Abstract
A quartz crystal adsorbent functionalized with two promising porphyrins (the 5,10,15,20-tetrakis(4-tolylphenyl)porphyrin and the 5,10,15,20-tetraphenylporphyrin) was applied for the investigation of the adsorption phenomenon of aluminum chloride, iron chloride, and indium chloride. The aim is to prove new insights about the appropriate adsorption materials for metalloporphyrin fabrication. The equilibrium isotherms were measured at five adsorption temperatures (from 290 to 330 K) through the microbalance (QCM) method. The discussion of the experimental observations indicated that the adsorption of the aluminum chloride and the iron chloride was performed via a monolayer process. On contrary, the participation of the chloride ions in the double-layer adsorption of the indium chloride was explained by the layer-by-layer process. Overall, the statistical physics modeling of the experimental curves indicated that the number of ions per adsorbent site
1. Introduction
In recent years, porphyrins have been studied as chemical sensor materials of various metals leading to the formation of metalloporphyrin complexes [1, 2]. The metal-porphyrin complexes have received increased interest in several biological and environmental processes such as stereochemistry study and molecular recognition [3–6]. Note here that the nature of the central ion has influenced the photophysical properties of porphyrins [7, 8]. For example, these tetrapyrrolic macrocycles are involved in the chlorophyll structure (magnesium-porphyrin complex) and the hemoglobin structure (iron-porphyrin complex), so they are known as pigments of life [8]. In addition, metalloporphyrin complexes were also used as ionophores in the development of potentiometric recognition sensors supported with their structural variety and coordination chemistry [7, 8]. In particular, the photosensitizing properties of aluminum(III)-porphyrin have promoted its use as potential ionophore of fluoride [9, 10]. Moreover, the aluminum(III)-porphyrin and the indium(III)-porphyrin have been potentially used as supramolecular building blocks [11] and numerous other areas (sensing, molecular recognition, polymerisation reactions, etc.) [12, 13]. Surprisingly, it was remarked that while several papers have been devoted to metalloporphyrin complexes including the metal centers zinc, rhodium, and cobalt [14, 15], little attention has been paid to the use of porphyrins as complexing compounds of aluminum(III), iron(III), and indium(III) (Figure 1) despite their contribution in the interesting fields indicated above. In fact, the adsorption of these three metals on two tested porphyrins (H2TPP and H2TTPP) was examined in the present investigation.
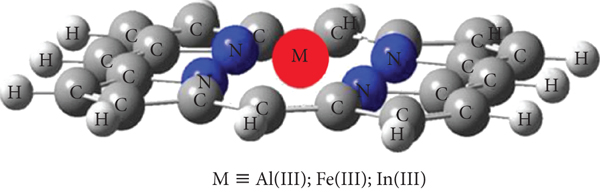
Chemical structure of the metalloporphyrin complexes.
In the literature, many approaches have been applied for the investigation of the adsorption mechanism of metals such as the sorption/reduction of heavy metal ions in environmental pollution management [16, 17]. Other experimental methods have been applied for the investigation of the adsorption mechanism [18–20], but in this work, the adsorption mechanisms of aluminum chloride, iron chloride, and indium chloride have been investigated by the QCM technique for many reasons. Firstly, this experimental method is a simple mass detector technique which requires the immobilization of porphyrins on the solid support of the quartz crystal. Then, the complexed mass of the metal into the macromolecule cavities can be controlled through the microbalance apparatus. Secondly, the metal-porphyrin complexes do not require an inert environment, and they are stable in the presence of water. Thirdly, the use of the QCM technique enables to plot the adsorption curves which describe the complexed amount of metallic ions on porphyrins at different temperatures. Lastly, the microscopic characteristics of the resulting film can be followed by easily fitting the experimental isotherms with analytical models [21].
According to the literature, the physical models were adopted by numerous authors to examine the adsorption problem. For example, the empirical expressions of the Langmuir and Freundlich models [22, 23] have been applied for the description of the isotherm curves, but they did not give a deep physicochemical analysis of the adsorption process. Importantly, the analytical adsorption models of the present investigation were established through the innovative statistical physics theory [24, 25] which provides the new vision about the metalloporphyrin description. Based on this idea, the fundamental aim of the modeling work of adsorption isotherms is to find the sufficient systematic model that can anticipate physical insights of porphyrin adsorption based on the physicochemical parameters of the statistical physics models [26].
2. Experimental Adsorption Isotherms
2.1. Materials
The aluminum(III) chloride (AlCl3), the iron(III) chloride (FeCl3), and the indium(III) chloride (InCl3) are the tested adsorbates in this paper.
The tetrakis(4-tolylphenyl)porphyrin (H2TTPP) and the tetraphenylporphyrin (H2TPP) are the porphyrins used in these experiments. They were synthesized based on a published method (the Adler-Longo strategy) [27].
Figure 2 shows the synthesis methods of porphyrins H2TPP and H2TTPP.

Synthesis methods of the 5,10,15,20-tetraphenylporphyrin (H2TPP) and the 5,10,15,20-tetrakis(4-tolylphenyl)porphyrin (H2TTPP).

H2TPP (Figure 2(a)) was synthesized through the reaction between propanoic acid (2 L) and benzaldehyde (40 mL). The synthesis of H2TTPP (Figure 2(b)) was performed by introducing 4-tolualdehyde (12.5 g) with the propanoic acid (110 mL). Note that the synthesis of the two tested adsorbents has been performed by following the same procedure (reflux for 30 min, allowing for cooling, filtration, and drying under vacuum for 3 hours). The dried solid mass of H2TPP was 9.76 g while the obtained mass of H2TTPP was 3 g. The two solid compounds of porphyrins were dissolved in chloroform giving two solutions with concentration of 2.9 10-2 mol·L-1.
2.2. Experimental Measurements
The experimental QCM setup is presented in Figure 3.

Experimental setup of the quartz crystal microbalance.
The QCM measurements were performed based on the piezoelectric quartz crystal [28, 29]. The crystal is a thin disc which is cut from the polished quartz (AT-cut). The fundamental resonant frequency of the quartz is 5 MHz, and the diameter of the disc is 2.54 cm [29]. A cleaning treatment was applied on the crystals by means of a Piranha solution at room temperature followed with rinsing with ethanol and drying with high purity nitrogen.
For the adsorption measurement, 60 μL of the adsorbents (porphyrins H2TPP and H2TTPP) was doped onto the clean crystal surface by the spin coating technique at 3500 rpm for 30 seconds. The functionalized crystals were dried at 393 K for 2 hours.
The functionalized crystal (adsorption cell) was placed in a Teflon probe, which was covered by a protective ring, and it was immersed in
where
At least 15 injections into the reactor were performed using a micropipette, then the variation of the resonant frequency (corresponding to the new adsorbate concentration in the reactor) was determined as follows:
with
2.3. Sauerbrey’s Equation
The deposited mass of the metallic ions on the surface after each injection was determined according to the Sauerbrey hypothesis which links the mass variation to the frequency change [30–32]:
where
In order to justify the application of the Sauerbrey equation in a liquid medium, the total frequency should be quantified according to the crystal properties. This allows writing the total frequency as follows [19, 33, 34]:
with,
Indeed, the influences of the temperature and the roughness on the total frequency should be neglected since the present investigation was carried out using polished crystals at a fixed temperature. The hydrostatic pressure influence was not taken into account according to Equation (2). Furthermore, low effects of the solution properties were noted, so they were not considered. Thus, the impact of the variation of the deposited mass is the dominant factor for the frequency change (
In fact, the adsorbed masses
2.4. Results of Experimental Measurements
The experimental adsorbed quantities of Al3+, Fe3+, and In3+ on H2TPP and H2TTPP are plotted in Figure 4 at 290-330 K.

Experimental isotherms of the six adsorption systems given at five temperatures (290-330 K): system (a) AlCl3-H2TTPP, system (a
Observing carefully the experimental data of the six adsorption systems, it is clear that the behavior of the isotherm curve depends on the type of adsorbate. The isotherms of aluminum chloride on H2TTPP and H2TPP (systems (a) and (a
In addition, comparing the performance of the six adsorption systems in terms of quantity, we can note the following order of adsorption performance:
3. Theory/Calculation
3.1. Statistical Physics Theory
The estimated values of the physical model parameters represent the key to understand the adsorption mechanism. The progress of this physical treatment is firstly seen against the well-known model of Langmuir (empirical model) [22]. The Langmuir model is developed by considering that one particle can interact with only one adsorbent site without taking account of the external factors. In general, this assumption led to wrong scientific conclusions. On the contrary, our statistical physics models correct this assumption by introducing a parameter defined by the number of adsorbed particles per adsorption site; it is noted in general by “
Based on the adsorption isotherm profiles, our statistical physics models can suggest various energies which are linked to different functional groups of the adsorbent surface contrary to the traditional equations of Langmuir [22] or Freundlich [23]. In the same direction, the statistical physics models are able to estimate the total number of the formed layer at all the reaction temperatures [24–26]. Note also that the multilayer adsorption of charged ions can be only carried out via a layer-by-layer (LBL) process [25, 36].
A general analysis of the adsorption data led to test two models: the monolayer model for AlCl3 and FeCl3 and the LBL multilayer model for InCl3. The modeling work is then arranged as follows: (a) it is worth mentioning the general methodology which allows developing the physical models expressing the adsorbed amount corresponding to the adsorbate concentration, (b) the statistical physics model can be applied and discussed on the experimental data, and (c) the choice of the adopted model led to describing the adsorption reaction via its parameters.
3.2. Advanced Adsorption Models
To establish the physical model expressions, the equilibrium of the adsorption reaction can be expressed via the next equation [25, 37]:
where
This system is characterized by the chemical potential (μ) and the temperature (
Concerning the adsorption via one energy level (
For the LBL adsorption process (double-layer or multilayer), two energies can be responsible for this process. Note that the adsorption of the first layer is characterized by the energy (
For the double-layer model:
For the multilayer model:
with
Then, the average occupation number of
In this modeling work, the analytical development can be carried out by introducing the chemical potential of the perfect gas (
where
The investigation of the adsorption problem can be also performed using the chemical potential (
Then, the adsorbed quantity
Finally, we get the analytical expressions of the six adsorption models:
The monolayer model of the ideal gas approach:
where
The monolayer model of the real gas approach:
where the energetic coefficient
The double-layer and multilayer models include two energetic coefficients involving two molar adsorption energies (
The double-layer model of the ideal gas approach:
where the energetic variables
The double-layer model of the real gas approach:
where
The multilayer model of the ideal gas approach:
where
The multilayer model of the real gas approach:
where
with
3.3. Adjustment of Adsorption Models with Experimental Isotherms
The six physical adsorption models were applied on the experimental isotherms by means of a numerical fitting program [42]. The mathematical fitting method is based on the Levenberg-Marquardt iterating algorithm using a multivariable nonlinear regression program [40, 43]. The criteria to adopt a descriptive model are the determination coefficient
Tables 1 and 2 show the numerical values of the error adjustment coefficients.
Values of the error adjustment coefficients
Values of the error fitting coefficients
According to Tables 1 and 2, the aluminum chloride isotherms (systems (a) and (a
Figure 5 illustrates the monolayer adsorptions of aluminum chloride and iron chloride and the double-layer adsorption of indium chloride on H2TTP and H2TPP.

Illustration of the monolayer adsorption of AlCl3 and FeCl3 and the LBL double-layer adsorption of InCl3 on the two porphyrins H2TPP and H2TTPP.

3.4. Analysis of Physicochemical Parameters
The mon-layer model (ideal gas), devoted for the description of the AlCl3 isotherms, was governed by three parameters (the number of aluminum ions per receptor site
All fitting parameters values are given in Tables 3 and 4. The study of the adsorption model parameters attributes interesting microscopic interpretations of the porphyrin complexation.
Values of the physicochemical parameters (
Values of the physicochemical variables (
3.4.1. Steric Interpretation
Basically, the parameters
The evolution of the two steric parameters versus temperature is reported in Figure 6.

Evolutions of the two steric parameters as a function of temperature: (a) evolution of the number of bonded ions per site

From Figure 6(a), the
The fitted values of
Furthermore, it is noted from Figure 4 that the temperature exerts exactly the same influence on the six complexation systems: once the temperature increases, the adsorption capacities increase. This is often explained from Figure 6 which shows that the values of the coefficients
3.4.2. Van der Waals Parameter Behaviors
It can be noticed that despite the AlCl3 and FeCl3 adsorptions being both monolayer adsorption processes, where there is no contribution of the chloride ions at the layer formation, the fitted values of
It can be concluded that the decrease of FeCl3 isotherms (Figures 4(b) and 4(b
Overall, it can be concluded that the use of aluminum chloride guarantees more stability during the metalloporphyrin formation.
The values of these two parameters for H2TPP and H2TTPP are summarized in Figure 7.

Variations of the adjusted values of the cohesion pressure
Generally, the interactions of the particles (lateral interactions) are characterized by the cohesion pressure
Furthermore, it is seen from Figure 7 that the parameter
3.4.3. Energetic Calculation
According to Equations (16), (18), and (20), the molar energies of the six adsorption mechanisms can be calculated by means of the energetic coefficients,
For aluminum chloride adsorption,
For iron chloride adsorption,
For indium chloride adsorption,
The values of the molar energies and their variations via the temperature are reported in Table 5 and Figure 8.
Values of the molar adsorption energies

Evolutions of the molar adsorption energies of the six adsorption systems as a function of temperature:
According to Table 5, for the indium chloride adsorption, it is obviously remarked that the calculated values of
Comparing the
It can be also observed that the molar energies rise with the expansion of the temperature (Figure 8). This can be interpreted by the endothermic behavior of the six studied processes.
Furthermore, the energies
In contrast, all the other adsorption mechanisms (FeCl3 and InCl3) take place via a physical process since they present adsorption energy values lower than 40 kJ/mol [42, 50, 51]. During physisorption, the adsorbed particles retain their individual properties and there is no change in the structure of the components of the adsorption system. The system is weakly energetic (low adsorbate-adsorbent interaction), so the presence of a desorption phenomenon is possible (e.g., FeCl3 adsorption). Moreover, the adsorbed layer can interact with other adsorbed layers and can act as a new template for the adsorption of the following layer (multilayer adsorption mechanism) such as the case of InCl3 in the present investigation.
4. Conclusion
In this work, the experimental QCM tool is used to control the adsorption of aluminum chloride, iron chloride, and indium chloride on porphyrins H2TTPP and H2TPP. By analyzing the experimental isotherms, the AlCl3-H2TTPP complex was found as the best adsorption system in terms of reproducibility since it showed the highest adsorbed quantities. Theoretically, statistical physical models were suggested for the analysis of the experimental results. The numerical investigation confirmed that the chloride ions contributed only in the adsorption of InCl3 (double-layer adsorption). The iron chloride adsorption was described by a monolayer model that considers the lateral interactions of the ions indicating the lowest stability of the formed iron-porphyrin complex. The physicochemical parameters of these models gave interesting microscopic insights about the complexation of the two porphyrins. Indeed, it was found that the six adsorption processes were governed by a multi-interaction process (
Footnotes
Data Availability
The data that supports the findings of this study are available within the article.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Acknowledgments
The authors extend their appreciation to the Deputyship for Research & Innovation, Ministry of Education – Kingdom of Saudi Arabia for funding this research through project number PNU-DRI-RI-20-016.
