Abstract
Phosphorus (P) is one of the key limiting factors for the growth of forests and their net primary productivity in subtropical forest ecosystems. Phosphorus leaching of the forest soil to the catchment and groundwater in karst region is the main source of water eutrophication. Strong P sorption capacity of minerals is generally assumed to be a key driver of P leaching in subtropical ecosystems which varies among different soil types. Here, we estimated P adsorption capacity of the O/A and AB horizon in both limestone soil and red soil of subtropical forests by fitting the Langmuir and Freundlich isotherm to investigate the potential environmental risks of P. The maximum P sorption capacity (
1. Introduction
Phosphorus is one of the key limiting factors for the growth of forest trees and their net primary productivity in natural forest ecosystems, and it exists mainly in the form of organic P (Po) and inorganic P (Pi) in primary and secondary minerals. Phosphorus that can be directly absorbed and used by plants is mainly phosphate in the ionic state [1]. Due to the sorption and fixation of P by soil minerals, phosphate accounts for only a few fraction of the soil P pool, resulting in a generally low availability of P in soils [2, 3]. This is especially the case in subtropical forest ecosystems, due to a high degree of soil weathering and more pronounced fixation of P by iron and aluminum oxides. Most of P in soil is fixed in a stable form that is difficult to be absorbed and utilized by plants, so P limitation in subtropical forest ecosystems is more severe than in other ecosystems [4]. The input of P in forest ecosystems is mainly from slow weathering of the parent bedrock [5] and in a shorter time scale from the decomposition of litter and mineralization of organic matter. The P output is mainly from the sorption and utilization of P by plants, P loss due to leaching, and surface loss due to surface runoff and soil erosion. Soil P fractions and available P are controlled by processes including precipitation-solubilization of phosphate, mineralization-immobilization by microorganisms, and adsorption-desorption of adsorbed Pi, which in turn affects the supply of soil available P and the risk of P leaching. Therefore, P transformation in forest soils is an important process in the P cycle, which has strong ecological and environmental implications. In different soil types, P fixation capacity is regulated by environmental physicochemical factors, and the surplus P may be leached into water with runoff, causing water eutrophication [6, 7]. Phosphorus speciation in soil determines P availability and P leaching. Soil DPS is an important predictor of soil P mobility and effectiveness, which integrates the soil P fixation capacity and P content, and can be applied to assess the soil P environmental threshold as well as to determine the risk of soil P loss [8, 9]. The larger the DPS, the higher the soil contains dissolved state, the lower the P sorption capacity, and the higher the risk of soil P leaching.
In this study, we investigated the P adsorption and potential environmental risk in limestone and red soils which are the main soil types in subtropical forests of China. The Langmuir equation and Freundlich equation were used to fit the P sorption curve, and
2. Materials and Methods
2.1. Study Area
The study area is located in Guilin City, South China, which is located at low latitude and belongs to the subtropical monsoon climate zone with mild climate, abundant rainfall, and basically the same season of rain and heat; the average annual temperature is about 19.1°C, the average annual rainfall is about 1887.6 mm, the average annual relative humidity is about 76%, and the average annual sunshine duration is about 1447.1 hours. In this study, Ludiyan (25°13
Physiochemical properties in soil of O/A and AB horizons of the red soil and limestone soil.
Note: (1)
2.2. Sample Collection
Three plots within
2.3. Soil Phosphorus Isothermal Adsorption
Eight soil samples of 2.0 g were weighed into 50 mL centrifuge tubes, and 25 mL of 0.01 mol·L-1 CaCl2 solution containing 0, 10, 20, 40, 80, 120, 150, and 200 mg P·L-1 of P was added, along with 2 drops of toluene to inhibit microbial activity. After shaking at room temperature for 24 h at 25°C, the samples were separated by centrifugation at 4000 r·min-1 for 15 min, and the supernatant used for analyzing the concentration of P was filtrated and aspirated. To develop the blue color, molybdenum blue reagent was added, and the volume was made up with deionized water. After 30 min, the absorbance was read at 880 nm using an enzyme-labeled instrument [10].
The phosphorus adsorption was calculated as follows.
The Freundlich and Langmuir equations were used to fit the soil P adsorption. In general, the Langmuir equation describes the physical sorption of a single molecular layer, while the Freundlich equation assumes that the sorption between the adsorbent molecules and the adsorbent should be a nonuniform sorption of multiple molecules. Both Langmuir and Freundlich equations are suitable for describing soil P isothermal sorption curves, but the fit varies depending on soil type.
The Freundlich equation is given by
The Langmuir equation is given by
The MBC is calculated by the following equation:
Soil phosphorus sorption index (PSI) is calculated by the following equation [11]:
The Mehlich-3 extractant was used to extract P (PM3), iron (FeM3), and aluminum (AlM3) from the soil to calculate DPSM3 [12]. The concentration of the soil-available P (POlsen), water-soluble P (PCaCl2), calcium-related P (PHCl), and iron-aluminum-related P (Pcitrate) was determined by using different extractants to calculate the corresponding DPSOlsen, DPSCaCl2, DPSHCl, and DPScitrate [12]. PCaCl2 is a water-soluble P that can mimic the salt status in soil solution, is positively correlated with bioavailable P in runoff or leachate water, and is an important indicator for assessing P leaching [3, 4]; PM3 and POlsen are both soil bioavailable P forms that are closely related to plant P uptake, and Pizzeghello et al. [13] found a significant correlation between the two P forms, where 58% to 98% of POlsen can be converted to water-soluble P [14], and the combination of POlsen and PCaCl2 can be used to evaluate P availability in environmental soil solutions [15]. Pcitrate is the P associated with Fe and Al and is an important pool for supplying bioavailable P in acidic red soils. Most of the PHCl is associated with calcium phosphate salts mainly including insoluble P such as octacalcium phosphate and decacalcium phosphate. In limestone soils, P sequestration is achieved mainly by precipitation with calcium phosphate or coprecipitation with carbonate [16].
Degree of phosphorus saturation (%) was calculated as follows [17–19]:
2.4. Statistical Analysis
The data in the study presented as
3. Results and Discussion
3.1. Soil Phosphorus Isothermal Sorption Curve
The isothermal sorption curves of both soil P were well fitted by Langmuir (

Fitting curve of (a) Langmuir and (b) Freundlich model for the O/A and AB horizons of the red soil (RS) and limestone soil (LS).

Parameter of adsorption isotherm fitting curve and PSI of the O/A and AB horizons of the red soil (RS) and limestone soil (LS).
3.2. Soil Phosphorus Adsorption Saturation
Extractable P concentrations with different extractants in red and limestone soils are shown in Figure 2. In both soils, the concentration of PCaCl2 was low (red soil: 1.56-1.72 mg·kg-1 and limestone soil: 2.55-2.58 mg·kg-1) (Figure 1); the PCaCl2 concentration in limestone soil was significantly higher than in red soil. In red soils, the concentration of PM3 (2.49-7.36 mg·kg-1) was higher than that in limestone soils (0.32-1.32 mg·kg-1); in the O/A horizon, the concentration of POlsen in red soils (4.99 mg·kg-1) was significantly higher than that in limestone soils (2.84 mg·kg-1), while there was no significant difference between the two soils in the AB horizon. In red soils, Pcitrate was the dominant P form, and in the O/A horizon, the content of Pcitrate (12.22 mg·kg-1) was not significantly different from PHCl (11.45 mg·kg-1), but significantly higher than other extractable P contents; in the AB horizon, the content of Pcitrate (10.08 mg·kg-1) was significantly higher than other extractable P contents. In limestone soils, PHCl was the dominant P form, and its content in the O/A horizon (9.66 mg·kg-1) was significantly higher than that of other extractable P. In both soils, except for Pcitrate in limestone soils, all other P fraction concentrations showed
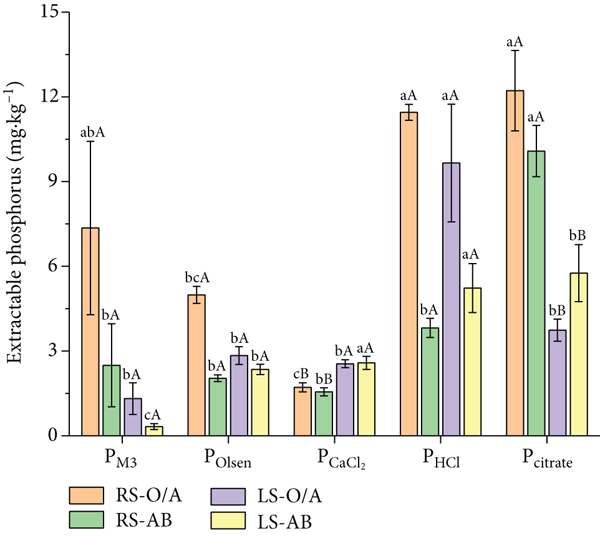
Distribution of extractable P in the bulk soils of O/A and AB horizons of red soil (RS) and limestone soil (LS). Different lowercase letters indicate significant differences (
In the O/A horizon, the values of DPSM3, DPSOlsen, DPSHCl, and DPScitrate in red soils (0.56%, 0.44%, 1.00%, and 1.06%, respectively) were higher than those in limestone soils (0.19%, 0.26%, 0.86%, and 0.26%, respectively), while the values of DPSCaCl2 (0.15%) were lower than those of limestone soils (0.23%); in the AB horizon, the values of DPSCaCl2, DPSHCl, and DPSOlsen were higher in limestone soils (0.25%, 0.50%, and 0.23%) than in red soils (0.16%, 0.39%, and 0.21%) (Figure 3).
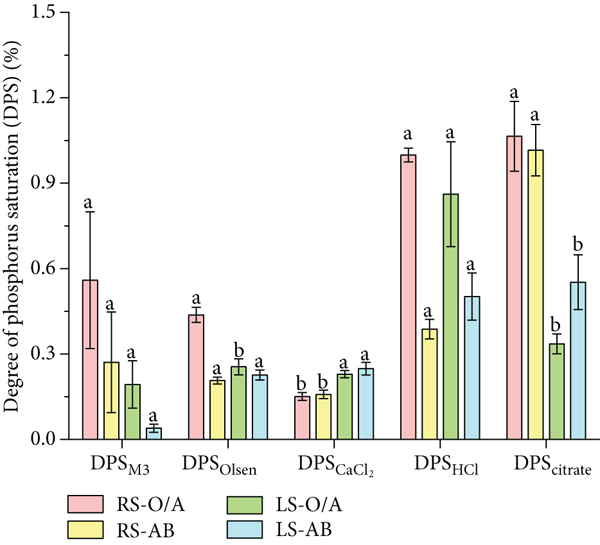
Degree of P saturation in the O/A horizon and the AB horizon of red soil (RS) and limestone soil (LS). Different lowercase letters indicate significant differences (
3.3. Sorption Characteristics and Release Risk of Soil Phosphorus
In this study, the
In the O/A horizon, PM3, POlsen, and Pcitrate had higher dissolved states in red soils, and in the AB horizon, limestone soils had higher levels of POlsen, PHCl, and PCaCl2. POlsen was at deficient to very deficient levels in both soils, indicating that bioavailable P was low. The DPS values in all soils in this study were less than 1.10%, which is lower than the DPS values (1.40% to 7.20%) in forest soils after revegetation of fire-burned sites [22, 23], indicating that the forest soils in this study are less disturbed by humans and have a lower risk of soil P leaching. Comparing the two soils, the values of DPSM3, DPSOlsen, DPSHCl, and DPScitrate in the red soil O/A horizon were higher than those in the limestone soil. In the AB horizon, all but DPScitrate and DPSM3 in the red soil were lower than those in the limestone soil (Figure 3). Combined with the small difference in PSI between the two soils, it indicates that the P fixation capacity of two soils was at a similar level. The DPS is considered as an indicator to identify soils with a high risk of P release [24, 25]; the magnitude of its value is influenced by a combination of soil physicochemical properties [26–28]. The threshold value is not uniform, and some studies have shown a significant impact on the quality of the water environment when the DPS is greater than 15.00% [29]. The others have shown that, when the DPS exceeds 25.00%, soils tend to have high levels of desorbable P and the risk of P leaching is greater [30].
4. Conclusion
Comparing the
Footnotes
Data Availability
All the data used to support the findings of this study are included within the article.
Additional Points
Highlights. Phosphorus (P) sorption and environmental risks of subtropical forest soils differ with lithology. The P sorption and buffering of red soils were higher than those of limestone soils. Compared to limestone soil, red soil has lower potential environmental risk of P leaching.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Acknowledgments
This work was supported by the Natural Science Foundation of Guangxi (Grant Numbers 2018GXNSFAA281350, 2020GXNSFAA238034, 2017GXNSFBA198162, and 2020GXNSFBA159029) and the Hundred Overseas Talents Introduction Plan of Colleges and Universities in Guangxi.
