Abstract
A novel material named Fe/Mn-C layered double hydroxide composite (Fe/Mn-C-LDH) was synthesized to remove arsenic from an aqueous solution. The removal performance of the composite toward arsenic ions was studied through the batch experiments. The experiment results showed that Fe/Mn-C-LDH exhibited a high adsorption capacity of 46.47 mg/g for As(III) and 37.84 mg/g for As(V) at 318 K, respectively. In addition, the investigation of the release of Fe3+ and Mn2+ in the process of arsenic adsorption revealed that the Fe/Mn-C-LDH exhibited better stability than Fe/Mn-layer double hydroxide (Fe/Mn-LDH) with fewer Mn2+ and Fe3+ releasing under the same condition. The BET results showed that the specific surface area of Fe/Mn-C-LDH decreased after adsorption of As (III) and As (V). Furthermore, the Density Functional Theory (DFT) calculation results proved that the adsorbent combining arsenic by T-site to produce a better adsorption effect for arsenic. Possessing better stability and adsorption capacity, Fe/Mn-C-LDH could potentially serve as a perfect adsorbent for arsenic removal from an aqueous environment. It would provide a promising approach for removing heavy metal from the aquatic environment in the future.
1. Introduction
As one of the highly poisonous and hazardous heavy metal elements in water, arsenic mainly occurred in the forms of As(III) and As(V) in the natural water environment [1–5]. As(V) is dominant in oxidizing conditions and mainly exists in the form of H2AsO4
- and HAsO4
2-, and As(III) principally as H3AsO3 [6]. Typically, As(III) is more difficult to be removed from wastewater or aquatic environment [6]. In order to ensure people’s health unaffected by arsenic, WHO stipulated that the maximum permissible limit of arsenic is 10 μg/L [7–9]. Therefore, it is urgent to develop a facile and highly effective method to remove arsenic from the aquatic environment efficiently. Adsorption, being considered as one of the most prospective approaches for removing arsenic, has the advantages of eco-friendly, high efficiency, and low cost [7, 10]. Many sorbent materials have been developed to scavenge arsenic from an aqueous environment [11]. Natural and synthetic materials in the literature mainly included activated carbon [12], activated alumina [13], biological adsorbents [14], and mineral adsorbents [15]. However, the poor performance in terms of adsorption capability and posttreatment separation have restricted their application. In recent years, the biochar and layered double hydroxides (LDHs) have drawn considerable attention in the scientific community because of their stability, large surface area, porous structure, and functional group [16], which have been widely applied as adsorbents in sewage treatment [17]. The arsenic adsorption capacity of biochar was heavily influenced by functional groups with a positive charge and ions on the surface because arsenic existed in the form of an anion in an aqueous solution [18]. LDH, as a kind of typical clay inorganic material [19, 20], and its special formula could be noted as [M1-x
2+Mx
3+(OH)2]x+(An-)x/n·yH2O, in which M3+ and M2+ are the divalent and trivalent cations, respectively.
Herein, scanning electron microscope (SEM), X-ray diffraction (XRD), Fourier transform infrared spectrophotometer (FTIR), X-ray photoelectron spectroscopy (XPS), and Brunner Emmett teller (BET) were applied to characterize the microstructure of Fe/Mn-C-LDH in this research. The adsorption capacity for arsenic was examined as well as the metal ion concentration in water solution after adsorption. Moreover, Density Functional Theory was applied to reveal the adsorption removal mechanism. All these provide a promising avenue for the application prospect of Fe/Mn-C-LDH to remove arsenic ions from the aqueous environment.
2. Method and Experiment
2.1. Materials
The details of the materials are listed in Text S1.
2.2. Method and Characterization of Fe/Mn-C-LDH
The barks of the bamboo pieces after drying were peeled off and soaked in 5% ammonia water for 6 h at 100°C. Followed by 2 h ultrasonication, the pieces were washed using ultrapure water and dried at 80°C. Afterwards, the pieces were roasted in a muffle furnace for 3 h, crushed, and sifted to obtain bamboo biochar powder. About 50 g bamboo charcoal powder and 250 mL concentrated nitric acid were added into a flask, boiled with stirring in an oil bath at 110°C for 6 h. After washing to neutral and dried, the bamboo charcoal powder was added into a beaker containing ultrapure water and stirred with a magnetic stirrer to obtain carbon/water mixture. The pH was adjusted to 10 with a mixed alkali solution of sodium hydroxide and sodium carbonate. Meanwhile, under violent stirring, the mixed metal chloride (
2.3. Characterization
The details of the characterization of Fe/Mn-C-LDH are presented in Text S2.
2.4. Adsorption Experiment
The adsorption experiment of arsenic by Fe/Mn-C-LDH was performed according to the controlled variable method. For kinetics study, 70 mg adsorbent was added into 50 mL arsenic solutions with a concentration of 5 mg/L, 10 mg/L, and 50 mg/L, respectively. The appropriate adsorption time intervals were regulated from 1 to 1440 min (
The adsorption capacity in the adsorption experiment could be calculated by Eq.(1) [27].
where
The Langmuir (Eq.(2)) and Freundlich (Eq.(3)) models have been used to simulate the adsorption isotherms [8, 28]. Temkin (Eq.(4)) isotherm represents the binding equilibrium between adsorbates and adsorbents [29].
The adsorption kinetics can be elaborated by the pseudo-first-order (Eq. (5)) and pseudo-second-order (Eq. (6)) kinetic models [31] to calculate the adsorption rate,
3. Results and Discussion
3.1. Characterization of Fe/Mn-C-LDH
3.1.1. SEM-EDS
The SEM-EDS images of raw biochar and Fe/Mn-C-LDH are shown in Figure 1. The results obviously suggested that raw biochar had a porous structure [32]. Also, the surface of biochar was covered with many particles, regarded as the flake-structured LDH distinctly observed in Figure 1(b) (Figure S1c).

SEM of C (a) and Fe/Mn-C-LDH (b).

The flake structure and layered structure observed in Figure S1d and Figure S1e were typical LDH morphologies. The EDS spectrum results (Figure S1f) showed that there contain 13.42% carbon in Fe/Mn-C-LDH (the specific content is shown in Table 1). Besides, the ratio of Mn to Fe was close to 2 : 1 in adsorbent, indicating that the Fe/Mn-C-LDH was synthesized successfully. It can be seen from Figure S1g and Figure S1h that after Fe/Mn-C-LDH adsorbs As(III) and As(V), the flake grains are flattened and piled up, resulting in agglomeration compared with that before adsorption.
The content of the element in Fe/Mn-C-LDH.
3.1.2. XRD
The powder XRD was used to characterize the Fe/Mn-C-LDH, with a scanning range of 10~80°. The C has a relatively wide shoulder peak at

XRD patterns of C and Fe/Mn-C-LDH.

3.1.3. XPS
The composition of Fe/Mn-C-LDH was analyzed by XPS before and after arsenic adsorption. The results are presented in Figure 3(a).
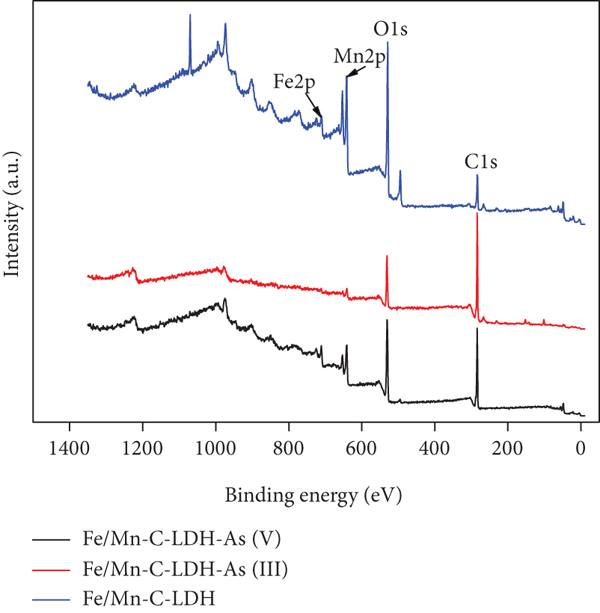
XPS spectra of survey (a), C 1s (b), As 3d (c), and O 1s (d) of Fe/Mn-C-LDH.


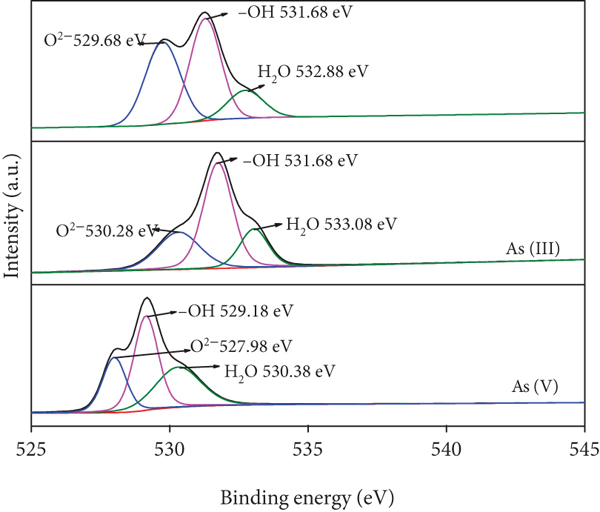
The peaks corresponding to C 1s, O 1s, Fe 2p, Mn 2p, and As 3d emerged obviously in the spectrum which indicated the existence of these elements in Fe/Mn-C-LDH. Typically, the peaks at 284.44 eV, 285.28 eV, and 288.56 eV were marked by the C-C, C-O, and C=O bonds, respectively [34], indicating the bond energy position change slightly after arsenic adsorption. The XPS spectrum of the As(III) after treatment exhibited two peaks at 43.65 eV and 44.58 eV. This shows that part of As(III) was oxidized to As(V) by Fe/Mn-C-LDH in the process of adsorbing As(III) [8]. However, in the process of adsorbing As(V), only one As 3d peak arose at 44.32 eV, which was ascribed to the As(V)-O bonding. There were three forms of O (O2-, -OH and H2O) in Fe/Mn-C-LDH presented in Figure 3(d). The change of bond energy position before and after arsenic adsorption illustrated that coordination exchange could have occurred between arsenate and functional groups of Fe/Mn-C-LDH.
3.1.4. FTIR
The FTIR spectrum of Fe/Mn-C-LDH and biochar before and after arsenic adsorption are presented in Figure 4. For biochar, the band at 3428.95 and 1629.61 cm-1 were emphasized to H-O scaling and bending vibration of interlayer water (δ H-O-H) [19]. The peaks at about 568.92 and 858.21 cm-1 were marked to the Fe-O and Mn-O vibration of Fe/Mn-C-LDH, demonstrating that Fe and Mn had been loaded on biochar; the absorption peaks of Mn-O and Fe-O shifted before and after adsorption, indicating that Mn-O and Fe-O were involved in the adsorption process [30, 35, 36]. The broad absorption band before arsenic adsorption at 1467.62 cm-1 was assigned to the antisymmetric stretching mode of CO3 2- groups [32]. Two peaks emerged at 1043.32 and 1039.49 cm-1 after arsenic adsorption, which was attributed to the asymmetric stretching vibration of the As-O bond [30].

The FTIR of C and Fe/Mn-C-LDH with As adsorption.
3.1.5. BET
Nitrogen adsorption-desorption isotherms at 77 K for Fe/Mn-C-LDH are revealed in Figure 5. According to the IUPAC classification, Fe/Mn-C-LDH followed type IV isotherm with H3 hysteresis loop indicating that Fe/Mn-C-LDH is layered materials. It can be seen from the diagram that the pore size of the material is mainly concentrated in the range of 2~50 nm, indicating that the material is mesoporous.
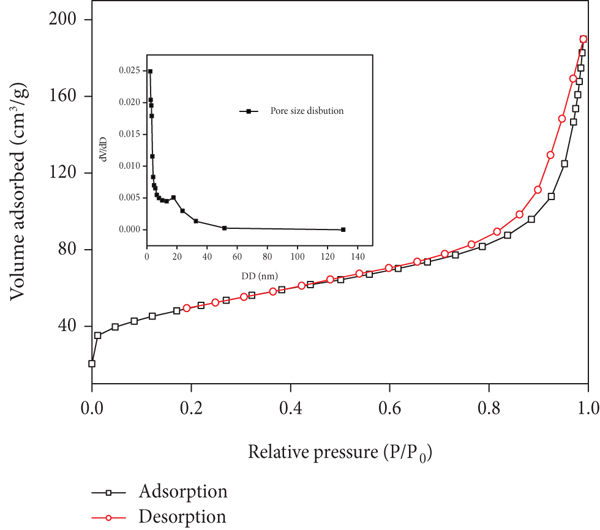
N2 adsorption-desorption curves of Fe/Mn-C-LDH.
According to the data in Table 2, Fe/Mn-C-LDH has a larger specific surface area than Fe/Mn-LDH, which is 170.51 m2/g and 82.61 m2/g, respectively, which proves that the introduction of biochar increases the specific surface area of the adsorbent; after the adsorption of As(III) and As(V), the specific surface area decreased, which shows that Fe/Mn-C-LDH is an adsorbent with excellent performance.
BET surface and pore volume of Fe/Mn-LDH before and after adsorption.
3.2. Effects
3.2.1. Initial Solution pH Value
The effect of pH on the removal of arsenic from aqueous solution using Fe/Mn-C-LDH is explained in Figure 6. The consequences illustrated that the effectiveness of As(III) removal was almost constant under acidic and neutral pH condition. Under the condition of

Influence of solution pH on As adsorption capacities on Fe/Mn-C-LDH.

3.2.2. Leaching of Metal Ions
The concentration of Fe3+ and Mn2+ in the supernatant of As(III) and As(V) adsorbed by Fe/Mn-C-LDH and Fe/Mn-LDH was determined through ICP-OES. The chemical stability of Fe/Mn-C-LDH was investigated by detecting the leaching concentration of Fe3+ and Mn2+ in samples at different pH value from 2 to 11. The results presented that there was no Fe3+ detected in the supernatant, and the concentration of Mn2+ is shown in Figure 7. Fe/Mn-C-LDH was stable when the initial pH was greater than 3 (Figure 7). In the process of experiment, the optimal dosage of Fe/Mn-LDH adsorbent was 0.03 g, while Fe/Mn-C-LDH was 0.07 g. Although the optimal dosage of the Fe/Mn-C-LDH was larger than Fe/Mn-LDH, the leaching of Mn2+ from Fe/Mn-C-LDH was less serious than the latter. This result indicated that the stability of Fe/Mn-C-LDH had been distinctly improved compared with Fe/Mn-LDH, mainly because the introduction of alkaline biochar would be served as an effective barrier to prevent/buffer the impact of H+ toward LDH nanoparticles [40].
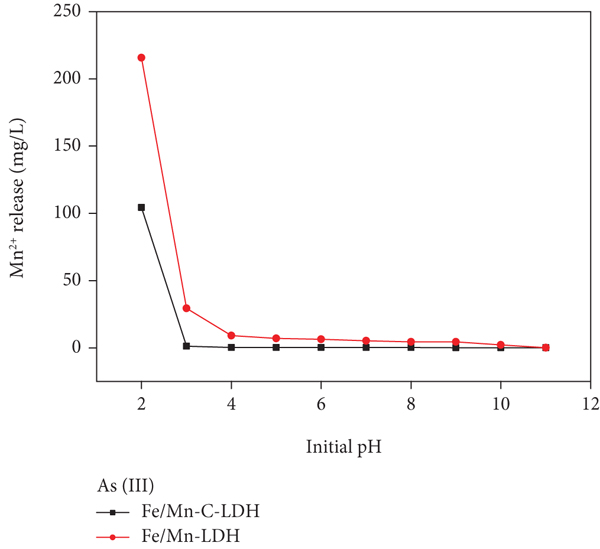
Mn2+ concentration in solution at different initial pH.

3.3. Adsorption Isotherms and Kinetics
3.3.1. Adsorption Isotherms
The adsorption isotherms of As(III) and As(V) on Fe/Mn-C-LDH at 298 K, 308 K, and 318 K are presented in Figure 8. The adsorption capacity of arsenic on Fe/Mn-C-LDH increased gradually with the initial arsenic concentration increment. The increase of solution diffusion and the decrease of viscosity were the reason of this tendency [41].

Adsorption isotherm for arsenic on Fe/Mn-C-LDH.
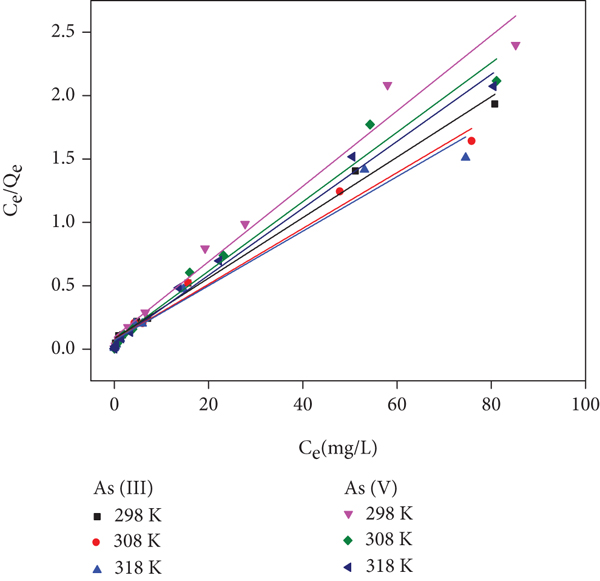
The related parameters of Langmuir (Figure 8(b)), Freundlich (Figure S8c), and Temkin (Figure S8d) isotherm models are shown in Table 3. The correlation coefficients (
Langmuir, Freundlich, and Temkin constants for arsenic on Fe/Mn-C-LDH.
The thermodynamic parameters of As(III) and As(V) adsorption were estimated by Gibbs free energy equation and linearized Van’t Hoff equation, which are calculated as follows:
Thermodynamic parameters of arsenic adsorption by Fe/Mn-C-LDH.
3.3.2. Adsorption Kinetics
The arsenic adsorption onto Fe/Mn-C-LDH at different time was performed (Figure 9(a)).
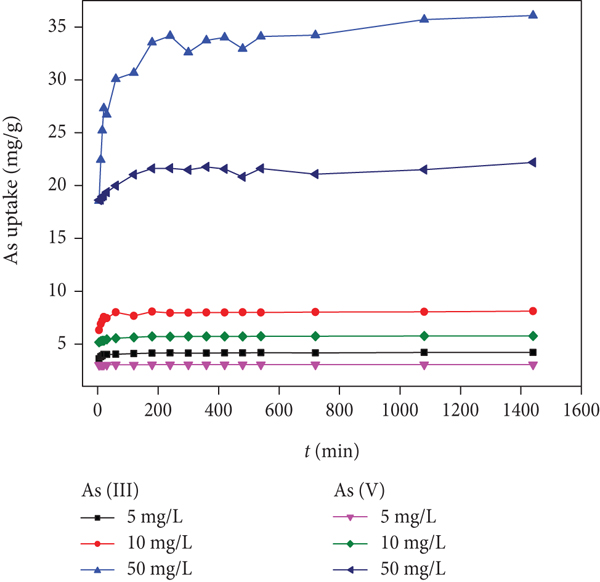
Kinetics of arsenic adsorption by Fe/Mn-C-LDH (a). Fitted adsorption isotherms (b).

The uptake of arsenic was a quick process at initial stage and then reached a plateau at 2 h. The loading capacity of As(III) was greater than As(V) under the same conditions; this is because in the solution, As(III) was effectively adsorbed to form an inner complex on the surface of the adsorbent, As(III) existed mainly as a neutral molecule (H3AsO3), which could exchange ligands with the plentiful hydroxyl groups on the surface of the metal laminates, and then As(III) was oxidized to As (V), but for As(V) adsorption, not the oxidized process. Comparing the experimental data with calculated values (Table 5), pseudo-second-order kinetic model with good correlation coefficient (
The pseudo-first-order and pseudo-second-order models for arsenic on Fe/Mn-C-LDH.
3.4. Comparison with Literature Data
Table 6 presents a list of different adsorbents that have been used to remove As(III) and As(V) from aqueous solutions. The comparison among the materials shows that Fe/Mn-C-LDH presents a favourable property. In addition, in the application of removing arsenic pollution in aqueous solution, Fe/Mn-C-LDH can be used as an efficient adsorbent for arsenic removal.
Comparison among different adsorbent materials for removal As(III) and As(V) from aqueous solutions.
3.5. DFT Calculation
The adsorption mechanism of arsenic on the Fe/Mn-C-LDH composite was theoretically predicated using DFT method. As shown in Figure 10, the parameters of the unit cell in the model were

Two density functional theory models of the materials.
Structure parameters in the models.
DFT functional calculations were performed with VASP (5.5.4). The binding energy was calculated by the following formulation:
Binding energy of the interaction between LDH and arsenic.
In the DFT structure of Fe/Mn-LDH and Fe/Mn-C-LDH, the optimized structure of As ion on T-site (directly above LDH) was observed [50]. As shown in Figure 10, the presence of Na+ has been observed in the structure of both materials; this is because of the ordered positioning of the cations in the layers and the local positive charge distribution; this mismatch of the spatial lead to a net negative charge, which needs to be compensated for by the intercalation of Na+ [51]. The T-site
4. Conclusions
In summary, Fe/Mn-C-LDH has been synthesized and applied to scavenge arsenic in aqueous environment. The pseudo-second-order kinetic model and Langmuir isotherm model can well explain the adsorption processes of arsenic. The adsorption performance of Fe/Mn-C-LDH was prominent at a comparatively wide pH range for both As(III) and As(V). There was no serious Mn2+ release from Fe/Mn-C-LDH in the pH range from 3 to 11, lower than the standard limit of GB 18918-2002 (2 mg/L). According to the calculation of Gibbs free energy and Langmuir model, the adsorption processes of arsenic were confirmed to be monolayer adsorption and exothermic reaction. XPS and FTIR researches attested that the exchange of functional groups and arsenic played an essential role in arsenic removal. The specific surface area of Fe/Mn-LDH is 170.508 m2/g far more than Fe/Mn-LDH, which proves that the introduction of biochar increases the specific surface area of the adsorbent. DFT results indicated that the adsorbate coordinated with the adsorbent by T-site. Our results demonstrated that the highly stable Fe/Mn-C-LDH might be a promising candidate for arsenic abatement.
Footnotes
Data Availability
The raw data used to support the findings of this study are available from the corresponding author upon request.
Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Nos. 21707024, 41763012, and 2019YFC0507502), the Guangxi Science and Technology Planning Project (Nos. GuiKe-AD18126018, GuiKeAA 17204047), the Guangxi Natural Science Foundation (2018GXNSFAA050044), Chinese Postdoctoral Science Foundation (No. 2019M650869), and Innovation Technical Innovation Center of Mine Geological Environmental Restoration Engineering in Southern Karst Area.
