Abstract
Antibiotics are emerging water pollutants that have attracted significant attention from the scientific community. Antibiotics are generally released via hospital effluents, industrial production waste, animal manure, and irrigated agricultural land. Antibiotic residues can harm all living organisms, with the most detrimental consequence being the generation of antibiotic-resistant microorganisms, commonly known as “superbugs.” Antimicrobial resistance is a concern to the healthcare community as it complicates the treatment of infections. Thus, the development of effective and economical technologies to remove antibiotics from the environment is necessary. Adsorption is a promising technology owing to its effectiveness and high operational feasibility, and carbon-based adsorbents are primitive materials that are particularly suited for antibiotic adsorption. Herein, an overview of the current state of antibiotic pollution will be summarised, including the adverse effects of different antibiotics and challenges associated with antibiotic removal. The adsorption behaviours of tetracycline (TC), quinolone, penicillin, and macrolides on carbon-based adsorbents (i.e., activated carbon, carbon nanotubes, and graphene-based materials) are reviewed. The interactions between antibiotics and carbon-based adsorbents, adsorption mechanism, and adsorption behaviour under different conditions are emphasised. In addition, the limitations of adsorption technology are highlighted to direct future research.
1. Introduction
Antibiotics are used to prevent and treat infectious diseases in humans and are extensively used in aquaculture and animal husbandry. [1] Antibiotic consumption has significantly increased, with reports that global antibiotic consumption rate increased 39% due to a rising defined daily dose of 65% between 2000 and 2015. [2] However, most antibiotics cannot be metabolised completely in humans and animals, allowing approximately 30 to 90% of the parent compounds to be discharged into the environment. [3, 4] Antibiotics have been detected in surface waters, groundwater, seawater, and soil. The dominant antibiotic residues of sulfonamides, macrolides, tetracycline (TC), and quinolones are detected in many surface waters. Table 1 shows the antibiotic residues detected in the aquatic environments of different countries. [4] The presence of excreted antibiotics in the aquatic environment promotes the development of antibiotic-resistant bacteria (ARB) and disturbs the ecological balance, posing an emerging threat to the ecosystem and human health [1, 3].
Antibiotic residues detected in the aquatic environments of different countries.
Recently, the spread of antibiotic resistance genes (ARGs) has rapidly increased along with the increase in antibiotic discharge into the environment. [1] ARGs persist in the environment and can be transferred between environmental bacteria and human pathogens. ARGs can eventually enter the food chain and affect human health. [1, 18] Antibiotic residues deteriorate drinking water quality by affecting the structural properties of metal ions. [19] Hence, the intake of antibiotic residues from the environment through eating and drinking has a potential biomagnification effect which may affect gut microbiota. It was reported that drug resistance-related diseases have caused >700,000 deaths per year, which may increase to 10 million per year by 2050 if the underlying causes are not addressed [4].
Conventional biological wastewater treatments are unable to remove antibiotic residues efficiently and require high energy consumption. [1, 2] Therefore, several antibiotic removal methods have been developed, including adsorption, ultrasonic cavitation, advanced oxidation technology, chlorination, electrochemical treatments, and membrane processes. [3] Adsorption is considered a particularly promising technology because of its ease of operation, high removal efficiency, universal application, and relatively low costs. [2, 3] This process can remove colour-forming organics, synthetic organic chemicals, heavy metals, and perchlorate. The interaction between the adsorbate and adsorbent is the driving force for capturing compounds of interest [4].
Various materials have been introduced for antibiotic removal from aqueous solutions, including carbon [20], polymeric materials [21], clay minerals [22], metals and their oxides [23], and chitosan [24]. Among these adsorbents, carbon materials have been widely applied to remove antibiotics because of their tunable surface functionalities, abundant pore structure, and high specific surface area. [25, 26] This review focuses on quinolone, penicillin, TC, and macrolide removal from aqueous solutions using carbon-based adsorbents including activated carbon as well as graphene-based, carbon nanotube-based, and biochar-based materials. The adsorption behaviour in terms of the mechanism, isotherm, and kinetics, along with influencing factors such as temperature and pH, is discussed herein.
2. Antibiotic Classes
Antibiotics are administered as therapeutics or prophylaxes to prevent and treat infections.[18] Antibiotics can be classified based on their chemical structure, mode of action, activity, administration route, and bacterial spectrum.[19] Antibiotics are commonly used in healthcare and veterinary medicine as inhibitors and biocides of infectious microorganisms. Additionally, they are used to raise livestock and as growth promoters in aquaculture, agriculture, and beekeeping.[3, 20] Antibiotic consumption has increased, and pathogen resistance to antibiotics has become a focal point of clinical and environmental research.[18] This paper will focus on TC, quinolone, penicillin, and macrolide antibiotics.
2.1. Tetracycline
TCs are broad-spectrum antibiotics derived from Streptomyces aureofaciens that are effective against Gram-positive and negative bacteria, allowing effectiveness against various bacterial infections.[3, 20] Based on their nature, elimination time, and dosage, TCs can be classified as TC, oxytetracycline (OTC), and chlortetracycline (CTC) [19], and their structures are shown in Figure 1. TCs are the most commonly used antibiotics worldwide because of their broad-spectrum activity, low toxicity, and low cost. [19, 22] However, they are unable to be metabolised completely in humans and animals. In addition, animal faeces as a plant fertiliser can lead to the notable persistence of residues in soil and aquatic environments.[21, 23] For example, the concentration of TCs detected in surface water in China was 482 ng/L, [20] while hospital wastewater exhibits TC concentrations of 100 μg/L and domestic wastewater contains concentrations closer to 1 μg/L. [19] In aquaculture farms in Malaysia, TC was most frequently detected (83%), with concentrations ranging from below the limit of quantification (LOQ) to 73 ng/L [24].
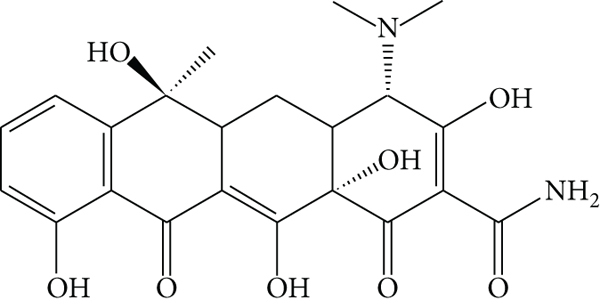
Structures of (a) tetracycline, (b) oxytetracycline, and (c) chlortetracycline.


2.2. Quinolone
First-generation quinolones, including nalidixic acid (NDA) and cinoxacin, were discovered in the 1960s. [25, 26] The core chemical structure of the quinolone family is a bicyclic structure related to 4-quinolone (Figure 2). [26] The first-generation quinolones exhibit a narrow spectrum of activity and are effective against Gram-negative bacteria in the treatment of uncomplicated infections.[25] The major drawback of first-generation antibiotics is their high inhibitory concentration and low serum concentration when administered.[26] Therefore, a new generation of fluoroquinolones (FQs) was introduced. The addition of a fluorine atom at the R6 position improved the activity spectrum and pharmacokinetic properties.[26] Ciprofloxacin (CIP), norfloxacin (NOR), and ofloxacin are well-known second-generation quinolones. To date, four generations of quinolones have been developed with activity against all Gram-negative organisms, including Pseudomonas sp. and Gram-positive organisms including S. pneumoniae, atypical pathogens, and anaerobic pathogens.[25] Newer quinolones are effective against various infections, including community-acquired pneumonia, intra-abdominal infections, pelvic infections, and sexually transmitted diseases.[25, 26] According to the review by Ahn et al., FQ is one of the five main classes of antibiotics detected in water samples from China, with a maximum concentration of up to 1000 ng/L [27].

The general structure of quinolone.
2.3. Penicillin
The first antibiotic, penicillin, was discovered by Alexander Fleming in 1928 and has saved millions of lives by preventing and treating various infections. Penicillin protects against infections by inhibiting transpeptidase, a crucial enzyme for cell wall synthesis and maintenance.[28] The

The general structure of penicillin.
2.4. Macrolide
Macrolides derived from Streptomyces are a class of weakly alkaline antibiotics which effective against Gram-positive and certain Gram-negative bacteria (Figure 4). Macrolide antibiotics include erythromycin (ERY), azithromycin, clarithromycin, tilmicosin, and tylosin. [3] The extensive use of macrolides in humans and animals poses a potential health risk due to bacterial resistance.[30] Azithromycin, clarithromycin, and ERY have been included in the European (EU) watch list of emerging concerns in the aquatic environment, [31] as they are frequently detected. For instance, up to 47 μg/L of ERY and roxithromycin was detected near swine farms and fishponds in the Haihe River Basin, China.[30] The presence of azithromycin with a concentration of 0.27 to 22.7 μg/L in municipal wastewater in the city of Zagreb has also been reported [32].

The structures of (a) tylosin, (b) carbomycin A, (c) spiramycin, (d) azithromycin, and (e) erythromycin [3].



3. Antibiotic Pollution
TCs are not likely to cause acute toxicity, but chronic toxicity may occur in nontarget organisms that are exposed to antibiotics in the environment over extended periods. [33] TCs can persist in the environment because of their high hydrophilicity and low volatility, facilitating the production of ARGs. Additionally, the persistence of ARGs in drinking and irrigation water systems for agriculture poses a risk of infectious diseases by influencing the human intestinal microflora.[22] The presence of tet (O)- and tet (S)-mediated TC resistance in bifidobacteria has been reported, illustrating that the human gastrointestinal tract may serve as a reservoir of TC resistance genes [34].
In addition, FQ can be easily transferred to the soil because of its excellent chemical stability, modifying soil strains of Salmonella typhimurium. These modified bacterial strains are genotoxic and can cause DNA damage to aquatic organisms. In addition, morphological deformities of higher plants can result from photosynthetic pathway interference by accumulated FQ.[35] Furthermore, where animal manure is utilised as fertiliser in an agricultural setting, the chances of releasing antibiotic residues into the environment are increased. Lillenberg et al. discussed the possibility of plants adsorbing antibiotic residues from the soil, showing that FQs (CIP and NOR) could be detected in lettuce, common barley, and cucumber. [36] This suggested that antibiotics in the environment can be transferred to the edible parts of crops, leading to another antibiotic resistance route via food consumption.
AMX in the environment can negatively affect aquatic organisms, with a prominent example of catalase activity of Danio rerio being inhibited by exposure to a high AMX concentrations.[35] Once antibiotic residues enter the human body, they interact with the microbiome, and the intestinal microbiota often changes due to exposure to broad-spectrum antibiotics. For example, the composition of Firmicutes increases, while that of Bacteroidetes is reduced. [37] Diseases, including colorectal cancer, pseudomembranous colitis, and intestinal disorders, could arise from an imbalanced intestinal microbiome [38].
Lastly, the presence of macrolide residues in the environment may have harmful effects on humans and adverse effects on the environment. [31] Vestergaard et al. revealed that insufficiently treated effluent from azithromycin production resulted in enriched resistance genes to macrolide antibiotics in the receiving river. This results in a higher chance for a pathogen to capture these resistance genes, increasing the risk of humans being exposed to resistant pathogens via the food chain, recreational activities, and water intake. [39] In addition, tilmicosin was found to affect the development of zebrafish embryos, including cardiac congestion and causing teratogenic effects. Apoptosis and oxidative stress have also been observed in embryos exposed to tilmicosin [30].
4. Challenges Associated with Antibiotic Removal
Wastewater treatment plants that have been developed to remove pollutants, including nitrogen and phosphorus, are generally unable to effectively remove antibiotics and hormones. [40] Biological treatment has been widely used in wastewater treatment owing to its low environmental impact, high cost-effectiveness, and robustness.[41] Typical biological treatments included activated sludge, anaerobic digestion, photodegradation, fungal treatment, biosorption, biodegradation, and stabilisation.[4, 42] These methods typically depend on nematodes, bacteria, or small organisms to transform organic contaminants in wastewater into simple substances and to break down organic pollutants into biomass via normal cellular processes.[4] Nevertheless, several challenges remain for antibiotic removal from aquatic environments using conventional biological wastewater treatment.
Antibiotic removal is often hindered by their physicochemical properties. Volatility is an important property that is defined by the Henry’s law constant (
In addition, antibiotic removal can be affected by operating conditions. Solid retention time (SRT) is a major factor that influences the removal of emerging pollutants from the aquatic environment, as it controls the diversity and size of the microbial community in wastewater. Generally, a high SRT value promotes microorganism proliferation in wastewater as well as increases and diversifies the microbial community.[42, 44] Enhanced removal of pharmaceutical compounds was found with an SRT of >26 days, whereas SRT of <8 days reduced removal efficiency. [44] Longer SRTs promotes slow-growing microorganism growth, including the nitrifying bacteria that are associated with antibiotic removal.[42] In addition, a short SRT of <15 days was found to reduce the efficiency of the activated sludge due to reduced sludge availability [43].
The effect of pH on antibiotic removal performance is associated with the inherent properties of the antibiotics. Antibiotic molecules exist in different forms depending on the solution pH and the acid dissociation constants (
5. Different Approaches for Antibiotic Removal
Antibiotic removal approaches can be broadly categorised as biological, chemical, or physical. Biological treatments rely on live organisms, including bacteria and nematodes, to break down organic pollutants through biological processes.[4] The biodegradability of antibiotics is a major aspect to be considered, as only biodegradable antibiotics can be successfully removed through biological techniques.[4, 41] A simple closed-bottle test is typically conducted to estimate the biodegradability of antibiotics. According to the Organization for Economic Co-operation and Development (OECD), an antibiotic is considered biodegradable when oxygen demand in the testing vessel is 60% higher than the theoretical value for 28 days.[4] The efficiency of antibiotic removal through anaerobic processes was investigated, showing a higher percentage of AMX (~80%) was removed compared to CIP and enrofloxacin (~38%).[46] This is likely because FQ biodegradability is lower than that of penicillins, so biological treatments are considered less effective at removing antibiotics with poor biodegradability.
Chemical treatments involve altering the chemical structure of pollutants via oxidation, reduction, electrolysis, or catalysis. By altering the chemical structure, chemical properties such as solubility and volatility can be changed to reduce the ability of pollutants to remain in the aqueous system[4]. Chlorination is the most conventional technique used in drinking water systems because of its cost-effectiveness. Various chlorine species, including hypochlorite, chlorine gas, and chlorine oxide, have been used in this process. Chlorine oxide is preferred over other species because of its selectivity and ability to react with micropollutants, while preventing the formation of carcinogenic trihalomethane by-products.[47] Navalon et al. investigated the antibiotic removal properties of chloride oxides at different pH values, showing that chloride oxide could oxidise AMX and cefadroxil, but not penicillin G.[48] However, with decreasing pH, the oxidation reactivity with penicillin was enhanced, while the reactivity with AMX and cefadroxil decreased because their chemical structures were altered by the varying pH. Chlorine oxide reacted with pollutants containing phenolic and tertiary amino groups, but not with molecules with functional groups including aromatic, hydrocarbons, 1° and 2° amine, aldehyde, and ketone groups.[49, 50]
Advanced oxidation is an alternative to conventional oxidation with chlorination that produces an extremely reactive hydroxyl radical (OH-). Hydroxyl radicals have a higher standard oxidation potential (
Physical treatments involve the removal of pollutants by van der Waals forces, hydrogen bonding, and dipole interactions.[4, 47] The reactant chemical structure is not altered during physical treatment and instead, in processes such as filtration, agglomerates form causing the physical state changes. [47] Other physical treatments include adsorption, filtration, coagulation, membrane treatment, and sedimentation.[4] The difference between physical and biological/chemical treatments is that physical treatment often does not involve pollutant breakdown. Instead, pollutants either separated or coagulated in a more concentrated form.[47]
Adsorption is the most commonly applied technique for removing pollutants because of its high capacity and design simplicity. It has been commonly applied as an efficient, effective, and economical technique for pollutant removal from wastewater for decades.[51] This process involves the mass transfer of chemical substances from the liquid phase to the solid phase. [4, 47] An advantage of adsorption over other techniques is that it does not produce secondary toxic by-products. Commonly used adsorbents are activated carbon (AC), graphene-based materials, and carbon nanotubes (CNTs).
6. Carbon-Based Adsorbents for Antibiotic Removal
To date, various adsorbents have been developed, and carbon-based adsorbents have been acknowledged as effective adsorbents for pollutant removal because of their high specific surface areas, strong interactions, and abundant pore structures. Common carbonaceous materials for antibiotic removal include AC, CNTs, graphene, and their composites (Figure 5). [52] The adsorption process and adsorption efficiency are greatly affected by the functional groups (-COOH, -OH, and -NH2) in carbon-based adsorbents. Electrostatic interactions involving attraction between the adsorbents and an oppositely charged adsorbate are the primary adsorption mechanism between AC and antibiotics. Furthermore, hydrophobic interactions,

Carbon-based adsorbents. Activated carbon, carbon nanotubes, graphene, and their composites are common carbonaceous materials that show good adsorption capability for antibiotic pollutant removal.

Adsorption mechanisms between carbon-based adsorbents and antibiotics. The types of adsorptive interaction include electrostatic interaction, hydrophobic interaction, hydrogen bond formation, and
6.1. Activated Carbon
AC is an activated black carbonaceous solid material with a large surface area, desirable pore-size distribution, and high adsorptive capacity. [23, 52] AC is a popular adsorbent used for the industrial-scale purification of water and air. Almost all carbon-based materials can be used to produce AC, but its properties depend on the material and activation method used. [3] Conventionally, AC is produced from petroleum, coal, lignite, and coke.[4, 52] Recently, different materials have been used to produce AC, including durian shells, coconut shells, olive stones, and wood.[53–56] Biochar (BC) and hydrochar (HC) have gained significant attention from researchers as alternative adsorbents to replace conventional activated carbon.[57] BC is a carbonaceous material produced via slow pyrolysis of biomass in an anoxic environment. HC is produced through hydrothermal carbonisation (HTC), a process wherein biomass is heated anaerobically in the presence of water.[57, 58] The difference between BC and HC is that dried biomass is used in the production of BC, whereas both dried and nondried biomass can be used in HTC [58].
Generally, differentiating between AC, BC, and HC causes confusion. Both BC and HC can be classified as AC after process “activation” where common activation methods for AC preparation can be classified as physical or chemical. During physical activation, the precursor material is activated after carbonisation at temperatures between 800 and 1000°C to prevent carbon losses. Chemical activation involves mixing activating agents with the raw material, followed by heating under an inert gas. This is a single process wherein carbonisation and activation occur simultaneously. Important advantages of chemical over physical activation including the short activation time, well-controlled activation reaction, and consistently high surface area attainment. [3, 23] To date, the adsorptive removal of antibiotics by AC has been studied extensively.
6.1.1. Adsorptive Removal of Antibiotics by AC
Some researchers have incorporated nanoparticles into AC to enhance its adsorption capacity. Zhou et al. studied the adsorption behaviour of TC from an aqueous solution using powdered activated carbon (PAC) in association with ferroferric oxide nanoparticles (FONP-PAC). Scanning electron microscopy (SEM) images showed that the synthesised nanoparticles were well distributed on the surface, pores, and channels of the PAC, with an average particle size of 20–50 nm. However, FONP inside the pores of PAC reduced the surface area of the adsorbent, resulting in a mesoporous structure. The adsorption behaviours of 1FONP-PAC were also evaluated and determined to be pH-dependent. Electrostatic repulsion occurred between TC and 1FONP-PAC at high pH (>7.7), as both existed in anionic form, whereas low pH (<3.0) resulted in weak electrostatic interactions. 1FONP-PAC demonstrated the highest adsorption efficiency of 140.2 mg/g at pH 3. Isotherm and kinetic data showed that the adsorption process of 1FONP-PAC followed the Freundlich and Elovich kinetic models. Strong interactions between TC and the adsorbent were observed, as indicated by the separation factors (
BC derived from pomelo peel and activated by potassium hydrochloride (KOH) was developed for the effective removal of TC from swine water. Increased pyrolytic temperatures increased the surface area and pore volume of BC. When activated by KOH, a significant enhancement of the surface area (2457.367 m2/g) and pore volume (1.14 cm3/g) of BC-KOH was observed by SEM analysis when compared with BC-400 and BC-600. The porous structure of the BC is closely related to its adsorption capacity, as BC-KOH exhibited the highest adsorption capacities of 124.95, 124.91, and 124.99 mg/g for TC, OTC, and CTC, respectively. However, when BC-KOH was tested in synthetic swine water, the removal efficiencies for TC, OTC, and CTC were 85.04, 82.17, and 96.64%, respectively. This indicated that other compounds in swine water affected the adsorption ability of the activated BC. The study found that a pseudo-second-order model was the best kinetic model, while the Langmuir model was the best isotherm model fitted to the adsorption. In addition, the highest adsorption capacity of the activated BC was obtained at pH 8.5, even though electrostatic repulsion occurred under these conditions. This indicated that besides electrostatic interactions, the possible adsorption mechanism of TCs onto the activated BC included
Zhang et al. reported the performance of PAC for the removal of 28 antibiotics from water. Penicillin G (PNG), oxolinic acid (OLA), NDA, and flumequine (FMQ) were studied at an adsorbent dosage of 20 mg/L and contact time of 120 min. PAC exhibited excellent removal efficiency towards all the tested antibiotics where 96.5–99.9% and 86.8–99.6% of antibiotics were removed from deionized and surface waters, respectively. The decreased removal efficiency in surface waters could be explained by the natural organic content competing with the antibiotics for PAC binding sites, and/or they may have interacted with the antibiotics and prevented them from approaching the pores. With increasing initial adsorbent concentration, the adsorption efficiency was enhanced. Increasing the adsorbent concentration results in a larger number of vacant active sites, enhancing the adsorption process. In terms of kinetics, the data fitted the best to the pseudo-second-order and Elovich models with
Li et al. studied the removal of CIP using tea leave-derived biochar (UTC) under different conditions by altering the initial antibiotic concentration (150–500 mg/L), solution pH (4–10), and temperature (30–60°C). The adsorption capacity of CIP at equilibrium increased from 32.9 to 146.9 mg/g when the initial CIP concentration was increased from 150 to 200 mg/L. A greater concentration gradient between the aqueous and solid phases generates a stronger driving force for antibiotic transfer from the aqueous phase into the solid phase. The point of zero charge (
The removal of CIP and AMX was studied using Prosopis juliflora-derived AC (PPJ), showing maximum adsorption efficiencies at pH 4.0 and 7.0, respectively, with a higher adsorption capacity was observed for CIP. The PPJ surface was negatively charged at
Gholamiyan et al. produced AC derived from almond shells and used Fe3O4 nanoparticles to synthesise magnetic activated carbon (MAC) for the adsorptive removal of ERY. Based on the SEM images, Fe3O4 nanoparticles that imparted magnetic properties were evenly distributed on the surface of the MAC. Similar to previous studies, the adsorption of ERY by MAC was pH-dependent, with a
Antibiotics adsorption by activated carbon. The adsorptive characteristics of the adsorbents are summarised. ND: not determined.
6.2. Carbon Nanotubes
Carbon nanotubes (CNTs) have shown great potential as absorbents for water remediation.[65] CNTs are carbon allotropes with an aromatic surface that rolls up to form a cylindrical structure (Figure 5).[66] CNTs exhibit high mechanical strength, low electrical resistivity, and high thermal conductivity owing to their unique structure. [67] CNTs can be grouped into two main types based on the number of cylindrical shells: single-walled CNTs (SWCNTs) and multiwalled CNTs (MWCNTs). [68, 69] SWCNTs are cylindrical with a diameter of approximately 0.42 nm with a graphite wall ring at both ends. In contrast, MWCNTs are characterised by concentric cylinders with a layer-to-layer spacing of 0.34 nm and diameter that varies from 2 to 25 nm.[52] The site density, purity, surface area, porosity, functional groups, and CNT type determines their adsorption behaviour. CNTs usually cohere with each other and aggregate in aqueous solution via van der Waals interaction between raw CNTs forma large bundles. This limits their application for removing pollutants and antibiotics as they are difficult to disperse homogenously in most organic and aqueous solutions.[70] In general, CNTs can be functionalized (e.g., -COOH and -OH) via different methods to improve their adsorption ability and properties.[66, 71] [69] The unique structure and functional groups on CNT surfaces allow them to interact with organic and inorganic compounds via different intermolecular forces.[66] Different CNT structures exhibit different adsorption processes because of the various available adsorption sites and variable access to adsorption sites.[72] In this regard, researchers have extensively studied the adsorptive removal of antibiotics by CNTs. CNT adsorbent characteristics and adsorption behaviour for antibiotics are summarised in Table 3.
Antibiotics adsorption by carbon nanotubes. The adsorptive characteristics of the adsorbents are summarised. ND: not determined.
6.2.1. Adsorptive Removal of Antibiotics by CNTs
Xiong et al. synthesised MWCNTs functionalized with MIL-53(Fe) and MWCNT/MIL-53(Fe) as adsorbents for antibiotic removal from aqueous solutions. A notable increase in the specific surface area and pore volume was observed when the MWCNTs were combined with MIL-53(Fe). Various mass ratios of MWCNT to MIL-53(Fe) in the composites (1, 5, 10, 20, and 30%) were synthesised, and MWCNT/MIL-53(Fe)-20% exhibited the greatest adsorption efficiency. This was attributed to the sharp increase in the pore volume, pore size, and surface area after MWCNT modification. The ionic strength and pH significantly influenced the adsorption ability where increased sodium chloride (NaCl) concentration resulted in decreased adsorption of TCs due to competition for active sites. In addition, reduced TC absorption was observed at higher pH, indicating electrostatic repulsion between the adsorbent and TCs. However, some TC adsorption was observed, illustrating that electrostatic interactions were not the only mechanism of adsorption, with others arising from
Ionic liquid-multiwalled carbon nanotube (IL-MWCNT) composite tablets were synthesised by Chen et al. [74] for the removal of TCs and heavy metals. The ionic liquid, N-butyl benzothiazole hexafluorophosphate ([C4Bth][PF6]), was loaded into MWCNT. Ethyl cellulose was selected as the diluent to prepare the composite tablet as it could increase the compression stress to >15 MPa and is stable in water when compared to microcrystalline cellulose. The factors influencing the adsorptive removal of TCs by the IL-MWCNT composite tablets were investigated. At low pH, the electrostatic interaction between the anionic form of TCs and the positively charged adsorbent increased the adsorption efficiency, reaching a maximum removal of 98.53% at pH 4.5. At higher pH, the TCs existed predominantly in anionic form and the adsorption efficiency decreased because of electrostatic repulsion between the like charges of the adsorbent and adsorbate. TCs have no net charge at pH 3.3–7.7 and no significant decrease in removal efficiency (>96%) was observed in this pH range. This showed that besides electrostatic interactions,
CNTs can be easily chemically modified to enhance their adsorption capacities.[65, 67] Therefore, hydroxylated (MH), carboxylated (MC), and graphitised (MG) MWCNTs were synthesised and compared with SWCNTs in terms of their CIP removal capacity.[69] SWCNTs with the highest surface area exhibited the highest adsorption capacity compared to the other modified MWCNTs. In addition, the optimum pH for CIP adsorption was 7.0 due to hydrophobic interactions, in agreement with the data reported by Ncibi and Sillanpaa.[75] However, while the hydrophobicity of MH was lower than that of MG owing to oxygen-containing functional group incorporation, the adsorption capacity of MH was higher than that of MG across all pH values, suggesting that other mechanisms contribute to the adsorption of CIP onto CNTs. FQ molecules can function as
Using sodium hypochlorite as an oxidising agent, Yu et al. studied the adsorption potential of MWCNTs with oxygen contents ranging from 2.0 to 5.9%. Sodium hypochlorite introduced phenolic hydroxyl groups to the surface of the MWCNTs. Increasing the surface oxygen enhanced the hydrophilicity and dispersibility of the MWCNTs, resulting in improved CIP adsorption onto the active sites. However, excessive hydrophilicity inhibited adsorption owing to the formation of surface water clusters on the MWCNTs. MWCNTs with an oxygen content of 4.7% exhibited the highest adsorption capacity of 209.6 mg/g. A further increase in oxygen content was associated with a slower rate of increase in the maximum adsorption capacity. This decline was attributed to
Balarak et al. investigated the effectiveness of SWCNTs for removing AMX. An increase in the initial AMX concentration resulted in an enhanced adsorption capacity and lowered adsorption efficiency. The lower adsorption efficiency was due to the lack of active sites available for the large number of AMX molecules in the concentrated solution. In contrast, increasing the SWCNT dose enhanced the adsorption efficiency owing to the large number of free active sites. However, the adsorption capacity was lower owing to the lack of active site saturation when the SWCNT dose increased and was related to the low ratio between the adsorbent and antibiotic. The adsorption of AMX could be completed within 45 min, removing 99% of the AMX at 323 K. When the SWCNT efficiency was studied at different temperatures, the maximum adsorption capacities at 278, 298, and 323 K were 108.84, 116.25, and 122.48 mg/g, respectively. The endothermic nature of the process was indicated by the increasing adsorption capacity with increasing temperature. The pseudo-second-order model described the adsorption well, with
The effective removal of ERY from aqueous solutions by MWCNTs was reported by Mostafapour et al. Based on SEM and transmission electron microscopy (TEM), the main external and internal diameter of MWCNTs used were 2.0–3.5 and 1.2–1.7 nm, respectively, with a specific surface area of 782.8 m2/g. The effects of contact time, temperature, mixing rate, and adsorbent dose on the adsorption ability towards ERY were investigated. A rapid increase in removal efficiency was observed during the first 30 min due to the large number of available active sites, followed by equilibrium at 75 min. When the temperature was increased, the removal efficiency increased, indicating that adsorption was endothermic. The highest removal efficiency (99.68%) and adsorption capacity (124.6 mg/g) were obtained at 318 K. The adsorption process exhibited a negative Gibbs free energy and positive entropy value, indicating that adsorption of ERY by MWCNTs is spontaneous with a high affinity for ERY. When the mixing rate was increased from 0 to 200 rpm, a significant increase in ERY adsorption efficiency was observed. The high mixing rate promoted contact between the adsorbent and ERY, increasing adsorption efficiency. An optimal removal of 98.9% was achieved at 200 rpm. Higher adsorbent dosages enhanced adsorption efficiency by providing more active sites, with an optimal dose of 1 g/L yielding a removal efficiency of 92.7%. A further increase in the adsorbent dosage beyond 1 g/L resulted in negligible increases in adsorption capacity due to the saturation of the active sites. The adsorption kinetics followed a pseudo-second-order kinetic model, indicating chemisorption. Using the intraparticle diffusion model, both intraparticle and film diffusion were shown to control adsorption rates. The rate-determining step was further analysed using the Boyd kinetic model, showing that film diffusion limited the adsorption rate of ERY. The removal efficiency of ERY by MWCNTs was 99.4% under optimal conditions, suggesting that MWCNTs are promising adsorbents for ERY removal [77].
6.3. Graphene-Based Materials
Graphene is a two-dimensional single layer sp2 hybridised carbon with a hexagonal aromatic ring structure (Figure 5). It is a promising adsorbent owing to its large surface area, electron-rich nature, electrostatic stacking properties, tunability with functional groups, and incorporation of multiple functional materials.[52] Graphene hydrophobicity allows it to interact with hydrophobic pollutants through van der Waals or
Graphene has a large surface area and high surface hydrophobicity which favours interactions with hydrophobic organic molecules. Surface modification of graphene can enhance its hydrophilicity and improve its ability to interact with a wider range of molecules. Researchers have combined nanomaterials with GO/rGO to further enhance their adsorption capacity for aqueous pollutants. Recently, interest in graphene-based nanomaterials has grown owing to their unique physicochemical properties, including high adsorption capacity, electroconductivity, good mechanical strength, and thermal stability. [74, 78] Metal oxide nanomaterials, including iron oxide and zirconium oxide, can enhance the removal efficiency for antibiotics.[79] Graphene-based materials can effectively treat water polluted with antibiotics as well as other organic and inorganic compounds.[80] Some innovative graphene-based adsorbents are reviewed in the following section.
6.3.1. Adsorptive Removal of Antibiotics by Graphene-Based Materials
Zou et al. composited α-Fe2O3 nanoparticles on rGO to form an α-Fe2O3/RGO hybrid sorbent for the removal of antibiotics (Table 4). The adsorptive capacities of α-Fe2O3/RGO-400 for TC, CTC, and OTC were determined to be 216.2, 180.8, and 98.4 mg/g, respectively, with adsorptive amounts of rGO for TC, CTC, and OTC of 102.1, 108.8, and 70.3 mg/g, respectively. This indicates that the α-Fe2O3 nanoparticles enhanced the adsorptive capability of rGO. In addition, the adsorption of TC and CTC was much higher than that of OTC, illustrating the relatively weak complexes formed between OTC and Fe(III). When tested at different pH values, the decontamination rate increased rapidly from pH 3.9 to 6.2 and decreased gradually when at
Antibiotics adsorption by graphene and their oxides. The adsorptive characteristics of the adsorbents are summarised. ND: not determined.
Miao et al. developed magnetic graphene oxide (MGO) to remove TCs (TC, CTC, and OTC) from aqueous solutions. The concentrations of TC, CTC, and OTC decreased rapidly in the first 400 min, followed by a gradual decrease, reaching a minimum concentration after approximately 8, 10, and 8 h, respectively. The increased adsorption capacity was mainly due to the high surface activity, large surface area, and full exposure of TCs to the MGO active sites. The maximum adsorption of TC, CTC, and OTC by MGO was 303.9514, 289.8551, and 141.4427 mg/g, respectively. For TC and OTC, the adsorption capacity increased from pH 2 to 7 and subsequently decreased at
Zirconium oxide (ZrO2) nanopowders with different structures, including monoclinic, tetragonal, and cubic, exhibited good performance for pollutant removal. Hao et al. developed nanocomposites of ZrO2 and rGO, ZrO2@rGO, consisting of pure monoclinic or tetragonal ZrO2 to enhance the adsorptive removal of OTC. The m-ZrO2@rGO and t-ZrO2@rGO samples were prepared, and their adsorption performances were investigated. The adsorption capacity for OTC by t-ZrO2@rGO (198.4 mg/g) was greater than m-ZrO2@rGO (177.9 mg/g). Both adsorbents showed a higher affinity towards OTC than TC or CTC due to the stronger complexation between OTC and ZrO2 with a high Zr-OTC stability constant. The influence of the contact time and pH on OTC absorption by m-ZrO2@rGO and t-ZrO2@rGO was studied. Increased adsorption was observed at 0–20 min due to the abundant active sites available. Negligible increases were observed after 20 min, indicating that the system achieved adsorption equilibrium. At low pH (
Zhu et al. compared the adsorption ability of graphene with coconut-derived GAC (CAC) for removing CIP and found that graphene outperformed CAC. The adsorption capacity of graphene was 323 mg/g, while CAC exhibited a maximum adsorption capacity of 217 mg/g. This can be explained by the greater specific surface area of graphene compared to CAC. In addition, up to 79.5% of the graphene pores were macropores. The large surface area and macropores facilitate the adsorption of macromolecules onto graphene active sites. The effect of the initial solution pH on CIP adsorption was investigated. When the pH was increased from 4 to 10, adsorption capacity decreased. Graphene existed in the negatively charged form in this range, with zero zeta potential at pH 3.9, whereas CIP was in cationic form below pH 6. At pH 6–8, CIP exists in the zwitterionic or neutral form. Therefore, when the pH increased, the interaction between graphene and CIP changed from electrostatic attraction to repulsion, rendering CIP adsorption less favourable. In addition to electrostatic interaction, shifting of the C=C bond absorption band in the Fourier transform infrared spectrum suggested a major role of
Yu et al. investigated the adsorption potential of alkali-activated graphene (G-KOH) for CIP removal. The adsorption capacity of G-KOH was higher than that of untreated graphene (G) by a factor of approximately 1.33 due to enhanced porosity and surface area. The surface area of G-KOH increased 3.7 times compared to that of graphene. The destruction of the graphitic structure of graphene after KOH treatment was responsible for the production of new micropores (0.056–0.209 cc/g), resulting in significant surface area increase. The oxygen atomic content of G-KOH increased after alkali activation treatment. The incorporation of oxygen-containing groups on the graphene surface promoted its dispersibility in aqueous solutions, exposed more active sites, and increased CIP adsorption. As CIP exists in different ionic forms at different pH values, the adsorption capacity varied over the tested pH range. At
The applicability of graphene-based materials in aqueous solutions can be improved by combining graphene, GO, or rGO with inorganic composite materials. Theoretically, the addition of magnetite can allow for easier retrieval of used adsorbents after treatment using a magnet, avoiding the need for centrifugation or filtration. Tang et al. synthesised an rGO/magnetite composite (RGO-M) in situ and investigated its adsorption capability for CIP and NOR. The surface morphology revealed that the monodispersed Fe3O4 microspheres were distributed homogeneously on the rGO surface, with an average diameter of approximately 130 nm. An absorption band at approximately 1440 cm-1 was observed in the infrared spectrum, indicating complex formation between the carboxyl functional group and Fe on the magnetic particles. This indicates that the Fe3O4 microspheres were covalently bonded to rGO. At room temperature (298 K) and pH 6.2, the maximum adsorption capacities were 18.22 and 22.20 mg/g for CIP and NOR, respectively. The adsorption was well described by the pseudo-second-order and Langmuir models. In addition, a good fit to the Temkin model illustrates the role of electrostatic interactions during adsorption. The involvement of electrostatic interactions was further confirmed when the electrostatic repulsion reduced the adsorption capacity at higher pH. In contrast,
7. Conclusions
Although antibiotics can improve public health and quality of life, their presence in the environment poses potential threats to human health. With rapid societal development, conventional activated carbon and graphene-based materials have demonstrated that carbonaceous materials can play an important role in the adsorptive removal of antibiotics. Activated carbon is the most commonly used adsorbent, but its adsorption capacity and selectivity can be further improved. Carbon nanotubes and graphene exhibit good adsorption capacities and have been widely applied to construct advanced adsorbents for antibiotic removal from aqueous solutions.
Although adsorption is an effective method for combating antibiotic pollution, a few limitations must be addressed to produce a better adsorbent.
High production and regeneration costs often limit the commercial application of ACs, CNTs, and graphene Disposal of secondary waste, including spent adsorbent and recovered antibiotics, remains a challenge Potential secondary pollution from carbon adsorbents modified with metals
In addition, most experiments were conducted under laboratory conditions wherein the antibiotic solutions are prepared without interferents, resulting in discrepancies when the adsorbent is applied to actual wastewater. Therefore, more concerted efforts are needed to produce practical, green, stable, and economically feasible carbonaceous adsorbents for antibiotic removal from aquatic environments.
Footnotes
Conflicts of Interest
The authors declare no conflicts of interest regarding the publication of this article.
Acknowledgments
This work was supported by the International Medical University under grant BP I-01-2021(09).
