Abstract
Magnetic composite fabricated from polyaniline and Fe3O4-hydrotalcite (Pan/MHT) was successfully applicated for removal of methyl orange (MO) from wastewater. The structure and properties of Pan/MHT were characterized by Fourier-transform infrared spectroscopy, scanning electron microscopy, X-ray diffraction, vibrating sample magnetometer, and Brunauer-Emmett-Teller adsorption isotherm. Adsorption kinetic results indicated that the adsorption process followed pseudosecond-order kinetic model (
1. Introduction
Dyes are widely used in many fields, such as textile, cosmetic, pigment, paper industry, dyeing, or tanning industry. In particular, textile industry wastewater contains a lot of dyes with large molecule structure including the azo group which is sustainable and difficult to decompose [1]. The presence of organic dyes is a main cause of environmental pollution problems because they are nonbiodegradable; so, an effective method is necessary to remove them from wastewater sources. It is estimated that 10-15% lost in the effluent during the dyeing process [2]. If they are not promptly treated strictly before releasing to the environment, they can destroy the ecosystem of aquatic organisms and threaten the self-cleaning capacity of the receiving source and the human life.
In recent years, there are many applications for treatment such as membrane filtration, redox, electrochemical, adsorption, and biological methods. Among them, adsorption is the popular method because of its efficiency and economy. The adsorbents which were widely used to remove azo dyes are either inorganic such as activated carbon [3–6], hydrotalcite [7–10], diatomite [11–15] and bentonite [16–19], or organic such as chitosan, alginate, polysaccharide, and polymer materials [20–31] and also hybrid and composite material [32–39]. However, the most challenge for the above adsorbents is the low adsorption capacity and efficiency. Therefore, it is necessary to research and develop new material with good adsorption capacity to treat water contaminated with organic dyes to meet economic efficiency and save costs.
Hydrotalcite (HT) with the general formula is
Although both polyaniline (Pan) and HT have high adsorption capacity and have been widely used in water treatment. The most important problem is the ability to recover and reuse them. Filtration is an easy method and is widely used but it’s not so effective and fast as using the magnetic field. When combined with a magnetic particle Fe3O4, the composite or hybrid from HT will have a magnetic property. As a result, the composite produces a constant suspension in an aqueous solution and is immediately separated from the external environment. On the other hand, modification of hydrotalcite including investigation of magnetic Fe3O4/HT nanocomposites (MHT) may have great potential for photocatalysis and environmental remediation [43].
As a conductive polymer, polyaniline (Pan) meets the requirements for environmental treatment applications as both an adsorbent and a photo catalyst according to the following characteristics: high adsorption area, flexible bandgap, and steady−state of activation [44]. Pan nanofiber is an effective adsorbent in studying the removal of methyl orange (MO) and black [45, 46]. Pan is also fabricated as a nanotube and nanoparticle, and they all have high efficiency for removal of MO and crystal violet as well as methylene blue [38, 47–51]. Pan nanoparticles give the highest adsorption capacity due to their high surface area of 1083 m2/g. Besides, it was used as an efficient adsorbent to remove for crystal violet, and methyl orange with the maximum adsorption capacity reaching 245 and 220 mg/g, respectively. In addition, Pan acts as a photo catalyst thanks to its electrical conductivity to increase protonation. Oxidation reactions occur on the catalyst surface, decomposition of the pigment produces intermediate compounds, and the final products are CO2, SO4 2−, NO3 −, and NH4 +. Therefore, the fast charge generation and slow charge recombination lead to enhanced photo catalytic activity.
In summary, both Pan and MHT have high efficiency in the removal of azo dye. However, studies on the adsorption applications of Pan are limited because Pan has a low surface area resulting in low adsorption efficiency. Therefore, many researches are carried out to composing Pan particles with other materials to enhance the adsorption capacity by various synthesis methods. Another way, many reports have shown that MHT material has been suitable for an adsorbent. So, the combination of Pan and MHT to create a Pan/MHT hybrid or composite material has great potential, and it is still not widely exploited for adsorption, especially in wastewater treatment to adsorp dyes (MO, Congo red, ...).
The structure of the hydroxide porous layer and the ability to exchange ions in MHT combined with Pan will give good capacitive properties and high environmental sustainability, because of the special structure that induces ion exchange, restructuration, and the large surface area, so that Pan/MHT can “trap” the MO dye molecules into its structure, recover MO molecules, and remove them from wastewater. In addition, by the restructuring ability, the Pan/MHT material can be desorped for reuse, easily recovered by magnets, saving costs.
For the above advantages of Pan/MHT composite, in this paper, we synthesized Pan/MHT composite with various mass ratios of
2. Experimental Section
2.1. Materials
All the used reagents (Zn(NO3)2.4H2O, Al(NO3)3.9H2O, FeCl3, FeCl2, NaOH, HCl, aniline, ammonium persulphate) were analytical grade reagents. Methyl orange (MO) is an azo dye, with the chemical formula C14H15N3O3S, and molecular weight is 305.35 g/mol and is often used as an indicator. In this work, MO was used to simulate industrial wastewater in order to evaluate the adsorption capacity of Pan/MHT.
2.2. Preparation of Magnetic Hydrotalcite (MHT)
MHT was preparated by coprecipitation method in the presence of Fe3O4 (Figure 1). An accurate amount of 0.1 g Fe3O4 was dispersed in distilled water (10 mL). A mixture of Zn(NO3)2.4H2O and Al(NO3)3.9H2O with Zn2+/Al3+ molar ratio of 2.0 was dissolved in 100 mL distilled water and then coprecipitated into the Fe3O4 dispersion. The pH of the reaction solution was kept at around

Schematic illustration of synthesis steps for MHT.
2.3. Preparation of Polyaniline/MHT (Pan/MHT)
Pan/MHTs were fabricated using in situ oxidative polymerization (Figure 2). An accurate amount of 0.2 g MHT was dispersed in a distilled water (40 mL) and alcohol (10 mL) mixture. 0.5 mL of Tween 80 5% was added to the mixture and then sonicated in 30 minutes to acquire a well−dispersed colloidal suspension. The desired amount of aniline dissolved in 25 mL of HCl 0.1 M. The mixture was stirred for 1 hour for complete mixing at 4°C. An accurate amount of APS (

Schematic illustration of synthesis steps for Pan/MHT.
2.4. Characterization of the Composite Pan/MHT
The Fourier transform infrared (FT−IR) spectra of Pan, HT, Fe3O4, MHT, 0.5 Pan/MHT, 1 Pan/MHT, and 2 Pan/MHT composites were obtained using the KBr method on a Bruker Tensor 27 spectrometer, operated in 400−4000 cm−1 region, at 2 cm−1 resolution, and for 32 scans.
Powder X−ray diffraction patterns of Pan, MHT, 0.5 Pan/MHT, 1 Pan/MHT, and 2 Pan/MHT composites were obtained using a Bruker D8 Advance diffractometer with CuKα radiation (1,5406 Å) at room temperature in the atmospheric pressure. Data were collected in the range of 2 theta from 5 to 70° with a step of 0.02° and a scanning rate of 1°/min.
The structure and morphology of MHT, Pan, and Pan/MHT composites were observed by scanning electron microscopy (SEM). SEM observations were carried out using a Thermo Scientific E−SEM instrument.
The magnetic property of MHT and series of Pan/MHT were analyzed with a vibration sample magneparameter (VSM) method using a Lake Shore Crytotronics device.
The specific surface area and average pore size of Pan/MHT and MHT were determined by nitrogen adsorption t 76 K. The Brunauer−Emmett−Teller (BET) method was employed for the corresponding calculation.
2.5. Batch Studies
The adsorption experiments were performed using the batch equilibrium technique in aqueous solutions at room temperature. Duplicate 20 mg Pan/MHT samples were equilibrated by shaking with 25 mL of MO solution with initial concentrations (
The adsorption of MO on Pan/MHT could be calculated from the difference between the initial concentration (
where
2.6. Desorption and Reusability of Pan/MHT
For the reusability study, 0.1 g of Pan/MHT was used repeatedly for the adsorption of MO. An adsorption process was performed by adding 0.1 g Pan/MHT in 150 mL of MO at 20 mg/L and shaking for 24 hours to equilibrium adsorption. After that, the Pan/MHT was separated from the MO solution by using a magnetic bar, and the MO solution was analyzed by UV-VIS spectrophotometry to determine the MO adsorption capacity.
The desorption process was performed by dispersing Pan/MHT previously used in the adsorption process in 100 mL of HCl 1.0 M which promotes MO desorption. The desorption was performed in about 1 hour and then separating Pan/MHT by using a magnetic bar. The MO concentration in HCl was also analyzed by UV-VIS spectrophotometry. The cycles of reuse of the same Pan/MHT sample were carried out for a total of 4 cycles. The adsorption capacity of MO and desorption MO concentration was calculated for each cycle of reuse.
3. Results and Discussion
3.1. Characterization of Composite Particles Polyaniline/Mag−HT (MHT)
3.1.1. Fourier Transform IR (FT−IR)
The FT–IR characteristic of MHT as spectrum in Figure 3 shows that a strong absorption band at 1381 cm–1 is assigned for elongation vibration –NO3 which is also present for the interlayers of MHT. The weak adsorption at about 1640 cm–1 is the vibration of –OH in the H2O structure attached on the interlayer. The wide band at about 3463 cm–1 is characteristic for elongated vibration of –OH groups from M–OH (Al–OH, Mg–OH, and Fe–OH), lattice, and alternating layer of water: 617, 447, and 409 cm–1 due to elongated vibration of M–O (Al–O, Zn–O and Fe–O). Thus, Fe3O4 has successfully combined with HT material to fabricate MHT hybrid, and the FT −IR spectrum is consistent with that of Sulistyaningsih et al. [52].

FT−IR spectrum of Fe3O4 (a), MHT (b), and HT (c).
The FT–IR infrared spectrum shown in Figure 4 clarifies the nature of the interaction between Pan and MHT in Pan/MHT materials. In the infrared (MIR) region, the peaks between 4000 and 1800 cm–1 are the prolonged vibration region of the O–H bonds in the hydroxyl group and N–H in the amine group. For MHT, the peak at 3447 cm–1 is the tensile vibration of –OH that binds the hydroxyl group surface in the structure. The adsorption peaks below 800 cm–1 at about 786 cm–1 and 618 cm–1 are characteristic for M–O bond (Al–O, Zn–O), and the peak at about 429 cm–1 is assigned for Fe–O.

FT-IR spectrum of polyaniline (a), 2 Pan/MHT (b), 1 Pan/MHT (c), 0.5 Pan/MHT (d), and MHT (e).
Pan characterized with the strong absorptions at 3439, 2941, 1569, 1495, 1301, 1130, and 875 cm−1, which was the evidence of the emeraldine form of polyaniline [53–55]. The vibration at about 3220 cm−1 is assigned for protonated radical cation N − H+·, and the vibration at about 3443 cm−1 is assigned for the N−H stretching. The characteristic band at 1515 cm−1 was assigned to both C=N and C−N stretching of quinoid ring structure, and the band at 1481 cm−1 was attributed to both C=C and C−N of benzenoid ring structure. All of these absorption peaks were characterized for the inplane bending vibration. The band at around 1301 cm−1 indicated the vibration of the C − N stretching of benzenoid ring, and the one at around 875 cm−1 was the C − H out of plane bending vibration parade-substituted benzene indicating polyaniline polymer formation. In addition, the strong band at 1130 cm−1 was assigned for the C − N=C group attached on the quinoid ring.
3.1.2. X-Ray Diffraction
The XRD of the MHT, Pan, 0.5 Pan/MHT, 1 Pan/MHT, and 2 Pan/MHT are measured in the range of 2θ from 5° to 70°, and the spectrums are shown in Figure 5. The XRD spectrum of MHT from Figure 3(e) has characteristic peaks of HT and some characteristic peaks of Fe3O4 at

X-ray diffraction spectrum of polyaniline (a), 2 Pan/MHT (b), 1 Pan/MHT (c), 0.5 Pan/MHT (d), and MHT (e).
The results from the figure show the characteristic diffraction peak of Pan at
3.1.3. SEM Images
The morphology of the material was observed by SEM images in Figure 6. Pan/MHT morphology was observed, in which the distribution of Pan and MHT differed markedly in the proportions. The structural morphology of MHT is a porous material consisting of multilayers, and Pan has the structure of a long carbon chain. The distribution of MHT for Pan is different when changing the mass ratio of Pan. This result is completely consistent with the results of structural analysis by significantly reduced X−ray diffraction.

SEM images of MHT (a), polyaniline (b), 0.5 Pan/MHT (c), 1 Pan/MHT (d), and 2 Pan/MHT (e).




3.1.4. Vibrating Sample Magnetometer (VSM) Analysis
The magnetic properties of MHT and series of Pan/MHT were investigated using a vibrating sample magnetometer at room temperature, and the magnetization curves of MHT and series of Pan/MHT were shown in Figure 7. The roughly coincident hysteresis curves show that the grain size of the MHT metal phase is very small, nanometers in size and is a superparamagnetic material. Because MHT is the hybrid material from HT with only 1 wt% Fe3O4 particle, the superparamagnetic characteristic of MHT is the low value of Ms and Mr that are
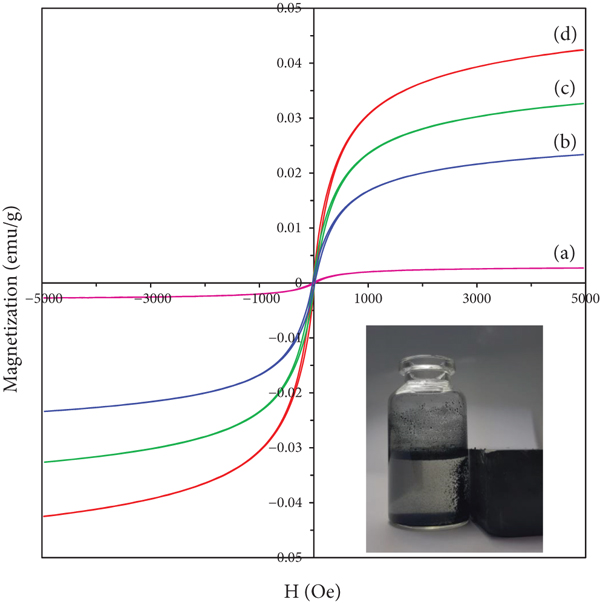
Magnetization vs. applied magnetic field for MHT (a), 2 Pan/MHT (b), 1 Pan/MHT (c), and 0.5 Pan/MHT (d).
Magnetic parameters of MHT and Pan/MHT composites.
The low
3.1.5. Bruanauer–Emmett–Teller (BET)
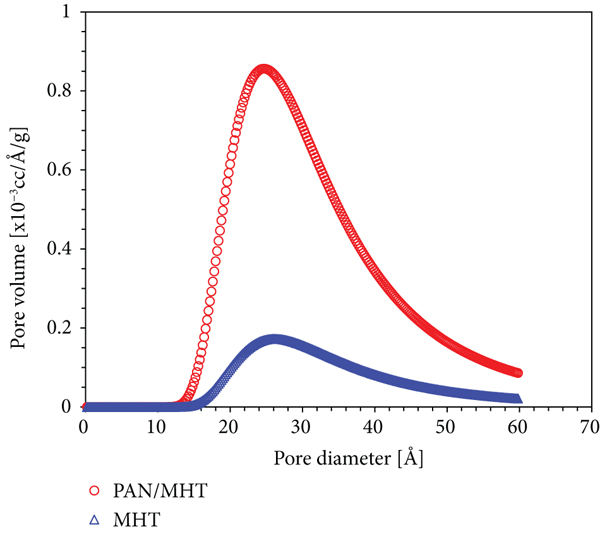
The surface area is well known to be important for adsorption performance. While the pore size of MHT and 2 Pan/MHT is the same which are 26.2 Å and 24.6 Å, the BET surface areas of MHT and 2 Pan/MHT were calculated to be 2.796 m2/g and 15.460 m2/g. The higher surface of 2 Pan/MHT provides more active sites for MO adsorption. This result is consistent with the morphology of MHT and Pan/MHT from the SEM images in Figure 8.
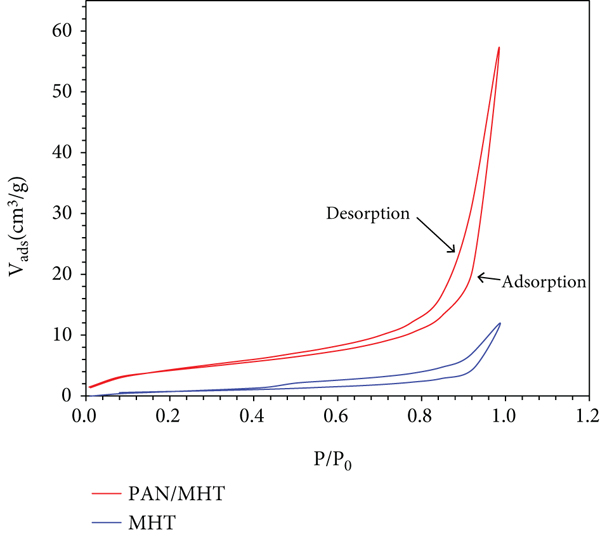
Nitrogen adsorption–desorption isotherms (a) and pore disstribution (b) of MHT and 2 Pan/MHT.
3.2. Effect of Contact Time and Kinetic Adsorption
3.2.1. Effect of Contact Time
Figure 9 shows the effect of contact time of 0.5 Pan/MHT, 1 Pan/MHT, and 2 Pan/MHT with 100 mL of 20 mg/L MO solution. The adsorption amounts of MO increased rapidly in the initial 30 minutes for all samples. Then, the adsorption amounts increased slighter in the next 30 minutes. It becomes slow to attained equilibrium time which is 120 minutes for 0.5 Pan/MHT, 1 Pan/MHT, and 240 minutes for 2 Pan/MHT in the same conditions. The difference of equilibrium time for MO adsorption obtained by various adsorbents can be explained by the MO anion adsorption on the adsorbents by surface and anion exchange mechanism. The adsorbent with the higher mass ratio of Pan/MHT, the more MO can be adsorbed. This result is due to the complex adsorption mechanism of Pan such as surface adsorption, anion−exchange, …, which is consistent with the research of Monika et al. and Wang et al. [58, 59].

Effect of contact time on MO adsorption onto 0.5 Pan/MHT, 1 Pan/MHT, and 2 Pan/MHT.
Kinetic modeling of the adsorption process provides a prediction of adsorption rates and allows the determination of suitable rate expressions characteristic of the possible reaction mechanisms. In this study, the most frequently used models, the pseudofirst−order, the pseudosecond−order, and the intraparticle diffusion model were tested.
Pseudofirst−order kinetics: the linearized from of the pseudofirst−order linearized equation is given as
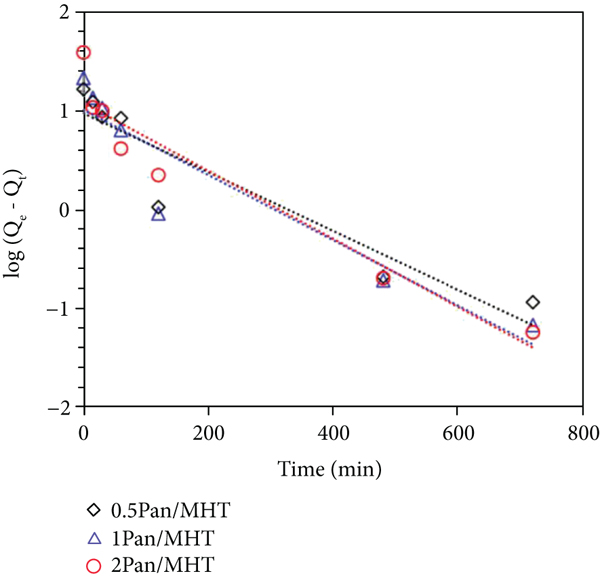
MO adsorption kinetics by 0.5 Pan/MHT, 1 Pan/MHT, and 2 Pan/MHT adsorbents. Pseudofirst−order model (a), pseudosecond−order model (b), and intraparticle diffusion model (c).


Pseudo−secondorder kinetics: the linearized form of the pseudosecond-order equation is given as
Intraparticle diffusion model: the intraparticle diffusion model assumes that film diffusion is negligible, and the only rate-controlling step is intraparticle diffusion in the adsorption process [60]. Hence, the adsorbed amount at time
The kinetic parameters obtained from the application of different models are shown in Table 2. The pseudosecond−order model is the most suitable in describing the adsorption kinetics of MO in all three Pan/MHT composites, based on the correlation coefficient (
Kinetic parameters of MO adsorption on Pan/MHT composite.
3.2.2. Effect of pH on the MO Adsorption
The result from Figure 11 shows that the adsorption efficiency of all three materials decreases gradually according to the pH value of the solution, the efficiency reaches the highest and most stable value at pH from 3 − 4, to pH 5, and the efficiency productivity started to decrease gradually. This is related to the surface area of the material, the presence of hydroxyl groups (OH−) on the surface of the material, and the exchange of protons (H+). This group is both capable of giving and receiving protons according to the pH of the solution when in solid−phase contact. The process of receiving protons on the surface takes place in an acid environment, while proton transfer occurs in an alkaline environment.

Effect of pH on MO adsorption onto 0.5 Pan/MHT, 1 Pan/MHT, and 2 Pan/MHT.
3.3. Adsorption Isotherms
3.3.1. Effect of Initial Concentration of MO on Sorption of MO
The effect of initial concentration on MO adsorption onto 0.5 Pan/MHT, 1 Pan/MHT, and 2 Pan/MHT is shown in Figure 12. The experiment was performed with a constant adsorbent dose of 20 mg/25 mL MO. The adsorption capacity of MO increased when increasing the initial MO concentration. The

Effect of initial MO concentration on MO adsorption onto 0.5 Pan/MHT, 1 Pan/MHT, and 2 Pan/MHT.
The Langmuir isotherm is used to describe the equilibrium between the surface of the solid and the solution and is valid for monolayer adsorption onto a surface with a finite number of identical sites. The Langmuir model equation is given as
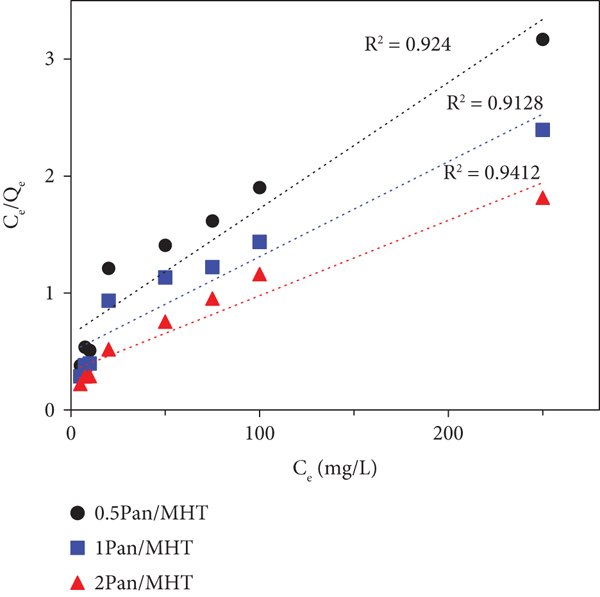
Adsorption isotherms of MO on Pan/MHT composites with Langmuir isotherm model (a), Freundlich isotherm model (b), and Dubinin-Radushkevich isotherm model (c).

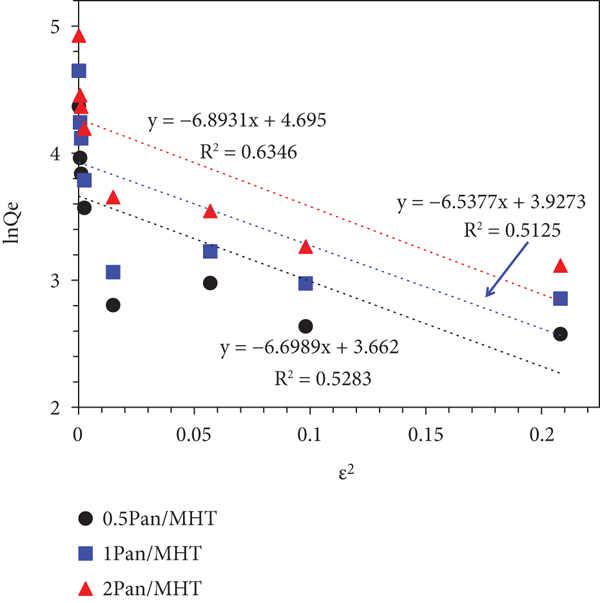
The Freundlich isotherm is an empirical equation that assumes that the adsorprion face becomes heterogeneous during the course of the adsorption process. The linearized form of the Freundlich model is given as
All the correlation coefficients,
Fitting parameters of adsorption isotherms for MO on Pan/MHT composites.
Dubinin-Radushkevich (D-R) isotherm is generally used to describe the sorption isotherms of a single solute system (Figure 13(c)). The D-R isotherm can also help to confirm that the adsorption process was chemisorption or physical adsorption [63]. D-R equation is represented as follows:
The D-R model is mainly used to estimate the average free energy of adsorption (kJ/mol)
3.3.2. Effect of 2 Pan/MHT Dosage on the Sorption of MO
The result from Figure 14 shows the adsorption efficiency (H %) when using a different dose of 2 Pan/MHT (0.01, 0.02, and 0.03 g) and shows the adsorption efficiency increases when increasing 2 Pan/MHT dose.

Effect of adsorbent dose in MO adsorption onto 2Pan/MHT composite.
For all 3 doses of 2 Pan/MHT, MO was immediately adsorbed on 2 Pan/MHT and after 30 minutes, the adsorption efficiency obtained 55.39%, 63.88%, and 65.97% according to use of 0.01, 0.02, and 0.03 g of 2 Pan/MHT, respectively. Then, the adsorption efficiency increase slightly and reached to equilibrium after 240 minutes with the adsorption efficiency obtained 64.43%, 68.59%, and 77.37% according to the use of 0.01, 0.02, and 0.03 g of 2 Pan/MHT, respectively. This result is expected because the increase of adsorbent dose leads to a greater surface area.
The FT–IR spectra of MO, 2 Pan/MHT, and 2 Pan/MHT after adsorption of MO are shown in Figure 15(a). MO is characterized by the peak at 2896 cm-1 for asymmetric CH3 stretching vibration; the peaks at 1520 cm-1 and 1421 cm-1 are assigned for C=C–H in plane CH bend; peak at 817 cm-1 is characteristic of the benzene ring. The peak at 1608.32 cm-1 is assigned for –N=N–, and the peaks at 1201 cm-1 and 1121 cm-1 are assigned for –C–N. The peaks at 697 cm-1, 623 cm-1, and 574 cm-1 are assigned for the stretching vibration -C-S-, and the peak at 1367 cm-1 is assigned for the stretching vibration S=O. These characteristics also exist on Pan/MHT materials after adsorption, demonstrating the presence of MO dye on the material, and this result is consistent with the announcement of Harikumar et al. [65].

FT-IR spectra of MO (red line), 2 Pan/MHT (blue line), and 2 Pan/MHT after adsorption of MO (—) (a), SEM images of 2 Pan/MHT (b), and 2 Pan/MHT after adsorption of MO (c).


SEM images of 2 Pan/MHT and 2 Pan/MHT after adsorption of MO (Figures 15(b) and 15(c)) clearly show the material surface before and after MO adsorption. The surface and pores of 2 Pan/MHT after adsorption of MO are covered with the MO (adsorbed components), and the result is the surface of the material more homogeneous than the original. It is mean that the material samples after adsorption have almost filled gaps.
3.4. Desorption of MO and Reusability of Pan/MHT
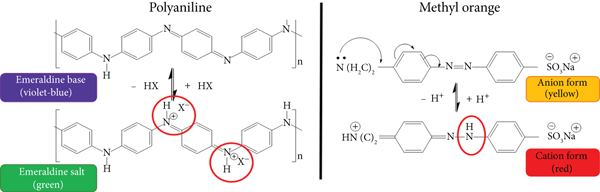
Another parameter that defines the functionality of adsorbent material is its capacity to be regenerated and reused in several adsorption and desorption cycles. For this reason, MO adsorption and desorption experiments were performed with Pan/MHT by using a 0.1 M HCl solution as an eluent. This eluent was chosen because in acid conditions, Pan is in emeraldine salt and is doped with H+ ions, and MO is in its cationic form [66, 67]. Therefore, due to electrostatic repulsions, the MO molecules would be desorbed from the composite structure.
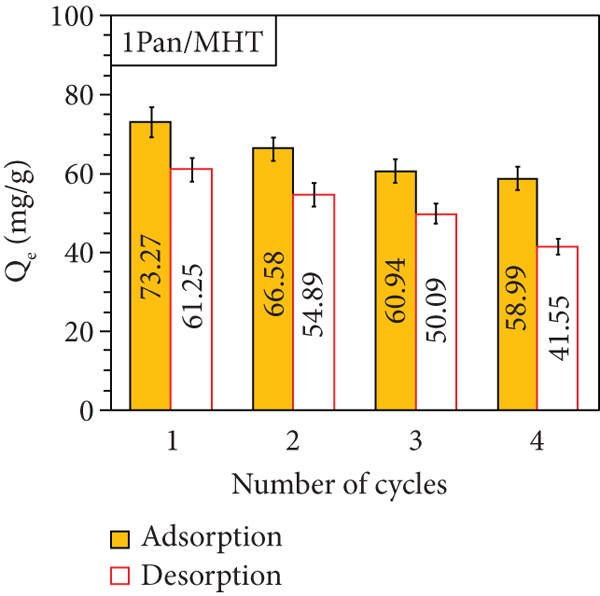
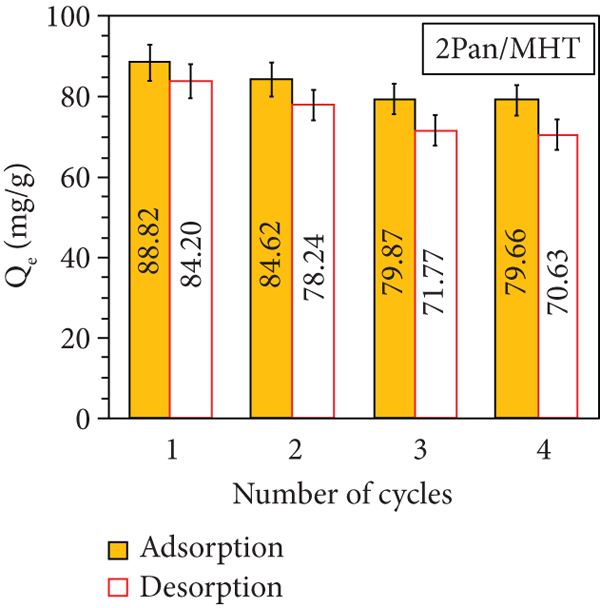
Figure 16 shows the adsorption and desorption efficiency of MO after 4 cycles. The result show that the MO adsorption decreases to 12.86%, 14.28%, and 9.16% according to using 0.5 Pan/MHT, 1 Pan/MHT, and 2 Pan/MHT. The MO desorption from 0.5 Pan/MHT, 1 Pan/MHT, and 2 Pan/MHT also decreases to 21.51%, 19.7%, and 13.57%, respectively. This can be due to the increase of Pan in a composite structure, which enhances both adsorption and desorption of MO on the adsorbent. Thus, Pan/MHT surface can be regenerated without a problem. Although MO is completely desorbed, the adsorption percentage decreases, and this behavior is because of the loss of Pan due to stirring and material management. Therefore, this material can be reused in multiple cycles if it wants to be coupled to a recirculation system.

MO adsorption and desorption cycles with 0.5 Pan/MHT (a), 1 Pan/MHT (b), 2 Pan/MHT (c), and the effect of solution pH on the reusability of 2 Pan/MHT for MO adsorption (d).

The results show that at a low pH 3 − 4, the MO adsorption capacity (H %) obtained over 80% after 3 cycles of reuse. When the solution
3.5. Mechanism of Adsorption
With all the results above, the process of adsorption, recovery, and desorption of MO on Pan/MHT was described as Figure 17(a). The adsorption and desorption mechanism of MO on Pan/MHT are the combination of chemisorption and physical adsorption, which can be illustrated in Figures 17(b)–17(d) and explained as below:
In solution, MO molecule is dissociated into M-SO3- with M is an organic part of dyeing molecule. Pan is a conducting polymer with NH+ group on full backbone that will interact with M-SO3-. The electrostatic interactions are preferably involved in MO adsorption onto Pan particles. Besides, the adsorption experiment studied on Pan/MHT was found to have followed the pseudosecond-order model which indicates the chemisorption mechanism of Pan/MHT The presence of MHT gives the good adsorption of MO because of the anion exchangeable. For this mechanism, the anion associated with the interlayer Pan/MHT is exchanged with MO anionic molecules in solution. It also means that the MO anion will be trapped into the interlayer structure, and it indicates the physical adsorption mechanical of Pan/MHT

Schematic illustration of the adsorption and desorption mechanism of MO on Pan/MHT.


4. Conclusion
The MHT material has been synthesized with 0.1 g Fe3O4 added during the coprecipitation process with the Zn2+/Al3+ salt mixture to create a magnetic MHT material. Then, a series of Pan/MHT composites were successfully synthesized with different mass ratios of 2 Pan/MHT was the best material with high surface area (15.46 m2/g), high Hc (18.56 Oe), and low Ms ( According to the MO adsorption kinetic studies, all of 0.5 Pan/MHT, 1 Pan/MHT, and 2 Pan/MHT composites are suitable with pseudosecond−order model, based on the correlation coefficient ( The The adsorption energy of all 0.5 Pan/MHT, 1 Pan/MHT, and 2 Pan/MHT is 0.273, 0.277, and 0.269 kJ/mol, and all of them are below 8 kJ/mol. This result indicates that the adsorption process of MO on three kinds of Pan/MHT composites is physical adsorption Pan/MHT was also investigated to reuse after desorption of MO in 0.1 M HCl, and the result shows that 2 Pan/MHT can be reused for 4 cycles with The mechanism of MO adsorption onto Pan/MHT composite is either of the surface adsorption or other types of interactions, such as electrostatic interaction, ion exchange, hydrogen bonding, and van der Waals forces
Footnotes
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
Conflicts of Interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Acknowledgments
This work was supported by Kurita Water and Environment Foundation (KWEF) for the award of KURITA Overseas Research Grant 2020 under Grant [20Pvn032-K43]. The authors thank to Institute of Chemical Technology for their warmly analysis equipment support.
