Abstract
Concerns have been increased regarding the existence of pollutants in environmental water resources and their risks to the ecosystem and human society. TiO2 photocatalyst is considered as an effective photocatalyst to remove the pollutants. Herein, the mesoporous TiO2-SBA-15 was prepared using the rice husk extract as the silica source. The fabricated nanocomposites were characterized using FTIR, small and wide angle XRD, Raman spectroscopy, UV-vis, BET surface area analysis, and HRTEM. The photocatalytic efficiency of the composites for the degradation of methylene blue (MB) has been evaluated under UV irradiation. Interestingly, due to the excellent dispersion of TiO2 on the wall of SBA-15 and good hydrophilicity, the nanocomposites displayed a good catalytic activity. The higher photodegradation performance was achieved by the composite containing 10 wt% TiO2 by which the MB was fully degraded within 15-20 min of irradiation. Besides, TiO2-SBA-15 could effectively inhibit the growth of Gram-positive and Gram-negative bacteria. These results offer a practical and economic approach in the environmental management industries.
1. Introduction
Rice husk (RH) is a byproduct of rice production [1–3]. RH contributes approximately 20-25 wt % of the whole dry weight of rice [4]. With the annual rice production of 489.1 million tons [5], about 120 million tons of RH were produced as the biomass waste. Due to its weak mechanical strength, poor nutritional value, and low bulk density, the use of RH is limited [6–8]. So far, the most popular RH disposal approach is the open field burning which leads to air pollution, greenhouse gas emissions, and a waste of energy [9]. However, RH contains a high amount of silica, which could be used as the raw material for producing value-added silicon-based materials with interesting texture and morphology. The implementations of such substances stemming from rice husks are very inclusive [10, 11]. In the past decade, this research area has been dramatically progressed and extended, driven by the worldwide emphasis on sustainable and renewable resources.
As a chemically inert material, porous silica plays a significant role in various applications including adsorption, sensing, and catalyst support and templating, owing to its high surface area, adjustable porosity, and surface properties [12–14]. Its application largely depends on its morphology and microstructure. Mesoporous silica can be artificially synthesized with a certain degree of control in its nanomorphology [15]. However, it usually involves a complicated process, for instance, high quality of the amorphous silica was produced by the multistep thermal reduction of crude silica. The need for high temperatures and high pressure under extreme acidic condition is energy-intensive with high impact on the environment [16]. Hence, it is important to develop an affordable, sustainable, and environmentally friendly technique for the fabrication of porous silica to satisfy the rising demand for its widespread applications.
With the rapid population and industrialization growth, water pollution from organic contaminants seems to have become a big worldwide problem that risks the ecological system and human health [17, 18]. To remove organic contaminants from water, several physical and biological treatment approaches have been used, including ultrafiltration, coagulation, and adsorption [19, 20]. However, their practical applications are restricted by the limited capacity to remove trace organic compounds and the costs involved in the replacement of media. Therefore, a highly efficient, environmentally friendly, and sustainable water treatment process is needed to remove organic pollutants from water.
Due to the moderate operating conditions, high catalytic activity, and environmentally safe, TiO2 photocatalyst has been an excellent candidate for the removing of organic contaminants from air and water [21, 22]. Although the use of TiO2 nanoparticles is valuable for scientific research, the size of the particles restricts their practical use in water treatment. It is difficult and expensive to recover and resue the TiO2 nanoparticles from a liquid environment. Meanwhile, complex water matrices could easily interfere with the surface property of the suspended fine TiO2 particles [23]. Therefore, the design and producing of nanoporous TiO2 framework have attracted some attention [24–26]. In such case, porous silica, as an optically transparent material, is an ideal candidate for supporting TiO2 used as the photocatalyst in an aqueous environment.
The primary target of the present work is to develop an effective, economical, and sustainable nanocomposite of TiO2 supported by mesoporous nanosilica using the rice husk as the sustainable raw material. The catalytic efficiency of the composites for the photocatalytic degradation of methylene blue (MB) was evaluated under UV irradiation. The unique mesoporous nanocomposite material displayed excellent photocatalytic performance in the degradation of MB and the bacteria inhabitation tests. The immobilized TiO2 also showed success in the recovering and reusing process with the potential for practical environmental treatment.
2. Materials and Methods
2.1. Materials
Sodium hydroxide (NaOH, ≥99%), hydrogen peroxide solution (H2O2, 30 wt %), titanium isopropoxide (TTIP, 99%), the nonionic surfactant of EO20PO70EO20, (Pluronic P123), ethanol (99%), and hydrochloric acid (HCl, 37%) were procured from Sigma-Aldrich Co., USA. MB (>96%) was supplied from Central Drug House (P) Ltd., India. H2SO4 (96%) used for silica extraction was obtained from Fisher Scientific. Rice husk was obtained by removing rice. Deionized water was utilized in the whole experiment. All materials were used without any further treatment.
2.2. Material Fabrication
2.2.1. Extraction of Nanosilica from Rice Husk
Silica was extracted from rice husk according to the previously reported procedure [27]. Briefly, 150 g of ground husk and 60 ml of concentrated H2SO4 were added in a glass bottle containing 1 L of deionized water. The mixture was digested at 80°C for 4 h under mechanical stirring. The precipitate was then collected by filtration. The obtained raw silica was calcinated at 800°C for 3 h to remove residual impurity. Finally, the produced silica was bleached using 10% H2O2 solution at room temperature for 3 hours.
2.2.2. Preparation of SBA-15
Mesoporous SBA-15 silica was synthesized using sodium silicate solution with Pluronic 123 as the structure-directing agent under acidic conditions. The Pluronic 123 solution was made by dissolving 4 g of Pluronic 123 in 120 g of HCl (2 M), followed by stirring at 40°C until Pluronic 123 was completely dissolved. Sodium silicate solution was produced by dissolving 1 g SiO2 from the rice husk in 230 M NaOH solution (3 M). The P-123 solution was gradually added to the sodium silicate solution at 40°C under intense stirring. The reaction between the P123 polymer micelles and the sodium silicate produced the emulsion after rapid mixing [28]. The mixture was continuously stirred for 24 h at 40°C, and the SBA-15 precipitates were formed. The solid was separated by filtration and rinsed several times with a mixture of water and ethanol. Finally, the solid sample was dried at 80°C under vacuum for 12 h. The collected SBA-15 was calcined for 6 h at 600°C to remove any organic residuals (Scheme 1).

The fabrication strategies for TiO2-SBA-15 nanocomposite from rice husk.
2.2.3. Preparation of TiO2-SBA-15 Nanocomposite
TTIP was used as the Ti precursor. In a typical synthesis, 1.0 g of dried SBA-15 was dispersed in 50 ml of ethanol followed by sonication for 60 min. Various amounts of TTIP (corresponding to 5, and 10 wt% TiO2) were then introduced to the suspension. The mixture was mixed for another 60 min until a clear solution was formed. Then, a certain volume of deionized water (30 ml) was introduced dropwise under vigorous stirring for 4 h to hydrolyze the TTIP to Ti (OH)4 species and to be deposited on the surface of SBA-15. The solid product was separated, rinsed several times with H2O and ethanol, dried overnight at 120°C, and finally calcinated for 4 h at 400°C to create the TiO2-SBA-15 nanocomposites [29, 30] (Scheme 1).
2.3. Material Characterization
Small and wide angle X-ray diffraction (XRD) measurements were carried out using an X-ray diffractometer (XPERT, PANalytical) with Cu Kα (
The optical property was measured with a UV-vis spectrophotometer (UV-2600, Shimadzu) equipped with diffuse reflectance difference spectroscopy attachment (DRDS). The sample was spread on a glass slide with a certain thickness,
The chemical oxygen demand (COD) was measured using an oxidation method in the presence of K2Cr2O7 [32].
2.4. Photodegradation Performance Measurements
In a typical photodegradation experiment, in double wall jacket glass reactor for temperaure controler, 50 mg of TiO2-SBA-15 photocatalyst was added in 100 ml MB solution with initial concentration (
2.5. Antibacterial Performance
The antibacterial performance of the fabricated materials was performed by the agar well diffusion approach. The bacterial isolates were cultured for 20 hours at 35°C in a medium of nutritional broth having 10 g/L NaCl, 3 g/L extract of yeast, and 8 g/L bactotryptone. Using sterile cotton buds, 100 μl of the suspensions of the cell was dispersed on a nutrient agar surface, and the agar was pierced with 8 mm diameter reservoirs and loaded with 300 μl of TiO2-SBA-15 aqueous solutions (5 mg/ml). The plates was incubated for 24 h at 35°C. The antibacterial performance of the nanocomposite was assessed by the size of the inhibition zone (growth diameter). The antibacterial performance of TiO2-SBA-15 nanocomposite with 10 wt % TiO2 against Gram-positive and Gram-negative bacteria is tested, and four different bacteria were selected for testing, including B. subtilis, B. cereus, Klebsiella pneumonia, and E. coli.
3. Results and Discussion
3.1. Material Characterization
In order to examine the TiO2 loadings in the composite of X%TiO2-SBA-15, elemental analysis is used, and the results are reported in Table 1. The results revealed that the TiO2 loadings in the composites range from 4.5 to 9.7 wt%.
Unit cell parameter and texture characteristics of pristine SBA-15 and TiO2-SBA-15 nanocomposite containing various TiO2 contents.
aTiO2 concentration in the gel. bTiO2 concentration in the calcined materials measured by ICP.
The small-angle XRD patterns for pristine SBA-15 and TiO2-SBA-15 nanocomposites with different TiO2 contents are presented in Figure 1(a). All the fabricated composites display well-defined XRD diffraction peaks related to the (1 0 0), (1 1 0), and (2 0 0) crystal planes of the SBA-15 structure with P6mm hexagonal symmetry [33]. The high intensities of these diffraction peaks indicate that good quality SBA-15 mesostructure was successfully produced. With the incorporating of TiO2, the diffraction peaks from the SBA-15 shift towards smaller angles, representing the increased lattice constant for the SBA-15. This finding suggests that some of the Si4+ were substituted by the Ti4+ since Si4+ has a radius of 0.040 nm, smaller than that of the Ti4+ (0.061 nm). The unit cell parameter (

(a) Low-angle XRD and (b) wide-angle XRD patterns of pure SBA-15 and TiO2-SBA-15 nanocomposite (JCPDS card no. 21-1272 (anatase TiO2) and ICDD # 00-001-0424 (silica phase)).

Figure 2 shows the nitrogen adsorption/desorption isotherms and pore size distribution of pristine SBA-15 and TiO2-SBA-15 nanocomposite with various TiO2 contents (5 and 10 wt.%). The measured properties of the synthesized materials are summarized in Table 1, including the specific surface area (
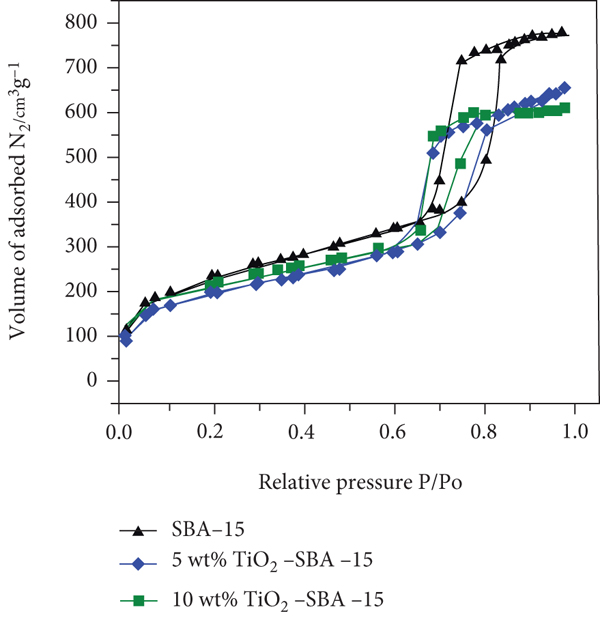
(a) Nitrogen adsorption/desorption isotherms and pore size distribution of pure SBA-15 and TiO2-SBA-15 nanocomposite containing various TiO2 contents. (b) Pore size distributions for SBA-15 and TiO2-SBA-15 (10 wt % TiO2).

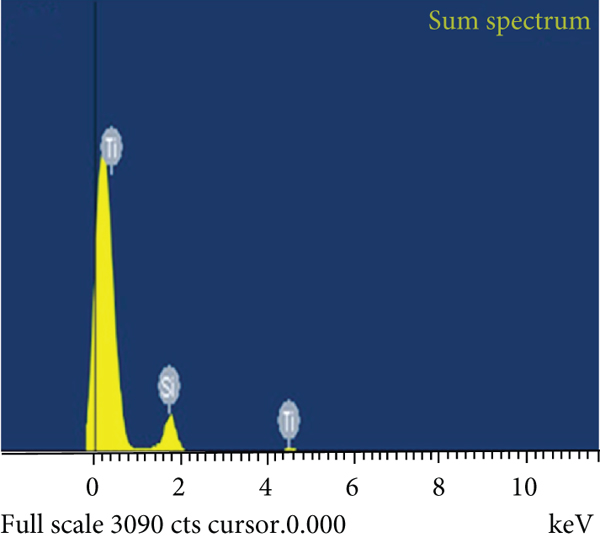
To verify the mesoporous structure of the SBA-15 after TiO2 coating, HRTEM images for TiO2-SBA-15 nanocomposite with 10 wt % TiO2 are displayed in Figure 3. HRTEM image of 10 wt % TiO2-SBA-15 nanocomposite with uniform one-dimensional mesochannels, parallel, and hexagona mesostructure has developed. A

(a) HRTEM images and (b) EDS of 10 wt % TiO2-SBA-15 nanocomposite.
The FT-IR spectra of synthesized SBA-15 and TiO2-SBA-15 nanocomposite with various TiO2 contents are depicted in Figure 4. There are bands representative of hydroxylated silica in the FTIR spectrum of SBA-15: 1074, 943, 804, 555, and 461 cm–1. The bands at 1074 and 804 cm–1 ascribed to the Si–O–Si bond’s asymmetric and symmetric stretching vibrations, respectively. The bands at 943 and 461 cm–1 relate to the Si–OH and Si–O–Si bonds’bending vibrations [36, 37]. The existence of a characteristic absorption band at 555 cm–1 attributable to Si–O–Si symmetric modes of the silicon-oxygen tetrahedral is a distinguishing characteristic of this spectrum. Bending vibrations of coordinately attached and adsorbed water molecules are shown by the bands at 3454 and 1622 cm–1. We find a decrease in the intensity of the absorption bands for stretching vibrations of the Si–O–Si bond in the spectrum of the composite with SBA-15 majority, and their maximum is moved toward smaller frequencies compared to the SBA-15 spectrum. We find a reduction in the intensity of the absorption bands for Si–O–Si stretching vibrations and a shift in their maximum to lower frequencies as the quantity of TiO2 in the composite increases. Simultaneously, with a high silica content in the composite, the absorption band corresponding to Ti-O vibration of octahedraon TiO6 vanishes. This could be linked to the transition of titanium atoms from octahedral to tetrahedral coordination when they substitute silicon in the SiO4 -4 tetrahedron [37]. We can only explain the absorption in this area as a result of the distinctive vibrations of Si–O–Ti bonds following suitable thermal treatment of the materials because the absorption bands for the Si–OH and Si–O–Ti bonds are near.

FTIR spectra of pure SBA-15 and TiO2-SBA-15 nanocomposite containing various TiO2 contents.
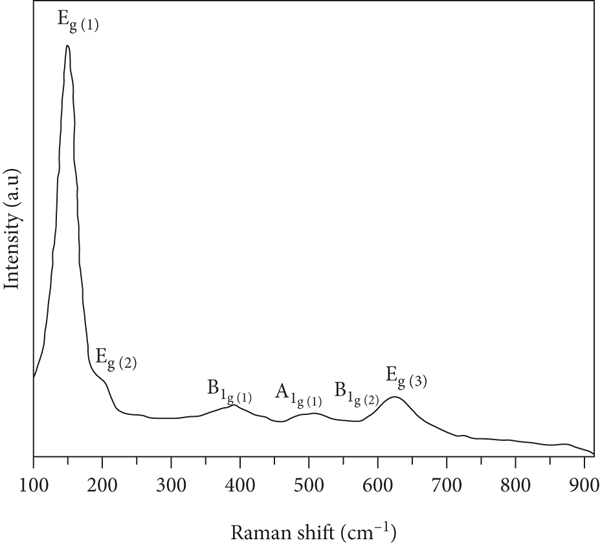
Raman spectroscopy was performed to explore the structural changes upon TiO2 deposition within the SBA-15 matrix and also to identify the phase of the developed titania. The Raman spectra of SBA-15 and 10 wt% TiO2-SBA-15 nanocomposite are displayed in Figure 5. SBA-15 in Figure 5(a) displays a band about 493 cm−1, which is ascribed to tetrasiloxane rings. The bands about 610 and 981 cm−1 could be attributed to cyclic trisiloxane rings and the Si–OH stretching vibration, respectively. Figure 5(b) shows the Raman spectrum of 10 wt % TiO2-SBA-15 nanocomposite. Four Raman active bands were identified at 149, 410, 510, and 633 cm-1, respectively, which are assigned to the Eg, B1g, A1g/B1g, and Eg bands associated with the anatase phase. Furthermore, the SBA-15 band is nearly undetectable, implying the presence of highly dispersed TiO2 species within the SBA-15.

Raman spectrum of (a) SBA-15 and (b) 10 wt % TiO2-SBA-15 nanocomposite.
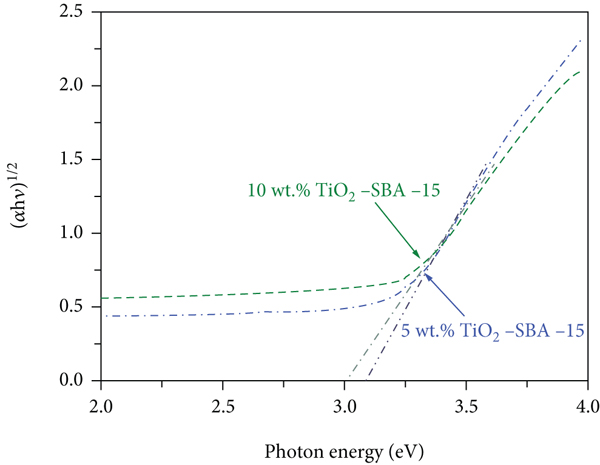
UV-vis absorption measurements were conducted to explore the optical properties of the TiO2-SBA-15 composites at different TiO2 loadings. The recorded spectra of the TiO2-SBA-15 nanocomposite with 5 and 10 wt % TiO2 are displayed in Figure 6. The TiO2-SBA-15 composites mainly absorb the UV, shown in Figure 6(a). The edge of the absorption shifted to a longer wavelength at higher TiO2 content [38, 39]. To determine the bandgap energies of the prepared nanocomposites, the corresponding Tauc’s plots are produced in Figure 6(b). Bandgap energies were obtained by extrapolating the linear part of the plot to the
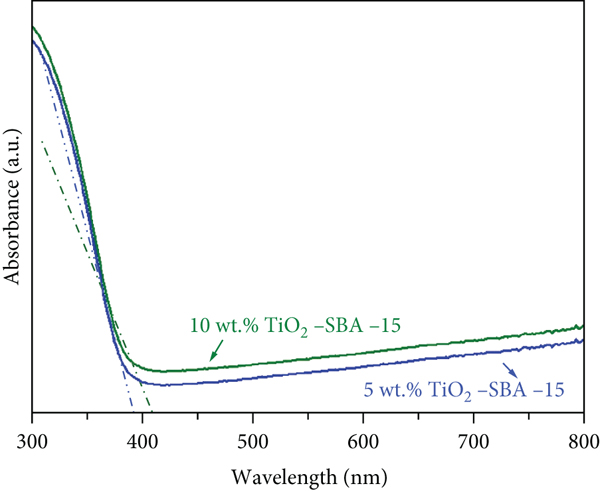
UV–vis absorption spectra (a) and Tauc’s profile for the band gap determination (b) of TiO2-SBA-15 nanocomposite containing various TiO2 contents.
3.2. Catalytic Performance
The dark adsorption experiments of MB over pure SBA-15 and TiO2-SBA-15 nanocomposite with 5 and 10 wt.% TiO2 at different time intervals are displayed in Figure 7. The adsorption saturations were achieved around 30 mins for all samples with the adsorption capacities of 11.8, 7.3, and 4.1 mg/g for pure SBA-15 and TiO2-SBA-15 nanocomposite with 5 and 10 wt.% TiO2, respectively. Hence, the photocatalytic decomposition experiments were delayed by 30 mins after mixing, allowing the adsorption equilibrium to be achieved. Owing to the large surface area of the mesoporous silica matrix, the SBA-15 displayed the highest adsorption capacity. However, after TiO2 incorporation, the adsorption capacity during the dark experiments was decreased compared to pure SBA-15, possibly due to the clogging and the decrease in surface area. The sample with 10 wt % of TiO2 offered the lowest adsorption capacity.

Time-dependent adsorption behavior of the MB on TiO2-SBA-15 nanocomposite containing various TiO2 contents under dark condition.
Under the UV irradiation, the MB photocatalytic degradation by the TiO2-SBA-15 nanocomposites with 5 and 10 wt % TiO2 was examined. The findings are displayed in Figure 8 and Table 2. To identify the effects of UV on the MB molecule, a control experiment using 100 ml MB solution under UV irradiation without catalyst was carried out, shown in Figure 8(a). Only a limited degradation of MB (< 2%) was observed. Hence, the MB is stable under UV irradiation. The photocatalytic performance under UV irradiation of the nanocomposites with different TiO2 loadings for the degradation of MB dye is also shown in Figure 8(a), plotted with the normalized MB concentrations as a function of reaction time. The higher photodegradation performance was achieved by the composite containing 10 wt% TiO2 by which the MB was fully degraded within 15-20 min of irradiation. To measure the reaction rate constant, a first-order kinetic model was used. Figure 8(b) shows the logarithm of MB relative concentration as a function of time. The degradation process obeys the first-order kinetic. The TiO2-SBA-15 photocatalyst with 10 wt % TiO2 gives an MB degradation rate constant of 0.139 min-1, which is about 4 times more efficient than that with 5 wt % TiO2 loading (0.033 min−1). Therefore, the rate constant was improved by the increase of the TiO2 loading. As a control experiment, 10% TiO2-SBA-15 was fabricated using SBA-15 (Sigma-Aldrich) as a support. A comparatively decline in the photocatalytic corresponding to rate constat (
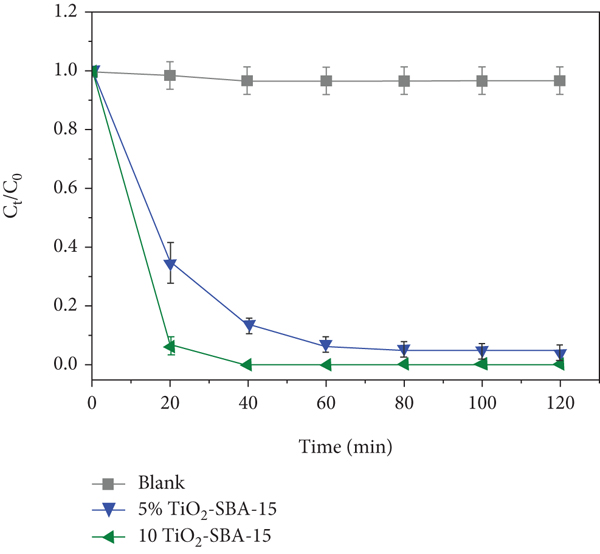
(a) Photocatalytic activity and (b) first-order kinetic curve under UV irradiation of TiO2-SBA-15 nanocomposites.

Chemical oxygen demand (COD) decline of MB with respect to time over the 10 wt% TiO2-SBA-15 nanocomposite.
Based on the abovementioned findings, TiO2-SBA-15 nanocomposite had an uniform pore distribution and large surface area, which aided the MB uptake and diffusion by the SBA-15 mesochanel, resulting in MB accumulation surrounding the TiO2 nanoparticles. This near vicinity between MB and the active centers is likely crucial to increasing the degradation performance. The nanocomposite particles were photoexcited that generate electron (e-) and (h). The electron participates in the reduction processes, whereas holes participate in the oxidation processes. The following equations display the mechanism of the photocatalytic reaction [47].
The involvement of active species was investigated using a trapping experiment to reveal the photodegradation mechanism of MB. Tertiary butyl alcohol (t-BuOH) was used to scavenge ·OH, while nitrogen bubbling was employed to scavenge ·O2− and h+. Figure 9 demonstrates that adding t-BuOH has minimal effect on MB degradation, indicating that ·OH is not involved in the photodegradation process, whereas the

MB degradation using the 10% TiO2-SBA-15 nanocomposite without scavenger and with scavengers under UV-light irradiation.
Furthermore, to ensure dye degradation, a chemical oxygen demand (COD) experiment was carried out. The COD test is a popular method for determining the organic strength of wastewater [32]. The test allows for waste assessment in terms of total oxygen needed for the organic matter oxidation to CO2 and water. A significant decrease in COD indicates degradation in carbon content in the sample and so indicates the extent of mineralization. Organic carbon is converted to gaseous CO2 as a result of degradation. The COD was substantially lowered (Table 2) as a result of the oxidative mineralization process, which involved the aromatic rings opening with the transitory synthesis of carboxylic acids, followed by the evolution of CO2 via the “photo-Kolbe” reaction.
With the use of a 10% TiO2-SBA-15 nanocomposite, the dye molecules might be totally mineralized, based on the considerable reduction in COD values.
One of the advantages of heterogeneous catalysis is the ability to recycle and reutilize the catalysts with a relatively simple procedure at low cost if the catalytic performance of the catalysts can be maintained. To evaluate the recyclability of the TiO2-SBA-15 with 10 wt % TiO2, the used catalyst was collected and immediately reused in the MB degradation without further treatment. The catalytic kinetic data for the consecutive five cycles are presented in Figure 10. The results show that the degradation performance values were 95.8, 95.7, 95.8, 91.7, and 89%. After the fifth degradation run, the ICP and surface area were tested. Obvious, no remarkable TiO2 is detected after the fifth run in reaction mixture, suggesting that the leaching process is not responsible for the decrease in the performance. However, the measured surface area after the fifth run for the nanocomposite is decreased (673 m2 g-1), which is the main reason for the decrease the catalytic degradation.
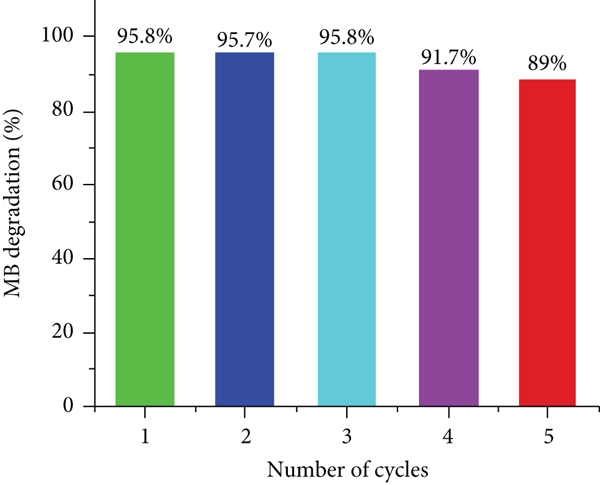
Durability of 10% TiO2-SBA-15 nanocomposites towards the photodegradation activity of MB.
The antibacterial performance of TiO2-SBA-15 nanocomposite with 10 wt % TiO2 against Gram-positive and Gram-negative bacteria is presented in Table 3. Four different bacteria were selected for testing, including B. subtilis, B. cereus, Klebsiella pneumonia, and E. coli. For both Gram-positive and Gram-negative bacteria, to acquire a better understanding of the antibacterial performance, a few control studies were carried out. The antibacterial performance of the nanocomposite-free control sample was tested under the identical conditions and found no significant differences. Besides, the antibacterial performance of the SBA-15 was also tested to enable the comparison with the nanocomposite prepared in this work. A comparatively decline in the antibacterial activity was achieved when using SBA-15. Particularly, the highest antibacterial activity was observed for the TiO2 deposited nanoparticles. Thanks to the presence of TiO2, the 10 wt% TiO2-SBA-15 nanocomposites display an enhanced antibacterial performance. Hence, the photocatalyst offered an effective universal antibacterial activity [48]. Bacteria possess enzymatic antioxidant protective mechanisms that limit lipid per oxidation as well as the impacts OH2 - and ·OH radicals. When such components are surpassed, a series of redox processes can cause the death of a cell by changing many critical components likes DNA, cell membrane, cell wall, and metabolism pathways. The mechanism of antibacterial performance of TiO2-SBA-15 nanocomposite was presented in Scheme 2.
Antibacterial activity of SBA-15 and 10 wt% TiO2-SBA-15 nanocomposite against Gram-positive and Gram-negative bacteria.

The antibacterial performance-based mechanism.
4. Conclusion
Mesoporous SBA-15 nanomaterial is successfully synthesized using sustainable, low-cost, and environmentally friendly rice husk extract as the silica source and used as the support for TiO2 nanoparticles. With 10 wt% loading of TiO2, the TiO2-SBA-15 nanocomposite showed higher efficiency in the reduction of MB within 15-20 min of irradiation and excellent antibacterial performance. This could be attributed to the excellent light absorption and limited density of surface defects. The degradation process of 5 and 10 wt % TiO2-SBA-15 obeys the first-order kinetic with rate constant of 0.139 min-1 and 0.033 min−1, respectively. The TiO2-SBA-15 nanocomposite also shows excellent recyclability with long lasting catalytic performance. Therefore, TiO2-SBA-15 is a potential excellent photocatalyst for practical application in environmental treatment.
Footnotes
Data Availability
The data used to support the findings of this study are included within the article.
Conflicts of Interest
The authors declare that they have no conflict of interest.
Acknowledgments
The authors extend their appreciation to the Deputyship for Research & Innovation, Ministry of Education in Saudi Arabia for funding this research work through the project number (UB-35-1442).
