Abstract
Magnetite-functionalized horse dung humic acid (HDHA) has been successfully prepared by the coprecipitation method, and the as-prepared adsorbent (MHDHA) has been applied as an easy-handling adsorbent for toxic Pb(II) in artificial wastewater. The MHDHA was characterized by Fourier transform-infrared spectroscopy (FT-IR), X-ray powder diffraction (XRD), transmission electron microscopy (TEM), energy dispersive X-ray (EDX), and vibrating sample magnetometer (VSM). The FT-IR study showed that the MHDHA had the characteristics peaks of HA and Fe-O stretching. The XRD analysis revealed that the MHDHA had the
1. Introduction
The immoderate release of contaminants into the aquatic environment is a primary global concern. Heavy metal contaminants are a severe threat to the life and development of humankind [1]. Lead is among the most hazardous pollutants that can cause health issues in human beings because it exhibits high toxicity and potential mutagenic and carcinogenic effects [2]. Therefore, the elimination of such contaminants from water has attracted growing attention. Several methods have been developed such as adsorption, organic treatment, photocatalyst, coagulation, ion exchange, membrane separation, solvent extraction, evaporative recovery, reverse osmosis, and chemical precipitation [3, 4]. However, though membrane separation is highly efficient, it has low economic viability and high maintenance price for large-scale applications. Precipitation and organic treatment are cost affordable, but they cannot reduce the pollutant below the desirable limit and produce a high amount of sludge. Photocatalysis commonly faces photoetching issues and quick electron-hole pair recombination and aggregation catalyst ion problems [5]. Although the adsorption technology has the disadvantages of difficulties in treating and parsing the posttreatment waste, adsorption is amongst the broadly applied technology in water and wastewater remediation. This technology has been identified as the promising, efficient, and extensively used fundamental method in the wastewater remedy processes, specifically hinging on its simplicity, economic viability, technical feasibility, environmental safety, and social acceptance [3, 5].
Numerous candidates of heavy metal adsorbents have been reported to accommodate the properties mentioned above, i.e., fly ash-zeolite to adsorb Cu(II), Fe(III), Mn(II), and Zn(II) [6]; green biowaste as Pb(II) and Cd(II) adsorbents [7]; modified activated carbon (AC) for Cd(II) removal [8]; and modified humic acid (HA) for various heavy metal removal [9–18]. HA seems a promising candidate because it is cheap and highly available and has a high adsorption rate and capacity [19]. HA is reported to possibly bind with various toxic metals, organic compounds, and radionuclides because it has an abundant reactive site dominated by carboxylic and phenolic groups [20]. However, utilizing original HA for adsorbent was time-consuming and a costly process because it needs centrifugation or filtration step from water post adsorption [21]. Besides, at
Unfortunately, the functionalized HA in the reported study derives from peat soils [10, 12–18]. Whereas peat soil is not a renewable source for HA because its formation takes a long time and peat soil exploration leads to the release of greenhouse gases from stored carbon in peat [22–28], alternatively, much organic biomass considered as waste, such as chicken manure [29], organic biomass [30–33], cow dung [34, 35], and horse dung [36–38], is recently reported as a source of renewable HA. Organic waste biomass is commonly used as fertilizer, except for horse dung, due to its rough texture and dense properties [39]. In addition, the existence of horse dung has a lousy effect on the environment because it is often scattered, creates a rotten smell, and is responsible for the appearance of nuisance animals [40]. The utilization of organic waste biomass, particularly horse dung as a renewable HA source, will overcome environmental problems caused by organic waste biomass and sustain the contaminant handling through the adsorption process.
Previous work on horse dung humic acid (HDHA) showed that the material has good removal uptake towards toxic heavy metal pollutants in water [36–38]. Therefore, it is fair to conduct further studies on the material. To our best knowledge, the incorporation of HDHA is still scarce. Hence, the purpose of the present study was to develop functionalized HDHA with enhanced stability, handling, and removal uptake towards toxic Pb(II) from artificial wastewater. In this study, the HDHA was incorporated with magnetite to produce magnetite-functionalized HDHA (MHDHA). Functionalization magnetite by peat soil HA has proven to enhance the adsorption handling, stability, and capacity; however, no document was found that investigated this for HA from the biological matrix as HDHA.
2. Materials and Methods
2.1. Chemicals
All chemicals, NaOH, HCl, HF, Pb(CH3COO)2, FeCl3.6H2O, FeSO4.7H2O, NH3, Ba(OH)2, and Ca (CH3COO)2, were of analytical grade produced by Merck® without further purification.
2.2. Adsorbent Preparations
Horse dung humic acid (HDHA) was extracted from 1 to 3 months of age of 50 mesh dry horse dung powder by the alkaline method [41] in the ambient atmosphere. Half a kilogram of dry horse dung powder was soaked into 5 L NaOH 0.1 M, and the mixture was stirred for 24 h. The filtrate of the mixture was separated by 3000 rpm centrifugation for 10 min. The filtrate was slowly added with HCl 0.1 M until the pH is approximately 1.0 and aged for 12 h to form two layers. Crude HDHA (solid layer) was separated by centrifugation at 5000 rpm for 15 min and dried at 50°C. The purification of crude HDHA was performed by soaking 10 g of dry crude HDHA powder into a 200 mL solution of 0.1 M HCl/0.1 M HF (1 : 3) in a polyethylene flask. The suspension was stirred for 24 h, and the solids were separated by centrifugation at 5000 rpm for 15 min as purified HDHA. Similar procedures were also applied for the extraction and purification of HA from Sumatra’s peat soils as a comparison to HDHA.
The synthesis of magnetite-functionalized HDHA (MHDHA) was conducted by a coprecipitation procedure based on Liu et al. [10] by dissolving 3.05 g FeCl3.6H2O and 2.10 g FeSO4.7H2O into 25 mL distilled water and heated up to 90°C. Two solutions, 5 mL NH3 25% and 0.25 g purified HDHA, were dissolved in 50 mL of NaOH 0.1 M, which were added sequentially and rapidly. The mixture was stirred at 90°C for 1 h and then cooled to room temperature. After 24 h, the black solids of MHDHA were rinsed with distilled water to neutral pH and dried in a vacuum oven at 50°C. Also, the bare magnetite was prepared similarly without HDHA addition.
2.3. Characterizations
Characterizations of extracted HDHA were conducted by UV-Vis spectroscopy (Shimadzu UV-1700 PharmaSpec) to analyze the humification degree, the functional group analysis by FT-IR (Shimadzu Prestige 21), and the total acidity. The total acidity (sum of carboxylic and phenolic contents) of HDHA was determined by the baryta indirect potentiometric titration method [41] by dissolving 50 mg of the sample into 20 mL Ba(OH)2 0.1 M. The suspension was shaken for 24 h. The filtrate was then separated from the solid titrated with standard HCl 0.5 M until the pH reached 8.4 (
The functional group analysis and crystal analysis of MHDHA were performed by FT-IR and XRD (Shimadzu XRD-6000) using Cu Kα radiation in the
2.4. Effect of pH and Ionic Strength
The effect of pH was studied by preparing a series of 25 mL of Pb(II) 25 mg/L that the pH was adjusted at 2.0, 3.0, 4.0, 5.0, 6.0, 7.0, and 8.0 by NaOH 0.01 M or HNO3 0.01 M addition. Into each prepared Pb(II) solution, 20 mg of the adsorbent was interacted and shaken for 2 h. After the sorbent was separated, the remaining Pb(II) in solutions was quantified with AAS. The sorbed Pb(II) was considered as the difference between the initial and final amounts of Pb(II) in the solution. The optimum pH was determined by the most adsorbed Pb(II) to the sorbent. Pb(II) analysis in the solution was conducted by atomic absorption spectroscopy (AAS) (PerkinElmer 3110).
The effect of ionic strength on Pb(II) adsorption was conducted by preparing a 5 series of 25 mL of Pb(II) 25 mg/L at optimum pH. Then, NaCl salt was added to each solution so that the final concentrations of NaCl were 0.005, 0.01, 0.05, 0.1, and 0.5 M. After 120 min stirring, the mixture was filtered and the filtrate was analyzed with AAS to obtain the adsorbed Pb(II).
2.5. Isotherm and Kinetics Sorption Experiment
The isotherm adsorption experiment was performed by contacting 20 mg sorbent with 25 mL of artificial wastewater which has various Pb(II) concentrations (20, 40, 50, 60, 80, 100, 150, 200, and 400 mg/L) for 180 min. The kinetics adsorption experiment was conducted by contacting 20 mg of sorbent with 25 mL artificial wastewater containing 25 mg/L Pb(II) for 5 min. A similar procedure was conducted with variations in contact time: 10, 20, 30, 40, 50, 70, 90, 120, and 180 min. An external magnet was used to attract MHDHA after the adsorption process. The residual Pb(II) in the solution was quantified by AAS.
2.6. Regeneration Studies
The reusability of MHDHA was checked by performing the five cycles of the adsorption-desorption process. The desorption of Pb(II) was performed using HNO30.1. M as a desorbing agent. The 50 mL of Pb(II) 50 mg/L was shaken together with 50 mg MHDHA for 120 min at 298 K. The Pb(II)-loaded MHDHA adsorbent was kept in the 100 mL conical flask (shaken for 120 min) with the desorbing agent (50 mL). The HDHA adsorbent was separated by an external magnet retraction, washed with deionized water, and again repeated for five cycles of the adsorption-desorption process. Metal recovery (%) was considered by the following equation [8]:
3. Results and Discussion
3.1. Characterization of HDHA
The successful HDHA extraction was indicated from high peaks at 250–300 nm and slopes with increasing wavelength with no specific peaks (Figure 1(a)). The smooth absorbance that decreases monotonously with the increasing wavelength between 250 and 800 nm of HDHA spectra is a characteristic spectrum of HA [43]. The steeper the slope indicates a high degree of humification [44]. There are three important regions to reveal the molecular properties of HA: at 280 nm, this indicates the transformation of lignin and quinone into HA, at 460–480 nm, this indicates organic material at the beginning of humification, and at 600-670 nm, this is an indication of a strongly humified material with a high degree of aromatic groups [45]. Then, based on the

(a) Characteristic UV-Vis spectra of purified peat soil HA (PSHA), purified horse dung HA (HDHA), crude peat soil HA (CPSHA), and crude horse dung HA (CHDHA). (b) Characteristic functional groups of PSHA, HDHA, magnetite, and MHDHA.

The high absorbance of peat soil HA (PSHA) at 250–300 nm with a sharp decrease clarifies a high degree of humification (Figure 1(a)). The lower ratio values of
Characteristic parameters (yield, ash content, total acidity, and UV-Vis absorbance ratio) of extracted C-HDHA and HDHA compared with C-PSHA and PSHA.
The FT-IR characterization of HDHA showed five characteristic peaks of HA, i.e., 3400 cm−1 could be assigned to hydrogen bonding of –OH groups; 2900 cm−1 is designated for the stretching of C–H aliphatic; 1720 cm−1 referred to C=O stretching of –COOH groups; 1625 cm−1 is ascribed to aromatic C=C, C=O of quinone groups, and aromatic rings formed by the hydrogen bond of O–H groups; and 1200 cm−1 was associated with O–H bending and C–O stretching of –COOH groups (Figure 1(b)). The Fe-O stretching vibrations of magnetite and MHDHA were significantly identified at 592 cm−1 [47]. In addition, the successful functionalization of HDHA on the magnetite surface was detected at ∼1625 cm−1 which indicated an interaction between C=O and the iron oxide surface (Figure 1(b)) [12]. The absorption band at 1400 cm−1 was strongly assigned to −CH2− scissoring, and the 1121 cm−1 band was assigned to the C-O stretching of –COOH [10]. The unobserved C=O stretching in MHDHA (Figure 1(b)) as free –COOH at above 1700 cm−1 suggested the attachment of HDHA to magnetite through ligand-exchange interaction between C=O and Fe3O4 surfaces [14]. Similarly, previous investigators reported that the linear thread-like polysaccharides could be strongly adsorbed by many oxide and aluminosilicate mineral surfaces [48].
The crystal properties of synthesized magnetite and MHDHA showed characteristic peaks with strong intensities emerging at

The diffractogram (a) and the magnetization hysteresis loop (b) of synthesized magnetite and MHDHA; TEM image of MHDHA and average particle size of MHDHA processed TEM image by free software ImageJ (inset picture) (c).


The magnetic properties, which were essential to ensure the separation process, were measured by graphing the hysteresis loop of synthesized bare magnetite and MHDHA (Figure 2(b)). The saturation magnetization (
Elemental analysis by EDX Spectroscopy revealed that the surface composition (
Elemental analysis by energy dispersive X-ray spectroscopy (EDX) of magnetite and MHDHA.
The point zero charged at varied pH (
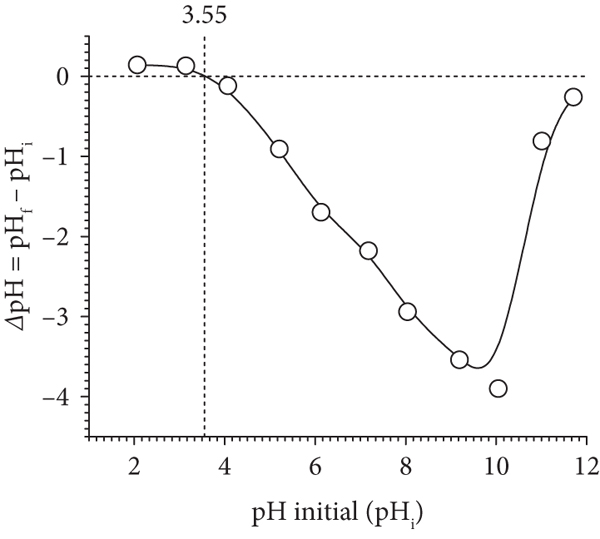
(a) Determination of the point of zero charge (
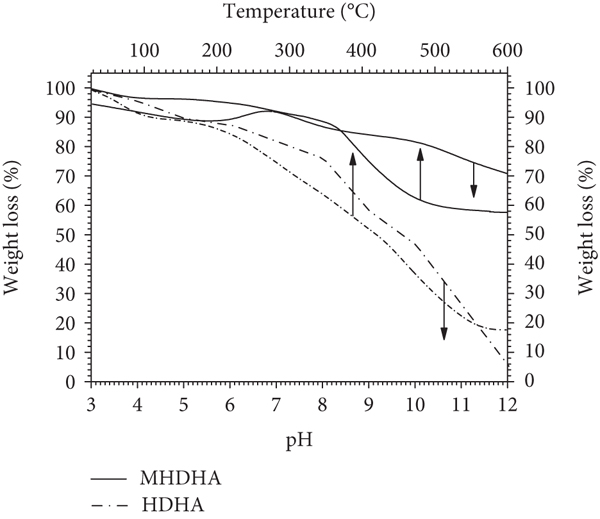
The comparison of the medium pH stability of MHDHA and HDHA in solid form is presented in Figure 3(b) (left
3.2. Effect of pH and Ionic Strength on Pb(II) Adsorption
pH is essential in adsorption studies because it affects the binding of Pb(II) to MHDHA. Medium acidity can affect the surface charge of the sorbent and the structure of the sorbate speciation. The adsorption of Pb(II) is intimately dependent on medium pH. The Pb(II) adsorbed was increased with increasing medium pH from 2.0 to 5.0 and decreased sharply when pH was above 5.0 (Figure 4(a)). This result was caused by the formation of solid Pb(OH)2 at

(a) Optimum pH of Pb(II) adsorption onto MHDHA; (b) % adsorption of Pb(II) onto MHDHA as a function of pH and ionic strength.
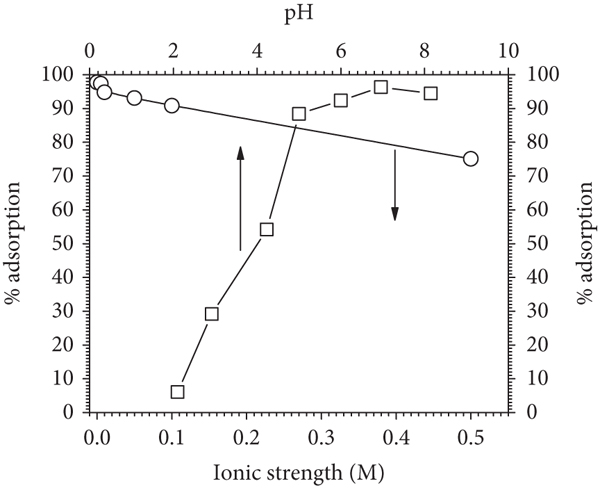
In percentage (%), at pH below the optimum pH, the adsorption was significantly affected by the pH. However, at pH 5.0–8.0, the % adsorption was relatively constant and more influenced by the ionic strength (Figure 4(b)). As shown, the high ionic strength has an effect of reducing the adsorption of Pb(II) on MHDHA. In literature, the low adsorption of divalent metal cations has been attributed to different factors [54]: (1) adsorbent active site blocking by salt, (2) repulsion between free positive-charge adsorbate and positively charged of adsorbent surfaces, (3) competition between positively charged species (H+ and Pb2+) and free adsorbate to the sorbent active sites, (4) lower formation of complexes/chelate with metal ions due to protonation of surface functional groups, and (5) combination of several of these factors. Considering the results, the adsorption of Pb(II) onto MHDHA seems to be affected by the first and third factors due to the ion exchange mechanisms between Pb(II) and MHDHA active sites. Previous works reported that the FT-IR spectra were applied to characterize the changes of the functional group in metal ion adsorption [55–57]. The FT-IR spectra of MHDHA before (Figure 1(b)) and after (Figure 5(c)) Pb(II) adsorption show the disappearing peaks at 1700 and 1200 cm−1 corresponding to the interaction between Pb(II) and carboxylate sites of MHDHA. The similar result of the FT-IR study on the Pb(II) interaction with the HA’s active site has been reported by researchers that indicate the ion exchange mechanism [9–11].

Regeneration study of MHDHA and pH change in each cycle (a); diffractogram of MHDHA (b); and FT-IR spectra after the first (MHDHA-Cy1), third (MHDHA-Cy3), and fifth cycle (MHDHA-Cy5) adsorption-desorption process (c); comparison of morphology and composition by SEM-EDX of original MHDHA (d); and that of MHDHA-Cy5 (e).




3.3. Adsorption Isotherm Study
To optimize the performance of MHDHA, it is essential to establish the isotherm parameters of HDHA. Various isotherm models have been used to comprehend the isotherm parameters (adsorption capacity, equilibrium constant, and adsorption energy), i.e., Langmuir by plotting
Figure 6(a) shows the effect of initial concentration (

(a) The removal uptake of Pb(II) as the function of initial concentration (


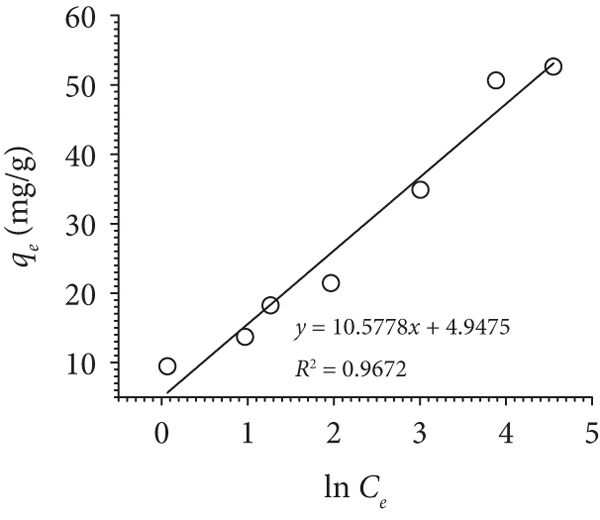

Isotherm parameters of Pb(II) adsorption onto MHDHA.
The comparison of the monolayer sorption capacity (
Comparison of Langmuir sorption capacity (
The important features of the Langmuir model can be expressed in terms of dimensionless constant sorption intensity,
The adsorption Pb(II) onto MHDHA was also evaluated by the Freundlich model to analyze the parameters associated with multilayer sorption. The value of
The Temkin isotherm explains the adsorbate-adsorbent interaction based on the assumption that there is an indirect relationship between the adsorption energy and the adsorbate-adsorbent interaction [60, 61]. As exhibited in Table 3, the value of the Temkin constant,
The D-R isotherm is possible to analyze the high degree of regularity [74]. The D-R theoretical isotherm saturation capacity,
3.4. Adsorption Kinetics Study
Investigation of the rate-controlling Pb(II) adsorption onto MHDHA was studied by the Lagergren pseudo-first-order (equation (6)) [77] and Ho pseudo-second-order (equation (7)) [78] kinetics models. In addition, the correlation of adsorption rate constant (
The adsorption rate of Pb(II) onto MHDHA was studied in time intervals of 5–180 min as shown in Figure 7(a). The Pb(II) ions were rapidly uptake in the first 20 min and slowly continue increasing up to 90 min reaction. The slow adsorption stage was caused by the smaller differences in concentration gradients that finally attained the equilibrium condition after 120 min. An increase in Pb(II) adsorption was observed in MHDHA due to the probability of Pb(II) occupying a vacant site on the adsorbent, where there was plenty of unsaturated vacant sites that resulted in rapid initial adsorption. However, as time continues, vacant sites get saturated therefore adsorption performance of MHDHA decreased equilibrium condition. The adsorbed Pb(II) at equilibrium was found to be 52.47 mg/g under the following experimental condition: 25 mL of solution at 298 K using an adsorbent dosage of 20 mg with an initial concentration of 25 mg/L at optimum pH 5.5.

(a) Profile of Pb(II) uptake onto MHDHA as a function of time and adsorption kinetics plot of 25 mg/L (0.12 mmol/L) Pb(II) onto 20 mg MHDHA by (b) Lagergren, (c) Ho, (d) proposed Santosa, and (e) proposed RBS kinetics model.




The application of experimental data to equations (5)–(8) and the calculated values of corresponding parameters are shown in Figures 7(b)–7(e), respectively, and Table 5. Compared to the Lagergren pseudo-first-order (Figure 7(b)), Santosa (Figure 7(d)), and RBS (Figure 7(e)) models, Pb(II) sorption kinetics were better described by the Ho pseudo-second-order model (Figure 7(c)), whose correlation coefficients (0.9999) were higher than those of the kinetics model. The calculation of
Kinetics parameters of Pb(II) adsorption onto MHDHA.
The low
3.5. Regeneration Studies
The regeneration studies represent the probable reusability of MHDHA as an adsorbent. The MHDHA adsorbent was only a loss of less than 15% of % adsorption after reuse for five successive cycles (Figure 5(a)). The adsorption-desorption percentage proves that the MHDHA possesses a virtuous regeneration ability. To strengthen the claims, the XRD (Figure 5(b)), FT-IR (Figure 5(c)), and SEM-EDX (Figures 5(d) and 5(e)) analyses show that there was no significant change in the crystallinity, functional group, morphology, and composition of MHDHA adsorbent, respectively, after five consecutive cycles of the adsorption-desorption process. The unobserved crystallinity damage implies that there is no significant change in the magnetic strength value and makes the HDHA remain easy and quick to separate by an external magnet. Therefore, the MHDHA is potentially efficient, economically effective, and easily separable adsorbent for lead-contaminated wastewater treatment.
The measurement of pH solution in each adsorption cycle showed that the pH was decreased constantly from optimum pH (
4. Conclusions
The green horse dung HA (HDHA) has been extracted and characterized by UV-Vis spectrophotometer, FT-IR, and total acidity. Functionalization of magnetite by HDHA has been successfully prepared by coprecipitation procedures based on FT-IR, XRD, TEM, SEM-EDX, TGA, and VSM characterization. The functionalization process improves the stability from 96% of original HDHA dissolved to less than 30% HDHA dissolved after magnetite functionalization at pH 12. Also, the MHDHA has only 10% weight loss compared with the original HDHA (88% weight loss) at 600°C. The MHDHA was easy to retract by an external magnet after the adsorption process within a few minutes with the 62.95 emu/g magnetic strength without additional process, i.e., centrifugation or separation by the filter paper. The adsorbed Pb(II) onto MHDHA was influenced by the pH, and the ionic strength with the adsorption rate was rapidly uptake in the first 20 min and attained equilibrium in 120 min. The kinetics data of the adsorption rate was agreed well with the Ho pseudo-second-order model. The removal uptake of Pb(II) ions increased when the initial concentration was increased and fitted with the Langmuir isotherm model. The adsorption capacity of the Pb(II) uptake was
Footnotes
Data Availability
The raw data was generated at the Department of Chemistry, Universitas Gadjah Mada. The data supporting the findings of this study may be made available from the corresponding author upon request.
Conflicts of Interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Acknowledgments
R.B. gratefully acknowledges the LPDP (Indonesia Endowment Fund for Education) BPI Program for the financial support of this work and the Ph.D. scholarship (KET-3/LPDP.3/2018). B.R. thanks Universitas Gadjah Mada (UGM) for partially financing this work through the RTA 2020 Program (Grant number: 607/UN1/DITLIT/DIT-LIT/PT/2020).
