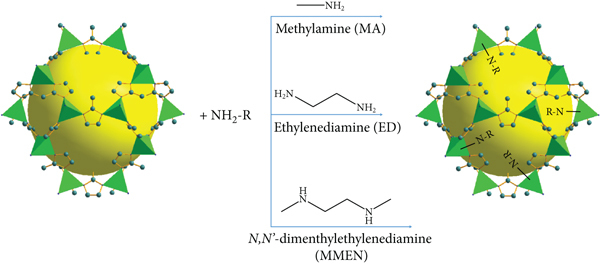
Preparation of amine-functionalized ZIF-8 using three different amines.

PXRD patterns of ZIF-8 and amine-modified ZIF-8 samples.
Figure 2 depicts the N2 adsorption-desorption isotherms at 77 K on amine@ZIF-8 materials. Obviously, the tested amine@ZIF-8 materials all presented typical type I behaviour, suggesting that there were mainly micropores in nature. Interestingly, hystereses were observed for the amine@ZIF-8 samples in the pressure range of 0.96-1.0. These hystereses were ascribed to the formed pores between the gaps of modified ZIF-8 particles, not owing to the mesopores.

N2 adsorption-desorption isotherms of modified ZIF-8 samples at 77 K.
Table 1 lists the parameters of porous structures for amine@ZIF-8. It can be seen that with the increase of MA, ED, and MMEN, the BET specific surface areas all showed a trend of first increasing and then decreasing. In addition, the total pore volume also presented the same trend as well as the micropore volume. Compared with the original ZIF-8, the BET specific surface areas of MA2-ZIF-8, MMEN2-ZIF-8, and ED1-ZIF-8 increased by 118.2%, 92.0%, and 29.8%, respectively. Furthermore, their total pore volume increased separately by 130.8%, 100%, and 48.7%. Moreover, the micropore volumes of MA2-ZIF-8, MMEN2-ZIF-8, and ED1-ZIF-8 raised separately by 110.8%, 75.7%, and 18.9%. Thereafter, the MA2-ZIF-8, ED1-ZIF-8, and MMEN2-ZIF-8 can be recognized as the three best samples among the amine@ZIF-8 materials, and they would be marked as MA-ZIF-8, ED-ZIF-8, and MMEN-ZIF-8, respectively, in the following adsorption experiments. However, it should be pointed out that the BET surface area of the as-made ZIF-8 was lower than that synthesized by Park et al. [54], but still in the range of the reported ZIF-8 (see Table S2). This, possibly, indicated that the prepared ZIF-8 in this work still contained some residual species in the cavities (e.g., CHCl3, DMF, or unreacted Hmim) which could not desorbed during the activated step. In addition, the different results between these three amine@ZIF-8 samples were probably owing to the different reactivities of amines. Therefore, the reason of the increases of the BET surface area and pore volume might be that, after the postsynthetic modification, some occlusive pores of the ZIF-8 were reopened, and more new pores were formed at meanwhile [45]. Nevertheless, when the loading amount of MA, ED, and MMEN added further, the surface areas and pore volumes begun to decrease because of hindrance effect of the introduced amines [55, 56].
Texture properties of the amine-modified ZIF-8 samples.
Figure S1 exhibits the DFT pore size distributions of ZIF-8 and amine@ZIF-8 materials. Apparently, for each MA-ZIF-8, MMEN-ZIF-8, and ED-ZIF-8 material, the main pore size was lower than that of the as-made ZIF-8. Figure 3 demonstrates the DFT pore size distributions for the best amine@ZIF-8 materials. It can be noticed that the main pore size of the original ZIF-8 is distributed at 10.87 Å, while those of the MA-ZIF-8, ED-ZIF-8, and MMEN-ZIF-8 are distributed at around 9.26 Å, suggesting that the introduction of an amine group would result in reduced pore size [47]. This might be due to the new formed pores with the introduction of MA, ED, and MMEN, respectively. The reduced pore size also indicated that there were mainly micropores in the amine@ZIF-8 frameworks.

DFT pore size distribution for ZIF-8 and amine-modified ZIF-8 materials.
Figure 4 illustrates the infrared spectra of amine@ZIF-8 materials. First of all, the infrared spectra of amine@ZIF-8 materials were basically similar to those of ZIF-8. Secondly, the peak intensities of MA-ZIF-8 and ED-ZIF-8 material at around 3136 cm-1 were significantly enhanced, and at 2960 cm-1 and 2930 cm-1, the intensities of the antisymmetric stretching vibration peaks of -CH3 and -CH2 increased, too. Thirdly, after the modification, a new N-H stretching vibration peak appeared at about 3136 cm-1, signifying the successful introduction of N-H bond into MMEN-ZIF-8, MA-ZIF-8, and ED-ZIF-8 materials. Fourthly, the antisymmetric stretching vibration peaks of -CH3 and -CH2 increased at around 2960 cm-1 and 2930 cm-1 after the modification, indicating that two groups -CH3 and -CH2 were introduced after modification. In summary, after ZIF-8 materials are modified with different organic amine groups, N-H bonds have been successfully introduced. The thermal stability of amine@ZIF-8 was evaluated by TGA experiment under N2 flow from 30°C to 800°C (see FIGURE S2). It can be evidently seen that the modified ZIF-8 materials all had better thermal stability than ZIF-8, while the MA-ZIF-8 was even stable up to 427°C.
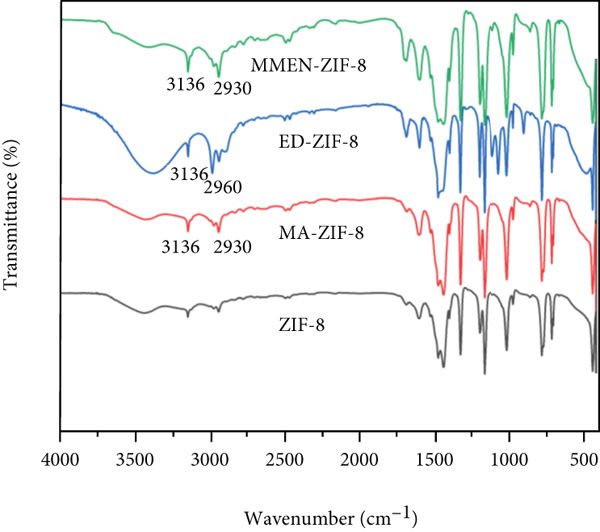
FTIR spectra of MA-ZIF-8, ED-ZIF-8, and MMEN-ZIF-8.
3.2. CO2 and N2 Adsorption on Amine@ZIF-8 at Ambient Temperature
3.2.1. CO2 Adsorption Isotherms on Amine@ZIF-8 Materials
Figure 5 exhibits the CO2 adsorption isotherms of amine@ZIF-8 materials at different temperatures (298 K, 288 K, and 273 K). Clearly, the CO2 adsorption capacities of amine@ZIF-8 materials increased dramatically at each temperature. At 298 K, their CO2 amounts adsorbed were in the following way:
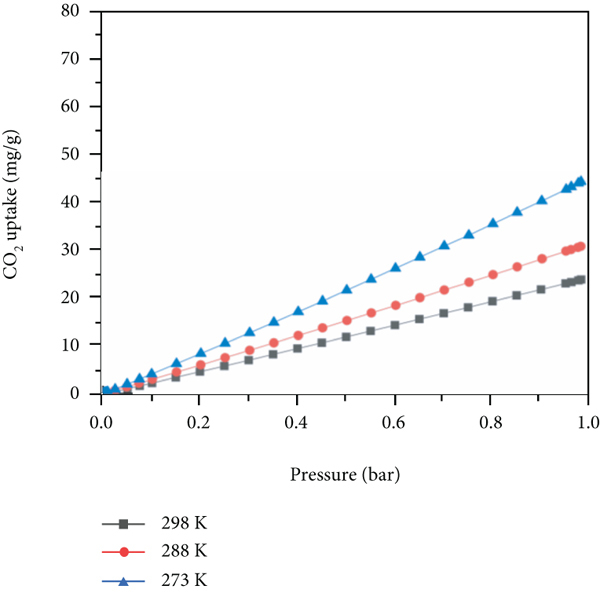
The CO2 adsorption isotherms on the amine@ZIF-8 materials at 273 K, 288 K, and 298 K. (a) ZIF-8; (b) MA-ZIF-8; (c) ED-ZIF-8; (d) MMEN-ZIF-8.



3.2.2. N2 Adsorption Isotherms on Amine@ZIF-8 Materials
Figure 6 displays the N2 adsorption isotherms on amine@ZIF-8 materials at different temperatures (298 K, 288 K, and 273 K). It can be observed that the N2 adsorption capacities of amine@ZIF-8 materials significantly increased. And, the order of their adsorption capacity for N2 is as follows:

The N2 adsorption isotherms on the amine@ZIF-8 materials at 273 K, 288 K, and 298 K. (a) ZIF-8; (b) MA-ZIF-8; (c) ED-ZIF-8; (d) MMEN-ZIF-8.



3.3. Adsorption Selectivity of CO2 vs. N2 on Amine@ZIF-8 at Ambient Temperature
The ideal adsorbed solution theory (IAST) developed by Myers and Praunitz was proved to be an efficient method to evaluate the adsorption equilibrium of gas mixtures and selectivity using single-component isotherms [45]. Before carrying out the IAST simulation, the single-component adsorption isotherms of CO2 and N2 were separately fitted by the dual-site Langmuir-Freundlich (DSLF) equation (see equation S1).
Figure 7 presents the single-component isotherms and the DSLF fitted ones. Obviously, the DSLF model can be applied favourably to fit the single-component adsorption data. From the fitting parameters listed in Table 2, it can be observed that the correlation coefficients
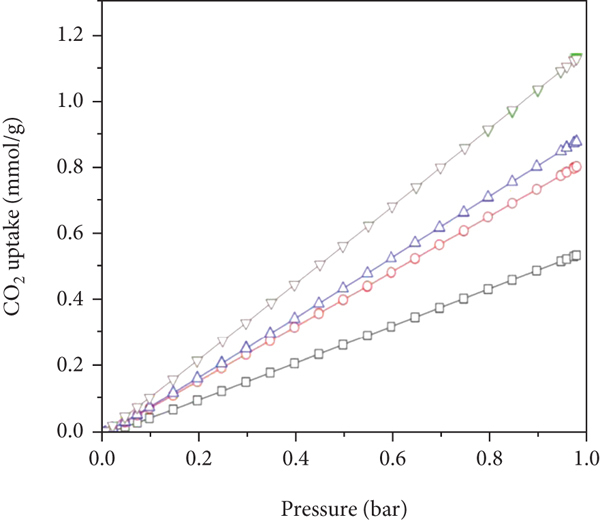
DSLF fitting of the CO2 and N2 isotherms on the amine@ZIF-8 at 298 K: (a) CO2; (b) N2.
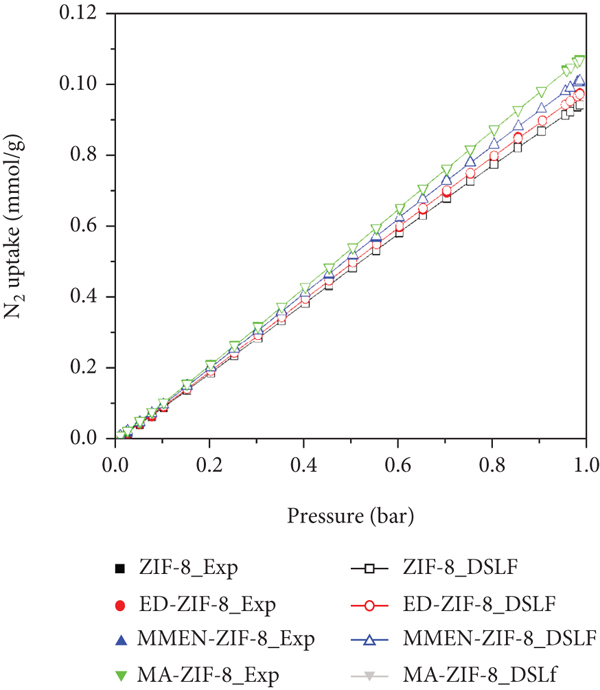
The fitting parameters of the DLSF model for the pure component isotherms of CO2 and N2 at 298 K.

The IAST-predicted selectivities for equimolar CO2 and N2 at 298 K as a function of total bulk pressure.
3.4. Isosteric Heat of Adsorption
The isosteric heat of adsorption (

Dependence of isosteric heat of adsorption on the amounts adsorbed of CO2 and N2 over the amine@ZIF-8 materials: (a) CO2; (b) N2.

3.5. Cyclic Adsorption-Desorption Performances of CO2 on MA-ZIF-8 Material
In order to assess the reusability of CO2 on the modified ZIF-8, we further performed the CO2 adsorption-desorption cycle test on MA-ZIF-8 material. Figure 10 presents the variation curves of CO2 uptake on MA-ZIF-8 material during four continuous cycles of CO2 volumetric adsorption and desorption experiments at 298 K. It can be seen that after four adsorption-desorption cycles, the CO2 desorption efficiency was higher than 99%, and only about 0.01 wt% CO2 remains on the adsorbent. This indicated that the adsorption of CO2 on the MA-ZIF-8 sample was reversible, and the MA-ZIF-8 sample had good regeneration performance. In addition, the shapes of the four adsorption-desorption cycle curves were analogous, denoting that MA-ZIF-8 was very stable and suitable for CO2 adsorption.

Volumetric recycle runs of CO2 adsorption-desorption on the MA-ZIF-8 at 298 K and 1 bar for adsorption and 1 mbar for desorption.
Figure 11 presents the six gravimetric CO2 adsorption-desorption cycles for MA-ZIF-8, where the desorption processes were evaluated at 125°C and 150°C, respectively. As it was noted, after each desorption process, an obvious decrease can be observed. Furthermore, when the desorption temperature increased from 125°C to 150°C, there was only a slight variation in the weight of MA-ZIF-8. For MMEN-ZIF-8 and ED-ZIF-8, similar phenomenon can be observed (see Figures S3 and S4). Therefore, the desorption temperature has little effect on the CO2 capture during cycles. It also can be concluded that 125°C is enough for the regeneration of amine@ZIF-8 materials.

Six gravimetric CO2 adsorption-desorption cycles on the MA-ZIF-8 at 298 K and desorption at 125°C and 150°C, respectively: (a) 125°C; (b) 150°C.

4. Conclusions
The as-synthesized ZIF-8 material was successfully modified with methylamine, ethylenediamine, and N, N
Footnotes
Data Availability
The data used to support the findings of this study are included within the article.
Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (21878122), the Pearl River S and T Nova Program of Guangzhou (201710010053), and the Zhengzhou Science and Technology Project (141PPTGG433).
