Abstract
A series of
1. Introduction
Antibiotics are widely used to control bacterial infections in medical, agricultural, and veterinary sectors [1]. Oxytetracycline hydrochloride (OTC) is a commonly used tetracycline antibiotic. Large amounts of OTC have been released into the environment due to its extensive use [2, 3]. It has a long half-life due to its naphthacene core, persisting in the environment for long periods of time. Its environmental residue, especially in water sources, being carcinogenic, and causing antibiotic resistance in bacteria, is dangerous for human health and the ecosystem [3, 4]. Along with antibiotic water pollution, air pollution has also increased in recent times. With rapid worldwide industrial development, large amounts of CO2 are released into the atmosphere, causing the greenhouse effect and global warming [5, 6]. Climate change, due to pollution, has caused severe meteorological phenomena, such as typhoons and floods. Thus, CO2 emission reduction and its environmental remediation are an urgent necessity.
Among different ways to solve these above pollution problems, photocatalysis is an attracted one in recent times [7–12]. The photocatalysis can oxidize OTC to nontoxic compounds [13] and reduce CO2 to useful chemical compounds, such as CH4 and CH3OH [14], reducing water and air pollution. It is a simple and green process, requiring only light irradiation for catalyst activation. The TiO2 photocatalyst is commonly used because of its high photocatalytic efficiency, good stability, and nontoxicity [15, 16]. However, it is only activated under 380 nm light irradiation. UV light intensity in the sunlight spectrum is approximately 5%; therefore, the TiO2 photocatalyst requires a UV light source, increasing the process cost. To overcome this drawback, numerous studies report reducing the TiO2 bandgap, activating TiO2 at longer wavelengths (such as visible light), or combining TiO2 with narrow bandgap semiconductors. In the first strategy, the TiO2 oxide is doped with nonmetallic elements (N, F) or transition metals (Fe, Co) [17–22]. In the second method, TiO2 is combined with other semiconductors, such as CuO, BiOBr, metal Au, Pt, or both [23–29]. When combining with an oxide-owned narrower bandgap energy, the new composite will be activated by a longer light wavelength, such visible light, and prevent electron-hole pair recombination. With metal deposition, the metallic phase conduction band attracts free electrons, reducing the recombination of electron-hole pairs.
Recently, graphitic carbon nitride (g-C3N4), a nonmetallic photocatalyst, has attracted immense interest [30–36]. Synthesized by facile and cost-effective methods, it exhibits high chemical and thermal stability. In particular, it exhibits potential for antibiotic-photocatalytic degradation in aqueous solutions and CO2 photoreduction [37, 38]. A low bandgap of 2.7 eV enables their visible-light activation. However, rapid recombination of photogenerated electron-hole pairs is a limitation of the g-C3N4 photocatalyst. The combining of g-C3N4 with other semiconductors to prevent this recombination is an interesting strategy [39].
Among the aforementioned photocatalyst improvement strategies, the construction of a heterojunction between two semiconductors is a promising method. In this structure, the photogenerated electron-hole pairs are separated into different zones, thus preventing recombination and enhancing photocatalytic activity. Further, the carrier transfer behavior has been altered by several photocatalytic mechanisms such as type-I, type-II, and, quite recently, the
In view of the above, this study demonstrates the coupling of TiO2 and g-C3N4 to obtain heterojunction photocatalysts,
2. Materials and Methods
2.1. Materials
Chemical compounds, melamine (C6H6N6) (Sigma-Aldrich), C16H36O4Ti (Sigma-Aldrich), CH3COOH (China), C2H5OH (China), TiO2 (Evonik P25), and oxytetracycline hydrochloride (OTC) (Sigma-Aldrich), of analytical purity were used as obtained.
2.2. Synthesis of g-C3N4 and N-TiO2-δ /g-C3N4 Composites
2.2.1. Synthesis of g-C3N4 and N-TiO2
Melamine calcination at 550°C, for 3 h, in nitrogen gas medium, was used to synthesize g-C3N4.
The nitrogen-doped TiO2 (N-TiO2) synthesis was inspired by the work of Viswanath et al. [46]. In this typical procedure, titanium (IV) butoxide in ethanol solution and melamine in hot water-ethanol (1 : 3 volume ratio) solution were mixed, then stirred for 24 hours and aged for 5 days. The obtained gel was dried and calcined at 400°C for 3 hours.
2.2.2. Synthesis of N-TiO2-δ /g-C3N4
Calculated amounts of g-C3N4, titanium (IV) butoxide, and acetic acid (1 : 30 in volume) were mixed in a 250 ml beaker, with 15 min magnetic stirring, and autoclaved at 140°C for 12 h. The resulting solution was centrifuged and rinsed several times with ethanol. The solid obtained was oven dried at 60°C for 24 h and calcined at 400°C for 3 h, under nitrogen gas. The
2.3. Photocatalytic Procedure
2.3.1. OTC Photodegradation
Into a beaker containing 100 ml 10 ppm OTC solution (C0), g-C3N4 or

Scheme of the photocatalytic reactor used for OTC oxidation (a) and CO2 reduction (b).

2.3.2. CO2 Photoreduction
Into a glass beaker (5 cm diameter) containing 15 ml deionized water, g-C3N4 or
2.4. Characterizations
X-ray diffraction (XRD) (Bruker D8), Fourier-transform infrared spectroscopy (FTIR) (8101M Shimadzu), N2 adsorption-desorption (TriStar 3000-Micromeritics), UV differential reflectance spectroscopy (UV-DRS) (Jaco V-530), transmission electronic microscopy (TEM, JEM 1400 – Plus Jeon), and X-ray photoelectron spectroscopy (XPS) (Thermo Scientific MultiLab 2000) were used to characterize g-C3N4 and
3. Results and Discussion
3.1. Structural Characterization
XRD patterns of

XRD patterns (a) and FTIR spectra (b) of g-C3N4 and

The light absorption abilities of the composites were analyzed by UV-vis DR spectra. Figure 3(a) shows the UV-vis DR spectra of g-C3N4 and
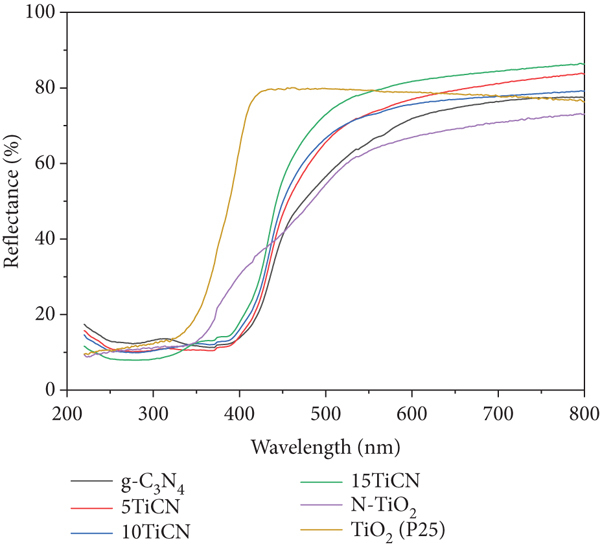
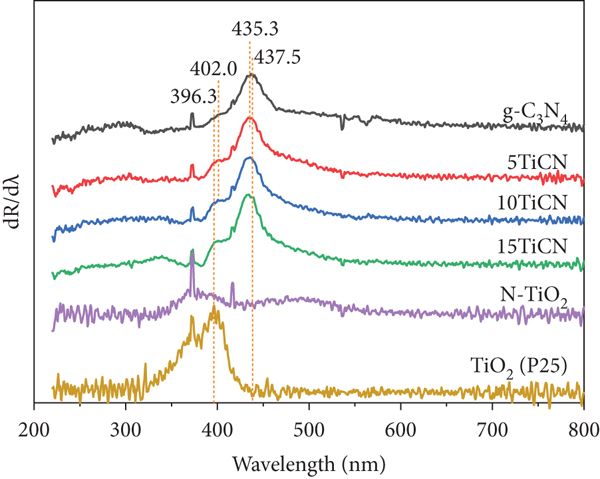
UV-vis DR spectra (a), energy bandgap determination by Kubelka–Munk function (b), and its corresponding differential curves (c) of g-C3N4, N-TiO2, and


The g-C3N4 and 5TiCN composites were selected to characterize its specific surface area, one of the important properties of heterogeneous catalysis. The results are presented in Figure 3(d). It is observed that two samples show the type-4 isotherms with H3 hysteresis loop, which indicate the presence of a mesopore. This is suitable with the obtained pore distribution curves. The BET surface areas are 18 and 20 m2/g for g-C3N4 and 5TiCN, respectively. Hence, the composite preparation did not seem to change the g-C3N4 structure.
The interaction between the TiO2 and g-C3N4 phases is better understood from the TEM images presented in Figure 4. It is observed that the TiO2 particles (dark areas in Figures 4(a) and 4(b)) were formed in various sizes and shapes. Some TiO2 particles were deposited on the g-C3N4 layer (e.g., position of cycle 1), while the others were covered by g-C3N4 multisheets, forming a core-shell structure (e.g., position of cycle 2), thereby enhancing the interaction surface between the TiO2 and g-C3N4 phases.

TEM images of 5TiCN.
The elemental composition and oxidation state of the catalyst influence catalytic performance. Therefore, the 5TiCN composite was characterized using XPS, as shown in Figure 5.

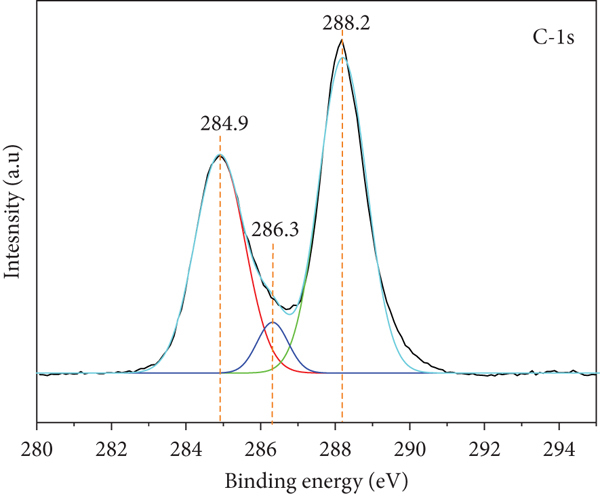
X-ray photoelectron spectra of 5TiCN: (a) survey spectra; (b) high-resolution spectra of Ti2p, (c) high-resolution spectra of O1s; (d) high-resolution spectra of N1s; (e) high-resolution spectra of C1s.
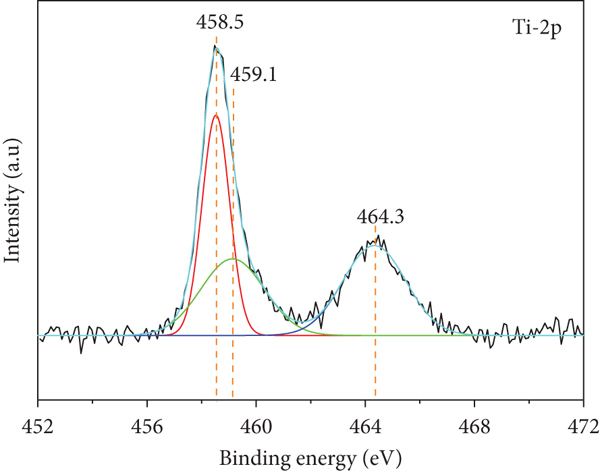


Three peaks at binding energies of 458.5, 459.1, and 464.3 eV, corresponding to orbitals Ti4+2p3/2, Ti3+2p1/2, and Ti4+2p1/2, respectively, were observed in the Ti2p high-resolution spectrum [50, 51]. As reported by Jia et al., binding energies of two prominent peaks (at 458.5 and 464.3 eV) exhibited 0.2 eV shifts compared to spectral peaks of pure TiO2. This could be due to the substitution of O2- by N3-, leading to the formation of N-Ti-O bonds [52]. This indicated N doping of TiO2 during composite synthesis, decreasing the TiO2 bandgap energy, leading to visible-light activation [17, 18]. The small peak of Ti3+2p1/2 confirmed oxygen vacancies in TiO2 [51]. Hence, TiO2-δ is a more accurate molecular formula than TiO2.
Deconvolution peaks for the O1s spectrum exhibited two peaks at 531 and 532 eV, assigned to O-H of absorbed water and the Ti–O bond [53], respectively. N1s spectrum exhibited three peaks at 398.6, 399.7, and 400.8 eV, ascribed to the sp2C of C-N=C, tertiary N of N-(C)3 group, and N-C=N bonds, respectively. The C1s spectrum exhibited three peaks at 284.9, 286.3, and 288.2 eV, corresponding to C-C, C-NH2, and N-C=N bonds, respectively [53, 54].
3.2. Evaluation of Photocatalytic Activity
3.2.1. Photooxidation of OTC
Before performing the photocatalytic test, the adsorption equilibria were carried out (Figure 6(a)). The results demonstrated that all composites reached rapidly the adsorption equilibrium after only about 15 minutes, while 60 minutes was required for N-TiO2. The calculation indicated that the equilibrium adsorption quantities of OTC at 60 min were 11%, 14%, 17%, 12%, and 88% for g-C3N4, 5TiCN, 10TiCN, 15TiCN, and N-TiO2, respectively. It remarked that there was a strong adsorption phenomenon of OTC on N-TiO2.

OTC adsorption equilibrium (a) on

The photocatalytic activity of the catalysts was investigated through OTC photodegradation and CO2 photoreduction. Figure 6(b) shows the photodegradation of OTC and UV-vis spectra of OTC solutions during test time, using 5TiCN. OTC concentrations were calculated from the absorbance intensity of UV-vis spectra at the 357 nm wavelength.
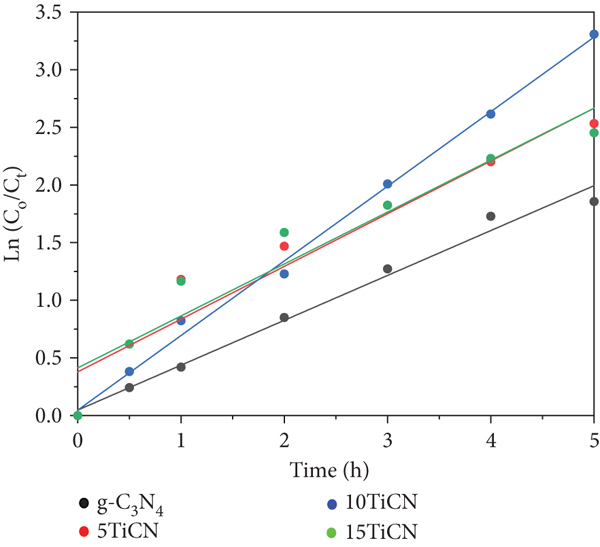
Figure 7 shows OTC photodegradation efficiency and kinetics. All composite photocatalysts exhibited excellent OTC degradation activity, with yields of 93%, 97%, and 92% for 5TiCN, 10TiCN, and 15TiCN, respectively. For N-TiO2, the adsorption phenomenon dominated, reaching 90% adsorbed OTC quantity after 1 hour of equilibrium, and the efficiency of OTC removal increased only ~4% when turning on the light for 5 hours. Hence, the photocatalytic reaction on N-TiO2 was negligible. This behavior was possibly due to the formation of the melon structure formed during the synthesis, besides the process of nitrogen doping on TiO2 [55]. The melon structure, which is not a semiconductor, could cover TiO2/N-TiO2 particles, preventing the photocatalytic process. A blank test (without catalyst) was also carried out for comparison. In the blank test, the OTC concentration decreased about 9% by photolysis. Hence, after 5 h light irradiation, the 10TiCN composite was slightly more active than those of 5TiCN and 15TiCN. It is noted that the adsorption phenomenon contributes a small part in the conversion calculation, only 14% in the case of 5TiCN as mentioned in the above adsorption equilibrium study. For these OTC conversion reactions, reaction kinetics were described by the following equation [56]:
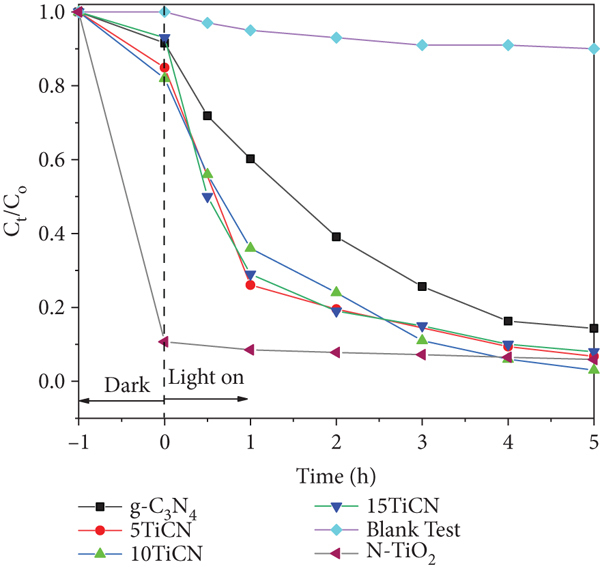
(a) Photodegradation efficiency of OTC on
There are no reports of OTC photodegradation on TiO2-g-C3N4 composites, while there are very few reports on tetracycline (TCL) photodegradation using such composites. Wang’s research group reported 20 ppm TCL solution photodegradation using composites, such as TiO2@g-C3N4 heterojunction, TiO2@g-C3N4 core-shell quantum heterojunction, and TiO2-x/ultrathin g-C3N4/TiO2-x direct
In general, the TiO2-g-C3N4 heterojunction structure improved photoactivity remarkably compared to those of bulk TiO2 and g-C3N4. OTC photodegradation yield reached up to 97%, with a 1.7 times higher rate constant with 10TiCN than with g-C3N4.
The influence of pH on the photoactivity was evaluated. The researches focused on 5TiCN (Figure 8(a)). After 5 hours of irradiation, the photoactivity at pH 11 and 5 was slightly improved, in comparison with pH 7. In the study of Yu et al., the isoelectric point of g-C3N4 was about pH 5 [61]. It means that the g-C3N4 surface charge is positive at

Photodegradation efficiency of OTC in function of pH (a) and photostability test on 5TiCN (b).
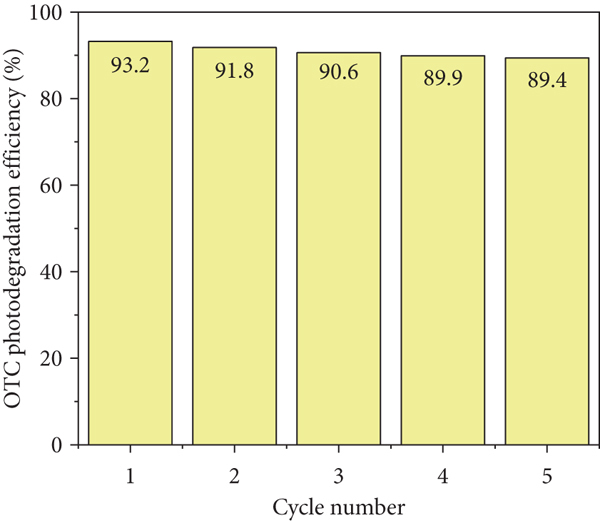
The stability tests were carried out for the 5TiCN composite (Figure 8(b)). The OTC conversion decreased from 93.4% to 89.4% after 5 cycles. This is the promising result in an application view.
3.2.2. Photoreduction of CO2
To investigate the dual photocatalytic behavior of N-TiO2-δ /g-C3N4 composites, they were used in the photoreduction of CO2 by H2O in the gaseous phase (Figure 9). Concentration of CH4 (the only product detected) was monitored to analyze the photocatalytic behavior of the synthesized composites. 5TiCN exhibited the highest activity, with 7.0 ppm CH4 concentration, followed by 10TiCN (5.2 ppm CH4), 15TiCN (4.3 ppm CH4), and g-C3N4 (0.9 ppm CH4). No product was detected in the N-TiO2 test. As mentioned in the OTC photooxidation results above, this low photoactivity of N-TiO2 could be explained by the formed melon structure that covered TiO2/N-TiO2 particles and possibly the lower CB potential position of N-TiO2 than the standard reduction potential of CO2/CH4 [55]. The detected CH4 concentration was quite low over g-C3N4. Thus, the test was performed three times, and average value was taken. Hence, the CH4 concentration was 7.8 times higher for 5TiCN (composite exhibiting maximum photocatalysis) than for pristine g-C3N4.

CH4 concentration on g-C3N4 and
There are very few reports on photoreduction using a photocatalyst formed by TiO2 and g-C3N4 [45, 63–69]. CO2 photoreduction in the gaseous phase [45, 63–65] and liquid phase [66–69] has been reported. Gas phase studies, with CH4 and CO products, indicate better photocatalytic activity for the TiO2-g-C3N4-combined photocatalyst, compared to bulk TiO2 and g-C3N4. Zhou et al. reported a 4 times higher CO formation on g-C3N4-N-TiO2 (14.73 μmol) than on P25 (TiO2) [63]. In liquid phase photoreductions, besides CH4 and CO, other oxygenated hydrocarbons (CH3OH, HCOOH, and CH3COOH) are formed. TiO2-g-C3N4-combined photocatalysts exhibit also higher photoactivity compared to single-phase TiO2 or g-C3N4. Badiei et al. reported an 11.3 μmol·g-1·h-1 CH3OH formation for g-C3N4@TiO2, which was 5 and 10 times higher than those for g-C3N4 and P-25 (TiO2), respectively [66]. Lu et al. reported a 283.9 μmol·h-1·g-1 CO formation for 2D g-C3N4/TiO2, which was 292.2, 6.8, and 5.7 times higher than those for TiO2, bulk g-C3N4, and mechanically mixed TiO2/g-C3N4, respectively [67]. These results cannot be compared due to different experimental conditions. However, all studies indicate higher photoactivity using TiO2-g-C3N4-combined photocatalysts. In this study, CH4 production on 5TiCN was 7.8 times higher than that on bulk g-C3N4.
3.2.3. Photocatalytic Mechanism
An outstanding photocatalytic activity was exhibited by the

Schematic illustrations of the energy band structures of N-TiO2-δ and g-C3N4 before contact (a), N-TiO2-δ /g-C3N4 heterojunction composite for OTC photooxidation mechanism (b), and CO2 photoreduction mechanism (c).
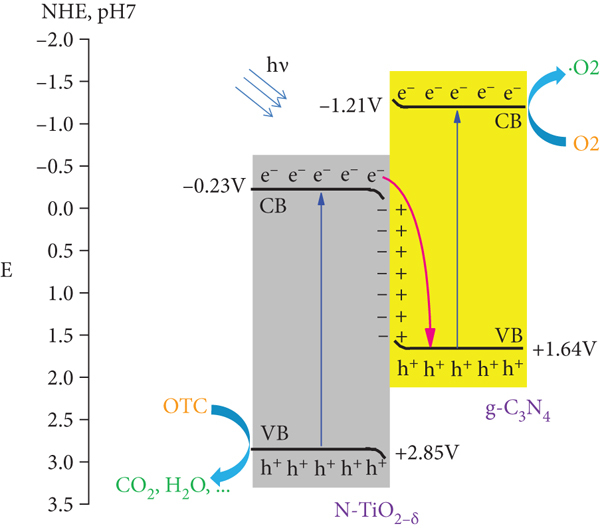

Note that the type-II and
The photogenerated electron-hole pair transfers are summarized as follows:
Then, for oxidation of OTC,
or for reduction of CO2,
4. Conclusions
Here,
Footnotes
Data Availability
The data used to support the findings of this study are included within the article.
Conflicts of Interest
The authors declares that there is no conflict of interest regarding the publication of this paper.
Acknowledgments
This research is funded by the Vietnam National Foundation for Science and Technology Development (NAFOSTED) under grant number 104.05-2017.39.
