Abstract
Based on the increasingly serious formaldehyde pollution, effective degradation of formaldehyde has become a practical problem that humans urgently need to solve. Among many treatment methods, activated carbon has the advantages of large specific surface area, high adsorption efficiency, and uniform pore size distribution. As a kind of clean photocatalytic material for formaldehyde degradation, titanium dioxide supported by activated carbon has become a research hotspot to develop adsorption-catalytic materials for formaldehyde degradation. In this paper, the research progress of activated carbon and its modification, the photocatalytic principle and modification of titanium dioxide, and TiO2/AC materials are reviewed. The results show that the pore size distribution gradient and acidic oxygen-containing functional groups of activated carbon play key roles in the formaldehyde adsorption process. TiO2 doped with metal ions and nonmetal ions can significantly improve the photocatalytic activity. The TiO2/AC material can greatly improve the photocatalytic rate and achieve the technical goal of efficient and clean degradation for formaldehyde.
1. Introduction
Relevant research shows that, with the rapid development of economic society, decoration materials are emerging one after another, and most of these materials (such as man-made panels and glue paint) contain pollutants such as formaldehyde [1, 2] which is also known as ant aldehyde, chemical formula HCHO, molecular weight 30.03, formaldehyde gas relative density 1.067, liquid density 0.815 g/cm3 (-20°C), melting point -92°C, and boiling point -19.5°C. In addition to decoration materials, the bedding (such as bedclothes and pillowcases), wardrobes, carpets, and air fresheners we use every day also contain more or less [3] formaldehyde which has a long release period. Indoor air pollution [4, 5] will occur when the cumulative amount of release exceeds a certain threshold. People spend 80% of their time indoors on average every day [6], so the emission of indoor air pollutants (such as formaldehyde, benzene, and radon) will seriously endanger people’s health [7].
Low concentrations of formaldehyde which is recognized as allergen source [8] can cause diseases such as eye redness, tracheitis, and asthma, and high concentrations can induce cerebral palsy [9]. Formaldehyde has been classified as a Category 1 carcinogen by international organizations (Word Health Organization and American Red Cross). In addition, formaldehyde can also cause varying degrees of harm to the nervous system, respiratory system, and immune system of children, the elderly, and pregnant women [10]. Formaldehyde has an inhibitory effect on the immune system and causes decreased neutrophil respiratory burst activity. Formaldehyde inhalation can affect the electropsychological function of the heart, resulting in bradycardia, abnormal or missing P waves, and changes in R-T interval. Apart from health effects, formaldehyde can also reduce people’s work efficiency [11].
James et al. and Amesj et al. [12, 13] measured that the emission factors of formaldehyde in rapeseed oil, soybean oil, and fried vegetables were 20.1 mg/kg vegetables, 18.6 mg/kg vegetables, and 12.4 mg/kg vegetables, respectively. In 2018, Zhao et al. [14] explored the emission rates of formaldehyde in the kitchen in the actual cooking process by using the mass balance equation of gaseous pollutants with natural gas as fuel. The research results showed that the emission rates of formaldehyde in the kitchen during the actual cooking process were
Summary of formaldehyde emission parameters for synthetic rubbers [15].
In the table,
In recent years, researchers have been working on new ways to remove formaldehyde. The removal method of formaldehyde pollution can be broadly divided into two kinds, one is the source of governance that does not use decoration materials or supplies which contain formaldehyde; the method is more convenient and does not need to put a specific material or process in indoor, but the technical means for the production of decoration materials without formaldehyde are limited, so it is not realistic to remove formaldehyde from the source. The second is posttreatment, such as ventilation method, plant absorption method, plasma purification method, adsorbent adsorption method, and photocatalyst degradation method. Due to the long release cycle of formaldehyde and being easily affected by the season, it is not applicable to take ventilation to remove formaldehyde in some cold areas. Green plants such as cactus, Chlorophytum, and aloe [16] can remove low concentration of formaldehyde; however, its normal growth will be affected or even withered in high concentration of indoor, so it has little effect on absorption of formaldehyde. The low-temperature plasma method, which refers to the use of ions to undergo a complex reaction with the molecular structure of formaldehyde to catalyze the degradation of formaldehyde, is an emerging method for removing formaldehyde. Studies [17] have shown that this method has the advantages of fast and efficient and can completely degrade formaldehyde into carbon dioxide and water, but it will produce O3 and CO and other substances, and the consumption of technology and economy is large, waiting for be further developed. The adsorption method uses the pore size distribution and surface functional groups of activated carbon fiber (ACF) [18], activated carbon (AC) [19], and other porous adsorbents to deal with pollutants such as formaldehyde, which has the advantages of easy material availability and convenient operation. Common adsorbents include alumina and zeolite molecular sieves. Photocatalytic degradation technology uses light of a certain wavelength to irradiate photocatalyst materials to produce active species, which participate in the reaction to degrade formaldehyde into carbon dioxide and water, and so produces the effect of air purification [20].
In this paper, the adsorption mechanism and modification of activated carbon, the structure of photocatalyst TiO2, the principle of photocatalytic reaction, and the load and modification treatment were reviewed, and the adsorption and degradation properties of activated carbon and TiO2/AC for formaldehyde were analyzed and compared, providing scientific guidance for the degradation of formaldehyde.
2. Activated Carbon and Its Modification
2.1. Adsorption of Formaldehyde over Activated Carbon
Activated carbon has the characteristics of large specific surface area, fast adsorption rate, and moderate pore size distribution, which has significant advantages [18] in adsorption and separation of formaldehyde pollutants. According to different adsorption methods, activated carbon adsorption is divided into physical adsorption and chemical adsorption. In the physical adsorption process, the pore size gradient distribution determines the adsorption capacity and rate. The large and medium pores play the role of transporting formaldehyde molecules, and the micropores have a huge specific surface area, which can provide enough places for pollutants to stay. The adsorption of formaldehyde over activated carbon is mainly realized by electrostatic force, covalent bond dispersion force, induction force,
Tang et al. [23] studied the influence of activated carbon micropores and mesopores on the adsorption performance of formaldehyde and found that the adsorption effect of activated carbon is positively correlated with the proportion of micropores; when the specific surface area of the mesopores increases, the time to reach adsorption equilibrium is shortened. Song [24] studied the adsorption of organic pollutants on activated carbon from the perspective of pore size distribution. He proposed that a better pore gradient distribution accelerated the internal diffusion of pollutants in the pore size of activated carbon and improved the adsorption capacity.
It can be seen from the research of Song that although activated carbon micropores can provide a huge specific surface area, transport pores such as macropores and mesopores are also required. The pore size gradient affects the adsorption capacity and rate.
In addition to exploring the effect of pore size distribution of activated carbon on adsorption performance, Lin et al. [25] studied the influence of surface functional groups on adsorption performance of coconut shell and woody activated carbon through dynamic penetration experiment. The experimental results showed that the coconut shell carbon had the highest adsorption performance for formaldehyde, and they believed that the coconut shell carbon had a higher content of phenolic hydroxyl and nitrogenous functional groups on its surface. Kowalczyk et al. [26] studied the reaction mechanism between functional groups on the surface of activated carbon and formaldehyde. Formaldehyde can not only form hydrogen bonds with functional groups but also oxidize with carboxyl and phenolic hydroxyl groups in pore size.
It can be seen from the previous studies that the content of functional groups on the surface of activated carbon determines the degree of chemical adsorption and oxidation reaction, the large specific surface area of micropores provides sufficient physical adsorption sites for pollutants, and the pore size distribution affects the internal diffusion ability of formaldehyde molecules. However, activated carbon is mainly dominated by physical adsorption, which is unstable and desorbs under certain pressure and temperature conditions, causing secondary pollution. Through the redox method, the number and type of chemical functional groups on the surface of activated carbon are changed, and the physical adsorption is transformed into physical-chemical synergy adsorption, which can effectively improve the adsorption of formaldehyde molecules on the surface of activated carbon.
2.2. Activated Carbon Modification
The increase of acid oxygen-containing functional groups on the surface of activated carbon can provide more chemisorption sites for formaldehyde which is polar molecule. The acidic functional groups can attract the free
He et al. [27] studied the effect of activated carbon modified by KMnO4 on the adsorption of formaldehyde through experiments. When the concentration of KMnO4 was 2%, activated carbon had the best adsorption performance for formaldehyde pollutants, and they believed that this phenomenon was attributed to the increased content of C=O and C-OH. In order to further explore the dual influence of KMnO4 concentration and heat treatment temperature on the modification of activated carbon, Jiang et al. [28] treated activated carbon with different KMnO4 concentrations and thermal temperatures, respectively, to explore the influence of modification on the formaldehyde adsorption performance of activated carbon samples. The study found that when the heat treatment temperature is 65°C and impregnation concentration is 0.08 mol/L, the adsorption capacity of formaldehyde molecule on modified activated carbon is the highest. However, when the concentration of KMnO4 is high, the pore size of activated carbon is blocked, and the adsorption capacity decreases.
From the research of Jiang and others, it can be seen that activated carbon is impregnated with higher concentration of KMnO4, and the specific surface and adsorption capacity are reduced to a certain extent, but after the reduction of KMnO4,
The polar adsorbents can be stably adsorbed on the surface of acidic modified activated carbon because of the increase of functional group content. Liu et al. [29] explored the formaldehyde adsorption performance of activated carbon modified by phosphoric acid and activated by nitrogen. The experiment showed that the formaldehyde adsorption capacity of modified activated carbon was 4.78 mg/g when the phosphoric acid mass fraction was 40%, and the nitrogen activation temperature was 550°C. Liu et al. [30] modified activated carbon with H2SO4, formaldehyde saturation adsorption capacity increased by nearly 50%, and desorption peak area and peak height increased to a certain extent. It can be seen from Liu et al.’s experiment that the formaldehyde adsorption capacity of the acidified activated carbon increases, which may be attributed to the synergistic effect of physical adsorption and chemical adsorption.
In addition to oxidation modification, surface physical modification of activated carbon is mainly to increase specific surface area and adjust pore size, and its distribution by physical and chemical means to achieve the purpose of changing surface physical structure of activated carbon. The surface physical modification of activated carbon is firstly modified by adding some activator in the preparation process of activated carbon, and secondly, it can be physically modified by microwave radiation technology instead of traditional heating technology, so as to improve its adsorption and catalytic performance [31, 32]. Microwave modification mainly uses microwave power, radiation time, and radiation temperature to modalize the surface structure and functional group content of activated carbon, which usually interacts with N2 and O2 to change the surface functional group content, so as to achieve the effect of improving the adsorption and catalytic performance of activated carbon. Jones et al. [33] conducted microwave radiation on activated carbon in O2 atmosphere and found that the surface carboxyl of oxygen-containing functional groups increased significantly, and the pore size increased significantly at higher power. Plasma modification technology is a kind of material surface modification technology developed rapidly in recent years. This technology which can produce large amounts of charged particles, excited state particles, photons, free radicals, and so on plasma in the gas medium (O2, N2, NH3, and CF4), using these high-energy plasma impacts the material surface to make the material surface physical and chemical properties change, with little damage to the material surface characteristics at the same time, so as to improve the specific surface area, pore size, pore volume, surface functional groups, and other related properties of the technology. Li et al. [34] used low-pressure nitrogen plasma to modify the surface of activated carbon, and the surface oxygen-containing acidic groups decreased with the increase of plasma power, while the nitrogen-containing basic functional groups increased accordingly.
Although the surface modification technology of activated carbon has made progress in many aspects, low cost, high performance, and simple environmental protection process are still the development direction of activated carbon modification in the future. Through the modification of activated carbon, its application has been involved in many fields, but most of it is still applied to simple adsorption process, lack of functional high-quality special activated carbon, especially as a catalyst, and catalyst carrier activated carbon. Optimizing the preparation process of activated carbon, developing activated carbon with high specific surface area and large pore volume, exploring the optimal adsorption conditions of activated carbon, reducing the consumption of resources, and improving the economic feasibility are also important research directions in the future.
2.2.1. Comment 6
Besides the activated carbon adsorption method, the photocatalytic degradation for formaldehyde by titanium dioxide is nontoxic and harmless, which has aroused the research of scholars at home and abroad.
3. Mechanism of Photocatalytic Degradation
According to the formula [35]
Common semiconductors and band gap widths.
Nano-TiO2 is a semiconductor with a discontinuous energy band structure [38] formed by the hybridization of Ti3d and O2p orbitals, which is composed of valence band (VB), condition band (CB), and energy gap (Eg).
German physicist Hertz discovered that photoelectrons will escape from the material only when Some of part electrons were captured by the defect site of titanium dioxide Some of the excited photoelectrons are captured by the hole, and energy is released in the form of heat, which reduces the separation efficiency of electrons and holes, thus affecting the photocatalytic efficiency.

Energy band theory schematic.
The photocatalytic degradation mechanism is shown in Figure 2 [43]. Photogenerated carriers migrate to different positions of the semiconductor under the action of electric field force, combine with O2, H2O, or OH- on the surface of TiO2 and react as follows, producing two active species -OH and -O2
-. These substances have strong oxidation capacity, equivalent to the high temperature of 3600 K. Pollutants such as formaldehyde can be catalyzed into carbon dioxide, water, or other inorganic small molecules.
The formation of electrons ( The formation mechanism of -O2
-
The formation mechanism of -OH Adsorb water molecules in the air to form Formed with the aid of the intermediate

Photocatalytic mechanism of TiO2.
The formation of
The formation of -OH is as follows:
Formed by reaction with OH-: Hole-electron pair recombination:
Yang [44] studied the intermediate species and reaction mechanism in the oxidation process of formaldehyde by physical methods such as TPD and ESR. He proposed that the reaction process of formaldehyde being oxidized into CO2 and H2O was as follows:
In addition to exploring the photocatalytic reaction mechanism of formaldehyde in experiments, Xie et al. [45] studied the connection relationship between reactants on the reaction potential energy plane and intermediate species at the level of 6-311++G(2df,pd) by using the B3LYP method and MP2 method of the density functional theory. Simulation shows that the intermediate is the isomer form of formic acid (HCOOH) (HOCOH).
According to the study of reaction mechanism by Yang et al.,
Because
The charge transport capacity is also an important factor affecting the photocatalytic efficiency [49]. Hoffmann et al. [50] studied the UV excitation process of TiO2 particles and other related issues. The photocatalytic reaction process and its characteristic time are shown in Table 3. The characteristic time of interfacial charge transfer is shorter than that of carrier generation, capture, and recombination. The characteristic time of interfacial charge transfer is shorter than that of carrier generation, capture, and recombination. Huang [51] believed that the recombination of photogenerated electrons and holes was much faster than charge transfer, which greatly reduced the number of photogenerated electrons and holes involved in the reaction. Therefore, effectively reducing the recombination of electrons and holes was also an important research content in the field of photocatalysis.
Primary processes and characteristic time domains for photocatalysis on TiO2 semiconductor.
4. Properties and Structure of TiO2
The three crystal structures of nano-TiO2 can be divided into rutile phase, anatase and brookite. The similarity between them is that they have the same basic components which is the TiO6 octahedron structure [52]. However, the crystal structure, arrangement, connection mode, distortion degree, band gap, and defect types are different.
Anatase TiO2 is octahedral common edge connection, while the rutile type is common edge and common fixed point connection, as shown in Figures 3 and 4 [53]. Rutile TiO2 is a stable crystal with fewer oxygen vacancies [54]. Anatase TiO2 has more dislocation and defects, so its distortion degree is higher than that of rutile phase, so anatase TiO2 has excellent catalytic performance [55]. The forbidden width of anatase TiO2 is about 3.2 eV, and that of rutile TiO2 is about 3.0 eV [56]. The structural parameters of anatase, titanite, and rutile TiO2 are shown in Table 4 [57], and it can be seen that their space groups, crystal cell parameters, and Ti-O bond lengths are different.

Simulations of anatase and rutile nanosized titanium dioxide. (a) Anatase type. (b) Rutile type.


A schematic diagram of nanosized titanium dioxide structure connection. (a) Coedge way. (b) Concurrent way.
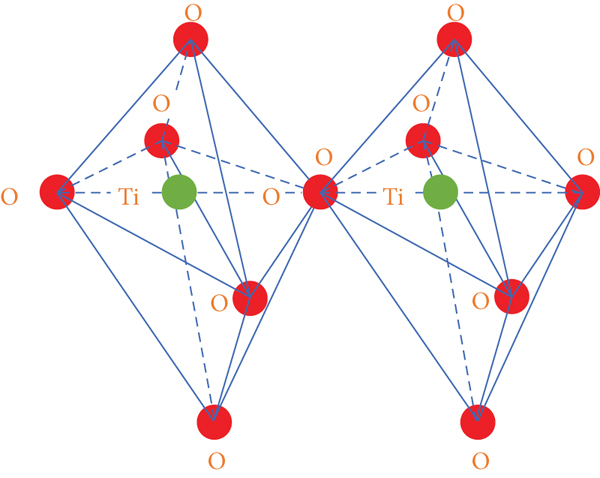
Crystallographic data of different TiO2 polymorphs.
Under the calcination condition of about 600°C, anatase transforms into rutile phase through continuous bond breaking and atomic rearrangement, and the phase transition temperature is affected by the size of titanium dioxide particles and ion doping, etc. [58]. The mixed phase of anatase, plate titanium, or rutile has good catalytic performance. For example, P25 (
5. Modification of TiO2
Zhu et al. [60] explored the influence of TiO2 irradiated with different wavelengths of light on the degradation effect of formaldehyde, and the study showed that the degradation rate of formaldehyde was about 29% without UV irradiation. Titanium dioxide which is a photocatalytic material has the advantages of safety, high efficiency, energy saving, good treatment effect, and no secondary pollution, so it can degrade formaldehyde and other toxic gases or liquids. However, titanium dioxide has the following disadvantages, so it has not been applied on a large scale in industry.
The disadvantages of nanoscale titanium dioxide in the field of photocatalysis are as follows [61]:
The dispersion of nanosized titanium dioxide is poor, which is prone to agglomeration. Therefore, the specific surface area and the contact area with formaldehyde molecules are reduced, affecting the photocatalytic performance The lifetime of electrons and holes is short, and the holes and electrons need to be continuously generated during the reaction process, so that they migrate to different positions in the semiconductor and participate in the reaction to produce active species TiO2 has a low utilization rate of light.
In summary, titanium dioxide has disadvantages of small particle size, poor dispersion, high electron-hole pair recombination rate, and low utilization rate of sunlight. In order to improve its catalytic performance, researchers have conducted a lot of exploration.
5.1. Deposition of Noble Metal
Zhu et al. [62] studied the effect of Au/TiO2 photocatalytic removal for gaseous formaldehyde, and the results showed that when the relative humidity and blue light intensity were 13% and 38.5 MW/cm2, respectively, the degradation rate of formaldehyde under light reaction was 77%, much higher than the conversion rate under dark reaction.
The principle of action on TiO2 modified by noble metal [63] is as follows:
Since the Fermi level of TiO2 is different from noble metals, a noble metal-TiO2 heterojunction can be generated when noble metals are deposited on TiO2. After the photogenerated electrons are excited by light, they will transfer from the surface of TiO2 to the surface of noble metals. A space charge layer will be formed when the Fermi level reaches the same level. The unbalanced charge distribution causes band bending to form the Schottky barrier, as shown in Figure 5 [64]. The excited electrons in the valence band can be captured by the Schottky barrier Lattice distortion is formed when noble metals are doped into the TiO2 crystal lattices, which hinders the recombination of electron-hole pairs to some extent and improves the photocatalytic performance of nano-TiO2.

The schematic Schottky barrier. In the figure, Ef represents the Fermi level, VB represents valence band, CB stands for conduction band,
When the noble metal is deposited on the surface of TiO2, it has little shielding effect on TiO2 and does not affect the specific surface area of TiO2. Studies have shown that when 10% Pt is deposited on the surface of titanium dioxide, the effect on nanosized titanium dioxide is only 6%. The recombination rate of electron-hole pairs will be increased if the noble metal is deposited in a large amount of titanium dioxide. According to the study [65–68], the noble metals deposited on the surface of TiO2 are mainly Pt and Ag. Huang et al. [69] found that Pd/TiO2 with 0.1% load of Pd could completely degrade formaldehyde into CO2 and H2O at room temperature.
5.2. Surface Photosensitization
Surface photosensitization [70] is a relatively simple method, and it is one of the effective ways to broaden the absorption spectrum of TiO2 to the visible region. There are two kinds of dye sensitization and compound sensitization.
Dye sensitization refers to the adsorption of organic dyes with an excited state potential more negative than TiO2 on the surface of TiO2 by physical or chemical means. After light irradiation, photogenerated electrons are generated and transferred to different positions and reacted with oxygen on the surface of TiO2. The mechanism of action is shown in Figure 6 [71]. Studies have shown that the photocatalytic activity of TiO2 modified by photosensitizer can be improved, and the utilization efficiency of photons can be improved by 30-80% [72]. Photosensitizers with certain stability include rosin, chlorophyll, eosin B, eosin thioneine, nori, etc. [73].

Mechanism of dye-sensitized titanium dioxide photocatalytic reaction.
Compound sensitization uses the combination of a semiconductor with a small forbidden band width and a titanium dioxide material to form a heterojunction. According to the working principle of the heterojunction, the electrons in the valence band of the TiO2 composite semiconductor are excited and transported to the TiO2 conduction band, as shown in Figure 7 [74]. Generally, a semiconductor with a similar band structure and a narrow band gap is chosen to composite with titanium dioxide. For example, CdS-TiO2, SnO2-TiO2, Fe2O3-TiO2, WO3-TiO2, and other semiconductor composites have been applied. The synergistic effect between the matched band structure of two components can broaden the spectral response range of the composite catalyst and improve the life of photocarriers to a certain extent. The construction of heterojunction is a new research direction in the field of photocatalysis [75–78]. The band diagram of CdS-TiO2 composite system is shown in Figure 8 [79]. The band gap of CdS is about 2.4 eV. CdS is excited first under illumination, and the photogenerated electrons are transported to the TiO2 conduction band to achieve photogenerated carrier separation [80]. Studies have shown [81] that CdS-TiO2 is usually used in experiments due to its good composite effect.

Schematic energy level diagram of TiO2 and CH3NH3Pb I3.
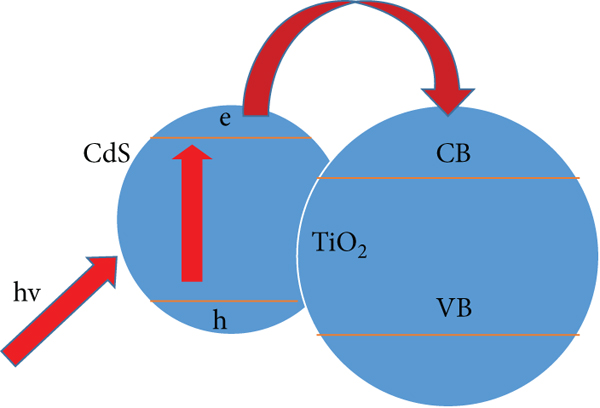
Energy band diagram of the CdS-TiO2 composite system.
5.3. Metal Ion Doping
The results [82] showed that the absorption spectra of nano-TiO2 doped with Fe3+ and Mn2+ could produce red shift and improve its photocatalytic activity. This discovery quickly led to the study of metal ion modification of pure titanium dioxide. Li [83] prepared Ag-doped TiO2 to degrade formaldehyde and found that when the dosage of Ag/TiO2 was 40 g, the degradation rate of formaldehyde could reach more than 93%. Kubacka et al. [84] studied the catalytic degradation of toluene by TiO2 modified by cations (such as V, Mo, and W) and found that photocatalytic activity of TiO2 modified by V and W is improved. Frindell et al. [85] studied the influence of doping with rare earth elements on TiO2 and found that the spectral response range of TiO2 was effectively broadened after modification by rare earth elements. Li et al. [86] found that TiO2 modified by Au/Au3+ has a good photocatalytic ability to degrade methylene blue, because the doping of Au3+ can broaden the spectral response range. Researchers [87–89] found that some metal ions such as 19 kinds of Mo5+, Fe3+, etc. can improve the photocatalytic activity of TiO2, while some were on the contrary, such as CO3+ and Al3+, which would accelerate the recombination of electrons and holes to a certain extent and reduce its photocatalytic activity. Huang [90] studied the modification of titanium dioxide by doping La and Ce, and the results showed that when the doping amount of La and Ce was 1.5 wt% and 0.5 wt%, respectively, the transformation of TiO2 from anatase to rutile phase could be greatly inhibited. Liu [91] studied the influence of doping amount of Fe3+ on formaldehyde degradation performance of TiO2, and the results showed that when doped with 0.5% Fe3+, formaldehyde degradation rate was up to 92.5%.
The action principle of TiO2 doped by metal ion is as follows [92]:
The position of Ti4+ is replaced by metal ions, causing lattice distortion. TiO2 produces more defects to capture free electrons, which improves the photocatalytic activity to a certain extent Metal ions are doped into TiO2, and they can be used as free electron acceptors to capture them, which hinders the recombination of carriers After doping with metal ions, TiO2 forms an impurity level [93], which reduces the band gap width to a certain extent and widens the spectral response range.
The following conditions which can improve the photocatalytic activity of TiO2 doped by metal ion should be met:
The shallow trapping potential wells for electrons and holes are formed in TiO2 which is doped with metal ions. For example, the energy level of Fe3+ is close to the valence band and conduction band of TiO2 [94], so TiO2 doped by Fe3+ forms a shallow trapping potential well, effectively capturing electrons and holes [95, 96]. In recent years, researchers have achieved outstanding results by doping [97, 98] titanium dioxide with rare earth elements. The reason is that the 5d vacant orbit of rare earth elements provides a place for electron migration Doping with metal ions similar to Ti4+ ion radii (74.5 pm) (for example, Fe3+, Co3+, Ni3+, and Cr3+ ion radii are 69.0 pm, 68.5 pm, 70.7 pm, and 75.5 pm [99], respectively). Because metal ions with similar ionic radius are more likely to replace Ti4+, the band structure of TiO2 is changed, the shallow trapping potential well is formed, and the photocatalytic activity is improved. There is a parabolic relationship between the concentration of doped metal ions and the photocatalytic performance [100, 101], so the photocatalytic activity can be improved only by doping metal ions with appropriate concentration.
5.4. Nonmetallic Ion Doping
Studies [102, 103] have shown that N-doped nano-TiO2 can broaden its spectral response range, which has aroused the attention of scholars at home and abroad to nonmetal ion doped TiO2. Li [104] believed that the formaldehyde degradation rate of
The action principle of TiO2 doped by nonmetallic ion is as follows:
When the oxygen position of TiO2 is replaced by nonmetallic anions, which can change the distortion of the TiO2 structure and reduce the recombination of electron-hole pairs to a certain extent, more electrons and holes can participate in the reaction [110] The hybridization of O orbital with nonmetallic ions (such as N3- and C4-) causes the conduction band of TiO2 to shift down, which reduces the band gap width of TiO2 and widens the spectral response range Nonmetallic anions enter the titanium dioxide lattice to replace the interstitial sites, forming a deep impurity level. On the one hand, the spectral response range is broadened; on the other hand, at the deep impurity level, electrons and holes will recombine. It has been proven that it is very important to solve this competitive relationship for improving the photocatalytic activity. N3-, P3-, C4-, and S2- are common nonmetallic anions which doped into titanium dioxide, among which N3- [111] has the best doping effect.
In summary, the main methods to broaden the spectral response range of nano-TiO2 and improve its photocatalytic performance include noble metal deposition, surface photosensitization, metal ion doping, nonmetal ion doping, etc.
6. TiO2 Loaded on Activated Carbon
Studies at home and abroad have found that nano-TiO2 supported on the carrier (such as silicon dioxide and activated carbon) to prepare TiO2/AC composite material can effectively solve the problem of difficult recovery of titanium dioxide powder and increase the photocatalytic activity of TiO2. Activated carbon can increase the local concentration around the catalyst, increase the contact area between pollutants and titanium dioxide, and completely degrade the intermediate products [112].
Huang et al. [113] studied the layer-by-layer deposition of photocatalyst TiO2 and activated carbon on electrospun fibers by electrospray and obtained the NF-P/C multilayer composite film. It was found that the reason why the composite film has a larger contact area with air pollutants is because the electrospun nanofibers with a large specific surface area serve as the carrier of the photocatalyst. At the same time, it is more convenient to access and prevent the powder from scattering. Wu [114] prepared ACF/photocatalyst-free composite materials, which combined adsorption and catalysis to accelerate the degradation of indoor pollutants such as formaldehyde. Sun et al. [115] studied the degradation effect of TiO2/ACF adsorption catalyst materials on formaldehyde, and the results showed that the degradation rate of synergistic materials to formaldehyde can reach 94.06%, and the adsorption capacity of activated carbon for formaldehyde is 60 mg/g, which is much higher than that of the single activated carbon.
From the study on the degradation of formaldehyde by the adsorption-catalytic synergistic material prepared by Wu et al., it can be seen that the degradation rate of HCHO can be greatly increased after the adsorption-mass transfer-degradation process. While the supporting body provides a reaction environment for the catalyst, the AC can be regenerated in situ.
Chen et al. [116] explored the effect of the pore structure and specific surface area of activated carbon on the catalytic performance of the adsorption-catalytic synergistic material, and the results showed that the photocatalytic activity of the synergistic material increased with the increase of the average pore size and specific surface area of the activated carbon. At the same time, Lu et al. [117] used the sol-gel method to support activated carbon with different pore sizes and specific surface areas on titanium dioxide to degrade toluene, the degradation rate of titanium dioxide supported by activated carbon with large specific surface area could reach 97%, and the deactivation time of catalyst was significantly delayed.
It can be seen from the research of Chen and others that the specific surface area and pore size of activated carbon have a significant impact on the catalytic degradation performance of the adsorption-catalytic synergistic material. Activated carbon with mesopores and macropores supported titanium dioxide can significantly improve the photocatalytic performance.
Luo et al. [118] studied the dispersibility of TiO2 in synergistic materials, and they found that titanium dioxide has excellent dispersibility, with an average particle size of about 40 nm, and the catalyst active sites are evenly distributed, so the synergistic performance is significantly improved. Wang [119] studied the dispersibility of titanium dioxide on the surface of TiO2/AC materials through an electron microscopy. They found that titanium dioxide is more uniformly supported on the surface of activated carbon, but there are also a small part of titanium dioxide agglomerated particles, which they believe the particles can reduce the free energy of the system through agglomeration to improve the load stability. Xing et al. [120] effectively controlled the agglomeration of catalyst particles by controlling the times of supporting TiO2 on the surface of activated carbon.
Ma and Li [121] explored the influence of nano-TiO2 loading amount on the degradation of formaldehyde by wood-based activated carbon/TiO2 composite materials. The study showed that the degradation rate of formaldehyde first increased and then decreased with the increase of nano-TiO2 loading amount. Li et al. [122] found that when the titanium dioxide loading concentration on the surface of activated carbon increased, the light transmittance would decrease, so the degradation rate of formaldehyde would decrease. At the same time, she also studied the repeated practicability of adsorption-catalytic synergistic materials. As shown in Table 5, when the TiO2/AC material was used for six times continuously, the degradation rate of formaldehyde still reached 92%.
The relationship between TiO2/AC usage times and formaldehyde degradation rate.
Hu and Deng [123] studied the position of titanium dioxide loaded in the pore size of activated carbon by scanning electron microscopy. By comparing the electron micrographs of the activated carbon surface before and after loading, they found that when the catalyst loading reaches a certain threshold, the transition pores of activated carbon would be blocked, thus affecting the adsorption performance.
According to Hu and Deng’s research on the effect of titanium dioxide loading on the degradation of adsorbates, when the loading amount reaches a certain threshold, TiO2 will block the macropores and mesopores of activated carbon, affecting the migration rate of pollutants and thus reducing the degradation efficiency. Therefore, the composite ratio of adsorbent and catalyst should be adjusted to achieve the best catalytic degradation activity.
Table 6 lists some previous research results. It can be seen that compared with conventional formaldehyde degradation system, TiO2/AC has significantly improved formaldehyde degradation effect.
Some previous studies on adsorption and photocatalysis.
7. Conclusions and Outlook
According to the different adsorption modes, activated carbon adsorption can be divided into physical adsorption and chemical adsorption. Better pore gradient distribution speeds up the internal diffusion of formaldehyde in the aperture of activated carbon, improves the physical adsorption capacity, and increases the number of acidic oxygen-containing functional groups on the surface of activated carbon is conducive to improving the chemical adsorption of formaldehyde polar molecules. TiO2/AC adsorption and catalytic comaterial prepared by TiO2 supported on activated carbon is conducive to solving the problem of difficult recycling of catalyst. Through the absorption and concentration of formaldehyde by activated carbon, it provides a good reaction environment for photocatalysis and improves the degradation rate. In the preparation process, metal ion/nonmetal ion doping modification, on the one hand, can effectively broaden the spectral response range and, on the other hand, to a certain extent, inhibits the recombination of electrons and holes. If modified TiO2 is loaded on the surface of activated carbon, the adsorption synergistic material will have great advantages in adsorption catalysis.
With the in-depth study of TiO2 mechanism, TiO2/AC materials with high removal efficiency, large adsorption capacity, low energy consumption, and selectivity will be prepared to improve the preparation level of adsorption-catalytic comaterials, and TiO2/AC materials will have broader application prospects.
Footnotes
Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Acknowledgments
The present work was supported by the National Natural Science Foundation of China (51906193) and the Basic Research Program of Natural Science in Shaanxi Province (2020JQ-039).
