Abstract
Due to the complexity of the internal structure of natural coal and its characteristic of multicomponent, the depth of its methane adsorption potential well is nonuniform, which makes it difficult to accurately evaluate the adsorption capacity of coal. Besides, in order to find out the factors affecting the depth distribution of potential wells in natural coal, this paper calculated the depth and number of potential wells during methane adsorption in coal according to the Langmuir adsorption kinetics process. Coal samples with different metamorphic degrees were tested and analyzed by infrared spectroscopy diffraction technology. The relationship between the structural parameters of functional groups in coal samples with different metamorphic degrees and the distribution of different depths of adsorption potential wells in coal samples was studied. Some main conclusions are as follows: The number of adsorption potential wells at different depths in natural coal with different metamorphic degrees has multipeak distribution characteristics. With the increase of the metamorphic degree of coal sample, the structures such as aliphatic branched chain structure and oxygen-containing functional groups in coal structure break, fall off, and deoxygenate. The relative content of aliphatic hydrocarbons is significantly reduced and condensed into aromatic hydrocarbons and aromatic ring structures. The different types and quantities of functional groups on the surface of coal samples lead to different forces between coal molecules and methane gas molecules, thus affecting the distribution of different depths of adsorption potential wells in coal samples.
1. Introduction
An adsorption potential well refers to the position on the surface of coal where the local potential energy is relatively low and methane can be adsorbed, whose depth is related to the mutual attraction between coal and methane [1–3]. Due to the heterogeneity of natural coal [3, 4], the difference of oxygen-containing functional groups and side chains on the pore surface of coal [5], and the fractal characteristics of pore surface morphology and other reasons [6, 7], the adsorption potential well on coal surface has obvious heterogeneity. Many scholars [8–11] have measured and calculated the potential well depth or the value of adsorption heat when the coal body is adsorbing methane gas by using physical experiments and molecular simulation methods. Results show that the value is concentrated in the range of 0-30 kJ/mol. Nie and Duan’s calculation [12] shows that the potential well depth on the coal surface is basically consistent with the measured data of adsorption heat. Previous experiments [13, 14] have shown that methane adsorption in coal is highly sensitive to temperature variation in the range of 0-270°C. Therefore, according to the existing research, we can speculate that the potential well depth on the surface of coal has obvious nonuniformity, and there are both deep and shallow depths of potential wells that can adsorb methane on the coal surface.
Structural chemistry of coal has been studied. It is believed that the main body of the coal structure is amorphous polymer highly crosslinked in three-dimensional space and the macromolecular structure of coal is composed of many basic structural units with similar but not identical structures. The core part of the basic structural unit is mainly the condensed directional ring. The periphery of the basic structural unit is connected with three alkyl side chains below carbon and various functional groups. The main functional groups are oxygen-containing functional groups, including hydroxyl, carboxyl, methoxy, aldehyde, and carbonyl. Because the atoms of chemical bonds and functional groups remain in a state of continuous vibration, their vibration frequency is similar to that of infrared light. Different chemical bonds or functional groups have different absorption frequencies and will be at different positions in the infrared spectrum. Thus, the information about the chemical bonds or functional groups in the molecule can be obtained. In the midinfrared region of the infrared spectrum, there will be absorption peaks in the wavelength or wavenumber of the absorbed light. The more the wavelength is absorbed, the higher the absorbance, and the stronger the absorption peak. If there are many different absorption peaks in the measured infrared spectrum, it indicates that the sample absorbs many different wavelengths of light. According to the position, strength, and shape of these absorption peaks, the content and distribution of the chemical structure in the sample can be estimated. Besides, different chemical bonds and functional groups in coal structures have different binding abilities with methane molecules. Therefore, in order to analyze the structural reactivity and adsorption performance of coal, it is necessary to comprehensively consider the distribution of aromatic condensation rings, oxygen-containing functional groups, and aliphatic side chains in the coal structure, as well as the variation law of their content with the change of coal metamorphism degree. The influence of functional groups in coal on the adsorption potential well can be explored in this way.
2. Calculation of Potential Well Depth of Coal Adsorption Methane
The essence of gas adsorption on the coal pore surface is the result of mutual attraction between molecules on the coal pore surface and gas molecules. The adsorption potential well is an attribute of the coal body, which refers to the adsorption energy when coal surface molecules and adsorbed gas molecules reach the most stable state. It is related to the diversity of coal pore surface morphology and functional groups. The potential well depth characterizes the stability of the adsorption system composed of coal molecules and adsorbed gas molecules. The deeper the potential well is, the greater the adsorption energy is, and the more stable the adsorption system is.
This experiment refers to the temperature range of natural coal reservoirs and the temperature range of coal-bed methane produced by heat injection. The experimental temperature is set between 30°C and 150°C, and the temperature interval is 20°C. There are 7 groups of isothermal conditions in total. Under each isothermal condition, referring to the gas pressure of natural coal reservoirs, a total of 11 adsorption pressure measuring points with a pressure interval of 0.2 MPa were set within the range of 0.2 MPa~2.2 MPa to carry out the isothermal adsorption experiments. According to the Langmuir equation based on adsorption kinetics, the adsorption capacity of different adsorption pressure sections and the corresponding adsorption pressure were calculated and analyzed by piecewise fitting. The pressure measuring points of each fitting pressure section were 3, and a total of 9 pressure sections were selected. When fitting the data, the Langmuir equation is deformed as follows:
In Formula (1),
In formula (2),
According to formula (1), the adsorption pressure
The
In the formula,
Through the statistical analysis of the calculation results of the potential well depth and the number of potential wells, the columnar diagram of the relationship between the potential well depth and the number of potential wells for methane adsorption on four coal samples can be obtained, as shown in Figure 1. The distribution proportion of potential well depth in coal samples with different metamorphic degrees is counted, and the results are shown in Table 1.

Relationship between potential well depth and number of potential wells for methane adsorption on coal samples.
Statistics of potential well depth distribution in different metamorphic coal samples.
3. Infrared Spectrum Experiment
3.1. Coal Sample Preparation
In this paper, four kinds of coal samples with different ranks were selected for infrared spectroscopy experiments, namely, anthracite, lean coal, coking coal, and lignite. Samples of each coal rank were selected from typical coal-rich areas in North China and Northeast China. Anthracite was taken from 9# coal seam of Sihe Coal Mine in Jincheng City, Shanxi Province; lean coal was taken from 3# coal seam of Sanyuan Zhongneng Coal Mine in Changzhi City, Shanxi Province; coking coal was taken from 8# coal seam of Malan Coal Mine in Taiyuan City, Shanxi Province; and lignite was taken from II# coal seam group of Lingdong Coal Mine in Manzhouli City, Inner Mongolia. The coal samples used in this section are 200 mesh (particle size less than 0.074 mm) powder coal samples, which are obtained by grinding after the potential well depth measurement experiment of each sample.
3.2. Experimental Method
The VERTEX 80v infrared spectrometer produced by German Beuker Company in the Mineral Processing Laboratory of Taiyuan University of Technology is used to test coal samples with different metamorphic degrees. The experimental equipment is shown in Figure 2. The test steps are as follows: (1) The dried coal samples are placed in an agate mortar and thoroughly ground. (2) The high-purity potassium bromide (KBr) was about 100 mg, and a small amount of coal sample was added. The mass ratio of coal sample to potassium bromide was1 : 100, and the mixture was fully ground to 200 mesh. (3) The mixed samples are loaded into the mold of the sample holder. The mold is pressed to 100000 N/cm2 by the tablet press for 1 min to ensure that the formed sample sheet is uniform and there is no damage or leakage. (4) After the infrared spectrometer is preheated, the measurement background is scanned, and the sample frame of the pressed coal sample sheet is placed in the sample room of the infrared spectrometer. The instrument scanned 16 times in total, and the infrared spectrum is obtained after the baseline correction.

VERTEX 80v infrared spectrum analyzer.
4. Experimental Result
4.1. Conventional Characteristics of Coal Samples
Before the quantitative analysis of infrared spectral parameters, the elemental analysis of each coal sample should be carried out according to GB/T476-2001. The analysis results are shown in Table 2. The distribution range of four samples on the whole vitrinite reflectance
Elemental analysis and maceral determination parameters of coal samples.
4.2. Infrared Spectral Characteristics of Coal Samples
Infrared spectra of samples with different coal ranks were obtained after calibrating the data by baseline, as shown in Figure 3. The infrared spectra of each sample were analyzed by peak fitting, and the absorption peak of the infrared spectrum was assigned according to the previous studies [19]. The whole infrared spectra were divided into four parts: the aromatic structure whose infrared wave number is between 700 and 900 cm-1, the oxygen-containing functional group between 1000 and 1800 cm-1, the aliphatic branched chain between 2800 and 3000 cm-1, and the hydroxyl functional group between 3000 and 3600 cm-1. As shown in Figure 4, taking anthracite samples as an example to carry out peak fitting, the fitting correlation coefficients are all greater than 0.99, and the peak distribution follows previous studies [20–22].

Infrared spectra of four coal samples after calibration.

Fitting curve of infrared spectrum of functional groups of anthracite.



4.2.1. Aromatic Structure Parameters
The value of

Relationship between aromatic carbon ratio of infrared parameters and metamorphic degree of coal samples.
In addition to the aromaticity of infrared parameters, AR index parameters were used to characterize the aromaticity of different coal samples and the rank of coal metamorphism degree. The statistical analysis results of the AR index are shown in Figure 6. It can be found that there is a positive correlation between aromatic AR index and coal sample metamorphic grade index (maximum vitrinite reflectance

Relationship between aromaticity of infrared parameters and metamorphic degree of coal samples.
The relationship between aromatic structure condensation degree (DOC) and metamorphism degree index of coal sample (maximum vitrinite reflectance

Relationship between aromatic condensation degree of infrared parameters and metamorphic degree of coal samples.
4.2.2. Fat Structure Parameters
The ratio of methyl to methylene strength (
In the formula,
The relationship between the ratio of methyl to methylene strength (

Relationship between infrared parameters of fat structure parameters and coal metamorphism.
4.3. Structural Parameters of Oxygen-Containing Groups
The structural parameter
Figure 9 shows the variation trend of the C=O group relative to the metamorphism index of the coal sample (maximum vitrinite reflectance

Relationship between infrared parameter
5. Analysis and Discussion
The adsorption of methane gas by coal samples is caused by “van der Waals” force between adsorbate and adsorbent molecules. The different types and quantities of functional groups on the surface of coal samples lead to different forces between coal samples and methane gas molecules, which further affects the distribution of different depths of adsorption potential wells in coal samples. The greater the molecular polarity of the functional groups on the surface of the coal sample is, the higher the force between the functional groups and the methane molecules, the deeper the adsorption potential well of the coal sample for the methane molecules, and the stronger the stability of the coal sample for the adsorption of the methane molecules.
The distribution of adsorption potential well of coal samples with different metamorphic degrees is closely related to the types and quantities of functional groups on the coal surface. Figure 10 shows the relationship between the structural parameters of infrared spectrum diffraction of coal samples with different metamorphic grades and the distribution of potential wells at different depths.

The relationship between infrared spectrum diffraction parameters and potential well depth distribution.
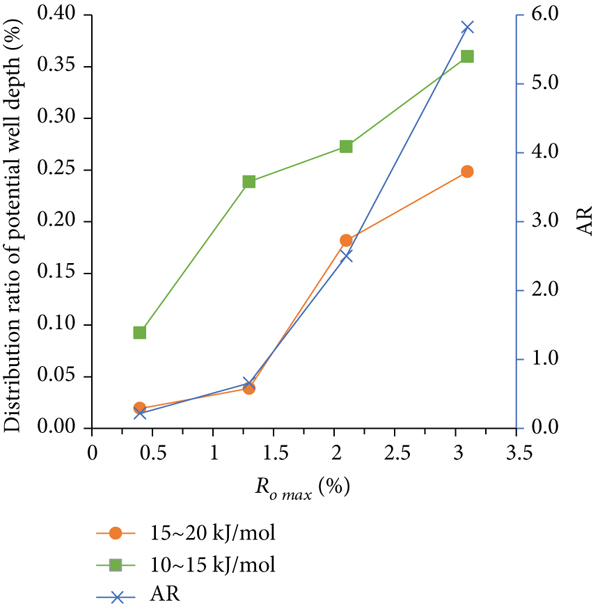

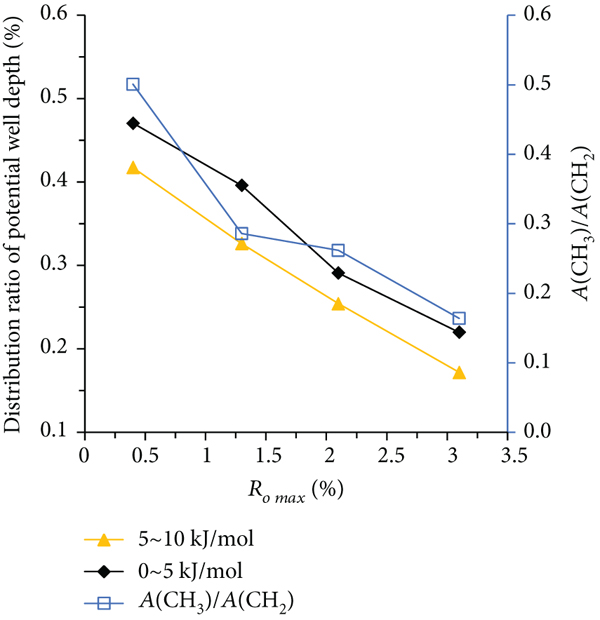

From Figures 10(a)–10(c), it can be seen that the aromaticity ratio, aromaticity, and condensation parameters of the coal sample structure show varying degrees of growth with the increase of coal sample metamorphism. This is consistent with the variation law of the distribution ratio of the adsorption potential well in the range of 10–15 kJ/mol and 15–20 kJ/mol with the metamorphic degree of coal sample, indicating that the aromatic carbon rate, aromaticity, and condensation degree parameters of the coal sample structure play a key role in the distribution ratio of the relatively high depth of adsorption potential wells. This shows that with the increase of the metamorphism degree of the coal sample, the degree of aromatization of the coal molecular structure increases, and the relative content of aliphatic functional groups decreases. Besides, the degree of condensation of the aromatic ring also increases, because in the process of coalification from low metamorphism to high metamorphism, oxygen-containing functional groups such as hydroxyl group and carboxyl group fall off [31]. As shown in Figures 10(a)–10(c), when the maximum vitrinite reflectance
6. Conclusions
In this paper, the number distribution of different depths of adsorption potential wells in natural coal with different ranks is calculated statistically. Four coal samples with different metamorphic degrees are tested and analyzed by infrared spectrum diffraction technology. The relationship between the structural parameters of functional groups of coal samples with different metamorphic degrees and the distribution of different depths of adsorption potential wells in coal samples is studied. The conclusions are as follows:
Through the statistical analysis of the calculation results of the potential well depth and the number of potential wells, the distribution diagram of the potential well depth and the number of potential wells of methane gas adsorbed by coal is obtained. Due to the diversity of natural coal components and the complexity of pore and fracture structure, the number of potential wells at different depths in natural coal with different metamorphic degrees has multipeak distribution characteristics. The distribution of adsorption potential well depth in coal samples shows different trends with the change of metamorphic degree With the increase of coal sample metamorphism degree, the aromatic carbon rate The different types and quantities of functional groups on the surface of coal samples lead to different forces between coal samples and methane gas molecules, thus affecting the distribution of different depths of methane adsorption potential wells in coal samples. The aromatic carbon rate
Footnotes
Data Availability
The raw/processed data required to reproduce these findings cannot be shared at this time as the data also forms part of an ongoing study.
Conflicts of Interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Acknowledgments
The authors would like to thank the National Natural Science Foundation of China (nos. 21373146) for financial assistance, and we thank Master Sun for polishing this manuscript.
