Abstract
A novel adsorbent prepared from the leaf powder of Teucrium polium L. (TPLLP) was characterized and its ability for adsorption of Cong red (CR) was inspected. Influences of CR concentration, adsorbent dosage, time of agitation, pH of solution, and temperature on the performance of this adsorption were also examined. Three models of kinetic along with three different isotherm models were applied for analyzing the empirical data of this adsorption. Additionally, the thermodynamic constants of were decided. The surface area, pore volume, pore size, and pHZPC of Zn/Cu-TPLLP were found to be 2.6436 m2.g-1, 0.013317 cm3.g-1, 527.393 Å, and 8.8, respectively. The achieved outcomes indicate the positive influence of temperature, concentration of CR in the range of 20 to 900 mg/L, adsorbent mass in the range of 0.005 to 0.02 g, time of adsorption from 0 to 120 min, and pH from 5.5 to 8.5. Models of the 2nd order with
1. Introduction
The dye of Congo red (CR) is known as typically anionic diazo dye having four different color-assisted functional groups [1], which has an IUPAC name of disodium 4-amino-3-[4-[4-(1-amino-4-sulfonato-naphthalen-2-yl) diazenylphenyl] phenyl] diazenyl-naphthalene-1-sulfonate [2] and has various molecular structures in the aqueous solutions that differ in their pH [2]; it is highly soluble in water and stable in air and light [3]. CR dye is used in laboratories as a pH indicator, histological dye, amyloidosis diagnosis, and for testing the contents of HCl in gastric [3]. This dye is also used in various industries like industry of textile due to its significant affinity to fibers of cellulose, papermaking, and industry of plastic, cosmetics, and pharmaceuticals [4, 5]. CR is a member of benzene compounds and contains in its structure aromatic amines; therefore, it will be converted to carcinogenic materials in the environment [3, 6]. Moreover, benzidine produced from metabolizing of CR poses hazard effects on human health such as irritation of gastrointestinal, eye, and skin [3]. Benzidine also causes clotting of blood, somnolence, and breathing problems [3]. Consequently, it is necessary to remove CR from sewage of those laboratories and industries before their discharge to the aquatic system. Thus, conventional biological methods have been established for decolorization of CR from the sewages of laboratories and industries, e.g., fungi of white rot [7], and foams of polyurethane and polypropylene [8] were applied for biodegradation of CR. It was reported that the biodegradation efficiency of CR is insufficient due to different aromatic structures and synthetic origin of this dye [9]. Therefore, other traditional techniques like electrocoagulation [10], coagulation by Fe3O4 [11], membrane filtration [12], and precipitation by chemical agents [13] were also utilized for removing of CR from water. The high cost and low removing performances of these methods prove that their usage is ineffective. Photocatalytic degradation of CR by activated hydrotalcites [14], cobalt ferrite nanostructures [15], nickel oxide nanoparticles [16],
Additionally, various sorbents such as activated carbon prepared from different materials [19–21], different clays [22] carbon loaded by nanoparticles of cobalt [23], magnetic nanocomposites of Co3O4 and SiO2 [24], binary oxide of iron zirconium [25], nanoparticles of MgO [26], and nanoparticles of SnO2 [27] were used to purify the sewages from CR. In comparison with other methods, the adsorption of CR by these adsorbents was found to be the best in terms of its efficiency, easy design, and operation [28]. Therefore, adsorption was selected in this study as the standard technique for removing of CR from synthetic aqueous solutions.
Although the nanoparticles of metallic oxides and activated carbon have a high adsorptive performance, their usages become limited due to the high costs of their production. Hence, several low or uncostly materials were applied as effective adsorbents for CR elimination from waters, e.g., hydrothermal-treated mushroom [1], powder of shrimp shell [2], tea waste [29], bottom ash and de oiled soya [3], and conjugates of sodium alginate-Chlorella polypyrrole-Chlorella[30]. The outcomes of these studies indicate that there are big differences between the adsorption performances of these cheap adsorbents towards CR. Therefore, other works should be carried out to investigate CR adsorption by other low-cost sorbents.
Teucrium polium L. (TPL) or germander is a herbal plant that has been utilized for more than 2000 years as traditional herbal remedies for their antifungal, carminative, antispasmodic, antipyretic, anti-inflammatory activity, and their ability to decrease high blood pressure [31]. This herbal plant belongs to Lamiaceae family that contains around 300 species and the flowering time of these herbs is between June and August. These species are abundantly distributed on hills and mountains in different locations such as Mediterranean countries, Southwest Asia, North Africa, and Europe [31, 32]. This plant is rich in flavonoids and commonly known in Saudi Arabia as Ja’adeh [32].
In the case of wastewater treatment, TPL has been used previously for preparation of Fe2O3 nanoparticles, which are used as a catalyst for photodegradation of methyl orange dye [33] and as an adsorbent for arsenic (III) removal from the aqueous solutions [34].
Despite the cheapness of this herbal plant and its availability in a considerable amount in many different countries, especially in Tabuk City and others in Saudi Arabia, there is no attempt that has been carried out till now to use the Teucrium polium L. leaf powder (TPLLP) as a new adsorbent for the removal of hazardous dyes from water or wastewater. Therefore, the main object of this work is to evaluate the adsorption efficiency of CR by using TPLLP as a novel, available, and cheap adsorbent. To achieve this aim, the leaf powder of this herb was modified by copper sulfide, zinc chloride (ZnCl2), and oxalic acid (HO2CCO2H); the modified and unmodified samples were characterized and the adsorptive performances of CR by these adsorbents were compared to select the best one. After that, influence of experimental conditions along with constants of kinetics, isotherms, and thermodynamic was examined for adsorption of CR by the best adsorbent prepared and tested in this work.
2. Materials and Methodology
2.1. Materials and Chemical Agents
The leaves of Teucrium polium L. (TPL) were supplied by a market located in Tabuk City, Saudi Arabia. Chemical reagents such as hydrochloric acid HCl (37%), sodium hydroxide NaOH with
2.2. Modification of TPLLP
The leaves of TPL were twice washed by distilled water, dried in an oven, and then converted to powder by electric grinder (TPLLP). A 100 g of TPLLP was refluxed with 1 L of 20%
Similar conditions and processes were applied for the mixture consisting of 100 g TPLLP and 50 g of CuS. The resulted adsorbent in this case was labeled as Zn/Cu-TPLLP.
A 100 g of TPLLP was also refluxed with 1 L of 20%
2.3. Characterization of Adsorbents
At a 10 kV accelerating voltage, the modified and unmodified TPLLP samples were scanned by SEM equipment to identify the surface morphology of these adsorbents. These adsorbents were also scanned by the instrument of FT-IR (Nicolet iS5 of Thermo Scientific FT-IR, USA) to detect the types of the functional groups on the surface of these samples.
The technology of BET (NOVA-2200 Ver. 6.11) was applied at 77.35 K for 22 h to estimate the surface area and porosity of these adsorbents. Furthermore, five solutions of 0.05 M Na2CO3 with different initial values of pH (2, 4, 6, 8, and 10) (
2.4. Adsorption Experiments
2.4.1. Determination of the Ideal Adsorbent
To identify the best and the most effective adsorbent prepared in this work towards CR, 20 mL of 100 mg/L CR solution was mixed with 0.03 g of raw leaf powder of TPL (TPLLP) in a 30 mL amber bottle. The sealed amber bottle was agitated by a shaker incubator for 30 h at 180 rpm and 27°C. Then, the mixture was filtered and the Jenway UV-6800 UV-Vis spectrophotometer was applied for measuring the balance concentration of CR in the filtrate at 500 nm. Similar experiments were carried out for the adsorption of this dye by the other three adsorbents (Zn-TPLLP, Zn/Cu-TPLLP, and Ox-TPLLP). Equations (1) and (2) were used for computing the CR percentage removal and amounts of CR adsorbed at equilibrium (
2.4.2. Influence of Experimental Conditions
The experiments of CR adsorption by the ideal adsorbent (Zn/Cu-TPLLP) were achieved in a batch system to examine the influence of the most important factors like Zn/Cu-TPLLP dosage (0.005-0.035 g), contact time (0-420 min), and pH (5.5-11.5) as the CR solution color will be changed to blue at pH less than 5, CR concentration (20-1000 mg.L-1), and temperature (27-57°C) on the adsorption. All of these experiments were carried out by mixing 20 mL of CR solution with the required amount of Zn/Cu-TPLLP in amber bottles (30 mL). Then, the sealed amber bottles were shaken at 180 rpm in a shaker incubator. After the required time for each experiment, the suspensions were filtered and the remaining CR concentrations were measured as mentioned in Section 2.2. The experimental conditions applied in this work are listed in Table 1.
Summary of the empirical conditions for adsorption of Congo red by Zn/Cu-TPLLP.
Equations (2) and (3) are used for the calculation of the amount of CR adsorbed at equilibrium (
2.4.3. Kinetic Studies
The experimental results of Section 2.3 for adsorption of 60, 150, and 300 mg.L-1 of CR solutions by Zn/Cu-TPLLP at 27°C and various times (0-420 min) were analyzed by the linear forms of three different kinetic models (Table 2) to determine the parameters of these models. Then, the obtained kinetic data were used for the investigation of the rate and mechanism of this adsorption.
Kinetic models applied in this work.
2.4.4. Isotherm Studies
The experimental data of Section 2.3 for adsorption of 20-1000 mg/L CR solutions by Zn/Cu-TPLLP (0.02 g) at a contact time of 22 h and various temperatures (27, 42, and 57°C) were analyzed by the linear equations of Langmuir, Temkin, and Freundlich isotherm models (Table 3) to evaluate the performance of this adsorption and to characterize the surface of Zn/Cu-TPLLP. Moreover, the values of
Isotherm models used in this work.
2.4.5. Thermodynamic Studies
The thermodynamic constants (
3. Results and Discussion
3.1. Adsorbent Characterization
Figures 1(a)–1(d) are the SEM images for adsorbent samples labeled as TPLLP, Ox-TPLLP, Zn-TPLLP, and Zn/Cu-TPLLP, respectively. In comparison between the image of an unmodified adsorbent (a) and images of the other three modified samples (b), (c), and (d), it can be detected that the surface of TPLLP is significantly changed due to the modification process, where most of folds in the modified adsorbent were collapsed and their structures became distracted. Moreover, the surfaces of the modified adsorbents contain many heterogeneous holes and pores which are supportive to the adsorption phenomena. Figure 1 also reveals that the density of the micropores on the adsorbent surface modified by ZnCl2 and CuS (Zn/Cu-TPLLP) is higher than that of the other three adsorbents.

SEM images of TPLLP (a), Ox-TPLLP (b), Zn-TPLLP (c), and Zn/Cu-TPLLP (d).



The spectra of FT-IR for unmodified and modified TPLLP are demonstrated in Figures 2. This figure reveals that the unmodified sample of TPLLP has seven peaks at 3335.93 cm-1 for stretching the hydrogen banded of O–H, 2923.91 cm-1 for C–H alkyl stretching, 2854.17 cm-1 for stretching of C–H, 1727.26 cm-1 for C = O stretching (aliphatic aldehydes), 1513.56 cm-1 for N-H (2°-amide) II band, 1158.35 cm-1 for stretching of C–O, and 1025.46 cm-1 for bending of C–H in-plane. Similar peaks with small shifting can be detected in the case of Zn-TPLLP and Zn/Cu-TPLLP samples (Figure 2). Figure 2 also demonstrates that three new peaks appeared in the case of Ox-TPLLP at 1616.92, 1318.54, and 780.67 cm-1. The presence of these various functional groups on the surface of these adsorbents implies the chemical activity of these adsorbents.

Spectra of FT-IR for TPLLP, Ox-TPLLP, Zn-TPLLP, and Zn/Cu-TPLLP.
According to the results of the BET surface analyzer (0.3806 m2.g-1, 0.001467 cm3.g-1, 151.065 Å), (0.7680 m2.g-1, 0.006674 cm3.g-1, 157.279 Å), (1.5875 m2.g-1, 0.007757 cm3.g-1, 400.197 Å), and (2.6436 m2.g-1, 0.013317 cm3.g-1, 527.393 Å) are the surface area, pore volume, and pore size of TPLLP, Ox-TPLLP, Zn-TPLLP, and Zn/Cu-TPLLP, respectively. This means that the surface area and pore characteristics of
Figure 3 depicts the relationship between the values of
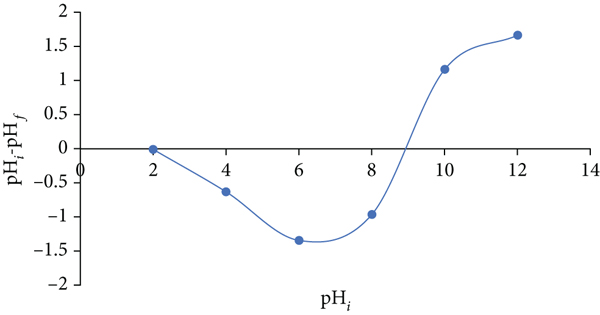
pHZPC of the adsorbent of Zn/Cu-TPLLP.
3.2. Results of Adsorption Experiments
3.2.1. Adsorbent Performance
The percentage removal of CR from aqueous solutions by TPLLP, Ox-TPLLP, Zn-TPLLP, and Zn/Cu-TPLLP adsorbents was determined to be 17.87, 45.11, 54.94, and 78.75 percent, respectively. Moreover, the values of 11.91, 30.08, 45.06, and 52.50 (mg.g-1) are the amounts of CR adsorbed at equilibrium (
This can be explained by the surface areas and pore characteristics of these adsorbents which are augmented in the same order (Section 3.1). The outcomes of this section confirm that Zn/Cu-TPLLP is the ideal sample that can be applied for CR adsorption. Thus, the other experiments of this work were conducted by using Zn/Cu-TPLLP only.
3.2.2. Influence of Zn/Cu-TPLLP Dose
To define the optimal mass of Zn/Cu-TPLLP that will be required for the adsorption of CR dye and to examine the dosage influence, the values of the % removal of CR from its solutions were plotted against Zn/Cu-TPLLP doses (Figure 4). This figure shows augmenting the values of CR
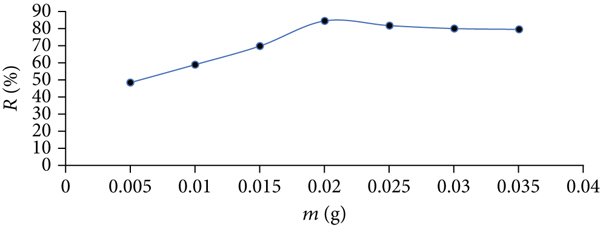
Influence of Zn/Cu-TPLLP dose on Congo red adsorption capacity.
3.2.3. Impact of pH
The process of adsorption is strongly affected by the solution pH of the adsorbate, as the adsorbent charge and ionization degree of the dye molecules depend on the value of pH. For this reason, the influence of this factor was considered in this work (Figure 5). As indicated in Figure 5,

Influence of pH solution on Congo red adsorption by Zn/Cu-TPLLP (
3.2.4. Kinetic Studies
Figure 6 illustrates the contact time impact on the adsorption of CR solution (60, 150, and 300 mg/L) by a fixed mass of Zn/Cu-TPLLP (0.02 g). According to this figure, there are three different adsorption stages, where the adsorption amount rapidly increased in the 1st stage (0-10 min), gradually increased in the 2nd stage (10-120 min), and almost was constant after 120 min (3rd stage). This proves that most of the sites were vacant at the first stage of this adsorption, and then, CR molecules gradually accumulated on these sites till fully occupied. The same outcomes were observed for CR adsorption by tea waste [29]. Although Figure 6 demonstrates that the equilibrium of this adsorption was achieved in 2 h, other experiments were performed at 22 h to ensure that all adsorption constants have been essentially studied at equilibrium.

Influence of contact time on Congo red adsorption by Zn/Cu-TPLLP (
Moreover, the slopes and intercepts of the plots of Figure 7 (kinetic model of the 1st order), Figure 8 (kinetic model of the 2nd order), and Figure 9 (kinetic model of intraparticle diffusion) were applied for computing the dynamic parameters of this adsorption. The calculated values were recorded in Table 4 for the first- and second-order models and in Table 5 for intraparticle diffusion model. In comparison between the parameter values of the first- and second-order models (Table 4), it can be seen that
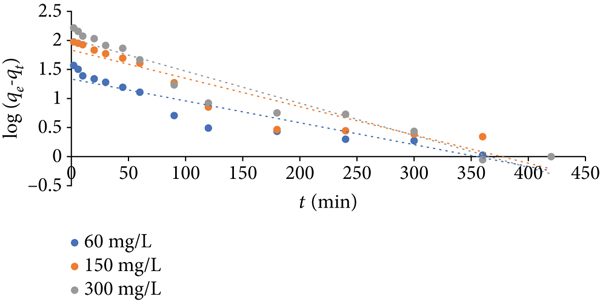
Kinetic model of the 1st order for Congo red adsorption by Zn/Cu-TPLLP (

Kinetic model of the 2nd order for Congo red adsorption by Zn/Cu-TPLLP (

Model of intraparticle diffusion for Congo red adsorption by Zn/Cu-TPLLP (
Parameters of the 1st- and 2nd-order kinetic models for adsorption of Congo red by Zn/CuS-TPLLP.
Parameters of the intraparticle-diffusion kinetic model for adsorption of Congo red by Zn/CuS-TPLLP.
Figure 9 reveals that each one of these plots is not linear for the total interval of time; none passed from the origin point and has two linear parts which have significant values of
3.2.5. Outcomes of Isotherms
The results associated with the influences of solution temperature and concentration on the performance of this adsorption are represented in Figure 10. The plots of this figure show that the adsorption of CR by Zn/Cu-TPLLP was positively influenced by the solution temperature because the CR solution viscosity and the movement energy of CR molecules decrease and increase, respectively, with rising temperature [43]. Figure 10 also shows that the values of

Influences of initial concentration and temperature on the adsorption of Congo red by Zn/Cu-TPLLP (
Furthermore, the analyzed experimental data of the isotherms of this adsorption are presented in Figure 11 (Langmuir model), Figure 12 (Freundlich model), and Figure 13 (Temkin model). Intercepts and slopes of the plots of these figures were used for calculation of the values of the isotherm factors recorded in Table 6. Langmuir model has the highest value of

Langmuir isotherm for adsorption of Congo red on Zn/Cu-TPLLP (
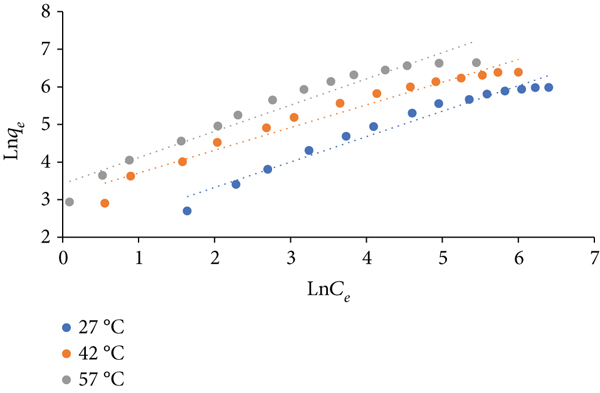
Freundlich isotherm for adsorption of Congo red on Zn/Cu-TPLLP (

Temkin isotherm for adsorption of Congo red on Zn/Cu-TPLLP (
Isotherm constants for adsorption of Congo red by Zn/CuS-TPLLP.
Moreover, high adsorption capacities of 526.32, 666.67, and 909.09 (mg.g-1) (Table 6) were obtained at the temperatures of 27, 42, and 57°C, respectively. This proves that Zn/Cu-TPLLP as a cheap and effective adsorbent will meet special interesting in the purification processes of wastewaters and water from CR.
3.2.6. Thermodynamic Outcomes
The values of
Thermodynamic constants for adsorption of Congo red by Zn/CuS-TPLLP.
3.3. Comparison with Other Low-Cost Adsorbents
The capacities of CR adsorption by Zn/Cu-TPLLP and other common low-cost adsorbents are given in Table 8. As shown, Zn/Cu-TPLLP, due to its significant chemical activity, has adsorption capacity higher than those of the common low-cost adsorbents used for removal of CR from the aqueous solutions before this work. Thus, the low cost of Zn/Cu-TPLLP is along with its higher adsorptive performance which confirms the importance and novelty of this adsorbent among the other low-cost adsorbents previously used for removing of CR from wastewaters.
Adsorbents used for adsorption of Congo red.
4. Conclusions
In this research, the leaf powder of Teucrium polium L. (TPLLP) was modified by oxalic acid, CuS, and ZnCl2. The modified and unmodified samples were characterized and tested for adsorption of Congo red (CR). The sample modified by the mixture of CuS and ZnCl2 (Zn/Cu-TPLLP) was the best (
Footnotes
Data Availability
Completely, data produced or investigated during this work were involved in this submitted article.
Conflicts of Interest
The authors confirmed that there are no probable conflicts of interest that impact the work reported in this paper.
Acknowledgments
The authors are very appreciative to the Deanship of Scientific Research at Tabuk University for funding the work through new faculty research/funding (Project No. S-0095-1441H).
