Abstract
The aim of this work was to investigate the adsorptive performance of the polypyrrole-based KOH-activated carbon (PACK) in the removal of the basic dye crystal violet (CV) using a batch adsorption system. The equilibrium data, obtained at different initial CV concentrations (
1. Introduction
Water pollution is a major environmental problem worldwide and requires urgent solutions. Industrial discharge is the main source of aquatic pollution [1] and results in the release of various physical, chemical, and biological pollutants, including suspended elements, dissolved gasses, mineral and organic solutes, and microorganisms [2]. The effluents from the paper, textile, leather, food, plastic, pharmaceutical, and printing industries typically contain large quantities of dyes that are used to color their products. Although many of the dyes used are not highly toxic, they cause visual pollution via aesthetic changes to the natural environment. Furthermore, they cause a reduction in light penetration into water, impinging on the photosynthesis of aquatic organisms. Nevertheless, many dyes have long-lasting hazardous and toxic effects on aquatic life, largely due to their high water solubility and nondegradability under natural conditions [3].
Crystal violet (CV), also known as gentian violet, is a synthetic cationic dye belonging to the triphenylmethane group. Compared with anionic dyes, cationic dyes are more toxic to mammalian cells due to their ability to interact with negatively charged cell membrane surfaces, which enables them to penetrate into cells and concentrate in the cytoplasm [4]. CV is used in a variety of applications, including textile, plastic, and paper coloring; biological staining; and veterinary and animal medicine. However, researchers have reported that CV is a mutagen and mitotic poison that is toxic to mammalian cells and may exhibit carcinogenic effects [5–8]. Moreover, it can cause irritation to the skin and digestive tract. In extreme cases, CV can cause kidney failure and severe eye irritation leading to permanent blindness [9, 10]. These harmful effects necessitate efficient strategies for the removal of CV (and similar toxic dyes) from water bodies.
Numerous methods have been developed for removing dyes from contaminated wastewater, including adsorption-based strategies, membrane filtration, photodegradation, coagulation, chemical oxidation, and biological processes [6, 11–13]. Among these, adsorption is the most common technique due to its numerous advantages, which include its superior performance, ease of operation, cost effectiveness, and the availability of necessary reagents. Furthermore, it has been widely demonstrated that activated carbon is the most effective adsorbent of the various applicable materials, which include natural clays, zeolites, biomasses, polymers, and polymer-based materials [14].
Nitrogen-doped polypyrrole-based activated carbons (termed PACK), which is applied here for the removal of CV as a model harmful cationic dye, have been demonstrated to be excellent adsorbents. Various studies have indicated their high capacity in the adsorption of gasses including nitrogen and carbon dioxide, toxic heavy metal ions such as Pb(II), and anionic dyes such as methyl orange [15–18].
The objective of the present study was to evaluate the efficiency of PACK in the removal of CV from synthetic wastewater. Its adsorption kinetics, isotherms, and thermodynamic parameters are investigated, discussed, and compared with those of other reported materials.
2. Materials and Methods
2.1. Materials
Pyrrole (Py, >98%), ammonium persulfate (APS, 98%), potassium hydroxide (KOH, 85%), sodium nitrate (NaNO3, 99%), and sodium hydroxide (NaOH, 98%) were purchased from Alfa Aesar, Karlsruhe, Germany. Hydrochloric acid (HCl, ~36%) and ethanol (EtOH, 99.5%) were obtained from Fisher Chemical, Loughborough, UK. CV (>90%) was provided by BDH Chemicals Ltd., Poole, England, UK, which was dried at 80°C for 2 h before use. All materials were used as received unless otherwise stated.
2.2. Adsorbent
PACK was prepared as described in the literature [15–18]. Briefly, HCl-doped polypyrrole (PPy) was prepared using an equimolar quantity (0.08 mol) of the monomer (Py) and the initiator (APS) in cold HCl (1 L, 0.1 M). The obtained material was washed and dried and then calcined under nitrogen at 650°C attained at 3°C/min for 2 h with four times the mass of KOH, the chemical activator, using a Carbolite MTF 12/38/250 tube furnace (Wolf Laboratories, UK). The PACK obtained was sequentially washed with 0.5 M HCl and then water until neutrality and dried.
The pH at which the surface net charges of PACK are zero (pHPZC) was determined by a pH drift method described elsewhere [19, 20]. Typically, a solution of 0.1 M NaNO3 was prepared from which batches of 15 mL were adjusted in the pH range 2–11 using 0.1 M HCl or 0.1 M NaOH solution and denoted as pHi. To each sample, 0.015 g PACK was added, and the suspension was shaken intermittently for 24 h at room temperature (
2.3. Adsorbate
The chemical structure of the adsorbate CV is shown in Figure 1. It is a well-known cationic dye with a color index number of C.I. 42555, a

Chemical structure of CV.
2.4. Adsorption Experiments
Adsorption equilibrium experiments were performed using 50 mL Eppendorf tubes at CV concentrations of 50, 100, 200, 300, and 500 ppm and temperatures of 25, 35, and 45°C, using 20 mg of the adsorbent (PACK), agitated at 150 rpm using a GFL 3017 digital shaker (GFL Gesellschaft für Labortechnik mbH, Burgwedel, Germany) for 24 h. Then, the adsorbent was filtered off, and the residual CV concentrations were measured. Kinetic experiments were conducted using 1 L Erlenmeyer flasks under the following conditions: 40 mg PACK, 500 mL CV solutions (15 and 45 ppm), 150 rpm agitation, and 25°C adsorption temperature. The residual dye concentrations were measured at 0, 5, 10, 30, 45, 60, 90, and 120 min, with at least two replicate concentration measurements being performed. All experiments were performed in duplicate without pH adjustment. However, the pH value of, e.g., 50 ppm CV solution analyzed at
2.5. Theoretical Calculations
The equilibrium adsorption capacity (
The adsorption mechanism was investigated by employing the pseudo-first-order (PFO), pseudo-second-order (PSO), and intraparticle diffusion (IPD) kinetics models proposed by Lagegren and Svenska [21], Ho and McKay [22], and Weber and Morris [23], respectively. The linear forms of these three models are given by Equations (3), (4), and (5), respectively.
The adsorption isotherm was modeled with the Langmuir [25, 26], Freundlich [27], Temkin [28], and Dubinin-Radushkevich (D-R) [29] linear form models, as given in Equations (7), (8), (9), and (10), respectively.
Conversely, the Freundlich isotherm, Equation (8), assumes multilayer adsorption and a heterogeneous adsorbent surface. The
The effect of heat on the adsorption system was determined using the thermodynamic parameters Gibbs free energy change (
3. Results and Discussion
3.1. Characterization
The essential features of the adsorbent PACK, including its structural and surface properties as well as its efficacies as an adsorbent of gasses, heavy metal ions, and anionic dyes represented by carbon dioxide (CO2), lead ions (Pb2+), and methyl orange (MO), respectively, have been reported by Alghamdi et al. [15–17] and Sevilla et al. [18] (Table 1). Furthermore, to obtain insight into the surface charge, the pH at which the net surface charge is zero (pHPZC) was estimated and found to be 6.35 (Figure 2). Thus, the surface is positively charged below pH 6.35 and negatively charged above pH 6.35. Moreover, the zeta potential at neutral pH (-30.6 mV, see Figure S1) indicated a negative surface which further supports the pHPZC result.
Properties of PACK.
BET: Brunauer–Emmett–Teller; SA: surface area; APD: average pore diameter; MO: methyl orange.

Point of zero charge for PACK adsorbent.
3.2. Effect of Contact Time and Kinetic Studies
Figure 3 shows the time profile of CV adsorption onto PACK for two different initial concentrations (15 and 45 ppm). Figure 4 shows a histogram representing the values of the adsorption efficiency (Re, %) as well as the capacities at the first adsorption phase (
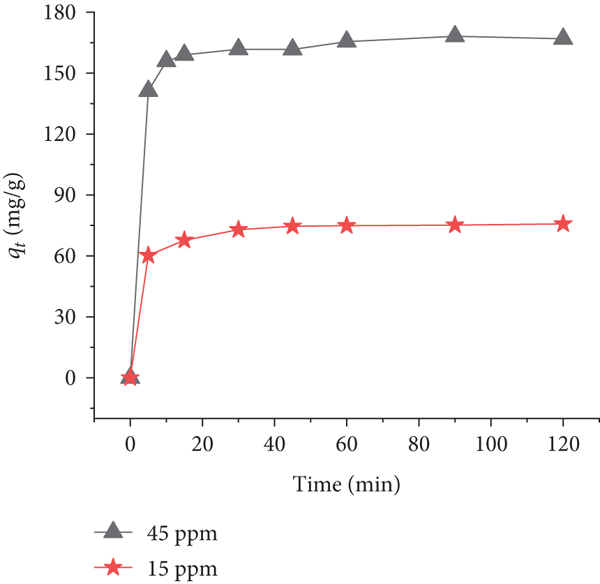
Effect of contact time on the adsorption of CV onto PACK.
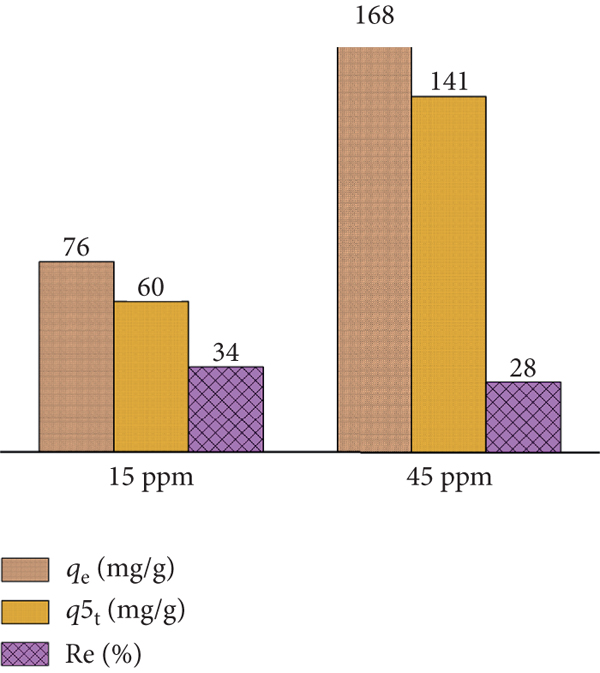
Graphical illustration of the adsorption of CV (
The mechanism involved in this adsorption system was investigated using PFO, PSO, and IPD kinetic models (Figures 5, 6, and 7), as described by Equations (3), (4), and (5), respectively. The corresponding kinetic parameters and correlation coefficients are presented in Table 2. As can be seen, the relatively higher value of the correlation coefficient, which is an important criterion in the determination of good fit, for the PSO model (

PFO kinetic plot for the adsorption of CV onto PACK. Conditions:

PSO kinetic plot for the adsorption of CV onto PACK.

Webber-Morris intraparticle diffusion plot for the adsorption of CV onto PACK (see Figure 4 for conditions).
Kinetic parameters for the adsorption of CV onto PACK.
3.3. Adsorption Isotherms
Adsorption isotherms generally describe the way in which an adsorbate interacts with an adsorbent when equilibrium is attained at a certain temperature. In this study, the adsorption system was evaluated at 25, 35, and 45°C, and the experimental data were fitted into four models expressed by Equations (7)–(10) (Figure 8), and the calculated parameters are presented in Table 3. The goodness of fit for the experimental data was found to be in the order

Langmuir, Freundlich, Temkin, and D-R isotherm plots for CV adsorption onto PACK.
Langmuir, Freundlich, Temkin, and D-R isotherm parameters for CV adsorption onto PACK.
3.4. Adsorption Thermodynamics
The values of Gibbs free energy (
Thermodynamic parameters for the adsorption of CV onto PACK.
Figure 9 is a schematic presentation for the adsorption mechanism in which π-π and ionic interactions are dominant. This is in line with the results predicted by the adsorption models discussed above. Thus, the physisorption mechanism is practically superior; however, chemical interaction involving strong ionic bonds and ion exchange cannot be ignored, a case that is evinced by the results of isotherms and thermodynamic parameter values, which are generally in the region of the physical process but closer to chemical limits. Moreover, the effect of the diffusion process in the adsorption mechanism is also expected at least in the last stage of adsorption.

Schematic presentation of the adsorption mechanism of the cationic dye crystal violet by PACK adsorbent.
3.5. Reusability
As the reusability of the adsorbent is of crucial importance in the economic development, efficient protocols for adsorbate recovery and adsorbent regeneration have to be established; however, common adsorbents vary greatly in their stability and applicability to be regenerated. Figure 10 illustrates the chemical regeneration efficiency calculated according to [5]

Desorption percentage of CV from PACK after three cycles using solvent mixtures (NaOH/acetone or HCl/acetone) or single solvent (NaOH or HCl; only cycle one presented).
The low desorption efficiency of a single solvent elution system (NaOH, HCl) may be due to ion competition, indicating high comparative affinity of CV dye toward PACK (Figure 10; only cycle 1 is presented). However, NaOH shows better desorption performance of CV than HCl, possibly due to electrostatic repulsion at pH above pHPZC [5].
When acetone was added to the above single solvents, the regeneration efficiency was enhanced. As shown in Figure 10, the desorption efficiency reduced about 4 and 11% when the binary eluents NaOH/Ac and HCl/Ac were used, respectively. The results generally indicate a total mass recovery of CV of about 72% after three cycles of the adsorption-desorption process. The reduced efficiency (28%) could be due to incomplete desorption of CV [42]. The results indicate the reusability of the adsorbent with possible regeneration enhancement; however, it necessitates further optimization of the regeneration conditions including solvents types, concentrations, solution pH, etc.
3.6. Relative Performance of PACK
For comparative purposes, the values of adsorption capacity,
Comparison of the CV adsorption capacities of various reported adsorbents.
4. Conclusions
Here, the adsorption efficiency of PACK for the removal of CV from aqueous solutions was investigated. Batch adsorption processes were performed to investigate the effects of contact time (0–120 min, and up to 24 h), initial CV concentration (50–500 mg/L), and temperature (25–45°C). The adsorption kinetics, evaluated using PFO, PSO, and IPD models, revealed that the PSO model provided a better fit (
Footnotes
Data Availability
Data that support the findings of this study are included within the article and supplementary information file.
Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Acknowledgments
The authors extend their appreciation to the Deanship of Scientific Research at King Saud University for funding this work through Research Group No. RGP-1438-040.
